e Reversible reactions Learning intention Learn how reversible

e) Reversible reactions Learning intention Learn how reversible reactions may reach equilibrium instead of completely converting reactants to products. Find out how dynamic equilibrium is defined in terms of reaction rates and concentrations of reactant and product.

Follow up questions 1. What is equilibrium? 2. What factors can effect equilibrium?



Learning Intention To be able to explain equilibrium Success Criteria • I can state key facts about equilibrium • I can show the point of equilibrium on a graph • I can state how temperature effects equilibrium

Dynamic equilibrium Reactants Products Chemical reactions which take place in both directions are called reversible reactions. The following is an example of a reversible reaction hydrogen and iodine reacting to form hydrogen iodide. H 2(g) + I 2(g)⇄ 2 HI(g) The equilibrium can be arrived at from different starting points. The position of an equilibrium does not depend on the starting position.
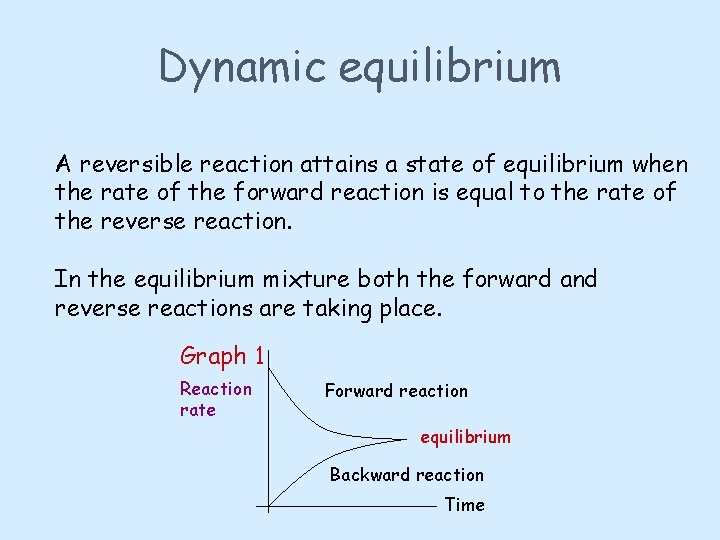
Dynamic equilibrium A reversible reaction attains a state of equilibrium when the rate of the forward reaction is equal to the rate of the reverse reaction. In the equilibrium mixture both the forward and reverse reactions are taking place. Graph 1 Reaction rate Forward reaction equilibrium Backward reaction Time

Dynamic equilibrium Since the rates of the forward and reverse reactions are equal, the concentration of reactants and products remain constant, though not necessarily equal. The system is said to be at dynamic equilibrium. Graph 2 Concentration Forward (reactants) Backward (products) Time

Position of equilibrium At equilibrium, the concentration of the products and the reactants will remain constant The concentration of reactants will probably not equal the concentration of the products.
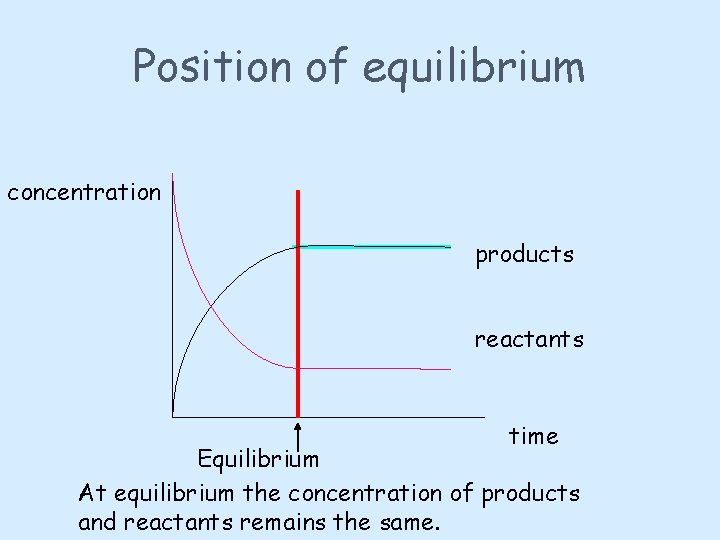
Position of equilibrium concentration products reactants time Equilibrium At equilibrium the concentration of products and reactants remains the same.

Equilibrium A reversible reaction can reach equilibrium in a closed system. N 2 + 3 H 2 ⇄ 2 NH 3 A reaction reaches equilibrium when the rate of the forward reaction equals the rate of the reverse reaction.

