e Research at Emory Phase I e IRB

e. Research at Emory Phase I: e. IRB Date: 4/27/06 2/26/2021 1

Table of Contents I. What is e. Research? II. Benefits of e. Research III. What is the e. IRB? IV. Access to e. Research V. Project Timeline VI. Training Opportunities VII. Roles in the IRB Review Process VIII. e. IRB Study Review Process IX. New Study Application Overview § § § Study Team Home Workspace New Study Submission Form Study Team Study Workspace Department and Division Head Study Workspace X. Training Opportunities 2/26/2021 2

What is e. Research? Our vision is for e. Research to streamline research submissions and processing at Emory University. • e. Research is a Web-based system for submission, workflow, approval and management of research information. • Initially, the system will be used for NEW research studies involving human subjects. • The system will assist the University in standardizing the method in which human research studies are submitted, processed and reviewed. • The system will be a tool to facilitate compliance with relevant government regulations and to bring transparency to the application review process. 2/26/2021 3
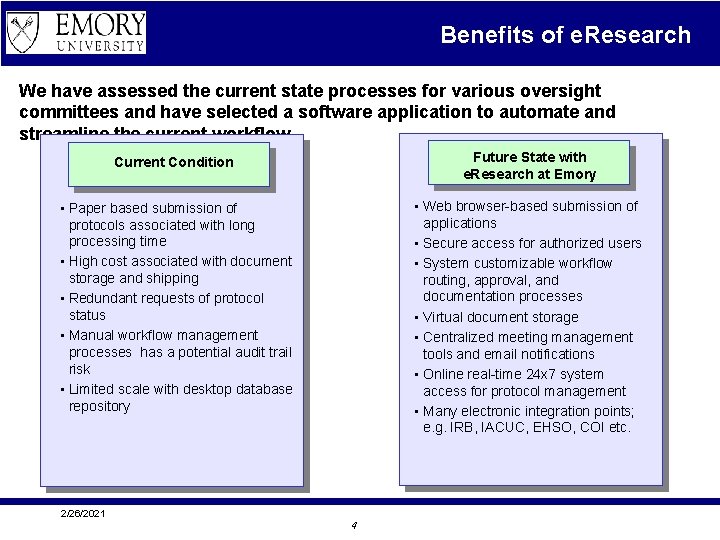
Benefits of e. Research We have assessed the current state processes for various oversight committees and have selected a software application to automate and streamline the current workflow. Current Condition Future State with e. Research at Emory • Paper based submission of protocols associated with long processing time • High cost associated with document storage and shipping • Redundant requests of protocol status • Manual workflow management processes has a potential audit trail risk • Limited scale with desktop database repository • Web browser-based submission of applications • Secure access for authorized users • System customizable workflow routing, approval, and documentation processes • Virtual document storage • Centralized meeting management tools and email notifications • Online real-time 24 x 7 system access for protocol management • Many electronic integration points; e. g. IRB, IACUC, EHSO, COI etc. 2/26/2021 4

What is e. IRB? e. IRB is the first module of the e. Research project and will be the vehicle for the IRB to streamline their business processes for handling NEW research studies with human subjects. • The IRB’s role will remain the same, the method in which they accept and manage protocol applications will change. • The system will be the tool used by the IRB to: § § § Collect study information Schedule study reviews and studies to IRB meetings Communicate study requests and status to the study team • The e. IRB module consists of 5 projects: • The study project will be the first project of the e. IRB module to be made available to the research community. § § § Study Continuing Review Amendment Reportable Events Termination 2/26/2021 5

Access to e. Research Due to HIPPA regulations login procedures to e. Research will be different than other Emory University applications. • The login id will be the same as the user’s network id. • The password will be different from the network id’s password. • The password must be a combination of 8 numbers and characters. • Passwords will expire every 90 days. • e. Research will adhere to Emory University’s network ID policies: http: //it. emory. edu/showdoc. cfm? docid=1086&fr=1070 2/26/2021 6

