ECHELON1 Phase III Trial of Firstline Brentuximab Vedotin

ECHELON-1: Phase III Trial of First-line Brentuximab Vedotin + AVD Chemotherapy in Advanced Classical Hodgkin Lymphoma Integrating New Hematology Findings Into Practice: Independent Conference Coverage of ASH 2017, * December 9 -12, Atlanta, Georgia *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. This activity is supported by educational grants from Abb. Vie; Astra. Zeneca; Celgene Corporation; Genentech; Janssen Biotech, Inc administered by Janssen Scientific Affairs, LLC; Jazz Pharmaceuticals; Novartis Pharmaceuticals Corporation; Pharmacyclics Inc; Seattle Genetics; and Takeda Oncology.

First-line Brentuximab Vedotin + Chemotherapy in Advanced c. HL (ECHELON-1): Background § Among pts with newly diagnosed advanced c. HL, up to 40% may develop relapsed/refractory disease following standard first-line ABVD therapy[1, 2] § Brentuximab vedotin: antibody–drug conjugate comprising CD 30 -directed m. Ab linked to microtubule inhibitor MMAE[3, 4] – Approved for c. HL after failure of auto. HSCT or ≥ 2 prior chemotherapy regimens and as consolidation post‑auto. HSCT for increased-risk HL[3] – Phase I trial reported a CR of 96%, 5 -yr FFS of 92%, and 5 -yr OS of 100% with BV + AVD in pts with newly diagnosed bulky stage IIA or stage IIB-IV c. HL [5, 6] – Most common AEs: neutropenia, peripheral neuropathy[3, 4] § Current phase III study compared efficacy, safety of first-line BV + AVD vs ABVD in pts with previously untreated advanced c. HL[7, 8] References in slidenotes Slide credit: clinicaloptions. com
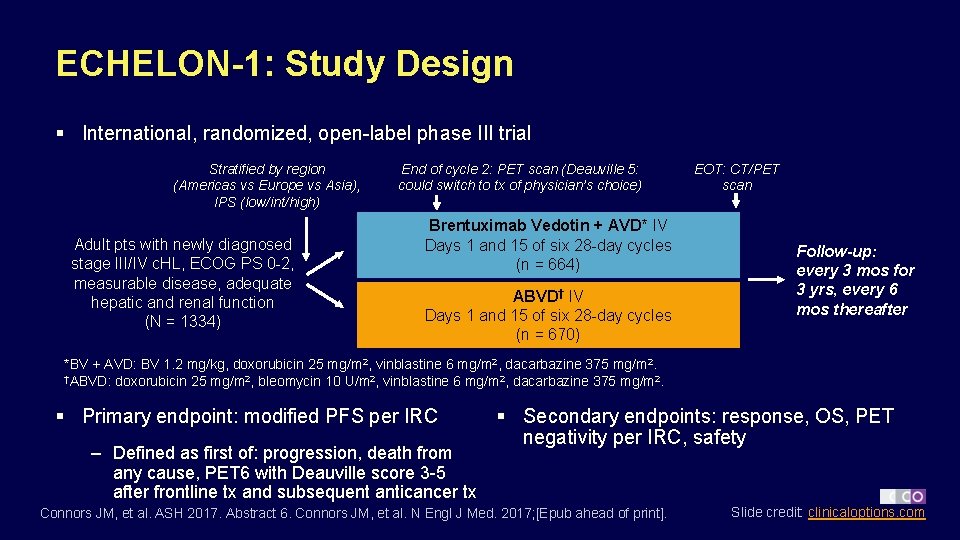
