E Neher Brainsignals Synaptic Transmission and Shortterm Plasticity

E. Neher Brainsignals, Synaptic Transmission and Short-term Plasticity. . and about Young Investigator Groups in Europe Ideas of the ERC and Excellence in Research Tallinn May 29 th, 2007

The Origin of ‚Bioelectricity‘: 1780 - 1800 Galvani Volta Lichtenberg
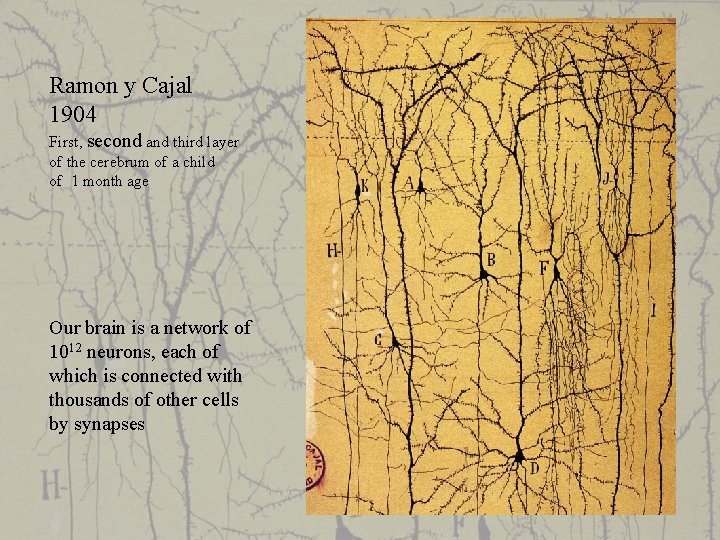
Ramon y Cajal 1904 First, second and third layer of the cerebrum of a child of 1 month age Our brain is a network of 1012 neurons, each of which is connected with thousands of other cells by synapses

Ramon y Cajal already predicted the direction of signal propagation - and was mostly right -
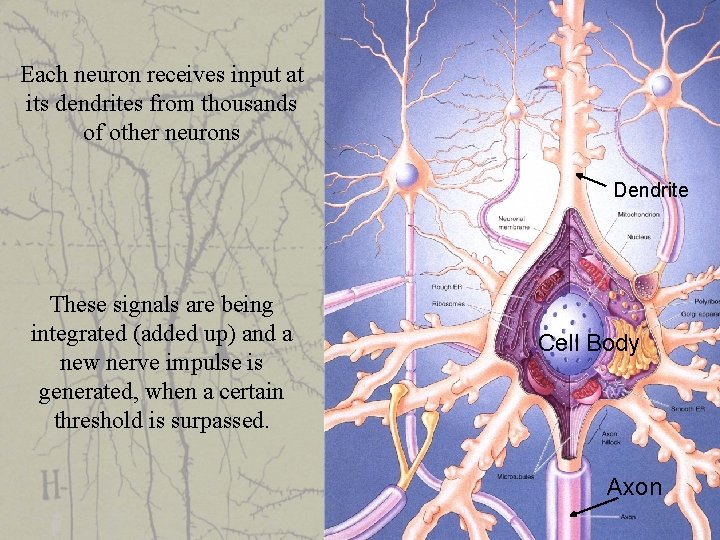
Each neuron receives input at its dendrites from thousands of other neurons Dendrite These signals are being integrated (added up) and a new nerve impulse is generated, when a certain threshold is surpassed. Cell Body Axon
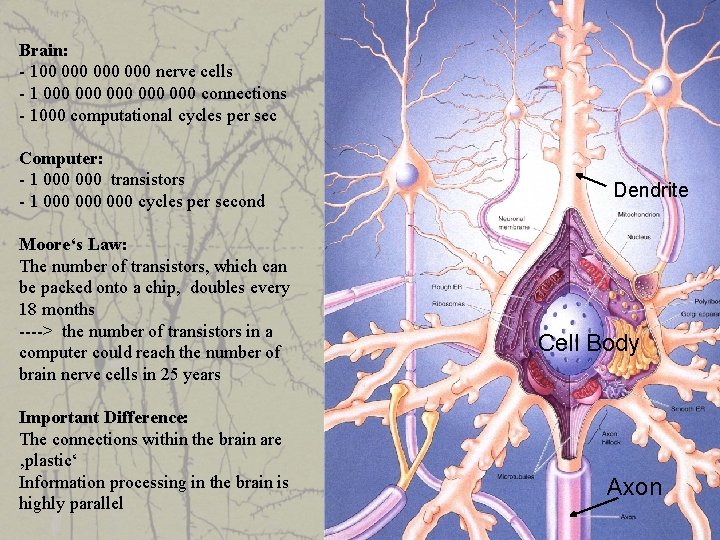
Brain: - 100 000 000 nerve cells - 1 000 000 000 connections - 1000 computational cycles per sec Computer: - 1 000 transistors - 1 000 000 cycles per second Moore‘s Law: The number of transistors, which can be packed onto a chip, doubles every 18 months ----> the number of transistors in a computer could reach the number of brain nerve cells in 25 years Important Difference: The connections within the brain are ‚plastic‘ Information processing in the brain is highly parallel Dendrite Cell Body Axon

