Early treatment of Clinically Isolated Syndrome CIS for












- Slides: 24

Early treatment of Clinically Isolated Syndrome (CIS) for prevention of Multiple Sclerosis (MS) CAWIN WONG MIZUBA, R 2 1/19/2015

Case Presentation 43 yo F with a PMH significant for possible ADEM after flu vaccination 1 yr PTA and HTN who presented with a 3 day history of vertigo, and 2 day history of diplopia and nausea. Exam: CN VI palsy of left eye with nystagmus Imaging: MRI brain: 12 mm enhancing lesion with T 2 hyperintensity in the left brachium pontis, second enhancing lesion in middle cerebellar peduncle CSF: Positive for oligoclonal bands. Cytology showing CD 45 bright T-cell lymphoid population.

Clinical Question P = Patient population Patients with CIS I = Intervention, prognostic factor, or exposure Immunonomodulating therapy C = Comparison Placebo O = Outcomes Delay conversion to multiple sclerosis Pub. Med – “multiple sclerosis”, “clinically isolated syndrome”, “immunomodulator”, “progression”

Lancet Neurol 2014; 13: 977– 86.

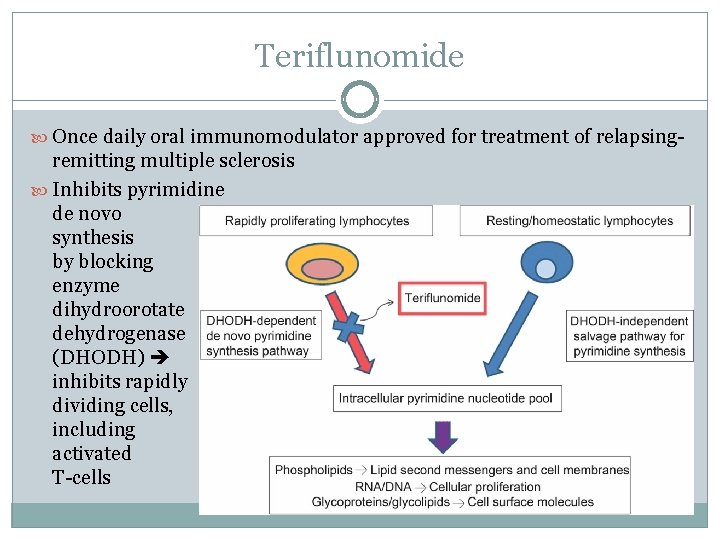
Teriflunomide Once daily oral immunomodulator approved for treatment of relapsing- remitting multiple sclerosis Inhibits pyrimidine de novo synthesis by blocking enzyme dihydroorotate dehydrogenase (DHODH) inhibits rapidly dividing cells, including activated T-cells

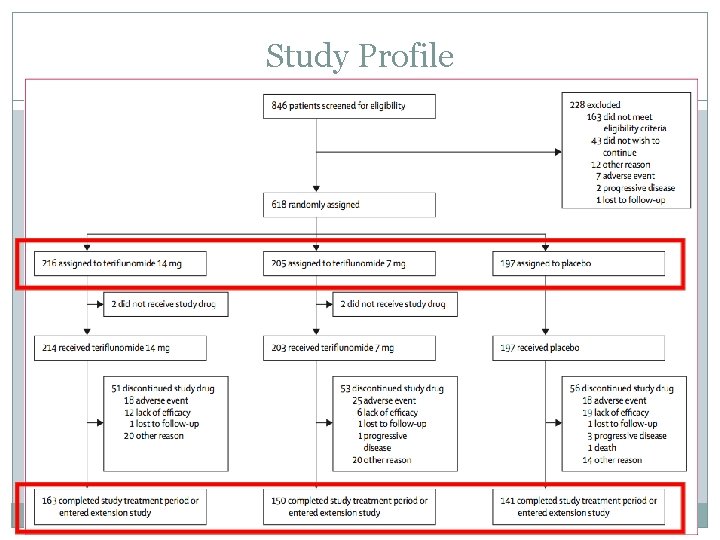
Study Design Randomized, double-blinded, placebo controlled, parallel-group study Recruited patients from 112 centers in 20 countries 108 week study with 14 scheduled clinic visits, MRI scans and labs completed at regular intervals. Optional extension study Subjects wanting to receive active treatment with teriflunomide after completion of 108 week study Pts with relapse and treated for at least 24 weeks