ECHELON-1: Study Design § International, randomized, open-label phase III trial Stratified by region (Americas vs Europe vs Asia), IPS (low/int/high) Adult pts with newly diagnosed stage III/IV c. HL, ECOG PS 0 -2, measurable disease, adequate hepatic and renal function (N = 1334) End of cycle 2: PET scan (Deauville 5: could switch to tx of physician’s choice) Brentuximab Vedotin + AVD* IV Days 1 and 15 of six 28 -day cycles (n = 664) ABVD† IV Days 1 and 15 of six 28 -day cycles (n = 670) EOT: CT/PET scan Follow-up: every 3 mos for 3 yrs, every 6 mos thereafter *BV + AVD: BV 1. 2 mg/kg, doxorubicin 25 mg/m 2, vinblastine 6 mg/m 2, dacarbazine 375 mg/m 2. †ABVD: doxorubicin 25 mg/m 2, bleomycin 10 U/m 2, vinblastine 6 mg/m 2, dacarbazine 375 mg/m 2. § Primary endpoint: modified PFS per IRC – Defined as first of: progression, death from any cause, PET 6 with Deauville score 3 -5 after frontline tx and subsequent anticancer tx § Secondary endpoints: response, OS, PET negativity per IRC, safety Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com
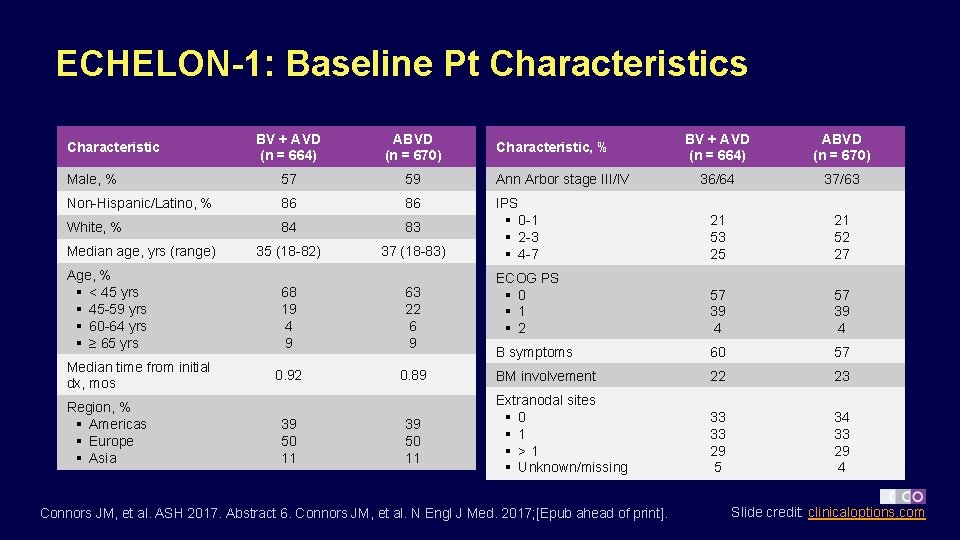
ECHELON-1: Baseline Pt Characteristics BV + AVD (n = 664) ABVD (n = 670) 36/64 37/63 IPS § 0 -1 § 2 -3 § 4 -7 21 53 25 21 52 27 ECOG PS § 0 § 1 § 2 57 39 4 B symptoms 60 57 BM involvement 22 23 Extranodal sites § 0 § 1 § >1 § Unknown/missing 33 33 29 5 34 33 29 4 BV + AVD (n = 664) ABVD (n = 670) Male, % 57 59 Ann Arbor stage III/IV Non-Hispanic/Latino, % 86 86 White, % 84 83 35 (18 -82) 37 (18 -83) Characteristic Median age, yrs (range) Age, % § < 45 yrs § 45 -59 yrs § 60 -64 yrs § ≥ 65 yrs Median time from initial dx, mos Region, % § Americas § Europe § Asia 68 19 4 9 63 22 6 9 0. 92 0. 89 39 50 11 Characteristic, % Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com
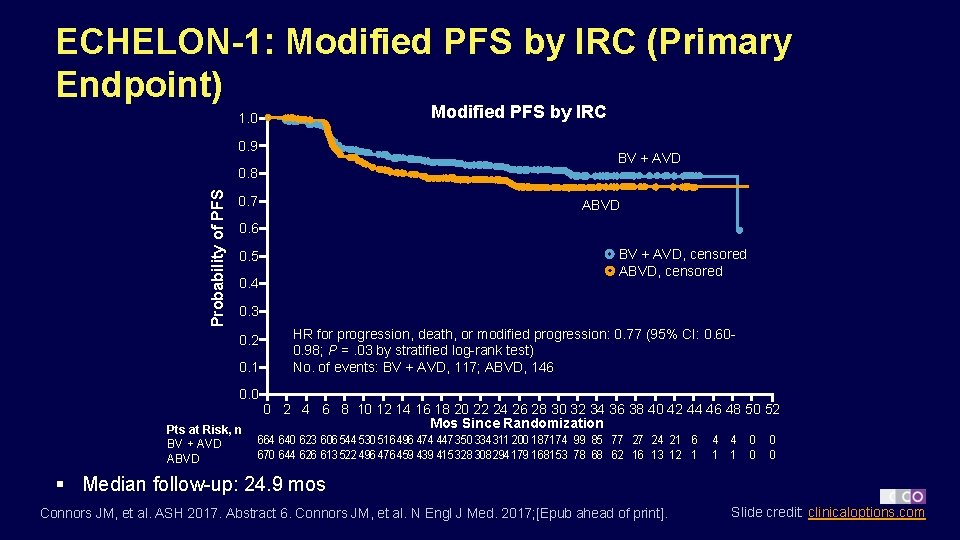