Synaptic Plasticity • • • Neuroscientists use the term ‚Plasticity‘ to describe the observation that synaptic strength changes constantly, depending upon use of a synapse Plasticity of synaptic connections underlies the complex information processing of the CNS Plasticity occurs on time scales of milliseconds to years Nature uses all possible mechanisms, to achieve a finely tuned regulation of synaptic transmission When we study synaptic transmission today, we not only want to understand the process of transmission per se, but also why synaptic strength changes in a usedependent manner
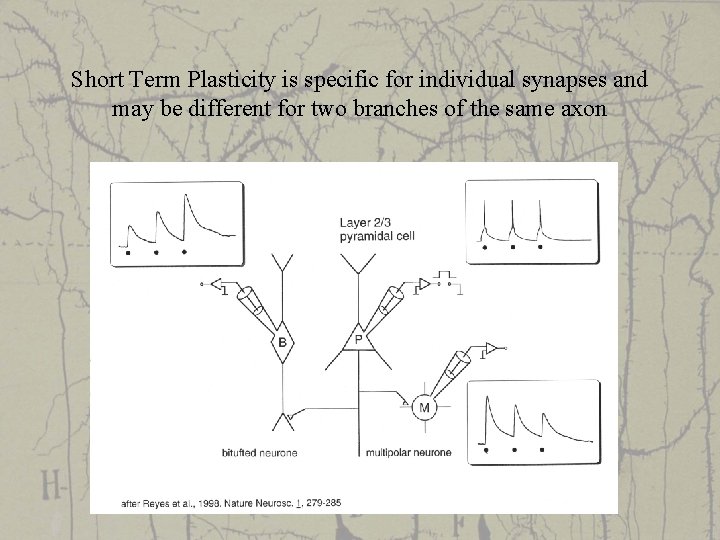
Short Term Plasticity is specific for individual synapses and may be different for two branches of the same axon

Short term depression is a key mechanism for a number of network properties • Sensory Adaptation (Chung et al. , 2002. Neuron 34, 437 -446) • Cortical Gain Control (Abbott et al. , 1997. Science 275, 220 -224) • Rhythm Generation (Senn et al. , 1996. Neural Networks 9 , 575 -588) • Network Resonance (Houweling et al. , 2002. J. Physiol 542, 599 -617) • Temporal Filtering (Fortune and Rose, 2001. TINS 24, 381 -385)
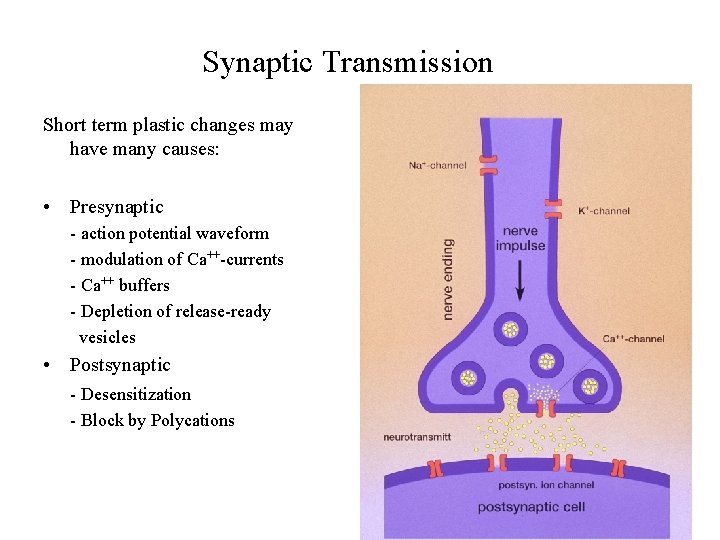
Synaptic Transmission Short term plastic changes may have many causes: • Presynaptic - action potential waveform - modulation of Ca++-currents - Ca++ buffers - Depletion of release-ready vesicles • Postsynaptic - Desensitization - Block by Polycations

