Ectocarpus siliculosus Filamentous cosmopolitan brown alga mostly from

Ectocarpus siliculosus • Filamentous, cosmopolitan brown alga – mostly from temperate seas • One of the best-studied seaweeds • The first fully-sequenced multicellular alga – ongoing genome annotation • > 300 fully-characterized strains in public-domain culture collections (CCAP, KU-MACC)

So what about Ectocarpus? • Ectocarpus accumulates iodine, but at much lower levels than Laminaria (data from Roscoff & Vernaison) • Genome annotation has revealed the presence of one vanadium haloperoxidase gene • X-ray absorption spectroscopy using synchrotron radiation (XAS) on Ectocarpus
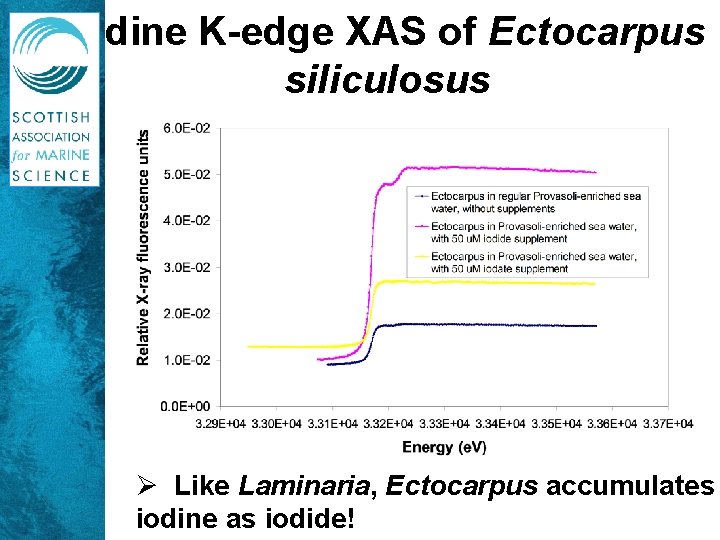
Iodine K-edge XAS of Ectocarpus siliculosus Ø Like Laminaria, Ectocarpus accumulates iodine as iodide!

Iodine metabolism in Ectocarpus • At present probably more questions than answers! • Does iodide have an apoplastic antioxidant function in Ectocarpus? • What does the genome tell us about antioxidant systems? • Does Ectocarpus produce iodinated / halogenated metabolites, are there iodo/halocarbons? • Ongoing work - NERC SOLAS grant to Lucy J. Carpenter (York) and FCK

Outlook and open questions • Mechanism of cell wall / apoplastic accumulation, storage of iodide? • How does the mobilization upon oxidative stress work? • Giant kelp / Macrocystis?

So what about Greece and the Mediterranean Sea? • Very little is known about halogen fluxes in Mediterranean coastal systems! • In particular, molecular iodine and halocarbon emissions from Mediterranean seaweeds are seriously understudied

So what about Greece and the Mediterranean Sea? • Some tropical/Mediterranean seaweeds are known to be strong iodine accumulators: Asparagopsis taxiformis (Rhodophyta) • Other candidates (known for strong halogen metabolism, albeit not iodine accumulation): Rhodophyta - Gracilaria sp. , Ceramium sp. , Dasya sp. , Laurencia sp. Phaeophyta – Ectocarpales, Fucales (Sargassum sp. ) • Deep-water kelp (Laminariales)?

Deep-water kelp in the Mediterranean Sea • Laminaria rodriguezii, mainly known from western / central Mediterranean • Not known from Greece so far – lack of exploration of relevant depth range (> 50 m)

Deep-water kelp forests in the tropics: An analogy to the Mediterranean Sea? Graham et al. , 2007: Deep-water kelp refugia as potential hotspots of tropical marine diversity and productivity. - PNAS 104 (42), 16576 -80

Deep-water kelp in the Mediterranean Sea • What are the implications for marine and atmospheric halogen chemistry in the region?

A place where you can smell the tide level… Roscoff, Brittany, France

Acknowledgements -- Peter M. H. Kroneck (University of Konstanz, Germany) -- Sonja Woitsch, Markus Weiller (dto. ) -- Lucy J. Carpenter and Carl Palmer (University of York, UK) -- Gordon Mc. Figgans (University of Manchester, UK) -- Philippe Potin (CNRS, Roscoff, France) -- Martin Feiters (University of Nijmegen, The Netherlands) -- Wolfram Meyer-Klaucke and Gerd Wellenreuther (European Molecular Biology Laboratory, Hamburg, Germany) -- Eva M. Boneberg (Biotechnologie Institut Thurgau, Switzerland, formerly at University of Konstanz) -- Alison Butler (University of California, Santa Barbara, USA) -- George Luther & Tim Waite (University of Delaware, Lewes, USA) -- Rafael Abela & Daniel Grolimund (Swiss Light Source, Paul Scherrer Institute, Villigen, Switzerland)

Thank you. Dunstaffnage Marine Laboratory, Oban, Argyll / West Highlands, Scotland