Project Timeline We are targeting early summer 2006 to implement the e. IRB module for new study submissions. Once a new study has been submitted, all subsequent activities related to that study will be done in the application. Apr ’ 06 May ’ 06 Jun ’ 06 Jul ’ 06 Aug ’ 06 Sept ’ 06 e. Research Intro Presentations Alpha Testing IRB Staff Training Paper Studies No Longer Accepted: July 5, 2006 Research Community Training Open Lab Sessions e. Research e. IRB module Go -Live: June 5, 2006 New Study Paper Submissions New Study Electronic Submissions Continuing Review Paper Submissions Continuing Review Electronic Submissions 2/26/2021 7 Oct ’ 06
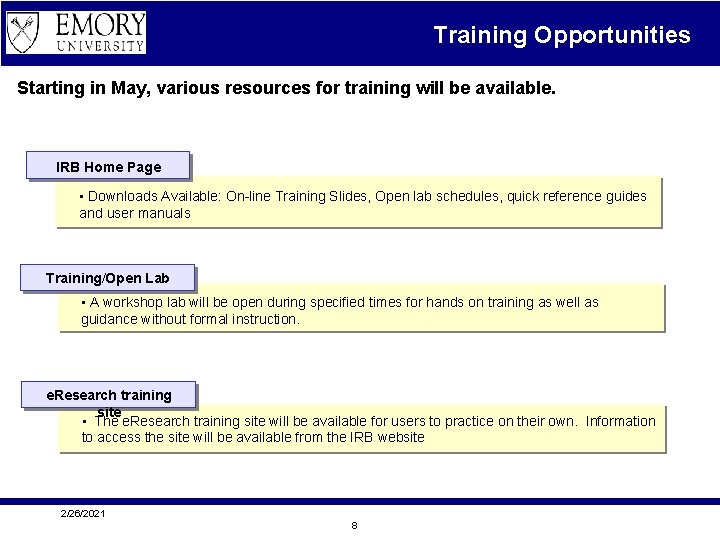
Training Opportunities Starting in May, various resources for training will be available. IRB Home Page • Downloads Available: On-line Training Slides, Open lab schedules, quick reference guides and user manuals Training/Open Lab • A workshop lab will be open during specified times for hands on training as well as guidance without formal instruction. e. Research training site • The e. Research training site will be available for users to practice on their own. Information to access the site will be available from the IRB website 2/26/2021 8
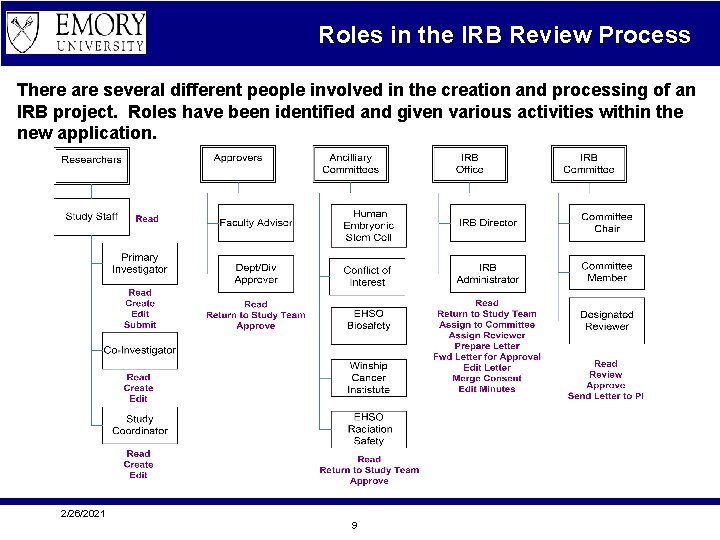
Roles in the IRB Review Process There are several different people involved in the creation and processing of an IRB project. Roles have been identified and given various activities within the new application. 2/26/2021 9
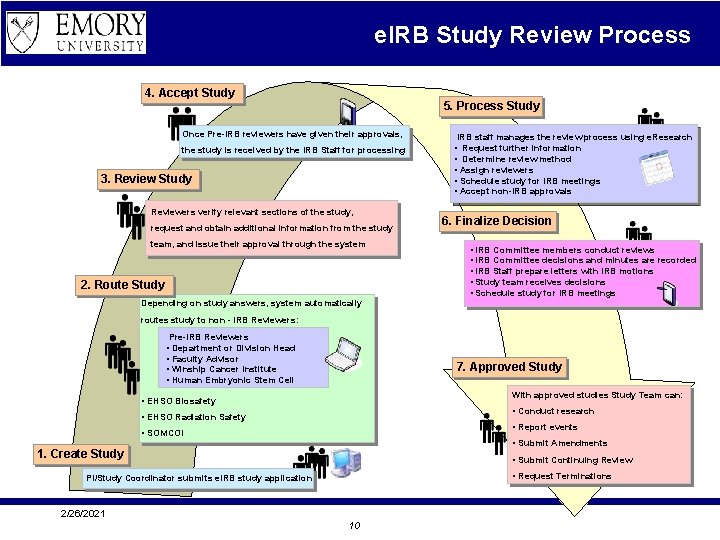
e. IRB Study Review Process 4. Accept Study 5. Process Study Once Pre-IRB reviewers have given their approvals, IRB staff manages the review process using e. Research the study is received by the IRB Staff for processing • Request further information • Determine review method • Assign reviewers • Schedule study for IRB meetings • Accept non-IRB approvals 3. Review Study Reviewers verify relevant sections of the study, request and obtain additional information from the study team, and issue their approval through the system 2. Route Study 6. Finalize Decision • IRB Committee members conduct reviews • IRB Committee decisions and minutes are recorded • IRB Staff prepare letters with IRB motions • Study team receives decisions • Schedule study for IRB meetings Depending on study answers, system automatically routes study to non - IRB Reviewers: Pre-IRB Reviewers • Department or Division Head • Faculty Advisor • Winship Cancer Institute • Human Embryonic Stem Cell 7. Approved Study With approved studies Study Team can: • EHSO Biosafety • Conduct research • EHSO Radiation Safety • Report events • SOMCOI • Submit Amendments 1. Create Study • Submit Continuing Review • Request Terminations PI/Study Coordinator submits e. IRB study application 2/26/2021 10

