ECCO 2009 GI Update Focus on EGFR Inhibitors

ECCO 2009 GI Update: Focus on EGFR Inhibitors in Metastatic Colorectal CA (m. CRC) Scott Berry Sunnybrook Odette Cancer Centree www. Oncology. Education. ca

Overview • EGFR Inhibitors in m. CRC – NEW DATA: – Pmab in combination with chemo data » 1 st Line with FOLFOX » 2 nd Line with FOLFIRI – Cetuximab in combination with CAPOX/FOLFOX - MRC COIN Trial – Updated cetuximab 1 st line data • CRYSTAL - updated • OPUS – updated www. Oncology. Education. ca

What did we know before ECCO 2009? www. Oncology. Education. ca
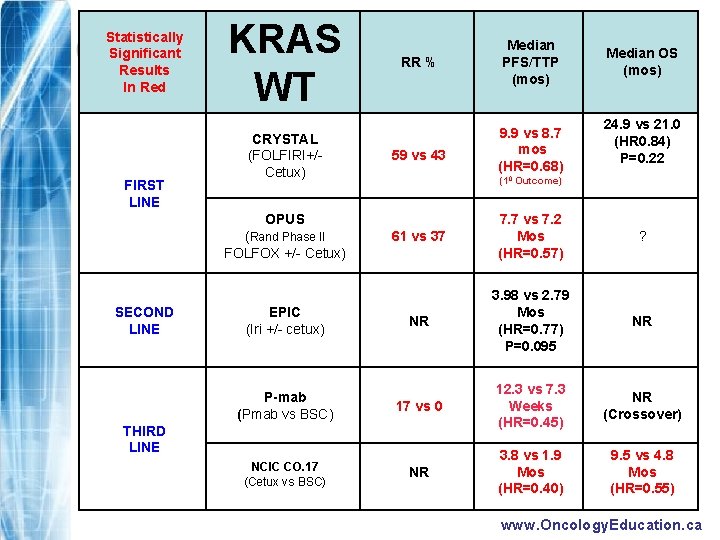
Statistically Significant Results In Red FIRST LINE KRAS WT CRYSTAL (FOLFIRI+/Cetux) RR % Median PFS/TTP (mos) 59 vs 43 9. 9 vs 8. 7 mos (HR=0. 68) SECOND LINE EPIC (Iri +/- cetux) P-mab (Pmab vs BSC) 61 vs 37 7. 7 vs 7. 2 Mos (HR=0. 57) ? NR 3. 98 vs 2. 79 Mos (HR=0. 77) P=0. 095 NR 17 vs 0 12. 3 vs 7. 3 Weeks (HR=0. 45) NR (Crossover) NR 3. 8 vs 1. 9 Mos (HR=0. 40) 9. 5 vs 4. 8 Mos (HR=0. 55) THIRD LINE NCIC CO. 17 (Cetux vs BSC) 24. 9 vs 21. 0 (HR 0. 84) P=0. 22 (10 Outcome) OPUS (Rand Phase II FOLFOX +/- Cetux) Median OS (mos) www. Oncology. Education. ca