Direction • The equilibrium position will be the same whether we start with only the products or only the reactants • Iodine dissolves in both cyclohexane and water/KI. • The experiment shows one boiling tube set up with 100% iodine in cyclohexane and one with 100% iodine in water/KI.
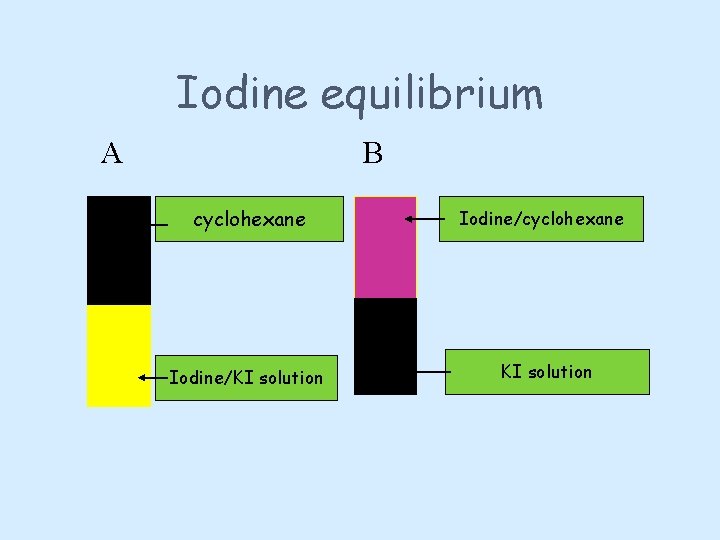
Iodine equilibrium A B cyclohexane Iodine/KI solution Iodine/cyclohexane KI solution

1. Using a spatula, place one iodine crystal in the KI solution and one in the cyclohexane. (Observe the different colours. ) 2. Add an equal volume of KI solution to the cyclohexane solution, without shaking. 3. Take the test tube with KI solution, and add an equal volume of cyclohexane, without shaking. 4. Watch for colour developing in the newly added solvent in each tube. 5. Cork both tubes and shake them gently, watching for changes in colour intensity in each tube. 6. Finally shake both tubes vigorously for 15 seconds, allow the two layers to settle out, then compare the colours in each solvent between both tubes. The situation you should have achieved is called an equilibrium. 7. Use a dropping pipette to remove the bottom layer (coloured potassium iodide solution) from one test-tube, then add the same volume of fresh potassium iodide solution to the cyclohexane layer and shake. 8. Repeat the removal of the coloured potassium iodide solution and addition of fresh potassium iodide solution two or three times. What happens to the amount of iodine left in the cyclohexane layer?
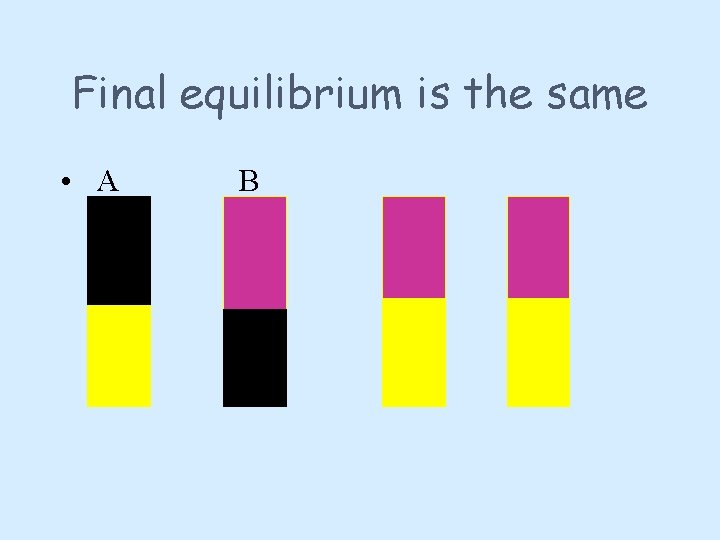
Final equilibrium is the same • A B

Copper sulfate equilibrium 1. Pour the copper sulfate solution into the conical flask. 2. Slowly add the acid down the side of the flask and swirl vigorously. 3. Carefully add the ammonia in the same way but initially without swirling. Wait and show the colour change. 4. Continue to add the ammonia with gentle swirling as the colour eventually changes to dark blue. 5. Reverse the reaction by adding acid in a similar fashion to the ammonia

f) Altering Equilibrium Position Learning intention Learn how chemists alter the position of equilibrium to increase product yield, by changing factors such as concentration, pressure, temperature and also how the use of a catalyst can ensure equilibrium is reached more quickly.