Study Team Home Workspace - Inbox The home workspace with the e. Research application displays all the e. IRB study submissions associated with the logged in user. The inbox tab contains submitted studies that require action by the user. 2/26/2021 11
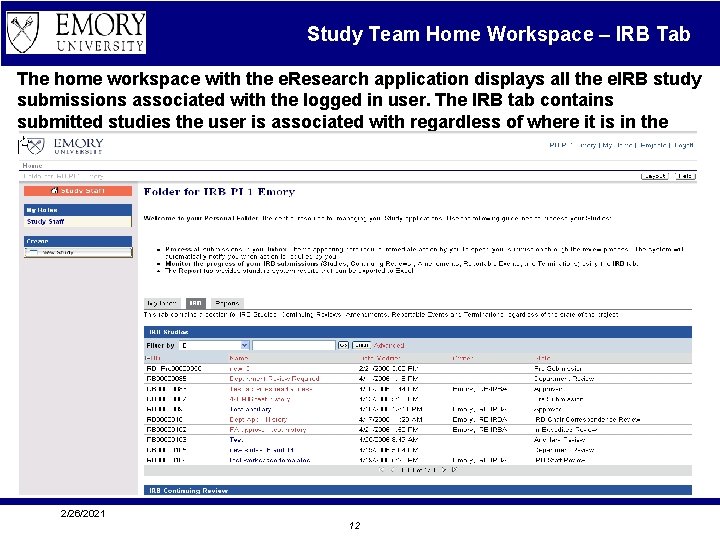
Study Team Home Workspace – IRB Tab The home workspace with the e. Research application displays all the e. IRB study submissions associated with the logged in user. The IRB tab contains submitted studies the user is associated with regardless of where it is in the process. 2/26/2021 12