What did we find at ECCO 2009? FIRST LINE www. Oncology. Education. ca
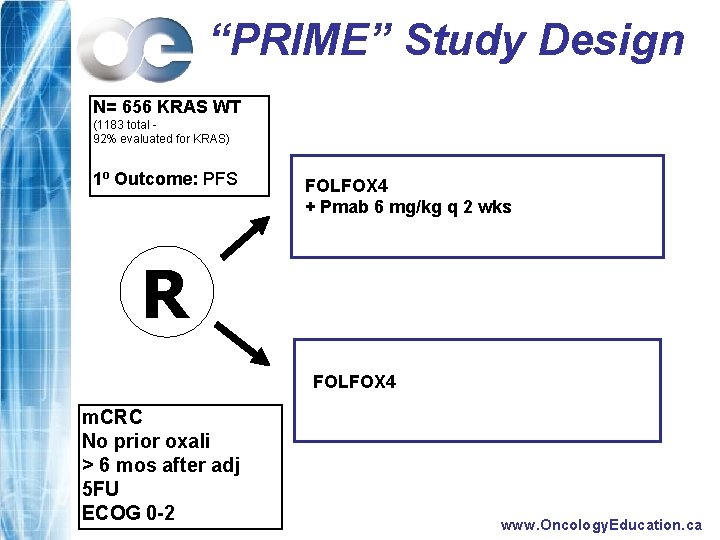
“PRIME” Study Design N= 656 KRAS WT (1183 total 92% evaluated for KRAS) 1 o Outcome: PFS FOLFOX 4 + Pmab 6 mg/kg q 2 wks R FOLFOX 4 m. CRC No prior oxali > 6 mos after adj 5 FU ECOG 0 -2 www. Oncology. Education. ca
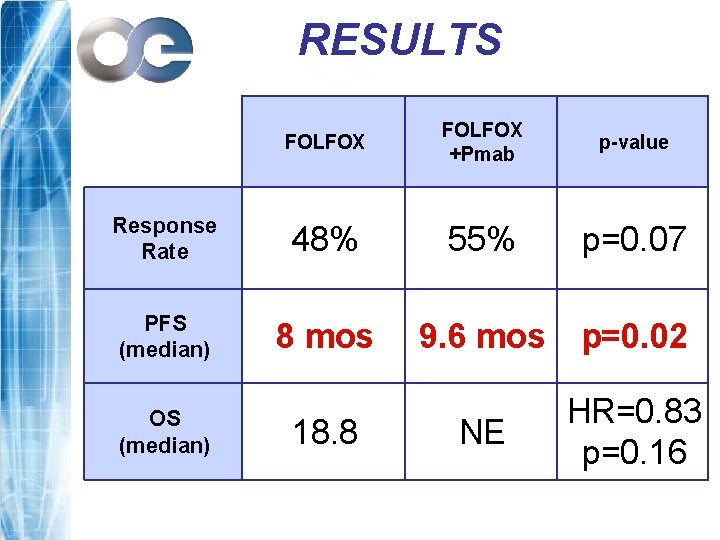
RESULTS FOLFOX +Pmab p-value Response Rate 48% 55% p=0. 07 PFS (median) 8 mos 9. 6 mos p=0. 02 NE HR=0. 83 p=0. 16 OS (median) 18. 8
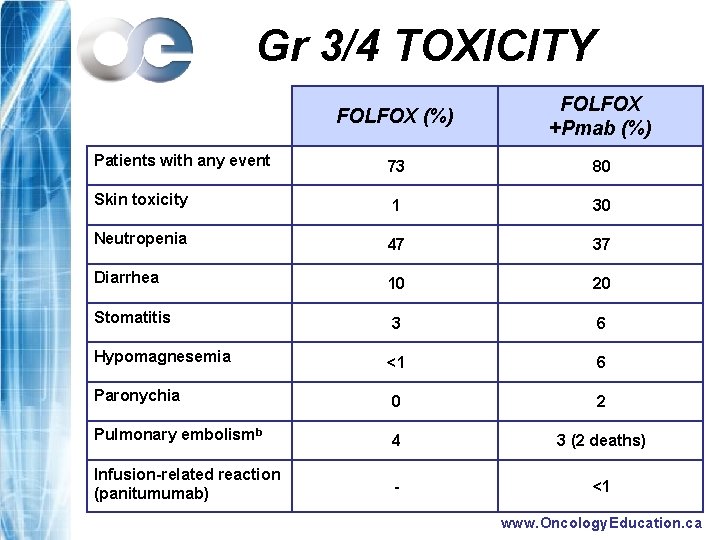
Gr 3/4 TOXICITY FOLFOX (%) FOLFOX +Pmab (%) Patients with any event 73 80 Skin toxicity 1 30 Neutropenia 47 37 Diarrhea 10 20 Stomatitis 3 6 Hypomagnesemia <1 6 Paronychia 0 2 Pulmonary embolismb 4 3 (2 deaths) Infusion-related reaction (panitumumab) - <1 www. Oncology. Education. ca

CRYSTAL Trial Updated 10 Endpoint= PFS R N=1217 EGFR expression via IHC A N D O MI ZE FOLFIRI + Cetuximab N=608 FOLFIRI N=609 * Cetuximab 400 mg/m 2 IV week 1 then 250 mg/m 2 IV weekly 1: 1 www. Oncology. Education. ca