In general, presynaptic terminals are very small, such that the study of neurotransmitter release is difficult. . However, it has been known for more than 100 years, that there are giant nerve terminals in certain regions of the brain Cajal: ‚Calices de Held, para refutar antineuronistas‘
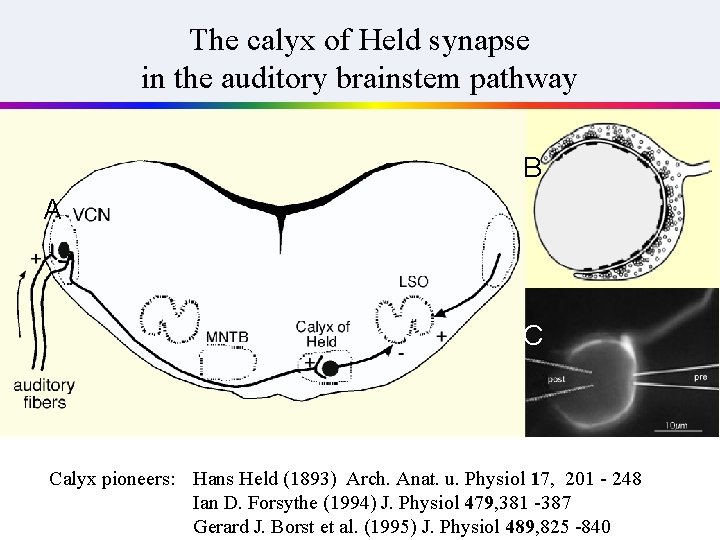
The calyx of Held synapse in the auditory brainstem pathway B A C Calyx pioneers: Hans Held (1893) Arch. Anat. u. Physiol 17, 201 - 248 Ian D. Forsythe (1994) J. Physiol 479, 381 -387 Gerard J. Borst et al. (1995) J. Physiol 489, 825 -840
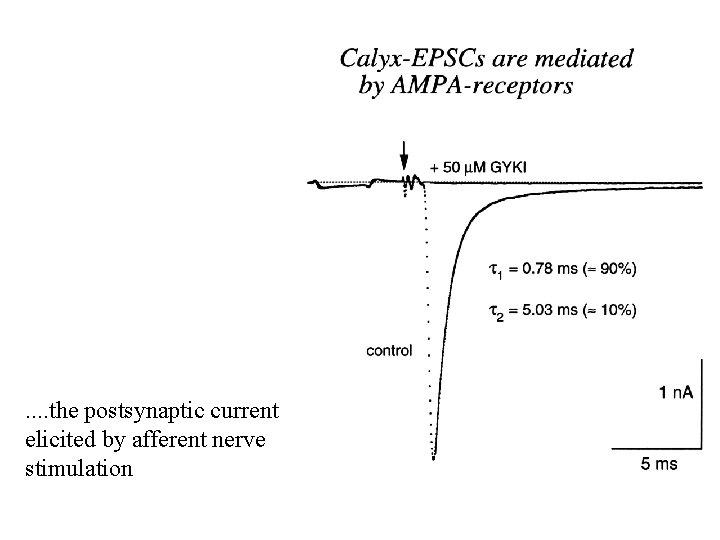
. . the postsynaptic current elicited by afferent nerve stimulation
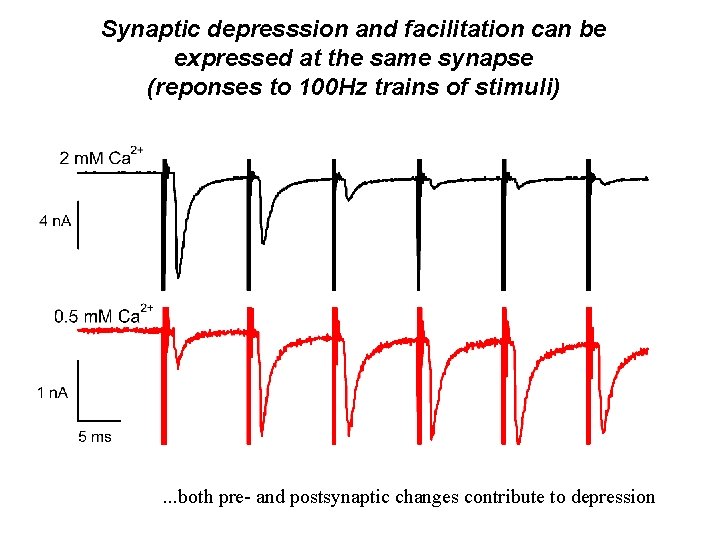