ECHELON-1: Modified PFS by IRC (Primary Endpoint) Modified PFS by IRC 1. 0 0. 9 BV + AVD Probability of PFS 0. 8 0. 7 ABVD 0. 6 BV + AVD, censored ABVD, censored 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 Pts at Risk, n BV + AVD ABVD HR for progression, death, or modified progression: 0. 77 (95% CI: 0. 600. 98; P =. 03 by stratified log-rank test) No. of events: BV + AVD, 117; ABVD, 146 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 32 34 36 38 40 42 44 46 48 50 52 Mos Since Randomization 664 640 623 606 544 530 516 496 474 447 350 334311 200 187174 99 85 77 27 24 21 6 670 644 626 613 522 496 476 459 439 415 328 308294 179 168153 78 68 62 16 13 12 1 4 0 1 0 0 0 § Median follow-up: 24. 9 mos Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com
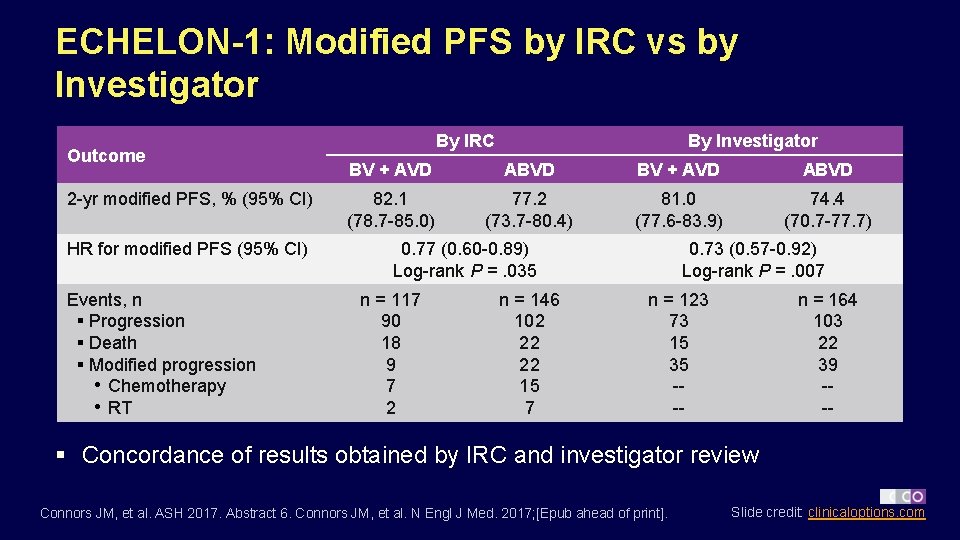
ECHELON-1: Modified PFS by IRC vs by Investigator Outcome 2 -yr modified PFS, % (95% CI) HR for modified PFS (95% CI) Events, n § Progression § Death § Modified progression • Chemotherapy • RT By IRC By Investigator BV + AVD ABVD 82. 1 (78. 7 -85. 0) 77. 2 (73. 7 -80. 4) 81. 0 (77. 6 -83. 9) 74. 4 (70. 7 -77. 7) 0. 77 (0. 60 -0. 89) Log-rank P =. 035 n = 117 90 18 9 7 2 n = 146 102 22 22 15 7 0. 73 (0. 57 -0. 92) Log-rank P =. 007 n = 123 73 15 35 --- n = 164 103 22 39 --- § Concordance of results obtained by IRC and investigator review Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com

ECHELON-1: Modified PFS by IRC in Key Prespecified Subgroups Event, n/N (%) Subgroup Overall Age: < 60 yrs ≥ 60 yrs < 45 yrs ≥ 45 yrs Region: Americas North America Europe Asia IPS: 0 -1 2 -3 4 -7 Stage III Stage IV B symptoms: yes no Extranodal sites: 0 1 >1 Sex: male female BV + AVD 117/664 (17. 6) 93/580 (16. 0) 24/84 (28. 6) 70/451 (15. 5) 47/213 (22. 1) 41/261 (15. 7) 38/250 (15. 2) 62/333 (18. 6) 14/70 (20. 0) 22/141 (15. 6) 57/354 (16. 1) 38/169 (22. 5) 40/237 (16. 9) 77/425 (18. 1) 77/399 (19. 3) 40/265 (15. 1) 40/217 (18. 4) 36/217 (16. 6) 39/194 (20. 1) 63/378 (16. 