Ecology BIO C 322 Biogeochemical Cycles Biogeochemical Cycles

Ecology (BIO C 322) Biogeochemical Cycles

Biogeochemical Cycles • Cycling of nutrients. – Reservoir pool (non-biological) – unavailable pool; – Cycling pool (exchanging rapidly b/w organisms & immediate environment) – available / exchangeable pool. • Gaseous type: Reservoir in atmosphere or hydrosphere. • Sedimentary type: Reservoir in earth’s crust.

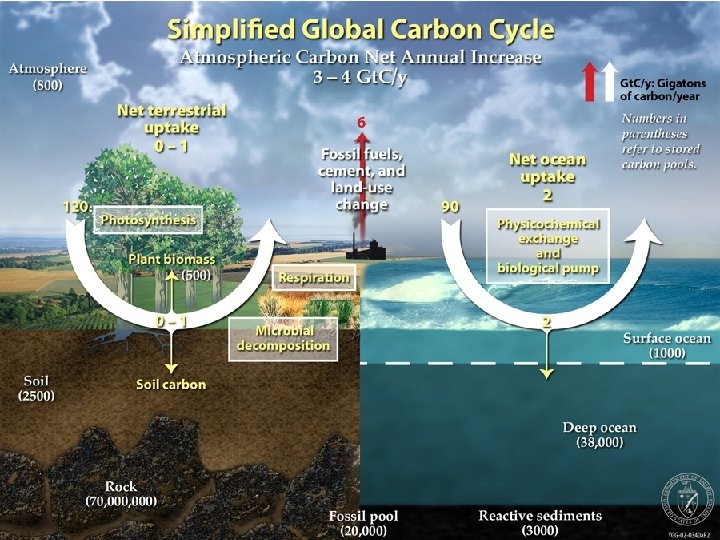
Carbon Cycle • Atmospheric C pool small. • Larger pool: C in oceans, fossil fuels (sediments). • Oceans effective buffers of atmospheric CO 2. • CO 2 + H 2 O ↔ H 2 CO 3 ↔ H+ + CO 32 • 2 other forms of C in atmosphere: CO, CH 4.

Nitrogen Cycle • Atmosphere main reservoir (78%). • Atmospheric N fixed (biofixation) by microbes or lightning. • N in plant/animal protoplasm broken down from org to inorganic forms by decomposers. • Organic N NH 4+ & nitrates Readily used by plants, or, • N returned back to atmosphere by action of denitrifying bacteria (Pseudomonas).
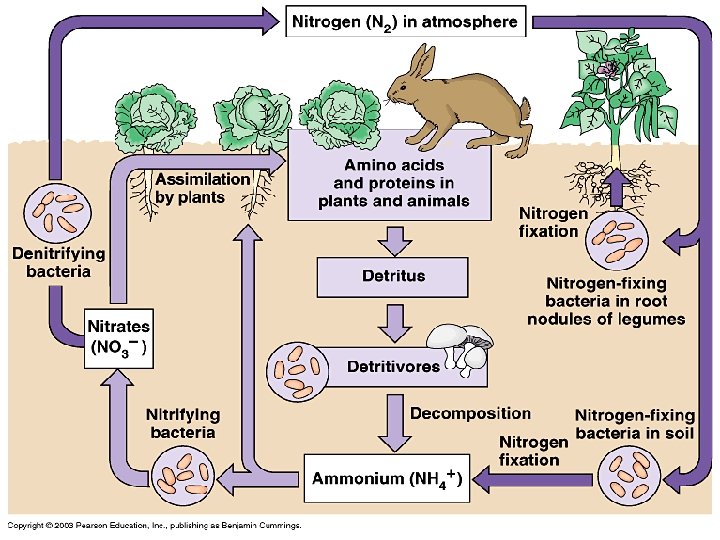

Nitrogen Fixation • Microorganisms: – Free living bacteria: Azotobacter (aerobic) & Clostridium (anaerobic). – Symbiotic nodule bacteria on legume plants: Rhizobium. – Cyanobacteria: Anabena, Nostoc. • Nitrogenase catalyzes splitting of N 2. • Endothermic process - Energy obtained from glucose/ATP.

Cyanobacterial Biofertilizers in Rice Agriculture • Aquatic fern Azolla contains symbiotic Nfixer Anabena. • Before planting rice seedlings, the fern planted. • Soil becomes N-rich. • Suitable for rice cultivation – S. E. Asia.