Synaptic depresssion and facilitation can be expressed at the same synapse (reponses to 100 Hz trains of stimuli) . . . both pre- and postsynaptic changes contribute to depression
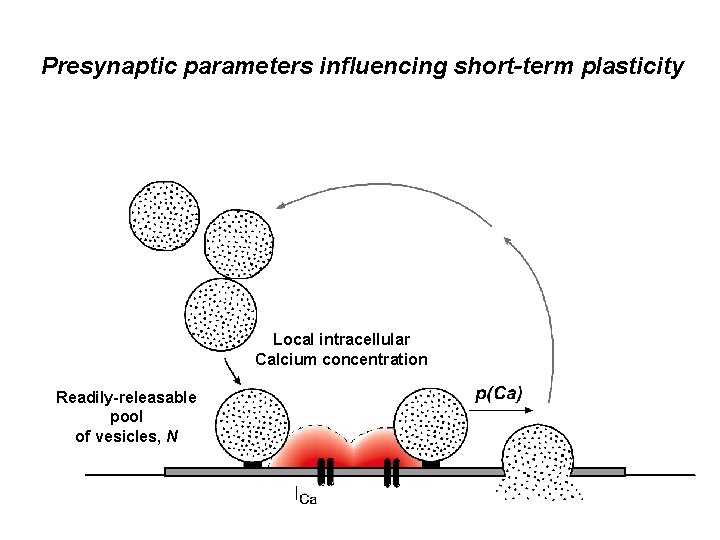
Presynaptic parameters influencing short-term plasticity Local intracellular Calcium concentration Readily-releasable pool of vesicles, N
![What is the microdomain [Ca 2+] needed for presynaptic vesicle fusion? What is the microdomain [Ca 2+] needed for presynaptic vesicle fusion?](http://slidetodoc.com/presentation_image/4566b54578986a863a342f783acebcba/image-16.jpg)
What is the microdomain [Ca 2+] needed for presynaptic vesicle fusion?
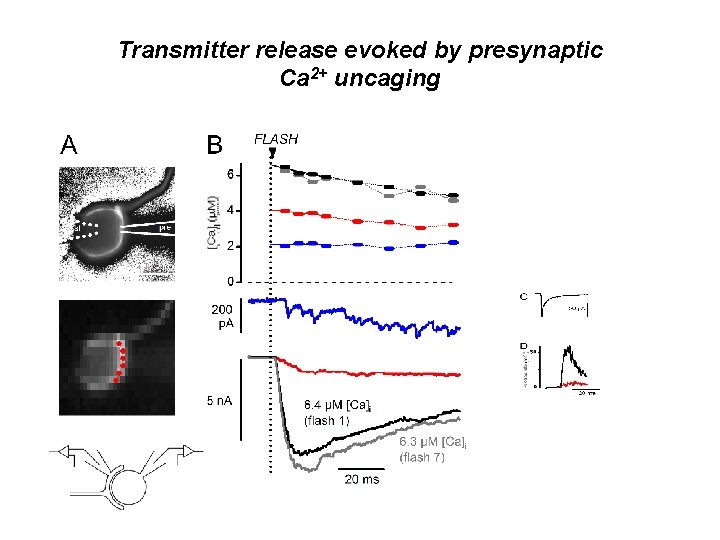
Transmitter release evoked by presynaptic Ca 2+ uncaging