Study Profile

Inclusion criteria Age 18 -55 yrs with clinically isolated syndrome (first acute or subacute neurological event consistent with demyelination eg. optic neuritis, spinal cord syndrome, brainstem or cerebellar syndromes), occuring within 90 days before randomization MRI showing at least two T 2 -weighted MRI lesions of at least 3 mm in diameter

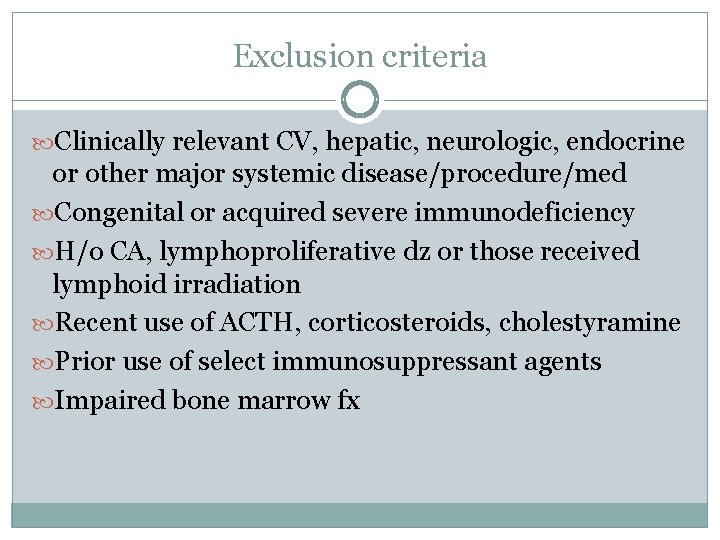
Exclusion criteria Clinically relevant CV, hepatic, neurologic, endocrine or other major systemic disease/procedure/med Congenital or acquired severe immunodeficiency H/o CA, lymphoproliferative dz or those received lymphoid irradiation Recent use of ACTH, corticosteroids, cholestyramine Prior use of select immunosuppressant agents Impaired bone marrow fx

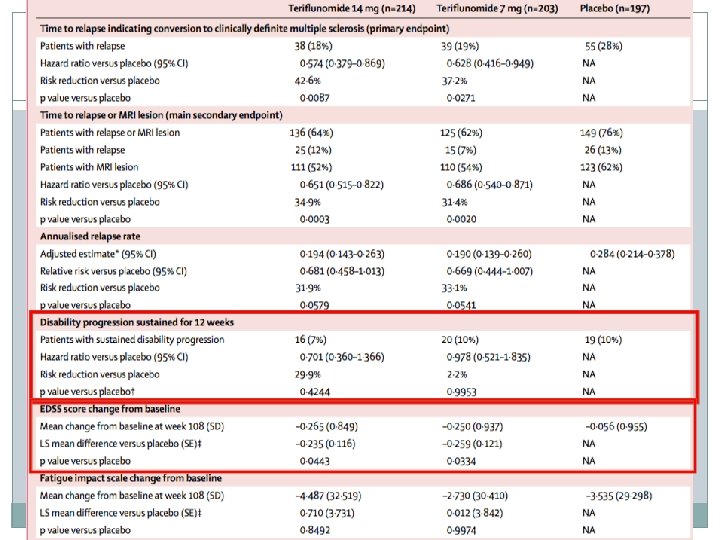
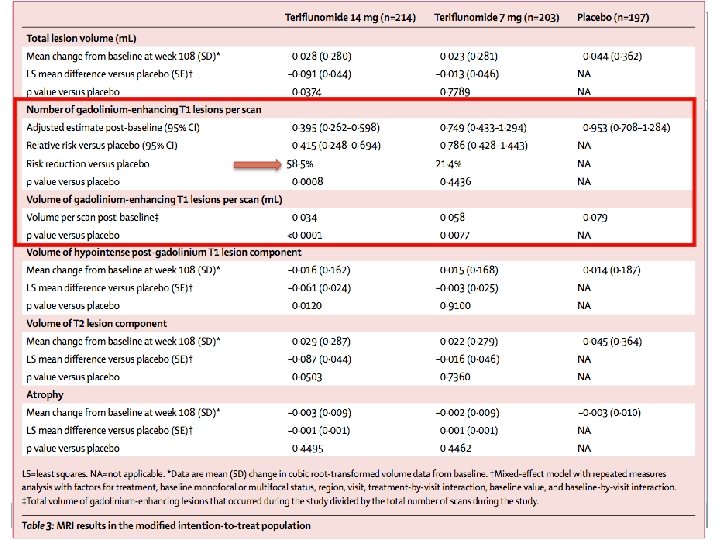
End Points Primary Time to relapse (conversion to clinically definite MS) Secondary Time to relapse or detection of new gadolinium-enhancing or T 2 lesions on MRI (whichever occurred first) Annualized relapse rate (# of relapses per patient year) Time to sustained disability progression Proportion of patients free from sustained disability progression Change from baseline in EDSS & fatigue impact scale total score Disease activity Volume of T 1 or T 2 lesion Safety and tolerability