9) 53/286 (18. 5) ABVD 146/670 (21. 8) 117/568 (20. 6) 29/102 (28. 4) 83/423 (19. 6) 63/247 (25. 5) 58/262 (22. 1) 57/247 (23. 1) 74/336 (22. 0) 14/72 (19. 4) 25/141 (17. 7) 68/351 (19. 4) 53/178 (29. 8) 43/246 (17. 5) 102/421 (24. 2) 94/381 (24. 7) 52/289 (18. 0) 39/228 (17. 1) 45/223 (20. 2) 57/193 (29. 5) 90/398 (22. 6) 56/272 (20. 6) 0. 1 HR (95% CI) 0. 77 (0. 60 -0. 98) 0. 73 (0. 56 -0. 96) 1. 01 (0. 59 -1. 73) 0. 73 (0. 53 -1. 01) 0. 86 (0. 59 -1. 26) 0. 65 (0. 44 -0. 97) 0. 60 (0. 39 -0. 90) 0. 83 (0. 59 -1. 17) 0. 91 (0. 43 -1. 93) 0. 83 (0. 47 -1. 48) 0. 79 (0. 56 -1. 13) 0. 70 (0. 46 -1. 07) 0. 92 (0. 60 -1. 42) 0. 71 (0. 53 -0. 96) 0. 74 (0. 55 -1. 01) 0. 79 (0. 52 -1. 20) 1. 04 (0. 67 -1. 62) 0. 75 (0. 48 -1. 16) 0. 67 (0. 44 -1. 00) 0. 71 (0. 51 -0. 97) 0. 86 (0. 59 -1. 26) 0. 5 1 HR Favors BV + AVD Favors ABVD Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com

ECHELON-1: Secondary Efficacy and Survival Outcomes BV + AVD (n = 664) ABVD (n = 670) CR* rate at end of randomized regimen 488 (73) 472 (70) ORR* at end of randomized regimen 569 (86) 553 (83) PET Deauville score 1 -2 after completing first-line tx 563 (85)† 537 (80)† PET Deauville score 4 -5 after completing cycle 2 § 1 -3 § 4 § 5 § Unavailable 588 (89) 26 (4) 21 (3) 29 (4) 577 (86) 28 (4) 30 (4) 35 (5) Endpoint, n (%) *Per IWG 2007 criteria. †P =. 03 per chi-square test. § Interim OS calculated with 67 out of 112 planned deaths for analysis – HR for BV + AVD vs ABVD: 0. 72 (95% CI: 0. 44 -1. 17; P =. 19) Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com