Starter

Le Chatelier's Principle “For a system in equilibrium, alteration of one of the factors (pressure, temperature or concentration) will cause the position of equilibrium to shift to reduce the effects of the imposed conditions”.

Le Chateliers Principle An equilibrium will move to undo any change imposed upon it. If the forward reaction is favoured we say the equilibrium has moved to the right. If the reverse reaction is favoured we say the equilibrium has moved to the left

Shifting the equilibrium position The proportion of products to reactants in an equilibrium mixture is described as the equilibrium position. A+B⇄C+D If the conversion of A and B into C and D is small the position of equilibrium lies to the left, or to the side of the reactants. If the equilibrium mixture is largely composed of C and D, the position of equilibrium lies to the right, or to the side of the products.

Poor question



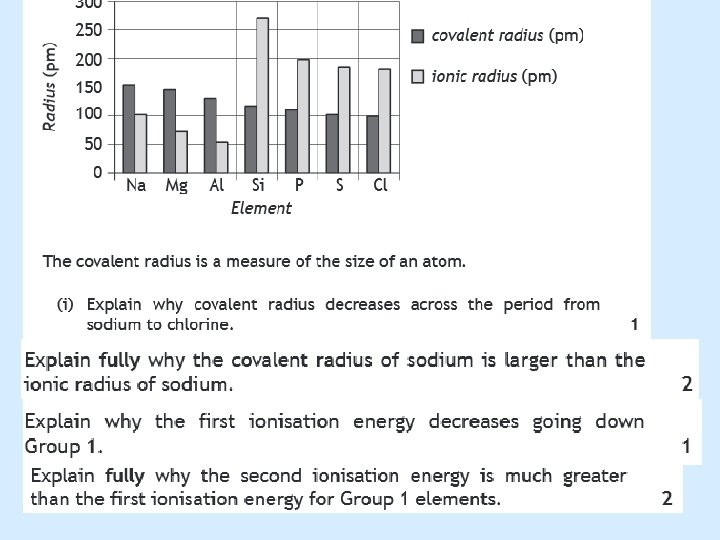
Answers given 1. The attraction between the positive nucleus and the negative electrons. 2. A measure of attraction a bond has for the electrons in a bond. 3. The attraction an atom has for electrons in a bond 4. Electronegativity is the force of attraction two covalently bonded atoms have for electrons.

Answers given 1. As more electrons shells are added the inner electrons shield the outer ones, the nucleus gets larger. 2. Because the number of energy levels increase, which decreases the nucleus’ pull on its outer electrons

Answers given 2 1. Going down group 7 boiling points increase as there are more/stronger intermolecular forces present. These intermolecular forces are known as London Dispersion Forces. Boiling point will increase as it is these forces that need to be overcome in order for a substance to boil.

Answers given 3 1. They increase as the number of electrons in the energy levels increase meaning there is more London dispersion forces between molecules, this means the atoms are more strongly attracted to each other so more energy is needed to be put in for the bonds to break.

Concentration may alter the position of equilibrium

Concentration may alter the position of equilibrium Consider the following reaction at equilibrium A+B⇄C+D An increase in concentration of A or B will speed up the forward reaction, thus increasing the concentration of C and D. A similar effect can be achieved by reducing the concentration of C or D.

Concentration Add 25 m. L of 0. 0020 M KSCN to a 100 m. L beaker. To this solution, add 25 m. L of deionised water. Note the colour of the solution. Add 5 drops of 0. 02 M Fe(NO 3)3 (aq) to this solution using a Pastuer pipet. Note the color of the solution. Label the test tubes A, B, C, D.
![Fe 3+ yellow + CNS - ⇄ [Fe. CNS]2+ red • Test tube A Fe 3+ yellow + CNS - ⇄ [Fe. CNS]2+ red • Test tube A](http://slidetodoc.com/presentation_image_h/e988609da6d9ccb544039f115829d607/image-33.jpg)
Fe 3+ yellow + CNS - ⇄ [Fe. CNS]2+ red • Test tube A will be a reference • Test tube B - add 10 drops of 0. 20 M Fe(NO 3)3 (aq) using a Pastuer pipet • Test tube C - add a scoop of KSCN (s) crystals. Note the colour change of the solution. • Test tube D - add a scoop of Na. H 2 PO 4 (s) crystals using a spatula. Note the colour change of the solution