OPUS: Rand Phase II Updated R A N N=338 D EGFR+ Metasatic CRC O MI ZE FOLFOX + Cetuximab N=170 FOLFOX N=168 * Cetuximab 400 mg/m 2 IV week 1 then 250 mg/m 2 IV weekly 1: 1 www. Oncology. Education. ca
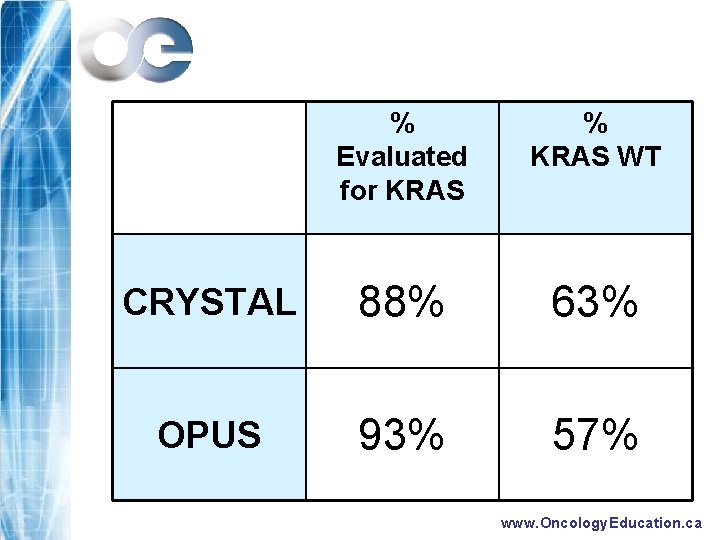
% Evaluated for KRAS % KRAS WT CRYSTAL 88% 63% OPUS 93% 57% www. Oncology. Education. ca
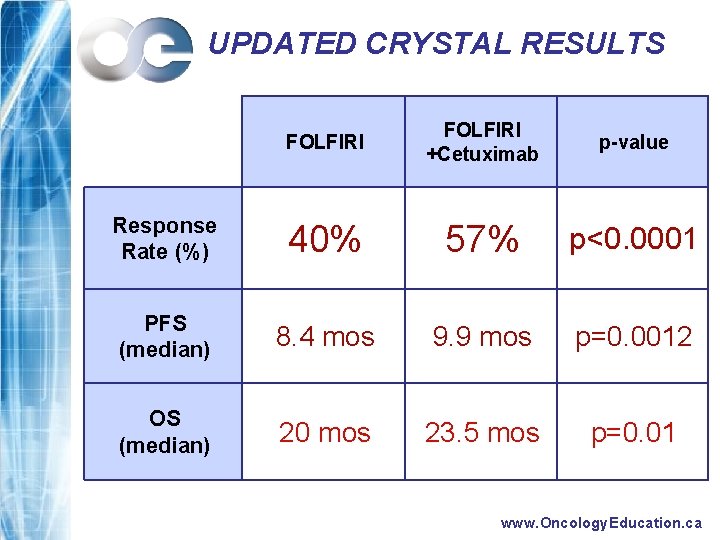
UPDATED CRYSTAL RESULTS FOLFIRI +Cetuximab p-value Response Rate (%) 40% 57% p<0. 0001 PFS (median) 8. 4 mos 9. 9 mos p=0. 0012 OS (median) 20 mos 23. 5 mos p=0. 01 www. Oncology. Education. ca
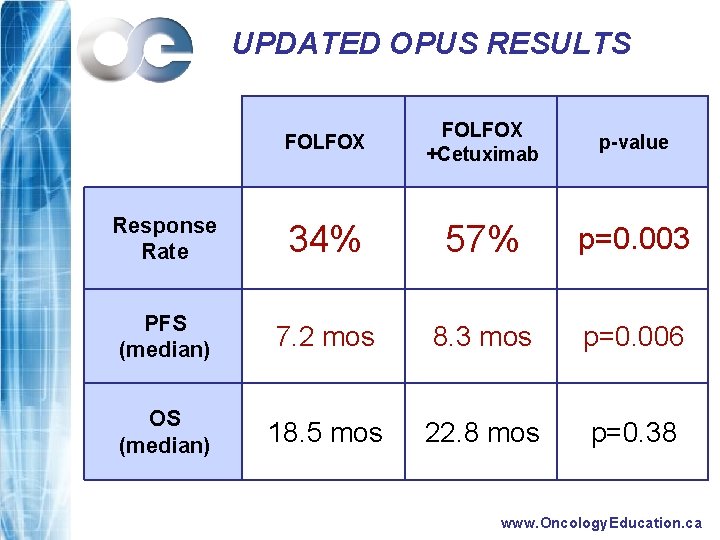
UPDATED OPUS RESULTS FOLFOX +Cetuximab p-value Response Rate 34% 57% p=0. 003 PFS (median) 7. 2 mos 8. 3 mos p=0. 006 OS (median) 18. 5 mos 22. 8 mos p=0. 38 www. Oncology. Education. ca