ECHELON-1: Subsequent Treatment BV + AVD (n = 662) ABVD (n = 659) 121 (18) 144 (22) Pts receiving systemic tx + RT, * n § Systemic tx received § RT received 80 52 111 52 Types of systemic tx ± RT received, * n § Chemotherapy § High-dose chemotherapy + transplant § Immunotherapy 66 36 10 99 54 16 RT only, * n 41 33 Subsequent Tx Pts receiving ≥ 1 subsequent anticancer tx, n (%) *Pts could receive ≥ 1 systemic tx ± RT. § Among pts receiving BV + AVD vs ABVD, 33% fewer received subsequent chemotherapy and 33% fewer received subsequent high-dose chemotherapy + transplant Connors JM, et al. ASH 2017. Abstract 6. Slide credit: clinicaloptions. com
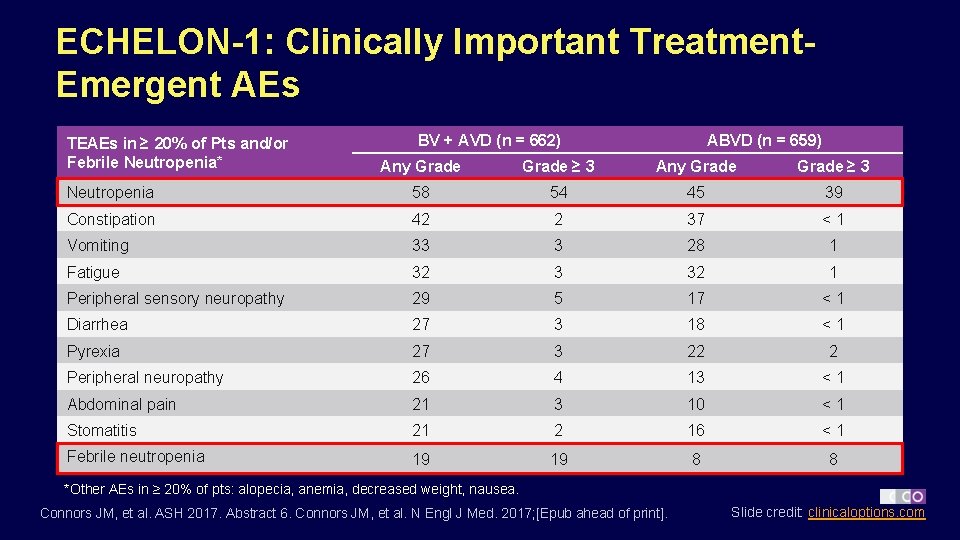
ECHELON-1: Clinically Important Treatment. Emergent AEs TEAEs in ≥ 20% of Pts and/or Febrile Neutropenia* BV + AVD (n = 662) ABVD (n = 659) Any Grade ≥ 3 Neutropenia 58 54 45 39 Constipation 42 2 37 <1 Vomiting 33 3 28 1 Fatigue 32 3 32 1 Peripheral sensory neuropathy 29 5 17 <1 Diarrhea 27 3 18 <1 Pyrexia 27 3 22 2 Peripheral neuropathy 26 4 13 <1 Abdominal pain 21 3 10 <1 Stomatitis 21 2 16 <1 Febrile neutropenia 19 19 8 8 *Other AEs in ≥ 20% of pts: alopecia, anemia, decreased weight, nausea. Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com
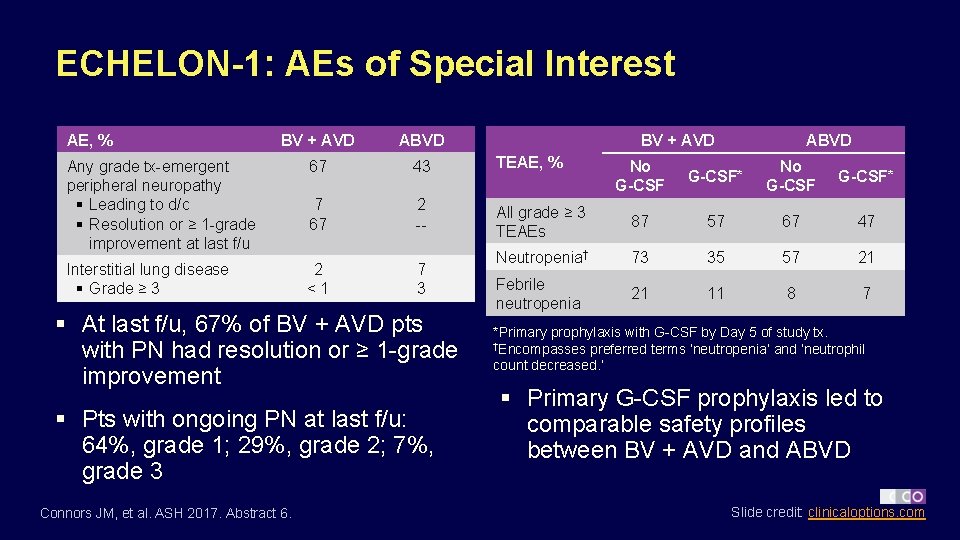
ECHELON-1: AEs of Special Interest AE, % BV + AVD ABVD Any grade tx-emergent peripheral neuropathy § Leading to d/c § Resolution or ≥ 1 -grade improvement at last f/u 67 43 TEAE, % 7 67 2 -- Interstitial lung disease § Grade ≥ 3 2 <1 7 3 § At last f/u, 67% of BV + AVD pts with PN had resolution or ≥ 1 -grade improvement § Pts with ongoing PN at last f/u: 64%, grade 1; 29%, grade 2; 7%, grade 3 Connors JM, et al. ASH 2017. Abstract 6. BV + AVD ABVD No G-CSF* All grade ≥ 3 TEAEs 87 57 67 47 Neutropenia† 73 35 57 21 Febrile neutropenia 21 11 8 7 *Primary prophylaxis with G-CSF by Day 5 of study tx. †Encompasses preferred terms ‘neutropenia’ and ‘neutrophil count decreased. ’ § Primary G-CSF prophylaxis led to comparable safety profiles between BV + AVD and ABVD Slide credit: clinicaloptions. com