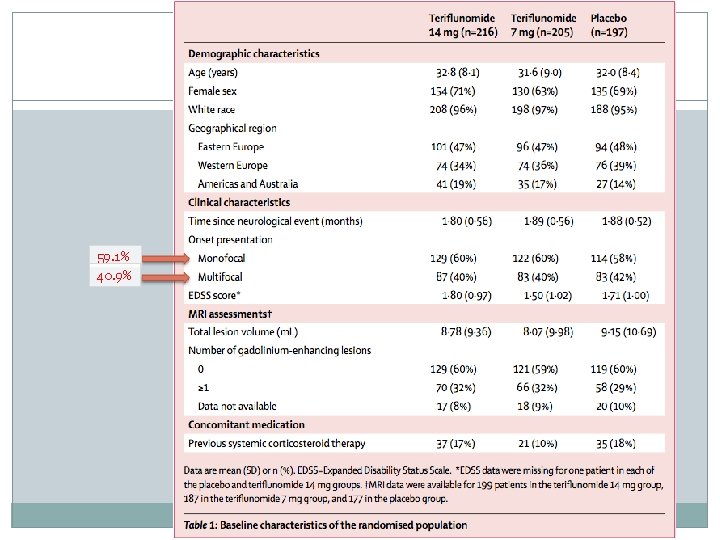
59. 1% 40. 9%

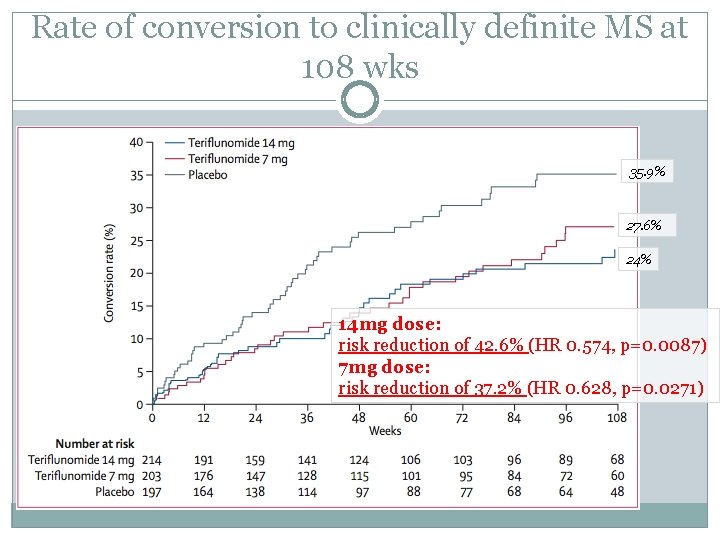
Rate of conversion to clinically definite MS at 108 wks 35. 9% 27. 6% 24% 14 mg dose: risk reduction of 42. 6% (HR 0. 574, p=0. 0087) 7 mg dose: risk reduction of 37. 2% (HR 0. 628, p=0. 0271)