MRC COIN Study Design N= 729 KRAS WT (80 % analyzed, 56% WT) 1 o Outcome: OS R First Line m. CRC PS 0 -2 CAPOX/ FOLFOX 66% RECEIVED CAPOX/ FOLFOX + CETUXIMAB www. Oncology. Education. ca
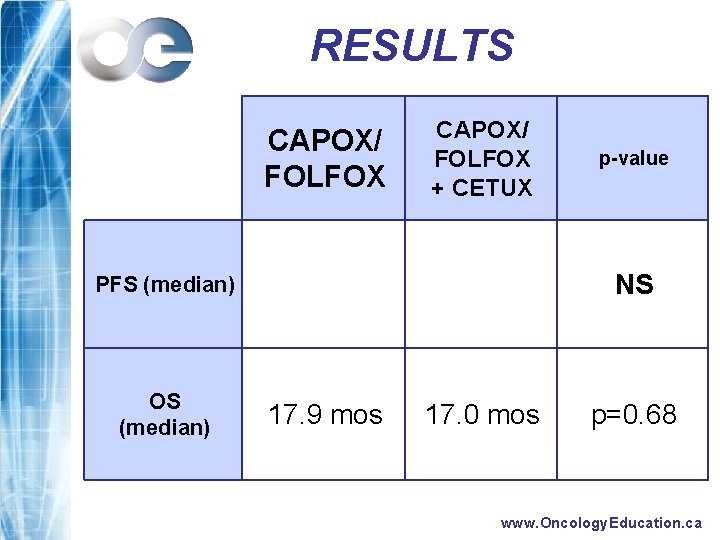
RESULTS CAPOX/ FOLFOX + CETUX NS PFS (median) OS (median) p-value 17. 9 mos 17. 0 mos p=0. 68 www. Oncology. Education. ca
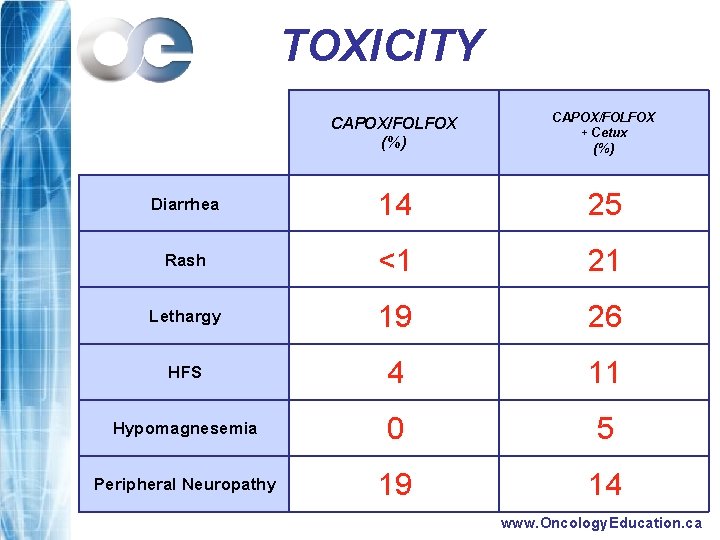
TOXICITY CAPOX/FOLFOX (%) CAPOX/FOLFOX + Cetux (%) Diarrhea 14 25 Rash <1 21 Lethargy 19 26 HFS 4 11 Hypomagnesemia 0 5 Peripheral Neuropathy 19 14 www. Oncology. Education. ca