New e. IRB Submission Application The new on-line submission form requires the same information to be input as on the old written form; however, it has smart-form capabilities and answers to certain questions will serve as prompts for additional information to be collected. Required Sections - Views Social/Population Views 1 Social Research 2 Study Population 3 Recruitment 2 Normal Volunteers 1 Study Identification 2 Required Reviews 3 Study Sites 4 Funding 5 Research Design 6 Research Type 7 HIPAA 8 IRB Miscellaneous Documents 9 Final Page Clinical Views 1 Biomedical Research 2 Banking 3 Clinical Trials - Device 4 Clinical Trials - Drugs 3 Minors 4 Students 5 Patients 6 Emory Employees 7 Women 8 Prisoners 1 Conflict of Interest 1 PHI 9 Cognitively Impaired 2 Human Embryonic Stem Cell 2 HIPAA 1 10 Non-English 3 EHSO-Bio Safety 3 HIPAA 2 11 Informed Consent Process 4 EHSO-Rad Safety 12 Data Safety Monitoring Plan External Committee Views 2/26/2021 13 HIPAA/PHI Views

New Study Identification View By clicking the New Study button on the home workspace, a submission form is created. The principal investigator field is pre-populated with the logged in user’s name. 2/26/2021 14
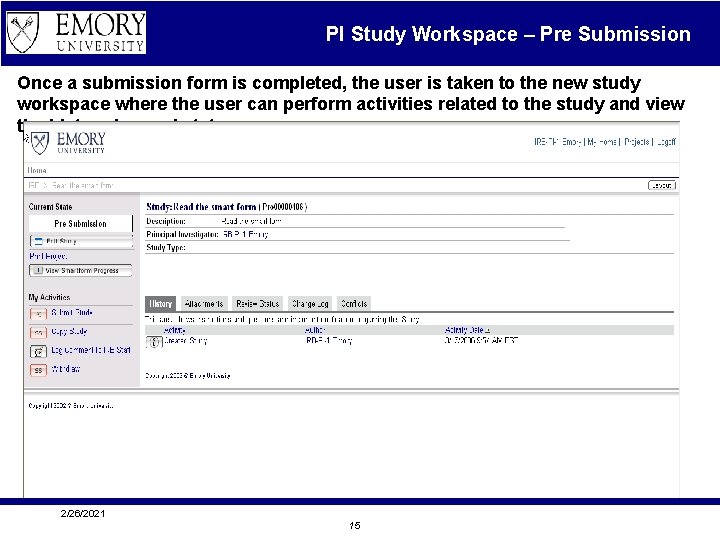
PI Study Workspace – Pre Submission Once a submission form is completed, the user is taken to the new study workspace where the user can perform activities related to the study and view the history log and status. 2/26/2021 15