Reserve slides follow
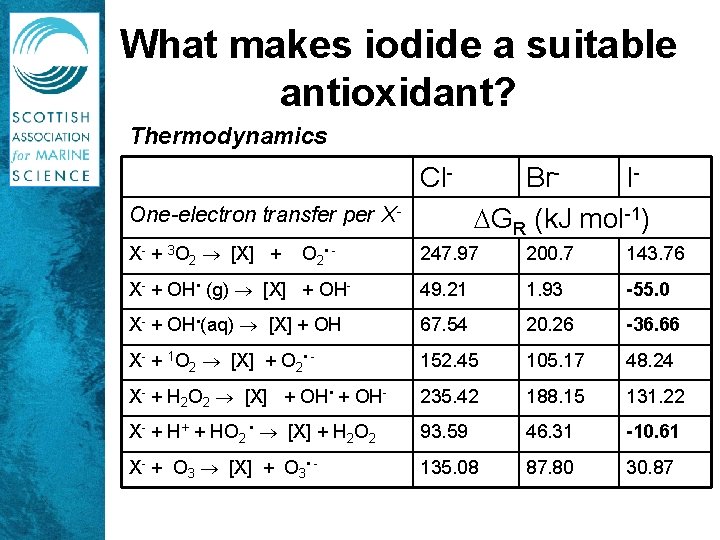
What makes iodide a suitable antioxidant? Thermodynamics Cl. One-electron transfer per XX- + 3 O 2 [X] + O 2 • - Br. I GR (k. J mol-1) 247. 97 200. 7 143. 76 X- + OH • (g) [X] + OH- 49. 21 1. 93 -55. 0 X- + OH • (aq) [X] + OH 67. 54 20. 26 -36. 66 X- + 1 O 2 [X] + O 2 • - 152. 45 105. 17 48. 24 X- + H 2 O 2 [X] + OH • + OH- 235. 42 188. 15 131. 22 X- + H+ + HO 2 • [X] + H 2 O 2 93. 59 46. 31 -10. 61 X- + O 3 [X] + O 3 • - 135. 08 87. 80 30. 87
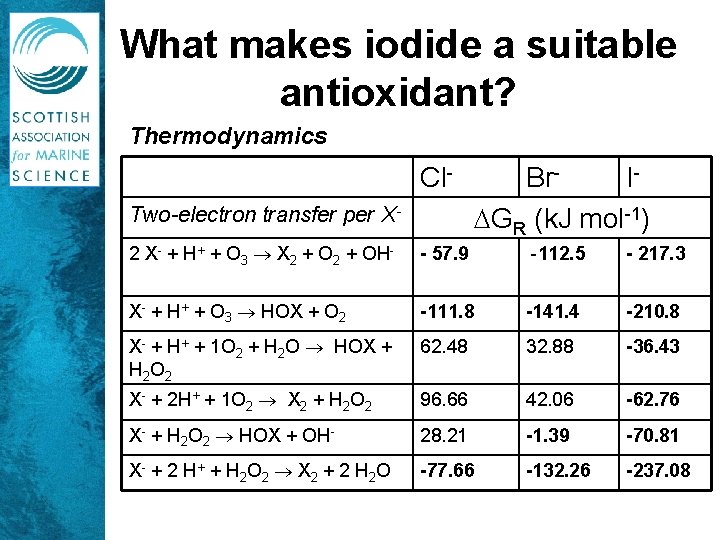
What makes iodide a suitable antioxidant? Thermodynamics Cl. Two-electron transfer per X- Br. I GR (k. J mol-1) 2 X- + H+ + O 3 X 2 + OH- - 57. 9 -112. 5 - 217. 3 X- + H+ + O 3 HOX + O 2 -111. 8 -141. 4 -210. 8 X- + H+ + 1 O 2 + H 2 O HOX + H 2 O 2 62. 48 32. 88 -36. 43 X- + 2 H+ + 1 O 2 X 2 + H 2 O 2 96. 66 42. 06 -62. 76 X- + H 2 O 2 HOX + OH- 28. 21 -1. 39 -70. 81 X- + 2 H + + H 2 O 2 X 2 + 2 H 2 O -77. 66 -132. 26 -237. 08

What makes iodide a suitable antioxidant? Kinetics Compound k 12 (M-1 s-1) O 3 reactions with IBr. Cl. Ascorbate Glutathione 1. 2 x 109 2. 48 x 102 < 3 x 10 -3 4. 8 x 107 2. 5 x 106 Singlet oxygen (1 O 2) reactions with I 1 x 108 (aprotic solvents) 8. 7 x 105 (p. H ~ 7) Br 1. 0 x 103 Cl 1. 0 x 103 Ascorbate 8. 3 x 106 Glutathione 2. 4 x 106 (in D 2 O, 310 K, p. D = 7. 4)

What makes iodide a suitable antioxidant? Kinetics Compound k 12 (M-1 s-1) OH radical ( OH) reactions with IAscorbate Glutathione Dimethyl sulfoniopropionate Dimethyl sulfide Dimethyl sulfoxide 1. 2 x 1010 1. 1 x 1010 1. 3 x 1010 (p. H = 5. 5) 3 x 108 1. 9 x 1010 6. 6 x 109 Superoxide (O 2 -) reactions with I 3 Ascorbate Glutathione 1 x 108 (no data available for I-) 2. 7 x 105 (p. H= 7. 4) 2. 4 x 105 (p. H= 7. 8)

What makes iodide a suitable antioxidant? Kinetics Compound k 12 (M-1 s-1) Hydrogen peroxide (H 2 O 2) reactions with I 0. 69 Br 2. 3 x 10 -5 Cl 1. 1 x 10 -7 Ascorbate 2 x 100 Glutathione 2 – 20 x 100 Glutathione peroxidase 6 x 107
- Slides: 19