STUDY COMMENTARY • Capecitabine Arm - Analyses not complete but: – Had much higher rates of non-haem toxicity leading to dose reductions and trial amendment - reduction of dose form 2000 mg/m 2 to 1700 mg/m 2 (Adams, Br J Cancer, 2009) – Hope to have updated analysis at ASCO GI 2010 further examining the issue of efficacy and toxicity of capecitabine cetuximab combination and impact on overall trial results www. Oncology. Education. ca

STUDY COMMENTARY • Median survivals in both study arms lower than other contemporary first line chemo /biologic trials – ? patient population • 9% > 75 • 8% ECOG 2 – ? access to subsequent line tx options www. Oncology. Education. ca

What did we find out at ECCO 2009? SECOND LINE www. Oncology. Education. ca
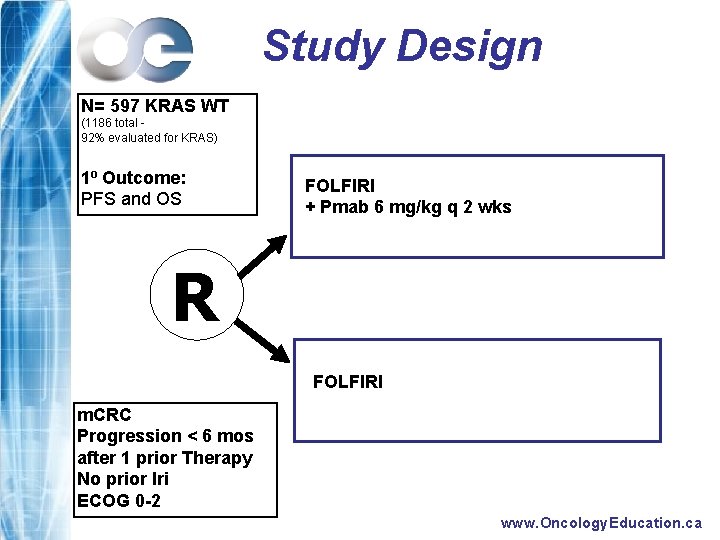
Study Design N= 597 KRAS WT (1186 total 92% evaluated for KRAS) 1 o Outcome: PFS and OS FOLFIRI + Pmab 6 mg/kg q 2 wks R FOLFIRI m. CRC Progression < 6 mos after 1 prior Therapy No prior Iri ECOG 0 -2 www. Oncology. Education. ca
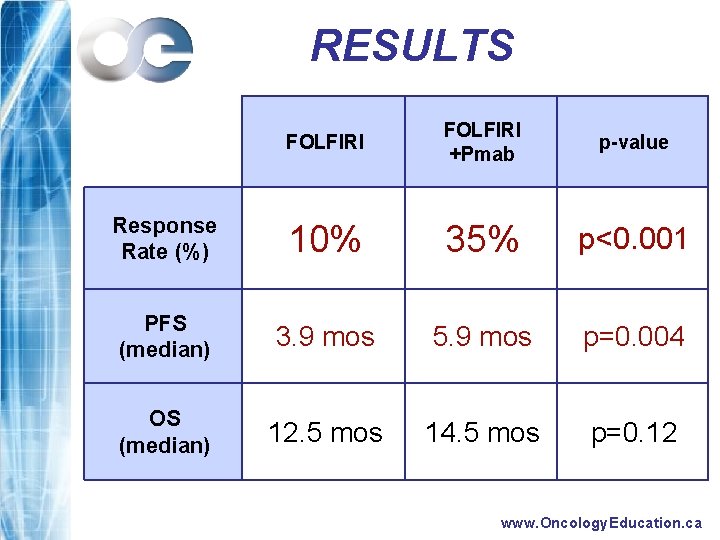
RESULTS FOLFIRI +Pmab p-value Response Rate (%) 10% 35% p<0. 001 PFS (median) 3. 9 mos 5. 9 mos p=0. 004 OS (median) 12. 5 mos 14. 5 mos p=0. 12 www. Oncology. Education. ca
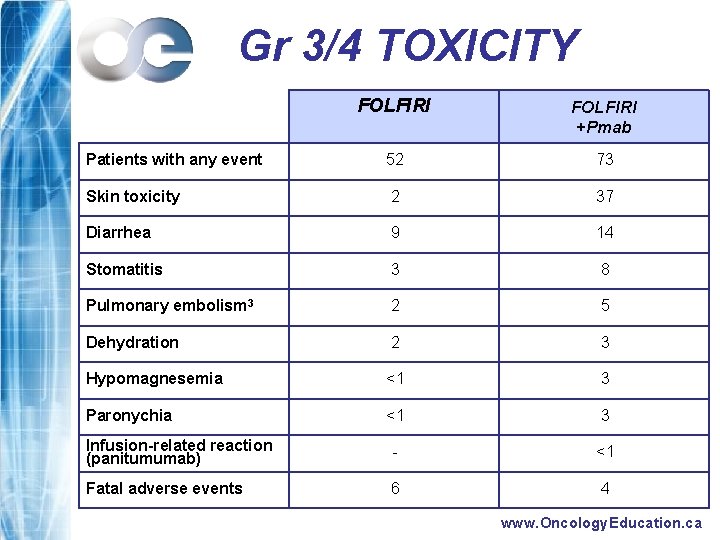
Gr 3/4 TOXICITY FOLFIRI +Pmab Patients with any event 52 73 Skin toxicity 2 37 Diarrhea 9 14 Stomatitis 3 8 Pulmonary embolism 3 2 5 Dehydration 2 3 Hypomagnesemia <1 3 Paronychia <1 3 Infusion-related reaction (panitumumab) - <1 Fatal adverse events 6 4 www. Oncology. Education. ca