Conclusions I • Ca++ uncaging allows one to establish a ‚dose-response-curve‘ - release-rate versus [Ca 2+] • During an action potential [Ca 2+] is postulated to rise to a peak of ≈ 20µM and 0. 5 msec width at the release site • Such high Ca++ concentrations are only obtained in microdomains around open Ca++ channels, which rapidly collapse, when channels close. . recent measurements by Bollmann and Sakmann Nat Neurosci. (2005), 8, 426 -34, in which short [Ca 2+] transients were produced by uncaging, show that only such short transients produce responses, which are similar to action potential-induced ones

Facilitation: Proposed Mechanisms • Residual Calcium (Katz and Miledi) • Extra Calcium Senor • Unblock of Polycations (postsynaptic) Question: Does Ca++ sensitivity change during pairedpulse facilitation
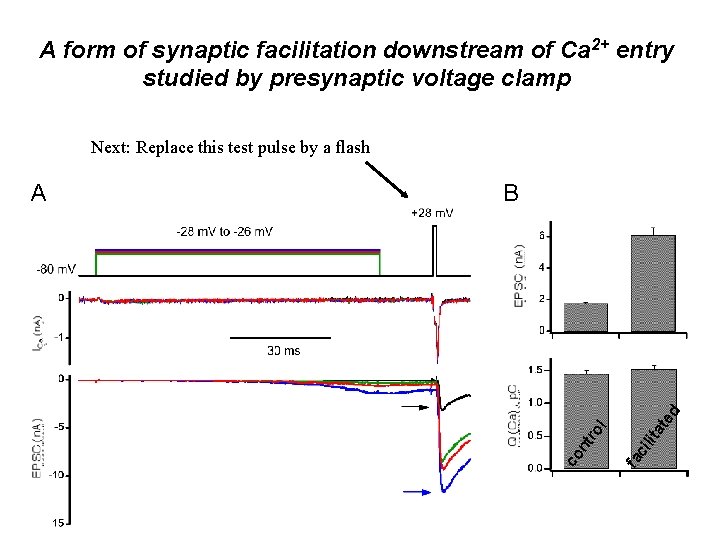
A form of synaptic facilitation downstream of Ca 2+ entry studied by presynaptic voltage clamp Next: Replace this test pulse by a flash ci lit fa ro l at e d B co nt A
![Is the Ca 2+ sensitivity of vesicle fusion increased during synaptic facilitation? Δ[Ca]i Is the Ca 2+ sensitivity of vesicle fusion increased during synaptic facilitation? Δ[Ca]i](http://slidetodoc.com/presentation_image/4566b54578986a863a342f783acebcba/image-21.jpg)
Is the Ca 2+ sensitivity of vesicle fusion increased during synaptic facilitation? Δ[Ca]i
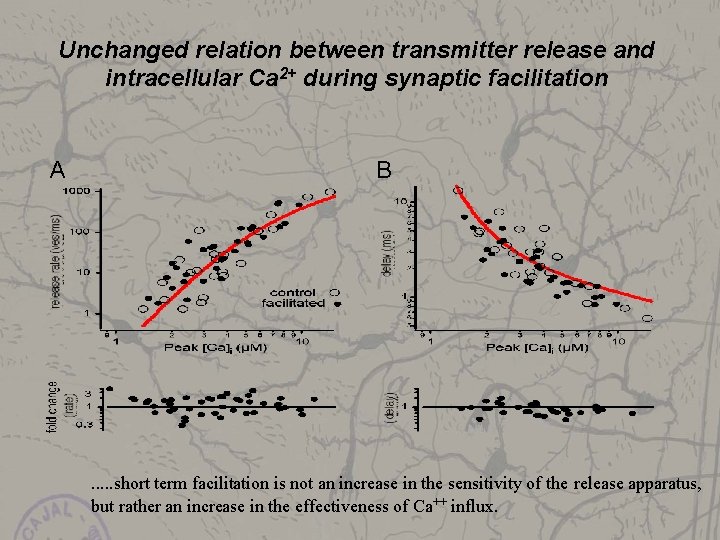
Unchanged relation between transmitter release and intracellular Ca 2+ during synaptic facilitation A B . . . short term facilitation is not an increase in the sensitivity of the release apparatus, but rather an increase in the effectiveness of Ca++ influx.

