ED Procedural Sedation Analgesia an evidencebased review for

ED Procedural Sedation & Analgesia: an evidence-based review for 2008 David Messenger, MD, FRCPC Emergency Medicine & Critical Care Queen’s University

Challenges to Evidence-Based PSA Practice • Very few RCTs • Multiple drugs commonly used in practice • Multiple dosing protocols for each drug or combination • Multiple relevant outcomes of interest: • Safety/Adverse effects • Procedural success rates • Recovery times • No consistent way of reporting outcomes

Importance of Evidence. Based PSA Practice • Risk of serious adverse events small, but nonexistent with all drugs used for PSA • Wide variability in practice leads to wide variability in rates of adverse events - studies necessary to establish best practice • Political challenges persist, particularly in U. S. , re. use of many of these drugs by non-anesthesiologists • Important to have rigorous evidence for the safety and appropriateness of these agents in the hands of EPs

2007: The Year of ED PSA Research • Several significant publications: • Handful of observational studies • First published case report of aspiration requiring intubation associated with PSA • 3 RCTs (virtually doubles the existing level I evidence!) • Major clinical practice advisory on fasting pre. PSA • Clinical Practice Advisory on propofol sedation

The Plan • Ask 4 practical clinical questions and review 4 studies from 2007 that address them: • What’s the best short-acting sedative agent? • If I give an analgesic, which one is best? • Should I give supplemental oxygen to patients during PSA? • What’s the best way to monitor patients for respiratory depression during PSA?

Question 1 • Which of the ultra-short acting sedatives is best for deep sedation in the ED?

Short-Acting PSA Agents • Propofol use for ED PSA well-supported by several studies • 28 published series, ~4000 patients • Pooled rate of hypoxemia 5. 8% (range 0 - 30%) • Pooled rate of assisted ventilation 2. 1% (range 0 - 22%) • 1 intubation • Variable dosing strategies, adjunct drug use, supplemental oxygen use

Short-Acting Agents • Etomidate • Widely used in U. S. for PSA, but less evidence than for propofol • Several observational studies of etomidate for ED PSA § Dose range 0. 1 -0. 2 mg/kg • One prior RCT in adults: § Fentanyl+etomidate vs Fentanyl+midaz for shoulder reduction § Shorter duration of sedation with etomidate

Etomidate vs Propofol Ann Emerg Med. 2007. 49(1): 15 -22

Etomidate vs Propofol • Enrolled healthy adult patients requiring ED procedural sedation • Randomized to either: • Etomidate 0. 1 mg/kg bolus, then 0. 05 mg/kg every 3 -5 minutes • Propofol 1 mg/kg bolus, then 0. 5 mg/kg every 3 minutes • Not blinded

Etomidate vs Propofol • Primary outcome: • Subclinical respiratory depression § ETCO 2 change from baseline of ≥ 10 mm. Hg § Sa. O 2 < 92% § Airway obstruction (defined as loss of capnograph waveform) • Secondary outcomes: • Airway events/interventions • Depth of sedation (BIS score & OAA/S) • Patient pain/recall/satisfaction • Powered to detect a 20% difference in subclinical respiratory depression • assuming 30% baseline rate in propofol group


Etomidate vs Propofol

Etomidate vs Propofol • Conclusions: • No significant difference in rate of subclinical respiratory depression • No difference in clinical events • Myoclonus seen more frequently with etomidate • ? responsible for lower rate of procedural success

Etomidate vs Propofol • Methodologic issues: • Drug dosing § Actual etomidate dosing was higher than protocol specified (mean dose 0. 15 mg/kg vs 0. 1 mg/kg) § ? impact of higher dose on outcome • Non-blinded study § Difficult given physical properties of propofol • No adjunct analgesic given with sedative § Despite many physicians’ clinical practice and previous RCT of etomidate for ED PSA (Burton, 2002)

Etomidate vs Propofol • Relevance to our practice: • Etomidate still only available through Health Canada SAP and not widely adopted for ED PSA here • No compelling evidence in support of using etomidate over other drugs for PSA in Canada • Propofol has an established track record, and appears to be the better agent


Question 2 • Unanswered question for both etomidate and propofol: • What’s the impact of adjunct analgesics on relative safety of each agent?

