EarthSpace Beginning with Science Lesson 4 Measurement Metric

Earth/Space: Beginning with Science

Lesson 4: Measurement
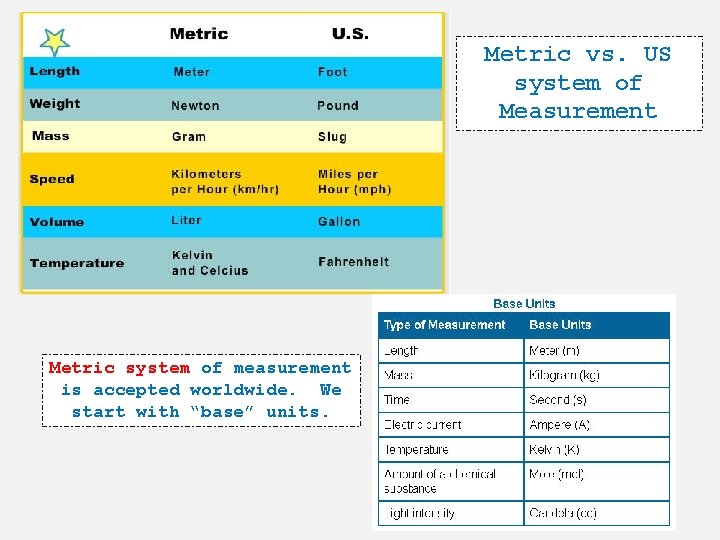
Metric vs. US system of Measurement Metric system of measurement is accepted worldwide. We start with “base” units.

Tools that we use are: Base metric unit = Gram (g)
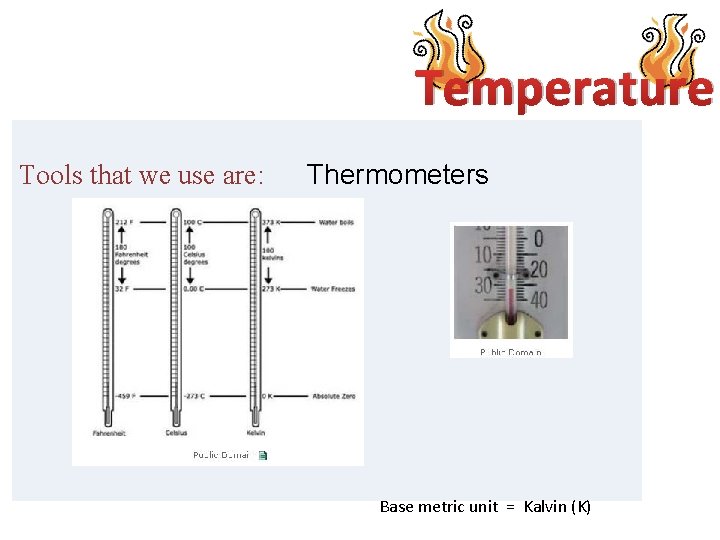
Temperature Tools that we use are: Thermometers Base metric unit = Kalvin (K)

Tool used could be a graduated cylinders. Base metric unit = Liter (l)

Length Tools used can be: Rulers, Meter sticks, Meter tapes, etc Base metric unit = Meter (m)
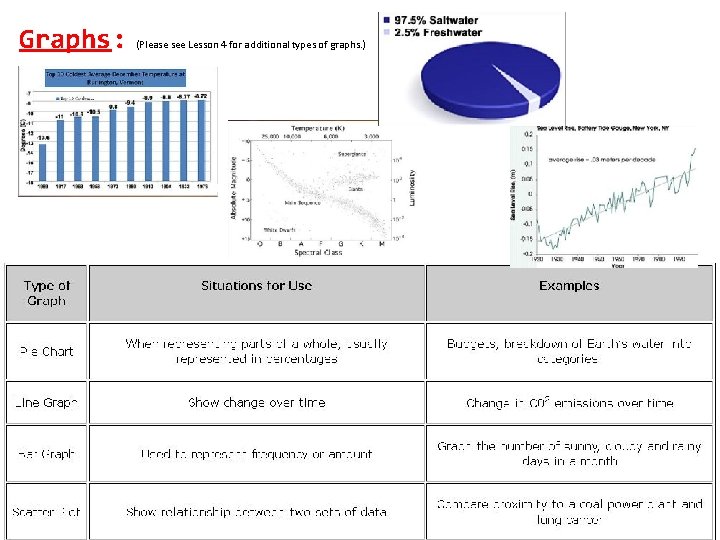
Graphs: (Please see Lesson 4 for additional types of graphs. )