Conclusions from Ca++ uncaging : • Ca++ uncaging allows to establish a ‚dose-response-curve‘ - release-rate versus [Ca 2+] • during an action potential [Ca 2+] rises to a peak of ≈ 20 u. M • the Ca++ sensitivity of the release apparatus does not change during short term facilitation
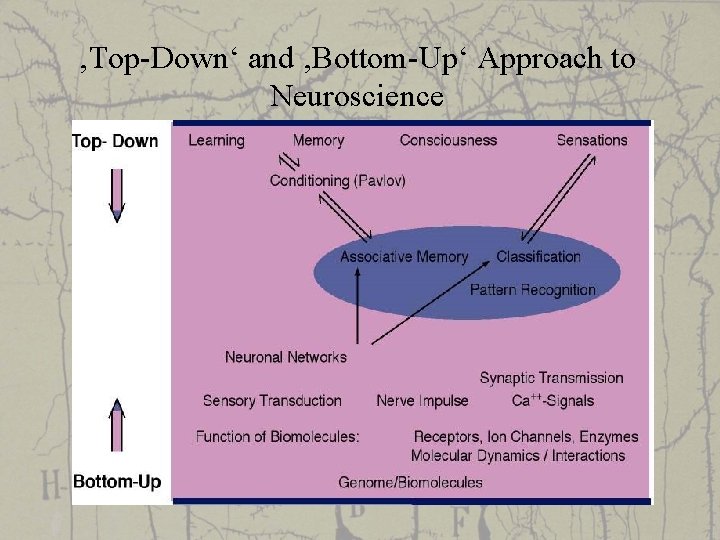
‚Top-Down‘ and ‚Bottom-Up‘ Approach to Neuroscience

ENI-Net: A European network, dedicated to the promotion of Young Investigators Its Goals: • Promote the independent research of Young Investigators (Career Development) • Intensify Collaboration and trainig • Stimulate Joint Activities, including applications to other programmes of the EU • Contribute to the establishment of the ‘European Research Area’

ENI-Net: A European network - the Alicante Meeting January, 2004 The participating Institutions commit themselves to provide laboratory space and infrastructure for at least 2 Young Investigator Groups Independence of Young Investigators is monitored by a ‘Steering committee’ Plans for: Yearly meetings, workshops, and exchange stimulating intensive collaboration Steering Committee of ENI-Net: Dr. David Attwell, London Dr. Carlos Belmonte, Alicante Dr. Christoph Mulle, Bordeaux Dr. Erwin Neher, Göttingen (Chair) Dr. Eva Sykova, Prague

ENI-Net: A European network, dedicated to the promotion of Young Investigators Application to Brussels for a Coordination Action in the Neurosciences (Sep. to Nov. 2004) : Twelve Institutes in 2004, now 18 48 Young Investigators approved by the Steering Committee 1. 2 Mio € granted through a CA for yearly meetings, workshops, and exchange
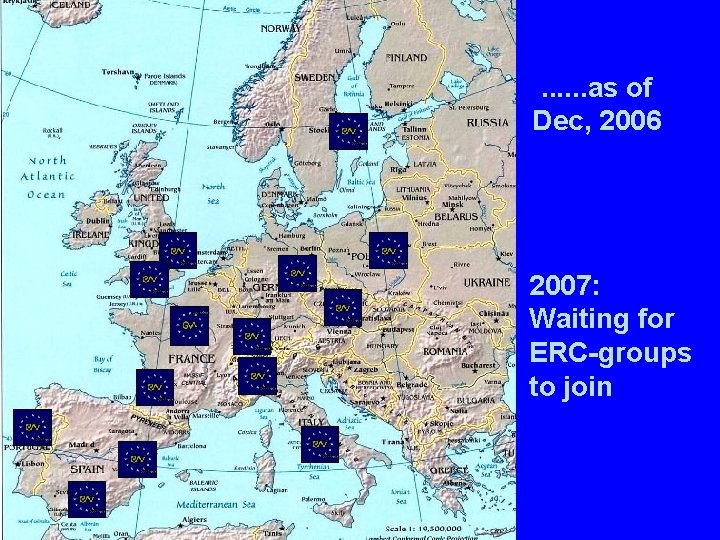
. . . as of Dec, 2006 2007: Waiting for ERC-groups to join
- Slides: 28