Summing Up: What do we know after ECCO 2009?
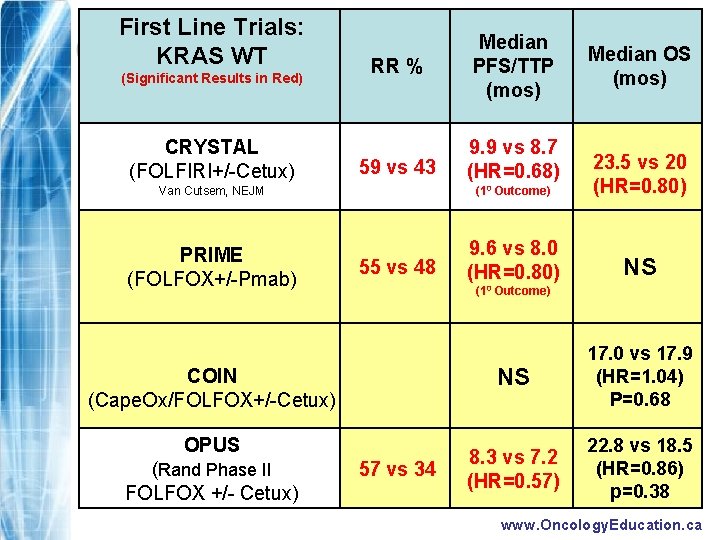
First Line Trials: KRAS WT (Significant Results in Red) CRYSTAL (FOLFIRI+/-Cetux) RR % Median PFS/TTP (mos) 59 vs 43 9. 9 vs 8. 7 (HR=0. 68) Van Cutsem, NEJM PRIME (FOLFOX+/-Pmab) 55 vs 48 (10 Outcome) 23. 5 vs 20 (HR=0. 80) 9. 6 vs 8. 0 (HR=0. 80) NS (10 Outcome) COIN (Cape. Ox/FOLFOX+/-Cetux) OPUS (Rand Phase II FOLFOX +/- Cetux) Median OS (mos) 57 vs 34 NS 17. 0 vs 17. 9 (HR=1. 04) P=0. 68 8. 3 vs 7. 2 (HR=0. 57) 22. 8 vs 18. 5 (HR=0. 86) p=0. 38 www. Oncology. Education. ca
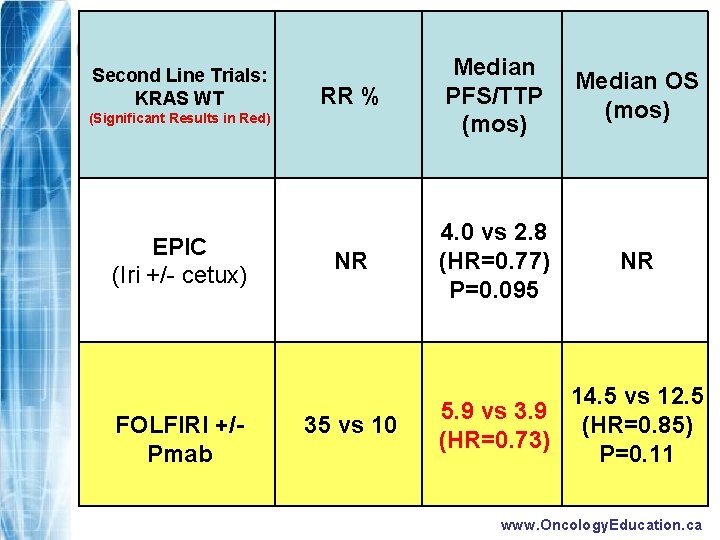
Second Line Trials: KRAS WT RR % Median PFS/TTP (mos) Median OS (mos) NR 4. 0 vs 2. 8 (HR=0. 77) P=0. 095 NR 35 vs 10 5. 9 vs 3. 9 (HR=0. 73) 14. 5 vs 12. 5 (HR=0. 85) P=0. 11 (Significant Results in Red) EPIC (Iri +/- cetux) FOLFIRI +/Pmab www. Oncology. Education. ca