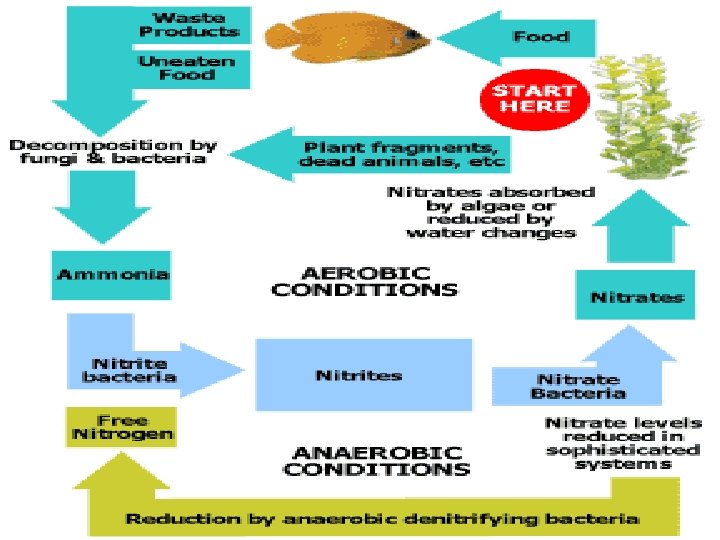
Ammonification / Mineralization • End result of N fixation: Incorporated into organic compounds. • Next, waste products (urea, uric acid) excreted or organisms die. • Next, acted upon by ammonifying organisms (bacteria, fungi) in soil.

• Ammonifying organisms decompose amino acids & other N-containing org compounds. • Production of inorganic NH 3 or NH 4+ (Ammonification). • Ammonification is an exothermic process; energy released used in maintenance of life processes by bacteria.

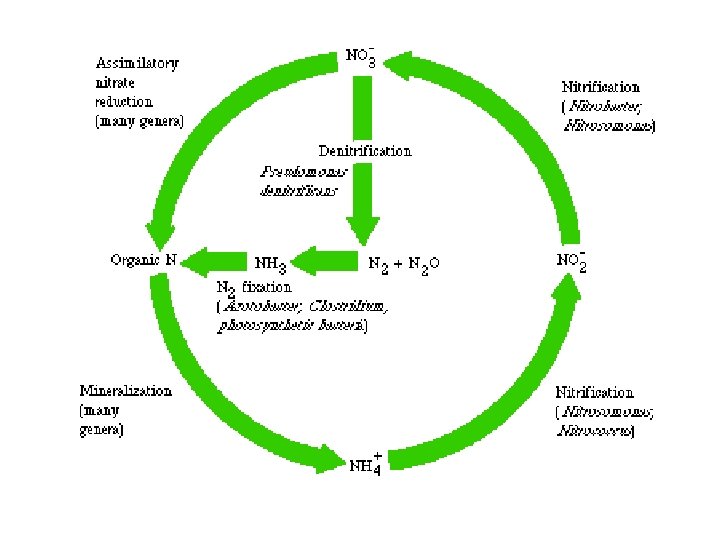
Nitrification – by Nitrifying bacteria • Conversion (by oxidation) of NH 3 or NH 4+ to nitrite (NO 2 -) or nitrate (NO 3 -). • Nitrosomonas: • 2 NH 3 + 3 O 2 2 NO 2 - + 2 H+ + 2 H 2 O • Nitrite is toxic to plants but rarely accumulates in soil. • Nitrobacter: • 2 NO 2 - + O 2 2 NO 3 • Both are chemosynthetic autotrophs, obtaining their energy from nitrification (exothermic). • Nitrate formed is easily absorbed by plants.

Denitrification • Reduction of nitrate to gaseous N 2, nitric oxide (NO) or nitrous oxide (N 2 O) (exothermic). • Effected by Pseudomonas, fungi, etc. • Evolved N 2 returned to atmospheric pool. • Occurs under anaerobic conditions (e. g. in poorly aerated soils). • Little denitrification in oxygen-rich surface waters of lakes and seas.

Nitrogen Too much… • Enrichment of an ecosystem with N opportunistic weedy species adapted to high nutrient conditions. • Consequence: reduced biodiversity.

Practice Concept • A low availability of soil nitrogen reduces not only net primary production (total production of plant tissues) but also the nitrogen concentration of the plant tissues that are produced. • The net effect is a lower input return of nitrogen in the form of dead organic matter. • Low nutrient (nitrogen) concentrations in the dead organic matter promote immobilization of nutrients from soil & water to meet the nutrient demands of decomposer populations. • This immobilization effectively reduces nutrient availability to the plants, adversely affecting primary productivity.