PI Study Workspace – Changes required If a submission is returned for changes, the user is taken to the new study workspace where the user can perform activities related to the study and view the history log and status. 2/26/2021 16
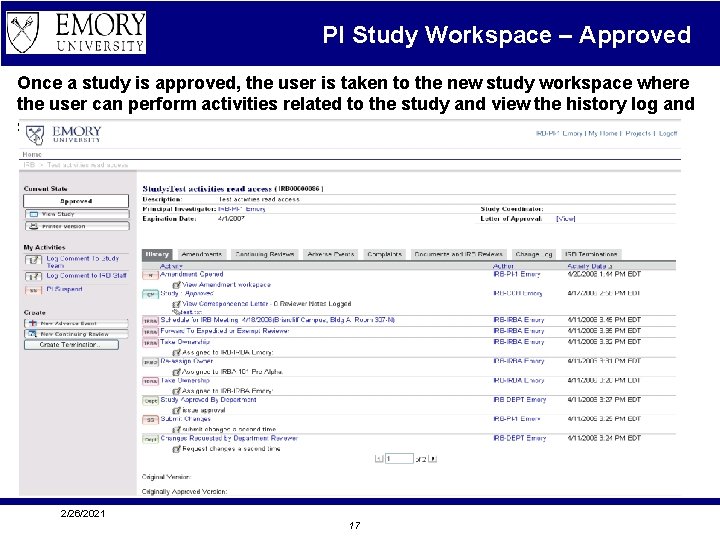
PI Study Workspace – Approved Once a study is approved, the user is taken to the new study workspace where the user can perform activities related to the study and view the history log and status. 2/26/2021 17
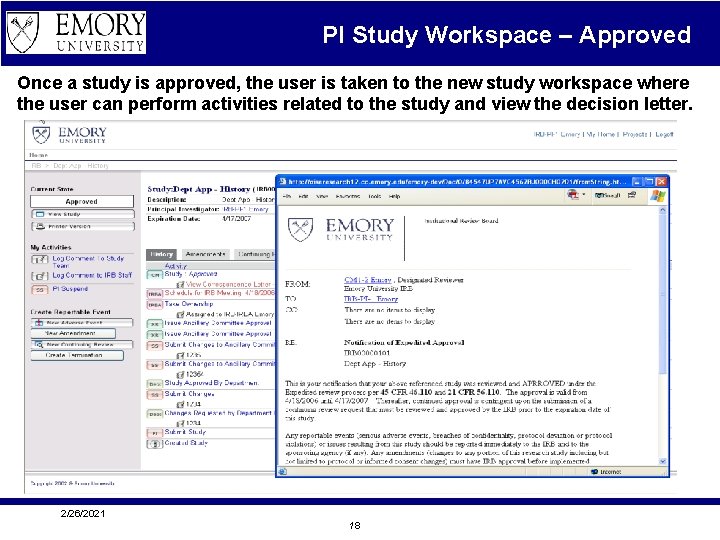
PI Study Workspace – Approved Once a study is approved, the user is taken to the new study workspace where the user can perform activities related to the study and view the decision letter. 2/26/2021 18
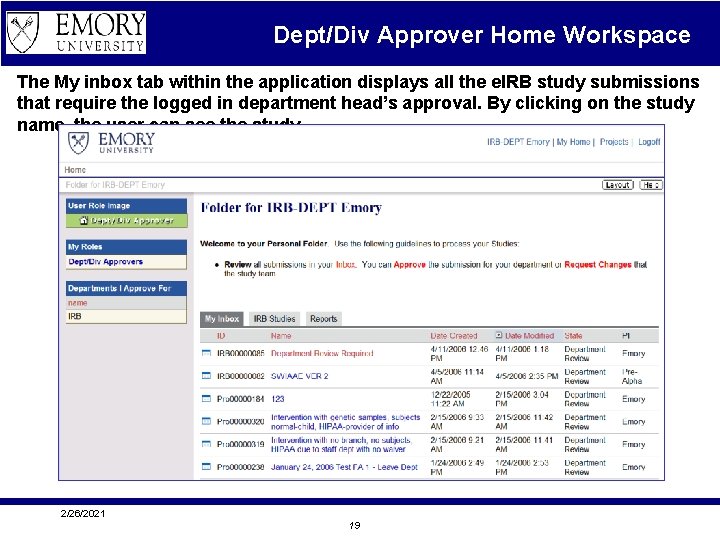
Dept/Div Approver Home Workspace The My inbox tab within the application displays all the e. IRB study submissions that require the logged in department head’s approval. By clicking on the study name, the user can see the study. 2/26/2021 19

Dept/Div Approver Study Workspace The study workspace displays details of the e. IRB study submission. The approver can view the study, request changes from the study team and issue the departmental approval. 2/26/2021 20

Open Lab Training Schedule Starting in May, various resources for training will be available. Training/Open Lab • A workshop lab will be open during specified times – • The 1 st hour will be instructor led training, The 2 nd hour will be an open lab • Monday 4: 00 PM – 6: 00 PM • Thursday 8: 00 AM – 10: 00 AM • Friday 11: 00 AM – 1: 00 PM Registration will be required To schedule group training, contact the IRB 2/26/2021 21

Contact Information e. Research at Emory Phase I: e. IRB e. Research@emory. edu Project Team: Tanya Sudia Robinson Ph. D Director, IRB/IACUC Office John B Wilson Jr. MBA/PMP Project Manager, AAIT Christine Gomez Sr. Desktop Consultant, IRB/IACUC Office 2/26/2021 22
- Slides: 22