Adjunct Analgesics with PSA • Is amnesia equivalent to analgesia? • Treating pre-procedure pain is important • With some procedures, patients experience the most painful stimulus while sedated § Fracture reduction § Incision/Drainage • Unclear if pain that isn’t remembered is important § Anesthetized patients have hemodynamic responses to pain § Oligoanalgesia -- ? sensitization of CNS, increased postoperative pain § Analgesics may reduce the amount of sedative required

Adjunct Analgesics with PSA • Inconsistent use of adjunct analgesics in studies of propofol & etomidate • 2007 Clinical Practice Guideline recommends propofol as a solo agent (Miner & Burton, 2007) § …based on several studies by a single investigator (who wrote the guideline) § NO RCT has compared propofol with and without an adjunct analgesic in the ED setting § Safety/efficacy • Remains an area ripe for investigation

Adjunct Analgesics • Fentanyl or morphine most commonly used • Major concern is risk of increased respiratory depression when opioids are used with sedatives • Ketamine also has analgesic properties, even at low doses (0. 1 - 0. 5 mg/kg) • An alternative to opioids?

Ketamine-Propofol for PSA • Growing popularity in Canada • Little ED evidence for/against its use in PSA • Review of non-EM literature demonstrates no consistent/convincing benefit to the combination § But very heterogeneous collection of studies • Potential benefits: • Ketamine has analgesic properties • Opposite hemodynamic effects - ? less hypotension • ? Less respiratory depression • Potential downsides: • Emergence reactions • ? Prolonged sedation compared to propofol alone

Ketamine-Propofol for PSA • Willman & Andolfatto. 2007. Ann Emerg Med. 49(1): 23 -30 • Prospective observational study of titrated samesyringe “ketofol” mixture • No control group • Mixture provided effective & apparently safe sedation in their patients • Difficult to rationalize simultaneous titration of two drugs with durations of action that differ by an order of magnitude

Adjunct Analgesics for PSA • Messenger et al. 2007. Acad Emerg Med. 14(5 s 1) [abstract] Low-dose ketamine versus fentanyl for analgesia during ED PSA with propofol: a randomized clinical trial • First ED RCT comparing adjunct analgesics administered with propofol PSA • Designed to assess safety (frequency of adverse events) as primary outcome • Hypothesis: ketamine will cause fewer and less severe adverse events than fentanyl

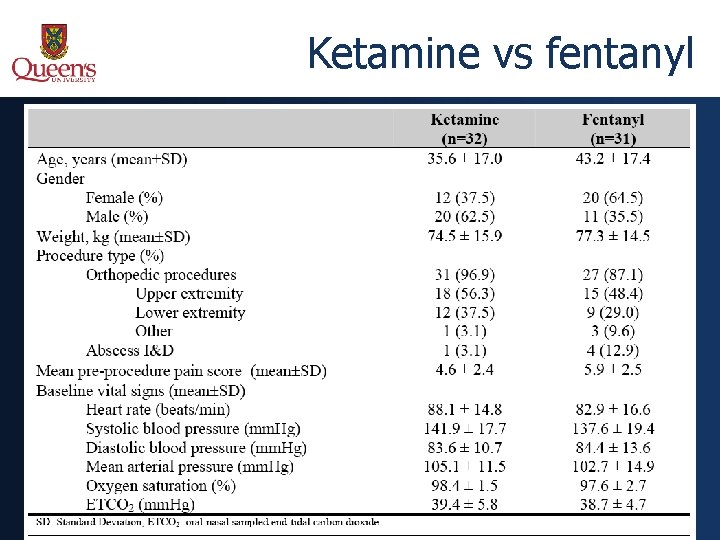
Ketamine vs fentanyl • Prospective, double-blind RCT • Attempted enrollment of consecutive patients • Inclusion Criteria: • Age 14 -65 years • ASA Class I-II • Orthopedic or minor surgical procedure • Exclusion Criteria: • • • Active cardiac, pulmonary, hepatic, renal disease Chronic opioid use/abuse Intoxicated History of psychotic disorder Weight > 130 kg Allergy to study medications

Ketamine vs fentanyl • 30 min washout period after opioids • Time = 0 (Study Drug Administered) • Ketamine 0. 3 mg/kg IV or Fentanyl 1. 5 µg/kg IV • Time = 2 min • Propofol 0. 4 mg/kg IV bolus • Propofol 0. 1 mg/kg IV q 30 s prn • Target: no withdrawal to trapezius squeeze

Ketamine vs fentanyl • 1: 1 nursing care • 2 physicians • Sedating MD • Operating MD • Continuous monitoring: • • ECG Sa. O 2 Oral/Nasal ETCO 2 NIBP at 3 -minute intervals • Supplemental O 2 not routinely administered unless desaturation < 92%1 1 ACEP Clinical Policy: Procedural Sedation and Analgesia in the Emergency Department, 2005