Concentration may alter the position of equilibrium Fe 3+ yellow + CNS - ⇄ [Fe. CNS]2+ red In B, the Fe 3+ ions are added. The equilibrium moves to use them up, favouring the forward reaction. The equilibrium moves right and the solution becomes more red

Concentration may alter the position of equilibrium Fe 3+ yellow + CNS - ⇄ [Fe. CNS]2+ red In C, CNS- ions are added. The equilibrium moves to remove these, favouring the forward reaction. The equilibrium moves right and the solution becomes more red

Concentration may alter the position of equilibrium Fe 3+ yellow + CNS - ⇄ [Fe. CNS]2+ red In D, the Fe 3+ ions are removed by precipitation with phosphate. The equilibrium moves to replace this, favouring the reverse reaction. The equilibrium moves left and the solution becomes more yellow

Temperature may alter the position of equilibrium

Temperature may alter the position of equilibrium Heating a reversible reaction at equilibrium shifts the reaction in the direction of the ENDOTHERMIC REACTION Cooling a reversible reaction at equilibrium shifts the reaction in the direction of the EXOTHERMIC REACTION The equilibrium will move to undo any change imposed upon it.
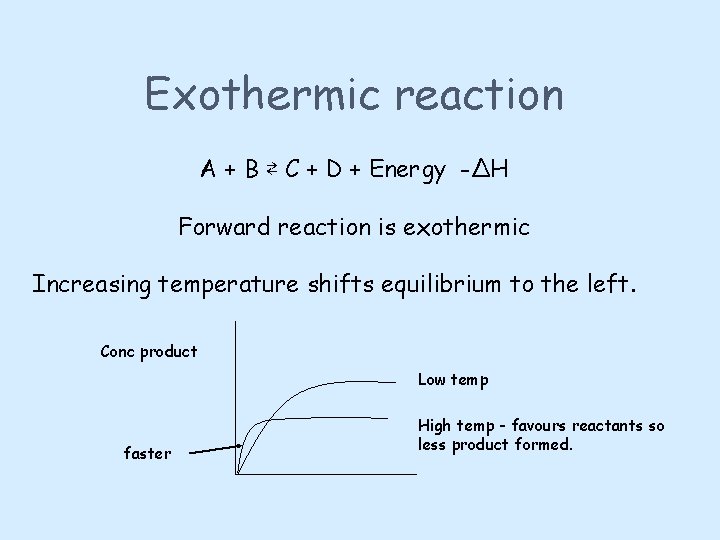
Exothermic reaction A + B ⇄ C + D + Energy -ΔH Forward reaction is exothermic Increasing temperature shifts equilibrium to the left. Conc product Low temp faster High temp - favours reactants so less product formed.
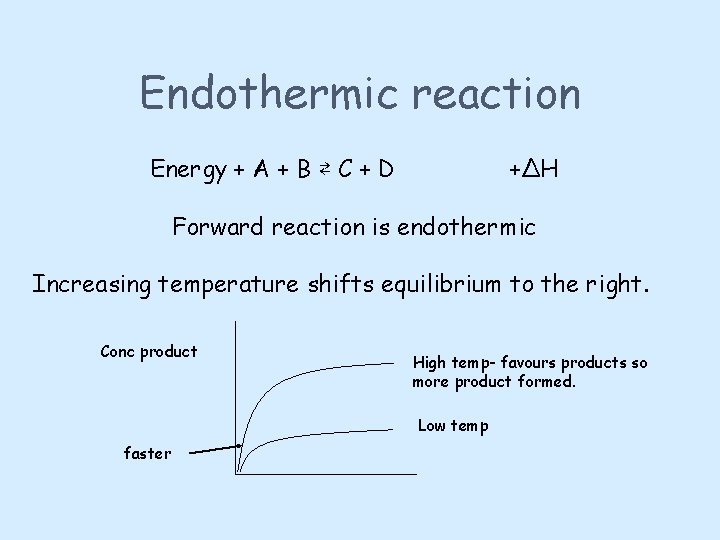
Endothermic reaction Energy + A + B ⇄ C + D +ΔH Forward reaction is endothermic Increasing temperature shifts equilibrium to the right. Conc product High temp- favours products so more product formed. Low temp faster
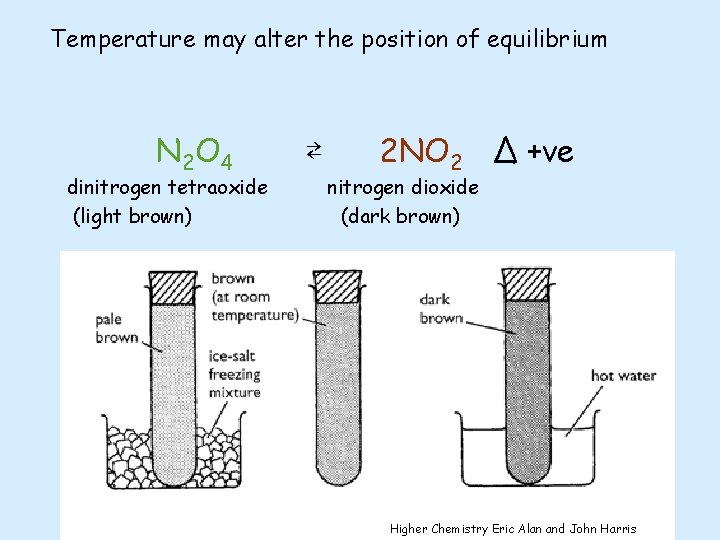
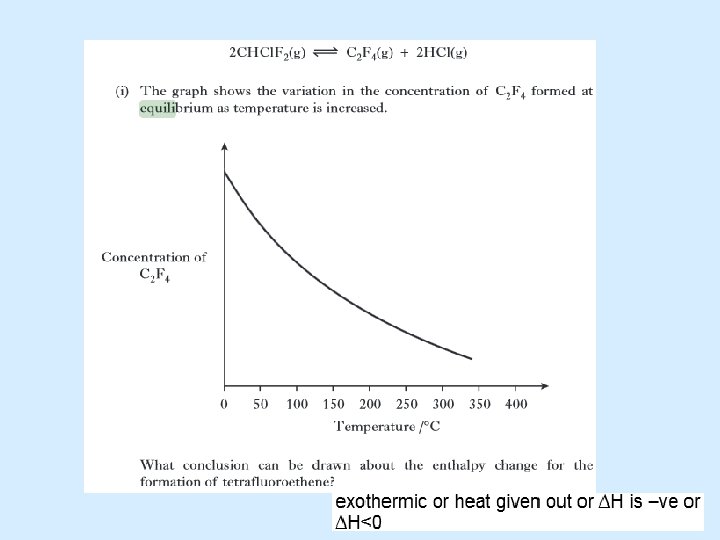
Temperature may alter the position of equilibrium N 2 O 4 dinitrogen tetraoxide (light brown) ⇄ 2 NO 2 Δ +ve nitrogen dioxide (dark brown) Higher Chemistry Eric Alan and John Harris