Lesson 5: Atomic Structure and Forces

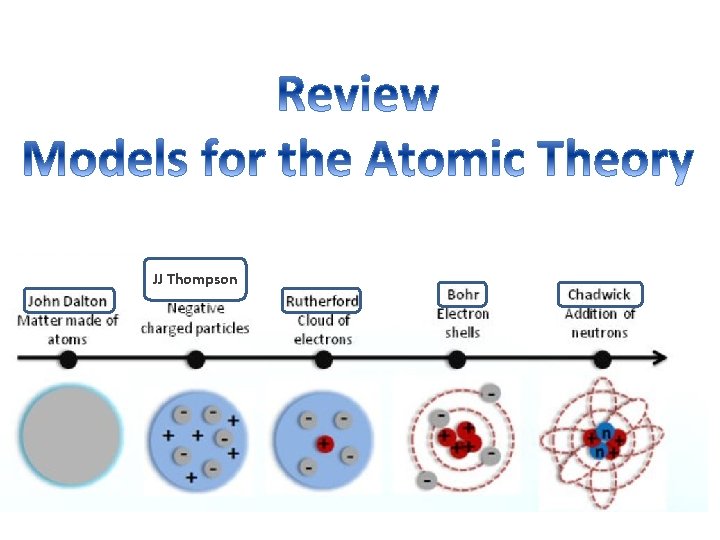
Review: Parts of an atom What are three sub atomic particles in an atom? 1. 2. 3. What charge does each particle have? (positive, negative, or neutral) 1. 2. 3. Where are they located? 1. 2. 3.

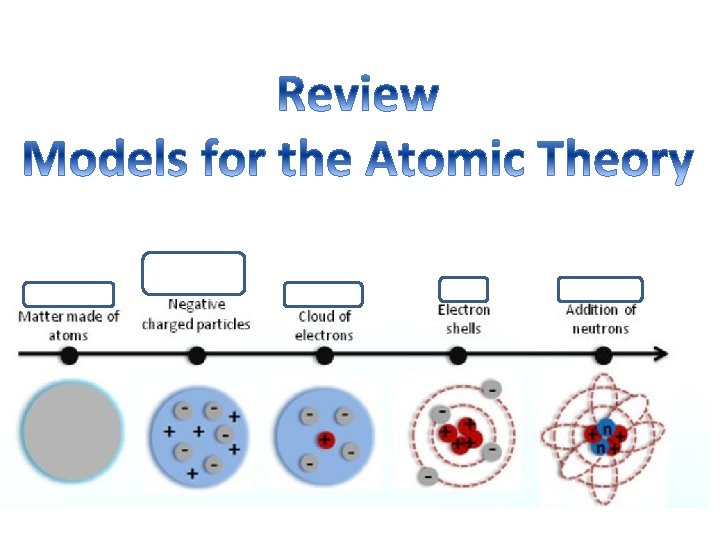
Review: Parts of an atom What are three sub atomic particles in an atom? 1. Protons 2. Neutrons 3. Electrons What charge does each particle have? (positive, negative, or neutral) 1. Proton = Positive (+) 2. Neutron = Neutral (0) 3. Electron = Negative (-) Where are they located? 1. Proton = Inside the nucleus 2. Neutron = Inside the nucleus 3. Electron = Outside the nucleus
JJ Thompson
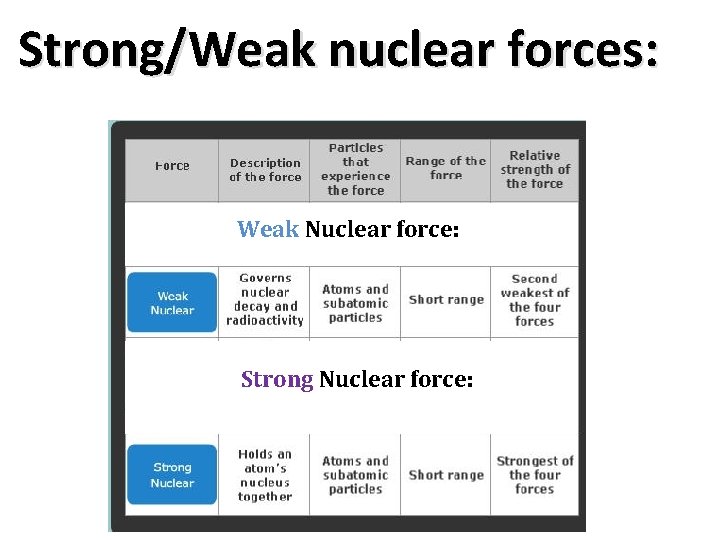
Strong/Weak nuclear forces: Weak Nuclear force: Strong Nuclear force:

What does the Atomic Number tell us? (what information)

Atomic Number – The number of protons in an atom. Atomic number: How many protons are in this atom. The number of protons also equals the number of electrons in an atom!

What does the Mass Number tell us?