Bottom Line for Canadian Medical Oncologists (1) – EGFR Inhibitors in m. CRC - Survival benefit now demonstrated: • in 3 rd line as monotherapy • with FOLFIRI + Cetux 1 st line • FOLFIRI + Cetux emerges as credible 1 st line option for KRAS WT patients » . . . but not a funded option in Canada at this time – NCIC CRC. 5 will address issue of which chemo + biolologic combination is superior first line » Chemo + Bevacizumab vs Chemo + Cetuximab » Double biologic arm dropped by intergroup www. Oncology. Education. ca
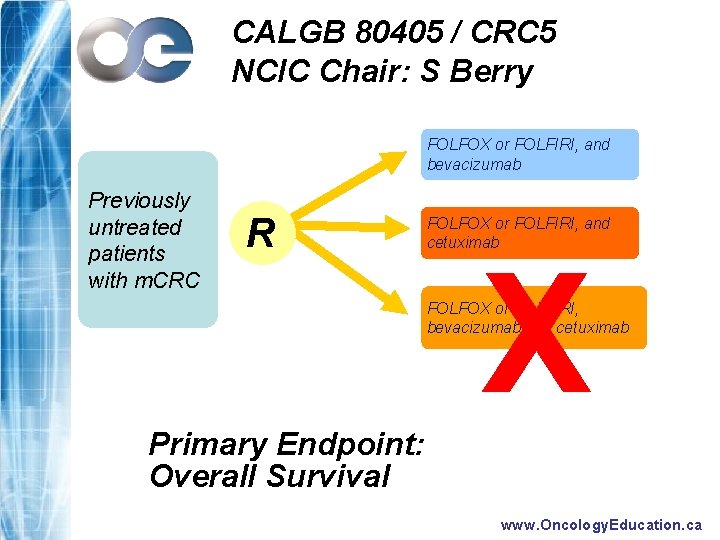
CALGB 80405 / CRC 5 NCIC Chair: S Berry FOLFOX or FOLFIRI, and bevacizumab Previously untreated patients with m. CRC R FOLFOX or FOLFIRI, and cetuximab X FOLFOX or FOLFIRI, bevacizumab and cetuximab Primary Endpoint: Overall Survival www. Oncology. Education. ca

Bottom Line for Canadian Medical Oncologists (2) • EGFR Inhibitors in m. CRC - PFS benefit seen with – FOLFOX + Pmab first line – FOLFIRI + Pmab second line – Further analyses of Pmab combos awaited • MRC COIN Trial – ? choice of chemo NB : preliminary signal re CAPOX + cetuximab - further information/analyses of efficacy and safety of COIN trial needed - hopefully available at ASCO GI 2010 www. Oncology. Education. ca
- Slides: 28