Temperature may alter the position of equilibrium N 2 O 4 dinitrogen tetraoxide (colourless) ⇄ 2 NO 2 Δ +ve nitrogen dioxide (dark brown) Increasing the temperature will cause the equilibrium to move to lower the temperature. The forward reaction takes in energy so the equilibrium moves to the right producing more NO 2 and less N 2 O 4. So the colour becomes darker. Decreasing the temperature will cause the equilibrium to move to raise the temperature. The reverse reaction gives out energy so the equilibrium moves to the left producing more N 2 O 4 and less NO 2. So the colour becomes lighter.

Demo Cobalt • http: //www. rsc. org/learnchemistry/resource/res 00000001/theequilibrium-between-two-coloured-cobaltspecies? cmpid=CMP 00005957 • For temperature and pressure

Concentration may alter the position of equilibrium A mixture of cobalt chloride and conc HCl sets up the following equilibrium: Co(H 2 O)62+ + 4 Cl- ⇄ Co. Cl 42 - + 6 H 2 O Δ +ve Adding extra Cl- ions forces the equilibrium to try to remove these. The forward reaction is favoured because this uses up chloride ions. The equilibrium has moved to the right so the solution becomes blue in colour.

Temperature may alter the position of equilibrium A mixture of cobalt chloride and conc HCl sets up the following equilibrium: Co(H 2 O)62+ + 4 Cl- ⇄ Co. Cl 42 - + 6 H 2 O Δ +ve If the temperature is increased the equilibrium will favour the forward reaction because that will lower the temperature. The equilibrium move to the right, therefore the solution becomes blue in colour. If the temperature is reduced the equilibrium will favour the reverse, exothermic reaction because that will increase the temperature. The equilibrium move to the left, therefore the solution becomes pinker in colour.
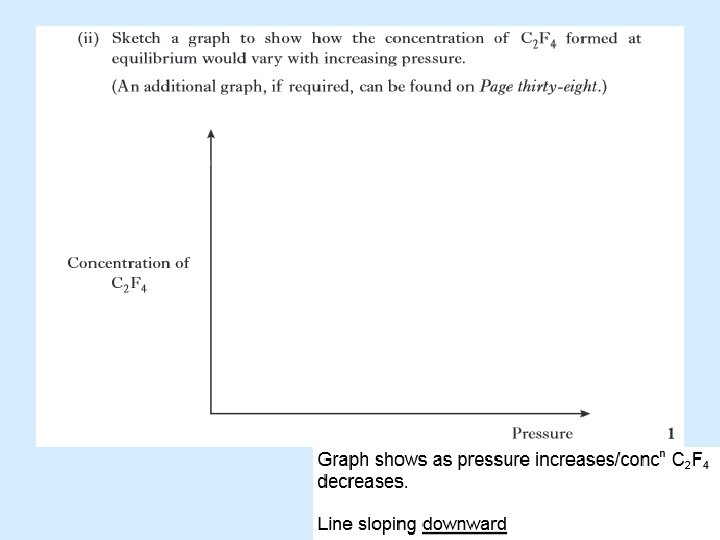




Pressure may alter the position of equilibrium

Pressure may alter the position of an equilibrium The pressure exerted by a gas is caused by the freely moving molecules bombarding the walls of the container. An increase in the number of molecules will result in an increase in pressure, assuming the size of the container is kept constant. An increase in pressure will cause the equilibrium to counteract this effect i. e. it will reduce the pressure. By favouring the side with less gas molecules.
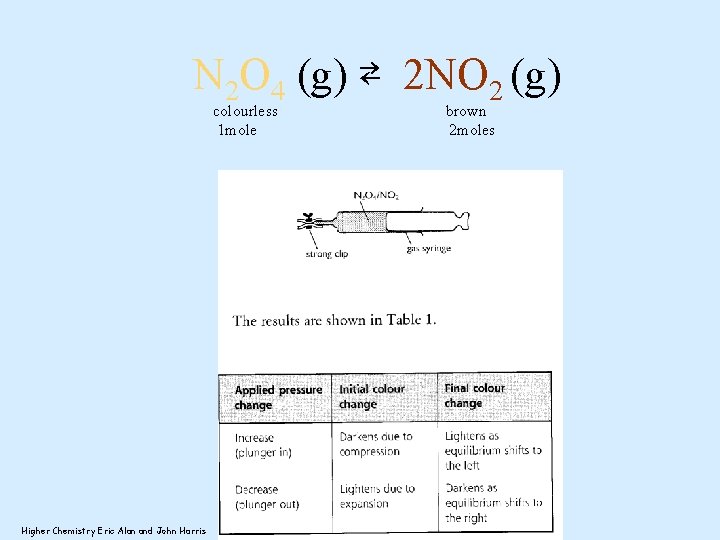
N 2 O 4 (g) ⇄ 2 NO 2 (g) colourless 1 mole Higher Chemistry Eric Alan and John Harris brown 2 moles

Pressure may alter the position of an equilibrium N 2 O 4 ⇄ colourless 1 mole, so fewer particles Lowers the pressure 2 NO 2 brown 2 moles Increasing the pressure will cause the equilibrium to move to decrease the pressure. The equilibrium will move to reduce the number of gas particles. The equilibrium moves to the left producing more N 2 O 4 and less NO 2 so the colour lightens.

Pressure may alter the position of an equilibrium N 2 O 4 ⇄ colourless 1 mole, so fewer particles 2 NO 2 brown 2 moles increase the pressure Decreasing the pressure will cause the equilibrium to move to increase the pressure. The equilibrium will move to increase the number of gas particles. The equilibrium moves to the right producing more NO 2 and less N 2 O 4 so the colour darkens.
Temperature and energy


Effect of chemicals not present in the equilibrium Dissolving chlorine in water produces the hypochlorite ion, Cl. O-, which has a bleaching effect. Cl 2 + H 2 O ⇄ 2 H + + Cl. O- + Cl-

Effect of neutralisation Cl 2 + H 2 O ⇄ 2 H + + Cl. O- + Cl- Adding an alkali will remove hydrogen ions from the equilibrium which will move to the right to replace them The bleaching effect will be increased.


