EC Directives for Medical Devices Active Implantable Medical



EC Directives for Medical Devices Active Implantable Medical Devices (AIMD) 90/385/EEC : 1 Jan. 1995 (主動式植入醫療器材) In Vitro Diagnostic Devices (IVD) 98/79/EC; 16, Dec. 2003 (體外診斷器材設備) Medical Devices Directives (MDD) 93/42/EEC : 13 Jun. 1998 (醫療器材)




醫療器材定義 Definitions for Medical Devices Used for— Diagnosis, prevention, monitoring, treatment or alleviation of disease; 對疾病之診斷,預防,追蹤。治療或減輕疾病之器材等,如聽診器 Diagnosis, monitoring, treatment, alleviation of or compensation for an injury or handicap 對於傷處或殘障部位之診斷、追蹤、治療、或修整等,如 ok繃 Investigation, replacement or modification of the anatomy or of a physiological process; 解剖或生理過程之檢查、換置或修正等,如手術刀、手術手套 Control of conception. 生育控制之器材,如避孕器、保險套

醫療器材分類 Medical Device Classification 考量醫療器材之設計及製造對人體可能帶來的危險程度,分類為: Class I – Low risk(i. e. examination gloves) Class lla – Low to medium risk(i. e. surgical golves) Class IIb – Medium risk(i. e. condoms, blood bags) Class III – High risk(i. e. contraceptive intrauterine devices) 分類原則: 非侵入式器材、主動式器材及其他特殊原則。 時間:暫時(<60分鐘)、短期(<30天)、長期(>30天) 類別界定: 由廠商自行決定。-可根據MDD的guidelines, 或是 93/42/EEC Annex IX 中之敘述來判斷。





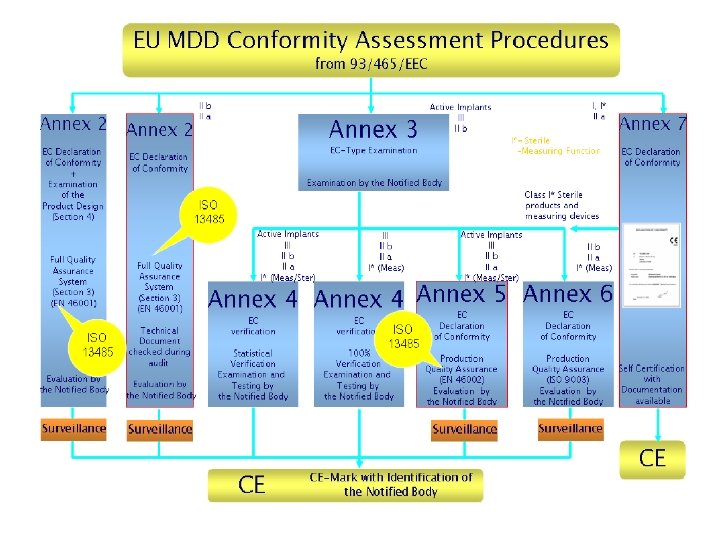
The conformity assessment modules & procedures (評核模式及程序說明) Module A > EC Declaration of Conformity / EC合格聲明 (Annex VII) Module B > Type-Examination/ EC型式試驗 (Annex III) Module D > Production Quality Assurance / 生產質量保證 (Annex V) / ISO 13485 Module E > Product Quality Assurance / 產品質量保證 (Annex VI) / ISO 13485 Module F > EC Verification/ EC驗證 (Annex IV) Module H > Fully Quality Assurance /全面質量保證體系 (Annex II) / ISO 13485

The Conformity Assessment Procedures Class I (等級I-一般醫療機器) General class I -> module A With sterile or measuring function -> module A+D or E or F Class lla (等級II – 管理醫療機器) Module A + D or E or F Module H - Class IIb (等級III – 高度管理醫療機器) Module H Module B + D or E or F Class III (等級III, IV – 高度管理醫療機器) Module B + D or Module H
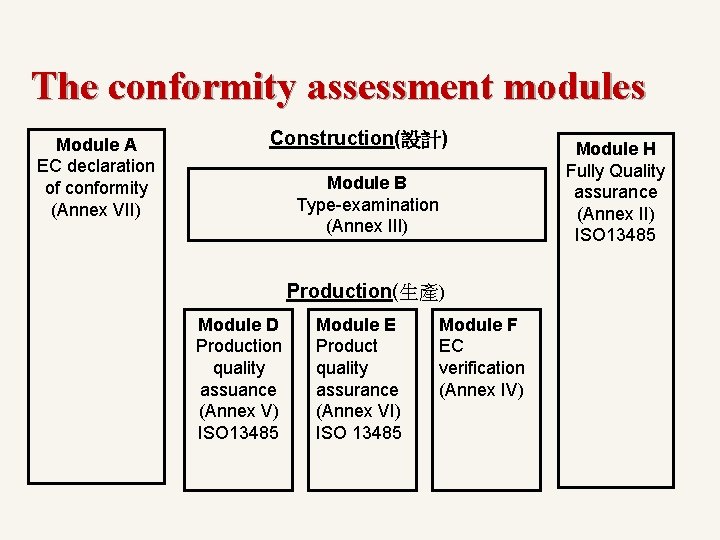
The conformity assessment modules Module A EC declaration of conformity (Annex VII) Construction(設計) Module B Type-examination (Annex III) Production(生產) Module D Production quality assuance (Annex V) ISO 13485 Module E Product quality assurance (Annex VI) ISO 13485 Module F EC verification (Annex IV) Module H Fully Quality assurance (Annex II) ISO 13485
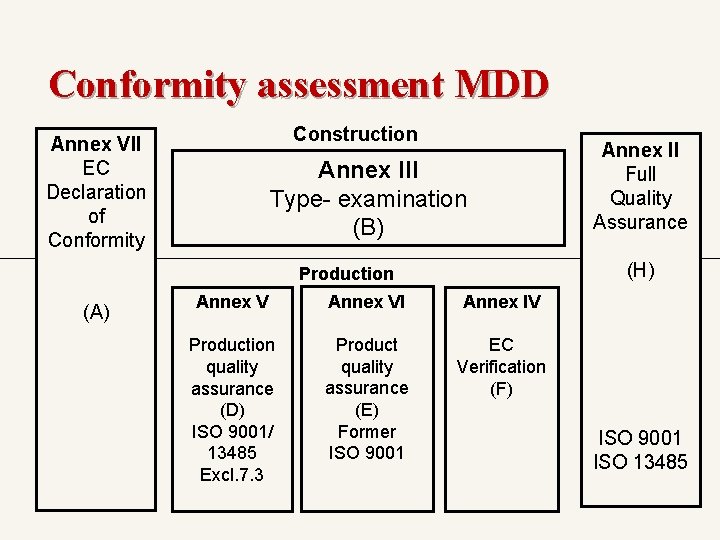
Conformity assessment MDD Construction Annex VII EC Declaration of Conformity Annex III Type- examination (B) (H) Production (A) Annex II Full Quality Assurance Annex VI Annex IV Production quality assurance (D) ISO 9001/ 13485 Excl. 7. 3 Product quality assurance (E) Former ISO 9001 EC Verification (F) ISO 9001 ISO 13485
- Slides: 17