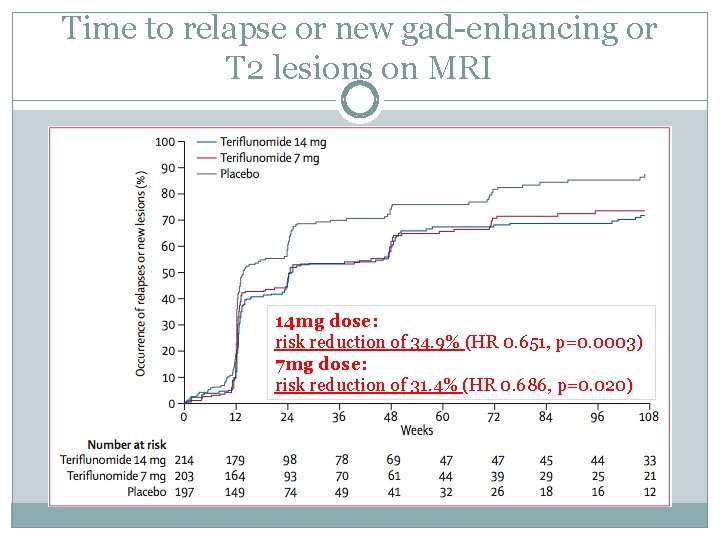
Time to relapse or new gad-enhancing or T 2 lesions on MRI 14 mg dose: risk reduction of 34. 9% (HR 0. 651, p=0. 0003) 7 mg dose: risk reduction of 31. 4% (HR 0. 686, p=0. 020)

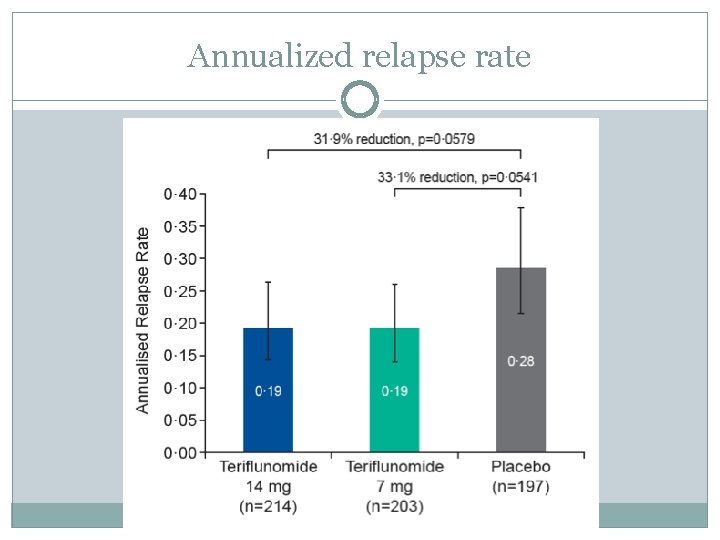
Annualized relapse rate


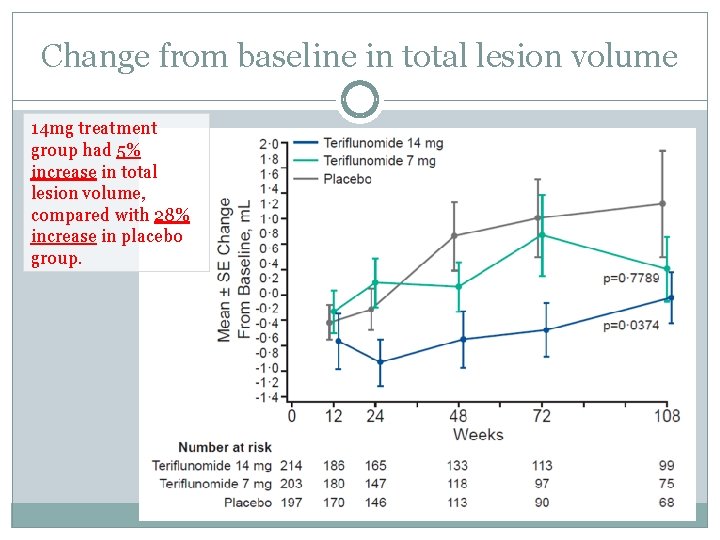
Change from baseline in total lesion volume 14 mg treatment group had 5% increase in total lesion volume, compared with 28% increase in placebo group.