Effect of precipitation Cl 2 + H 2 O ⇄ 2 H + + Cl. O- + Cl- Adding silver nitrate will remove chloride ions from the equilibrium as the precipitate silver chloride is formed. The equilibrium will move to the right to replace them so the bleaching effect will be increased.


Effect of acid Cl 2 + H 2 O ⇄ 2 H + + Cl. O- + Cl- Adding an acid causes the equilibrium to move to use up H+ ions. The equilibrium moves to the left producing more toxic Cl 2 This can be fatal and accidents caused by mixing bleach and acid are not unusual.

Starter

Catalysts • Catalysts increase the rate at which an equilibrium is formed but do not effect the equilibrium position. • The rate of the forward AND reverse reactions are speeded up equally.

7 principles of design process • • Availability of feedstocks Cost of the feedstock Sustainability of the feedstock Opportunities for recycling Energy requirements Marketability of by-products Product yield (or atom economy)

The Haber Process • How does the Haber process illustrate the 7 principles of design for an industrial process? • How is the equilibrium manipulated to move as far to the right as possible? Give 3 ways. • How is the effect of temperature on rate and on yield balanced?
Haber process The Haber Process combines nitrogen from the air with hydrogen derived mainly from natural gas (methane) into ammonia. The reaction is reversible and the production of ammonia is exothermic. http: //www. rmtech. net/uses_ of_ammonia. htm
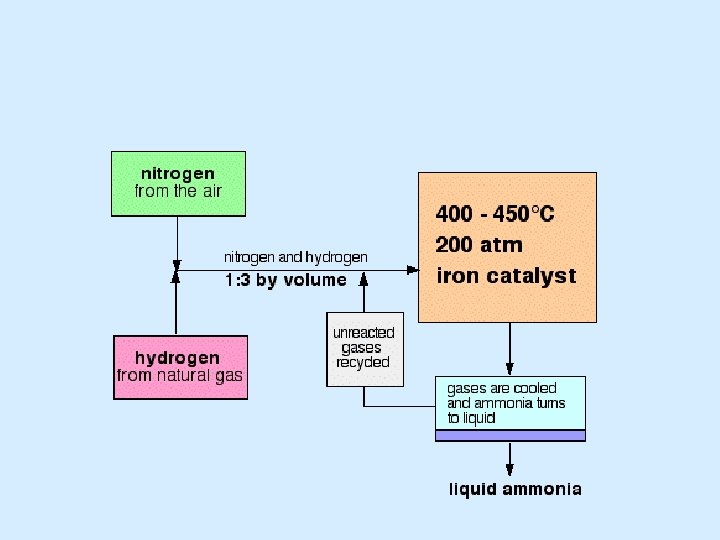

The Haber Process and equilibrium • N 2 + 3 H 2 ⇄ 2 NH 3 - ΔH • In the reactor, ammonia is removed after each cycle of the gases through the reactor. What effect would that have on the equilibrium position?

The Haber Process and equilibrium • N 2 + 3 H 2 ⇄ 2 NH 3 - ΔH • Increasing the temperature would speed up the rate of reaction, but what would happen to the amount of ammonia produced in the equilibrium? • The reacting gases are pumped around the reactor at a suitable ‘flow rate’

The Haber Process and equilibrium You need to shift the position of the equilibrium as far as possible to the right in order to produce the maximum possible amount of ammonia in the equilibrium mixture. The forward reaction (the production of ammonia) is exothermic. According to Le Chatelier's Principle, this will be favoured if you lower the temperature. The system will respond by moving the position of equilibrium to counteract this - in other words by producing more heat. In order to get as much ammonia as possible in the equilibrium mixture, you need as low a temperature as possible. However, 400 - 450°C isn't a low temperature!

The Haber Process and equilibrium The lower the temperature you use, the slower the reaction becomes. A manufacturer is trying to produce as much ammonia as possible per day. It makes no sense to try to achieve an equilibrium mixture which contains a very high proportion of ammonia if it takes several years for the reaction to reach that equilibrium. You need the gases to reach equilibrium within the very short time that they will be in contact with the catalyst in the reactor. The compromise 400 - 450°C is a compromise temperature producing a reasonably high proportion of ammonia in the equilibrium mixture (even if it is only 15%), but in a very short time.