Ketamine vs fentanyl • Composite primary outcome: • Frequency of cardiorespiratory adverse events, graded by severity, using a 4 -point ordinal scale (none, mild, moderate, severe) • Each subject scored based on most severe adverse event

Ketamine vs fentanyl

Ketamine vs fentanyl • Secondary outcomes: Frequency of specific adverse events Cumulative propofol doses Times to recovery MDs’ rating of of sedation and analgesia Patients’ rating of recall, remembered pain, and overall satisfaction • 90% power to detect 3 -fold reduction in odds of an adverse event ( =0. 05) • • • Estimated total sample size: 124 subjects

Ketamine vs fentanyl • Trial terminated early after interim analysis of first 61 subjects completed • 63 patients enrolled prior to termination of enrollment
Ketamine vs fentanyl

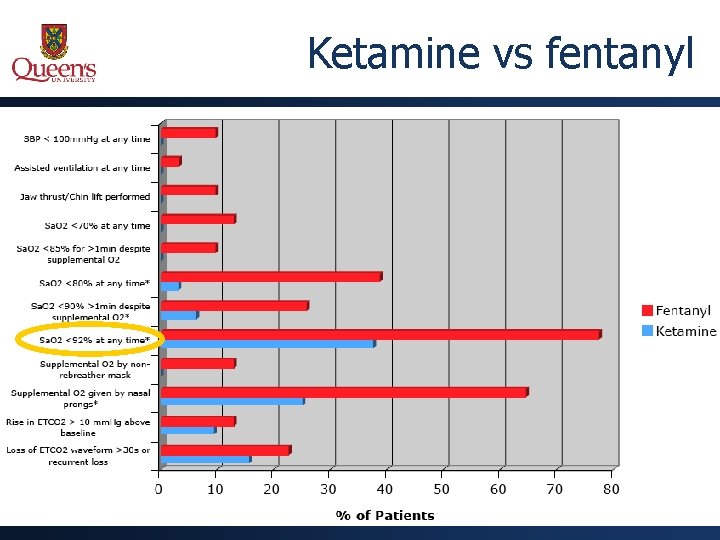
Ketamine vs fentanyl § p <0. 001 by Cochrane-Armitage Trend Test • Overall odds ratio 5. 1 (95% CI 1. 9 -13. 6)
Ketamine vs fentanyl

Ketamine vs fentanyl • Trend towards higher sedating propofol dose in ketamine group • 1. 5 mg/kg vs. 1. 1 mg/kg • Difference = 0. 4 mg/kg (95%CI 0. 0 -0. 7 mg/kg) • Higher mean propofol dose to maintain sedation in ketamine group • 0. 74 mg/kg vs. 0. 36 mg/kg • Difference = 0. 38 mg/kg (95%CI 0. 46 -0. 66 mg/kg)

Ketamine vs fentanyl • No differences observed with respect to: • • • Time to optimal sedation Duration of procedure Time to recovery MDs’ ratings of sedation and analgesia adequacy Patients’ ratings of recall, remembered pain and satisfaction • No emergence reactions observed

Ketamine vs fentanyl Limitations: • No comparison to propofol alone • Results apply only to drug doses studied • New Adverse Event Scale as primary outcome • No other validated rating of clinical adverse event severity • Results consistent using other comparators: § Frequency of individual adverse events § MDs’ ratings of adverse event severity • No supplemental oxygen given • ? Exaggerated number of adverse events

Ketamine vs fentanyl Conclusions: • Marked safety difference • Fewer adverse events at all severity levels in ketamine group, despite higher cumulative propofol doses • Fentanyl-propofol combo should be used with caution • No difference in efficacy • Similar recovery times • Similar MD and patient satisfaction • Ketamine appears to be the better choice if you’re going to use an adjunct analgesic with propofol

Question 3 • Should I routinely give patients supplemental oxygen during PSA?

Supplemental O 2 for PSA • Background: • Maintenance of spontaneous breathing a key goal of PSA • Transient hypoxemia may be frequent § As high as 30 -40% in some PSA studies • Most studies have used supplemental O 2 inconsistently… § “at discretion of treating MD”

Supplemental O 2 for PSA Study # 3 Ann Emerg Med. 2007. 49(1): 1 -8.