Nitrogen & Photosynthesis • Rate of photosynthesis related to leaf nitrogen conc. • Rubisco & chlorophyll N-rich molecules.

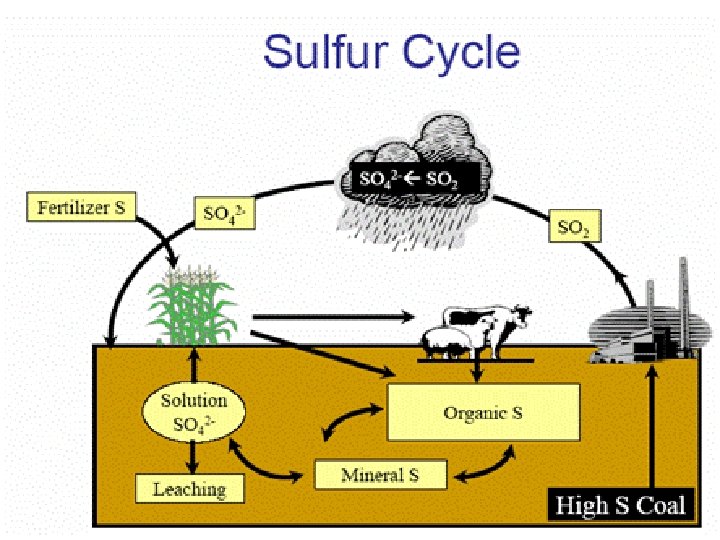
Sulphur cycle - Sedimentary Nutrient Cycle • S is an imp component of some amino acids. • Uptake of sulphate (SO 42 -) through plant roots. • Inorganic sulphates: Ca. SO 4, Na 2 SO 4. • Organic sulphates: Amino acids released by decomp of dead organisms or excreted matter. • Under anaerobic conditions, sulphate reduced to elemental S or H 2 S by Desulfovibrio.

Sulphur in Sediments • Present as ferrous sulphide (Fe. S), ferric sulphide (Fe 2 S 3, pyrite), calcium sulphate (Ca. SO 4). • Pyrite is an important source of Fe and S for organisms in sedimentary deposits such as marshes.

Importance of S Cycle • Photosynthesis mainly releases O 2 in the atmosphere. • But denitrifying and sulphate-reducing bacteria are also important. • Release of the large amount of oxygen bound up in nitrates and sulphates.

• High conc. of H 2 S in deeper portions of aquatic ecosystems detrimental to higher life forms. • But some sulphur bacteria (chemosynthetic autotrophs) depend on H 2 S to fix CO 2. • 6 CO 2 + 12 H 2 S C 6 H 12 O 6 + 6 H 2 O + 12 S • In deep sea hydrothermal vents (70 -100ºC), H 2 S production is due to sulphate reducers.

Sulphur in Atmosphere S in atmosphere comes from: • Decomposition of organic matter; • Combustion of fossil fuels; • Ocean surfaces; • Volcanic eruptions. • SO 2 SO 3 H 2 SO 4