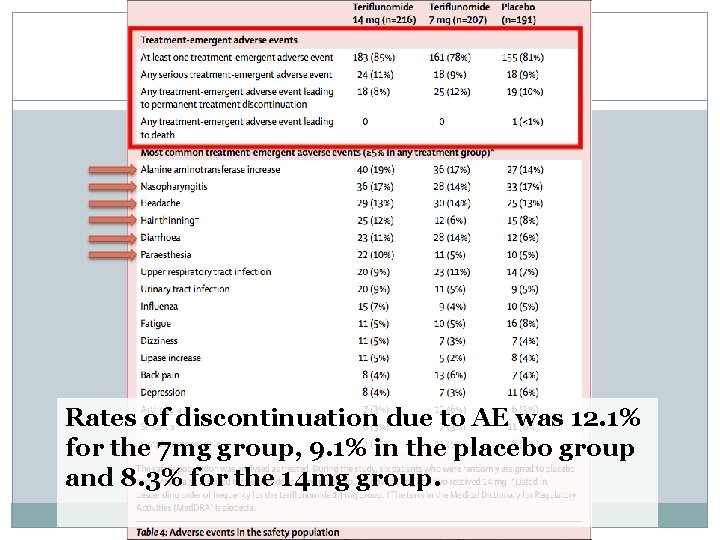
Rates of discontinuation due to AE was 12. 1% for the 7 mg group, 9. 1% in the placebo group and 8. 3% for the 14 mg group.

Adverse events Mean changes from baseline in lymphocyte and neutrophil counts <14% in all treatment groups Incidence of serious infections low in all tx groups No reports of malignancy in either tx group Unexpected pregnancies despite requirement to use reliable contraception 6 subjects randomly assigned to placebo also received teriflunomide at some point

Study Conclusions At 2 years, people taking Teriflunomide at the 14 mg dose were significantly less likely to develop definite MS when compared with placebo. 14 mg dose was associated with significant reduction in occurrence of relapses or MRI lesions and significant reduction in total lesion volume and number of gadenhancing T 1 lesions. Rise in ALT was the most frequent AE among treatment groups although did not reach statistical significance when compared with placebo.

Limitations Trial stopped by funder 3 mos early due to 2010 revisions of Mc. Donald criteria enabling earlier diagnosis of MS Early cessation of the study reduced statistical power in the interpretation of findings for annualized relapse rate and sustained disability progression

Conclusion Randomized, double-blinded, placebo controlled, parallel-group study, used intention to treat analysis No head trial with other oral immunotherapies and more established injectable drugs (eg. interferon beta, glatiramer acetate) Need longer term follow up for tracking disease progression

Questions? Thank you for your attention!

References Miller et al. Oral teriflunomide for patients with a first clinical episode suggestive of multiple sclerosis (TOPIC): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Neurol 2014; 13: 977– 86. Leist et al. Effect of oral cladribine on time to conversion to clinically definite multiple sclerosis in patients with a first demyelinating event (ORACLE MS): a phase 3 randomised trial. Lancet Neurol. 2014 Mar; 13(3): 257 -67. doi: 10. 1016/S 1474 -4422(14)70005 -5. Epub 2014 Feb 4. UTD. Clinically isolated syndromes suggestive of multiple sclerosis. National MS Society. http: //www. nationalmssociety. org/About-the. Society/News/Positive-Results-Announced-for-Aubagio-in. Phase-II.