Supplemental O 2 for PSA • RCT design: • Blinded, randomized trial • Oxygen 2 lpm by n/c vs compressed air 2 lpm during sedation with fentanyl/midaz • Continuous Sa. O 2 and ETCO 2 monitoring • Primary outcome: • Oxygen desaturation <90% • Powered to detect 20% reduction in hypoxemia (assuming baseline rate of 30%)

Supplemental O 2 for PSA • 80 patients included in analysis • Drug doses & other baseline characteristics similar between groups • Frequency of hypoxia: • Room air group: 5/36 • O 2 group: 6/44 • Effect size 0%, 95%CI -15% - 15%

Supplemental O 2 for PSA • Secondary analyses: • Defined “respiratory depression” as any one or more of: § Sa. O 2 <90% § ETCO 2 >50 mm. Hg or absolute change from baseline of ≥ 10% § Loss of ETCO 2 waveform • No difference in RD between O 2 and control group § 45% vs 52%, effect size 7% (95% CI -29% - 15%)

Supplemental O 2 for PSA • Methodologic problems: • Did an interim analysis of data after 80/96 planned patients enrolled § In order to meet abstract submission deadline… • Lower incidence of hypoxemia (13. 9% vs anticipated 30%), so study ended early § Study originally powered to detect a 20% reduction, underpowered to detect a smaller difference • Limitations: • Only studied one drug combination for moderate sedation • ? Applicability of results to more potent sedatives

Supplemental O 2 for PSA • Conclusions: • No observed difference in hypoxemia when patients given routine O 2, but underpowered to show small difference • WHO CARES? ? ? • Does giving oxygen have a downside?

Supplemental O 2 for PSA • Patients may develop respiratory depression well before the Sa. O 2 drops • Hypoventilation, apnea, obstruction • MDs often miss RD prior to onset of hypoxemia • Deitch study: they missed it every time • Giving O 2 may further delay recognition of RD • Hypoxemia may take longer to develop • Patients may require more aggressive interventions to correct/treat RD if its recognition is delayed

Supplemental O 2 for PSA • Significance of respiratory depression and hypoxemia during PSA unclear • Complications with PSA are extremely rare, but do occur • Recent Canadian case report of aspiration requiring intubation after ED PSA (Cheung et al. , 2007) • EP’s should strive to minimize potential risks to patients at all times • Prevention, early recognition and early treatment of respiratory events should be a primary focus of physicians performing PSA

Question 4 • Is there a better way to monitor patients’ respiratory status during sedation than just the Sa. O 2?
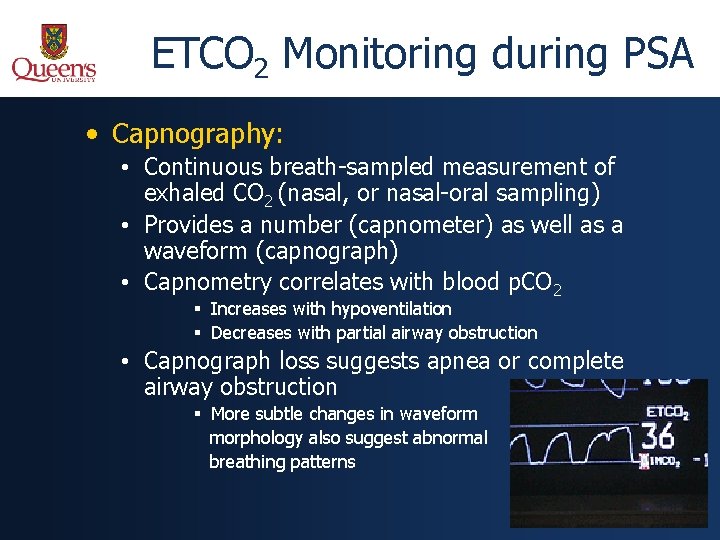
ETCO 2 Monitoring during PSA • Capnography: • Continuous breath-sampled measurement of exhaled CO 2 (nasal, or nasal-oral sampling) • Provides a number (capnometer) as well as a waveform (capnograph) • Capnometry correlates with blood p. CO 2 § Increases with hypoventilation § Decreases with partial airway obstruction • Capnograph loss suggests apnea or complete airway obstruction § More subtle changes in waveform morphology also suggest abnormal breathing patterns

ETCO 2 Monitoring during PSA • Growing literature suggests that capnography may be a valuable respiratory monitoring tool during PSA • This study among them…

ETCO 2 Monitoring during PSA Ann Emerg Med. 2007. 49(1): 9 -13

ETCO 2 Monitoring during PSA • Prospective convenience sample of 125 children sedated with propofol for fracture reduction • Monitored ETCO 2 via nasal sampling • All patients given 1 lpm O 2 by N/C • Outcomes of interest: • Hypoxemia (Sa. O 2 <90%) • Hypercarbia (ETCO 2 >50 mm. Hg or increase of 10 mm. Hg from baseline) • Apnea (loss of ETCO 2 waveform >30 s) • Airway interventions