ECHELON-1: Deaths § Fewer deaths observed with BV + AVD vs ABVD during treatment and follow-up – Among BV + AVD pts, 6 out of 7 neutropenia-associated deaths occurred in those who had not received G-CSF primary prophylaxis Deaths BV + AVD ABVD During tx, n (%) § Due to neutropenia or associated complications, n § Due to pulmonaryrelated toxicity, n 9 (1) 13 (2) 7 -- -- 11 During f/u, n (%) 19 (3) 26 (4) Connors JM, et al. ASH 2017. Abstract 6. Connors JM, et al. N Engl J Med. 2017; [Epub ahead of print]. Slide credit: clinicaloptions. com

ECHELON-1: Conclusions § In pts with previously untreated advanced c. HL, first-line BV + AVD showed significantly improved modified PFS by IRC vs ABVD – Risk of PD, death, or need for further anticancer tx reduced by 23% (HR: 0. 77; log-rank P =. 035) – Similar results obtained by investigator review § BV + AVD associated with higher rates of PN, neutropenia but lower rates of interstitial lung disease, pulmonary toxicity–related deaths vs ABVD – At last f/u, 67% of BV + AVD pts with PN had resolution or ≥ 1 -grade improvement – Primary prophylaxis with G-CSF led to comparable safety profiles between BV + AVD vs ABVD § Investigators conclude that bleomycin-free BV + AVD is a more effective first-line therapy than ABVD with a manageable safety profile for pts with untreated advanced c. HL – Recommend primary prophylaxis with G-CSF for all pts receiving BV + AVD Connors JM, et al. ASH 2017. Abstract 6. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASH 2017! Short slideset summaries of all the key data Additional CME-certified analyses with expert faculty commentary on all the key studies in: § § § Leukemias Lymphomas/CLL Myeloma Nonmalignant hematology Hot topics in hematology clinicaloptions. com/oncology
- Slides: 14