Mass Number – The total number of protons and neutrons in an atom. Mass number (or atomic mass): total number of the protons + the neutrons.
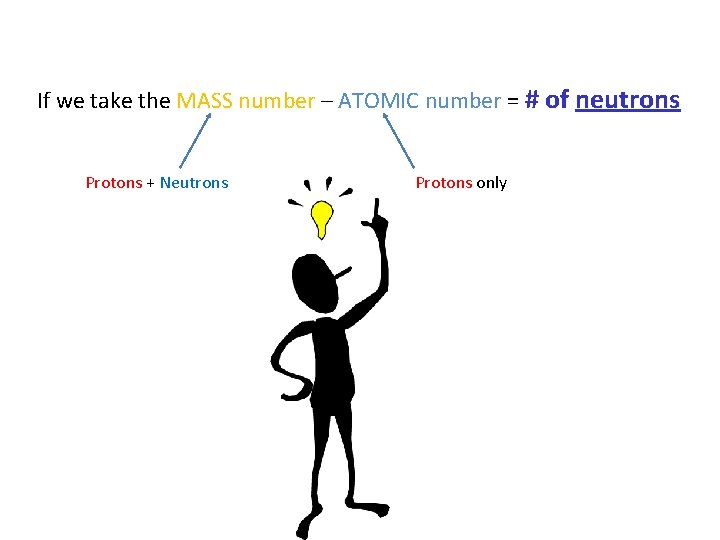
If we take the MASS number – ATOMIC number = # of neutrons Protons + Neutrons Protons only

• Number of protons changes the identity of an atom. • Number of electrons changes the properties of an atom. • Number of neutrons can affect the properties of an atom also.
What happens in an Ionic Bond?

Ionic Bond Atoms give up an electron or receiving an electron; becoming “ions”. Elements that are involved in ionic bonds are “I, I, I” elements.

What happens in a Covalent Bond?

Covalent Bond Covalent like CO-operate (sharing). A bond that forms by atoms sharing electrons.
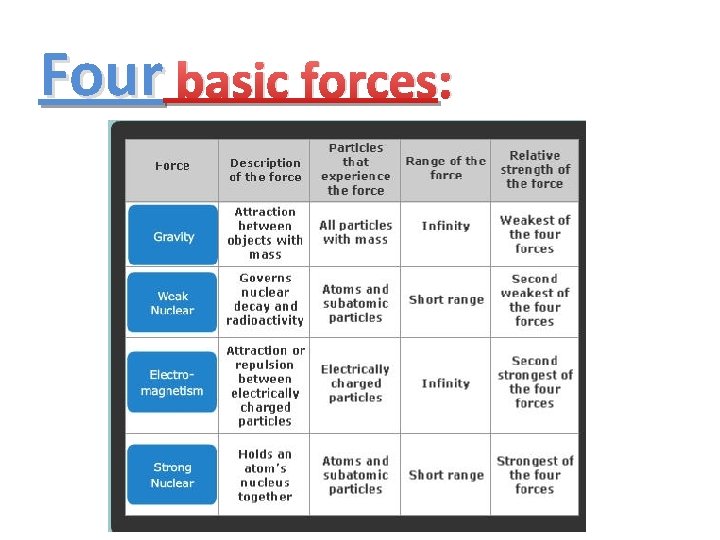
Four basic forces:


Lesson 6: Matter and Energy

What are the four states of matter?

The four states of matter are: 1. ) Solid 2. ) Liquid 3. ) Gas 4. ) Plasma

Properties of Matter What are the two different types of properties used to describe matter? 1. 2.

Properties of Matter What are the two different types of properties used to describe matter? 1. 2. Physical properties Chemical properties

List 2 examples of physical properties:

Physical properties Characteristics of a substance that can be measured without changing the identity of the substance. Examples: Appearance (Like: color or shape) Odor (smell) Melting point and boiling point Density Hardness Ductility and Malleability (ability to change shape) Solubility Electrical and thermal conductivity Magnetism

Chemical properties Characteristics of a substance that describe the ways it new materials. can react to form Flammability (reacts with oxygen) Reaction with acids and bases Reacts to water Decomposition (break down) caused by light or heat Tendency to corrode (rust)

Physical Changes A change that does not create a new substance. The shape, form, or appearance of a substance may have changed, but the substance is still the same. Paper Now I’ve torn (ripped) paper It’s changed… but not into anything new. It’s still paper.

How do we know something goes through it?

How do we know something goes through it? When something changes into something different, and we can’t get it back the way it was.

Chemical Changes The wood has changed into something “new” (ash) and you can’t get it back the way it was.

What are some things we can “observe” that tell us a chemical change occurred?

Things we can “observe” that tell us a chemical change occurred: 1. Change in color 2. Produces a gas 3. Heat 4. Light

A new substance being formed, is an example of what type of change? Physical or Chemical?

“A bike will rust if left outside”

What are three ways that heat is transferred?

Heat is transferred to the environment in three basic ways: radiation conduction convection

What is the Law of Conservation of Mass?

Law of Conservation of Mass Matter cannot be created or destroyed. When something goes through a physical or chemical change at the end of the change there is still the same amount of matter as you started with. (It might be different, but the total amount is still the same. )

Law of Conservation of Energy Any form of energy can be transformed to any other form of energy without a loss of net energy.

Energy is added to the water… particles move around more… the water changes to a different state of matter.


- Slides: 52