ETCO 2 Monitoring during PSA • Capnography change preceded clinical detection of adverse events in 11/14 cases

ETCO 2 Monitoring during PSA • Similar study in adults Burton et al. , Acad Emerg Med. 2006. 13(5): 500 -504 • 60 PSA encounters in 59 patients • Defined abnormal capnography as: § ETCO 2 change from baseline of ≥ 10 mm. Hg (up or down) § ETCO 2 level ≤ 30 or ≥ 50 mm. Hg • All patients given O 2 2 lpm by N/C

ETCO 2 Monitoring during PSA • 20/60 encounters had predefined “acute respiratory events” observed § § Sa. O 2 <92% increased O 2 due to apnea, hypoventilation or desat BVM, airway insertion repositioning, patient stimulation, reversal agent • Abnormal ETCO 2 findings in 17/20 • ETCO 2 change preceded event in 14/20 (70%)

ETCO 2 Monitoring during PSA • Both studies suggest a benefit to ETCO 2 monitoring for early detection of adverse respiratory events • Performed better than clinician observation and oximetry monitoring in patients who received supplemental O 2 • What about patients breathing room air?
![ETCO 2 Monitoring during PSA • Messenger et al. CJEM 2007. 9(3) [abstract] • ETCO 2 Monitoring during PSA • Messenger et al. CJEM 2007. 9(3) [abstract] •](http://slidetodoc.com/presentation_image_h2/b6f59e62208376c700e70db68cd67add/image-59.jpg)
ETCO 2 Monitoring during PSA • Messenger et al. CJEM 2007. 9(3) [abstract] • Prospective observational study nested within RCT of analgesic adjuncts to propofol sedation • 63 patients breathing room air • Observed ETCO 2 changes relative to oxygen saturation § Hypoxemia = Sa. O 2 <92% § Abnormal ETCO 2: § ETCO 2 >50 mm. Hg § Rise/Fall of ≥ 10 mm. Hg from baseline § Absent waveform >30 s

ETCO 2 Monitoring during PSA • Hypoxemia observed in 36/63 patients • Abnormal capnography observed in 30/63 patients • Loss of waveform: 12/30 • ETCO 2 >50 mm. Hg: 6/30 • ETCO 2 rise ≥ 10 mm. Hg: 7/30 • ETCO 2 fall ≥ 10 mm. Hg: 11/30

ETCO 2 Monitoring during PSA Oxygen Desaturation Abnormal ETCO 2 YES NO YES 21 9 NO 15 18

ETCO 2 Monitoring during PSA • Conclusion: • ETCO 2 abnormalities do not appear to precede oxygen desaturation in patients breathing room air

ETCO 2 Monitoring during PSA • Research still to be done: • Does ETCO 2 monitoring actually help reduce the frequency of adverse respiratory events? • What are the ETCO 2 changes most likely to predict adverse respiratory events? • ETCO 2 monitoring: • Will likely evolve into standard of care for ED PSA… momentum is strong • For physicians familiar with its use and interpretation, likely will be useful for helping reduce adverse events • Use it if you have it (with supplemental O 2); otherwise, keep patients on room air and observe patient and pulse oximeter closely

Summary • Evidence supporting PSA practice is increasing, but much remains to be done • Need more studies focusing on: • Comparison of drugs and drug doses • Prevention and early detection of respiratory depression

Our 4 Questions: • Which short-acting Agent is best for ED PSA? • Propofol • Which adjunct analgesic is safest when given with short-acting sedatives? • Pick ketamine over fentanyl

Our 4 Questions: • Should I give my patients supplemental oxygen? • Not if you want to detect respiratory depression early…. • Unless…. • How can I better monitor my patients’ breathing during PSA? • Consider capnography in patients if you give supplemental oxygen


Summary • Etomidate does not appear to offer any advantage over propofol for ED PSA • Stick with the white stuff… • Ketamine is a safer adjunct analgesic for propofol than fentanyl • Beware opioid-propofol combinations for PSA • Supplemental oxygen may not reduce the frequency of hypoxemia, and may only serve to delay the recognition of respiratory depression during ED PSA • Consider keeping your patient on room air if no other respiratory monitoring device is used • ETCO 2 monitoring appears to identify RD prior to adverse respiratory events in preoxygenated patients • Ideal practice appears to be to provide O 2 and incorporate capnometry into PSA monitoring
- Slides: 68