The Haber Process and equilibrium • N 2 + 3 H 2 ⇄ 2 NH 3 - ΔH • What would be the effect of increasing the pressure? • What might be the drawbacks, both technical and economic of a high pressure?

The Haber Process and equilibrium N 2 + 3 H 2 ⇄ 2 NH 3 - ΔH Notice that there are 4 molecules on the left-hand side of the equation, but only 2 on the right. According to Le Chatelier's Principle, if you increase the pressure the system will respond by favouring the reaction which produces fewer molecules. That will cause the pressure to fall again. In order to get as much ammonia as possible in the equilibrium mixture, you need as high a pressure as possible. 200 atmospheres is a high pressure, but not amazingly high.

The Haber Process and equilibrium Increasing the pressure brings the molecules closer together. In this particular instance, it will increase their chances of hitting and sticking to the surface of the catalyst where they can react. The higher the pressure the better in terms of the rate of a gas reaction.

The Haber Process and equilibrium The compromise 200 atmospheres is a compromise pressure chosen on economic grounds. If the pressure used is too high, the cost of generating it exceeds the price you can get for the extra ammonia produced.

The Haber Process • The catalyst is iron, which is cheap. • What is the effect a catalyst on a reversible reaction?

The Haber Process and equilibrium In the absence of a catalyst the reaction is so slow that virtually no reaction happens in any sensible time. The catalyst ensures that the reaction is fast enough for a dynamic equilibrium to be set up within the very short time that the gases are actually in the reactor.
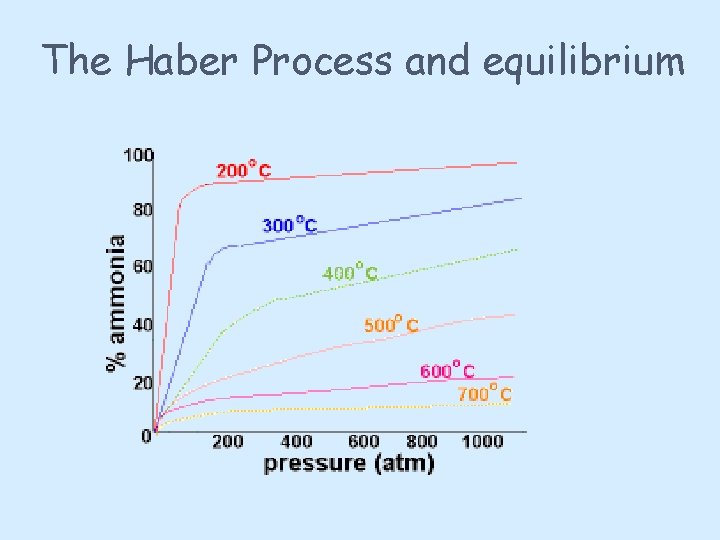
The Haber Process and equilibrium

The Haber Process and equilibrium When the gases leave the reactor they are hot and at a very high pressure. Ammonia is easily liquefied under pressure as long as it isn't too hot, and so the temperature of the mixture is lowered enough for the ammonia to turn to a liquid. The nitrogen and hydrogen remain as gases even under these high pressures, and can be recycled


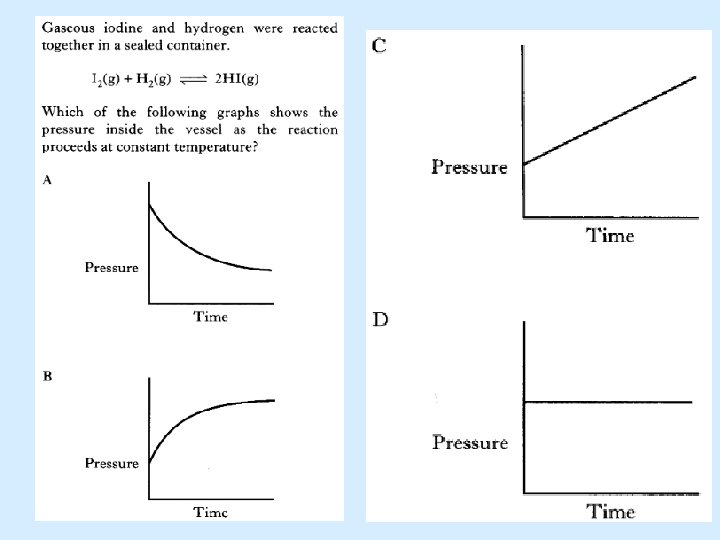
D
- Slides: 85