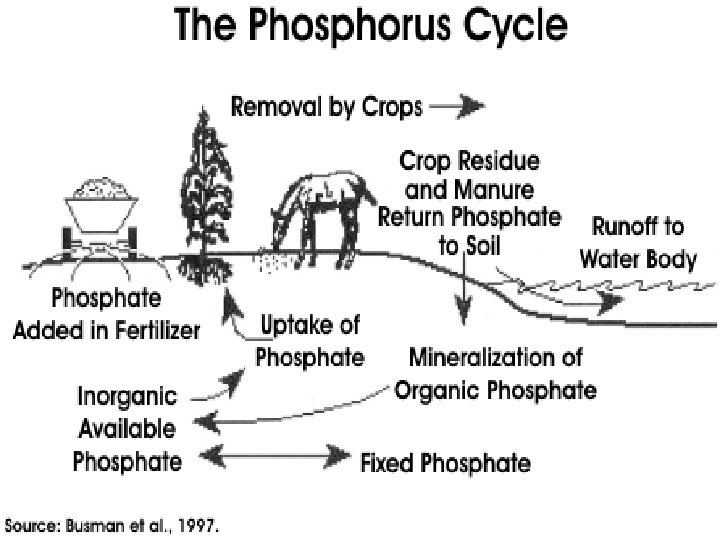
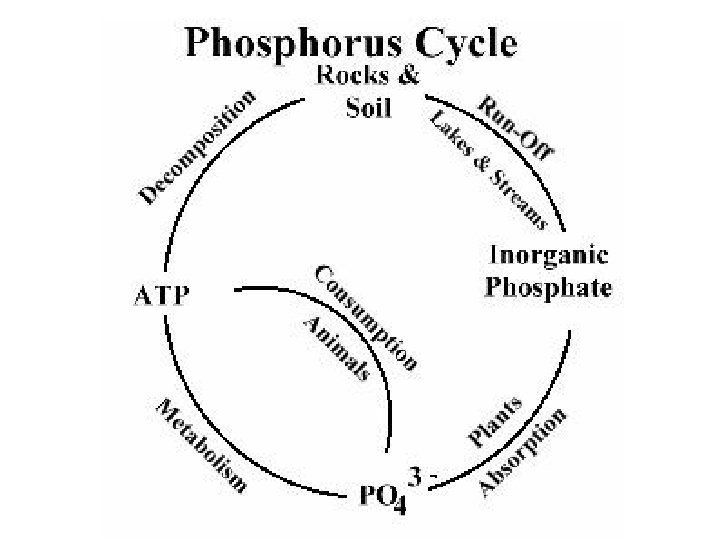
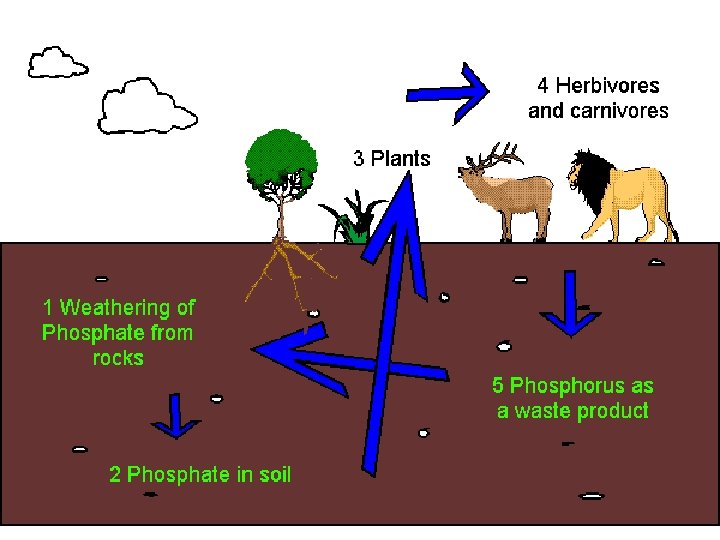
Phosphorus Cycle • P is an important constituent of nucleic acids, phospholipids, etc. • Organic phosphate comes from bones, teeth, dead organic matter, excretion. • Inorganic phosphate comes from erosion and mining from sediments. • Seabirds transfer P from sea to land (guano deposits or seabird droppings).
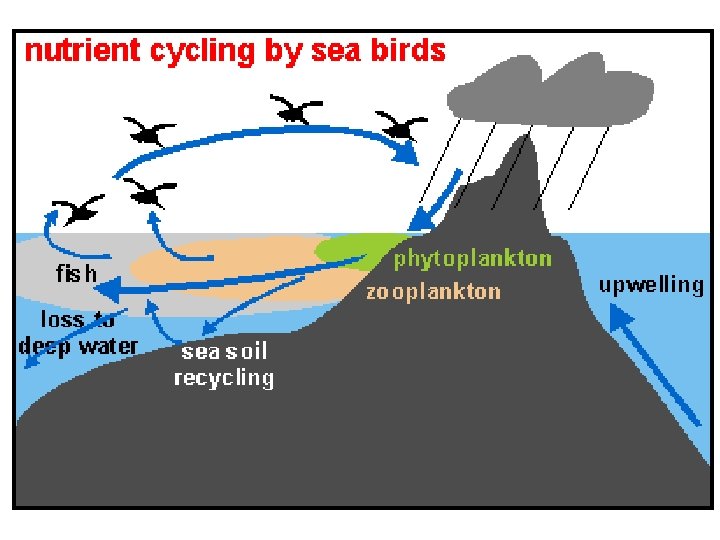

Guano Deposits by Sea Birds

• Sedimentation involves binding of P with cations (Ca, Fe, Mg) to form insoluble salts. • Sediments are important source of P for aquatic organisms. • Carbon, Nitrogen, Sulphur cycles have atmospheric phases. • But P cycle is mainly sedimentary. • Both S & P get precipitated by binding cations, & lost to sediments for extended time periods.
- Slides: 30