E coli Whats next HOST Bill Mc Dowell

E. coli: What’s next?

HOST Bill Mc. Dowell Editorial Director, Meatingplace MODERATOR Mike Fielding Editor, Meatingplace

POLL QUESTIONS Will the “Big Six” be declared to be product adulterants? • Yes • No Should they be? • Yes • No

Non-O 157 Shiga Toxin Producing E. coli: Biology, Detection and Significance Mohammad Koohmaraie, Ph. D. CEO – Meat Division IEH Laboratories & Consulting Group Lake Forest Park, WA

Presentation Outline • • • Nomenclature O 157 vs. non-O 157 Will Non-O 157 STECs get adulterant designation? Detection – FSIS Approach Prevalence Summary and conclusions

Nomenclature E. coli O 157: H 7
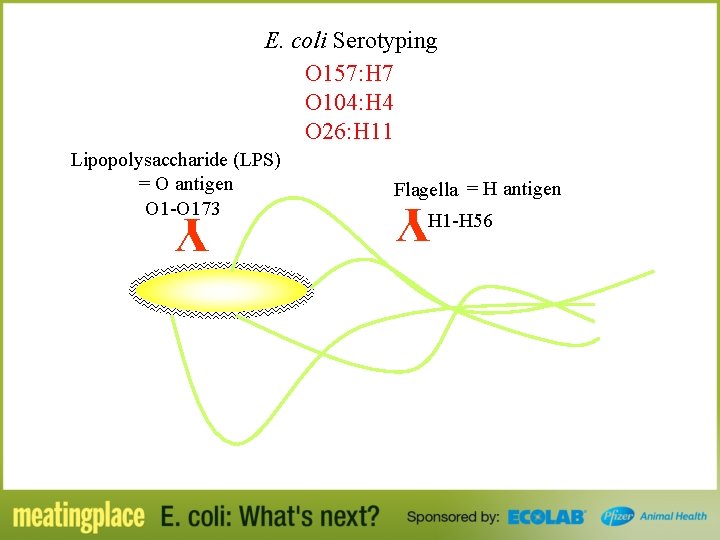
E. coli Serotyping O 157: H 7 O 104: H 4 O 26: H 11 Flagella = H antigen Y Lipopolysaccharide (LPS) = O antigen O 1 -O 173 H 1 -H 56 Y

Pathogenicity • Not all E. coli strains are pathogenic. • To be pathogenic, a strain must have the necessary properties to cause disease in human. • These properties are called “virulence factors. ” • Exactly what virulence factors are required is unknown.

Pathogenicity • E. coli can cause human disease when they possess stx 1 or stx 2. • Individuals infected with strains producing stx 2 are more likely to develop severe disease than those infected with strains carrying Shiga toxin 1. • It is commonly thought that E. coli must contain stx 1 or stx 2 and eae (intimin) and its substitutes to have the highest chance of causing disease in humans – of course there always exceptions.

Shiga Entro Toxin producing Hemorrhagic Escherichia Coli STEC EHEC p. STEC

Top Non-O 157 Serotypes (CDC) – – – O 26 O 111 O 103 O 121 O 45 O 145 22% of non-O 157 STEC 16% of non-O 157 STEC 12% of non-O 157 STEC 9% of non-O 157 STEC 7% of non-O 157 STEC 5% of non-O 157 STEC
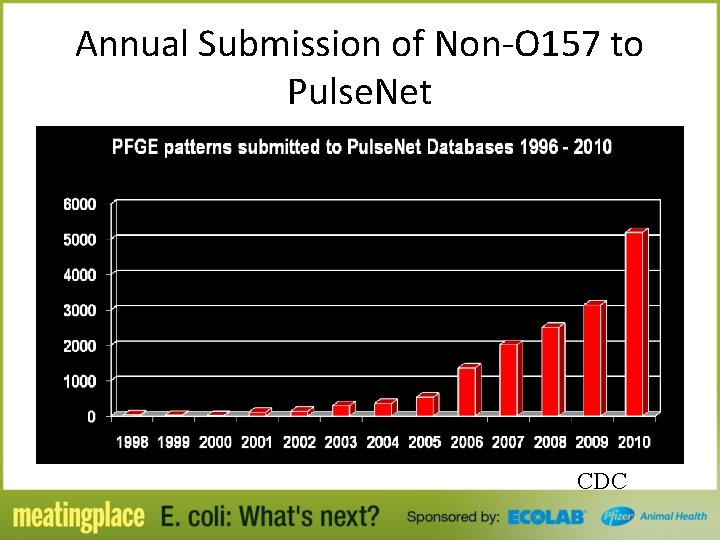
Annual Submission of Non-O 157 to Pulse. Net CDC

E. coli O 157 vs. Non-O 157 E. coli

Sequence of events in E. coli O 157: H 7 infection E. coli O 157 ingested 3 - 4 days non-bloody diarrhea, abdominal cramps 80% 1 - 2 days bloody diarrhea 92% resolution Mead. Lancet 1998 5 -6 days 8% HUS CDC
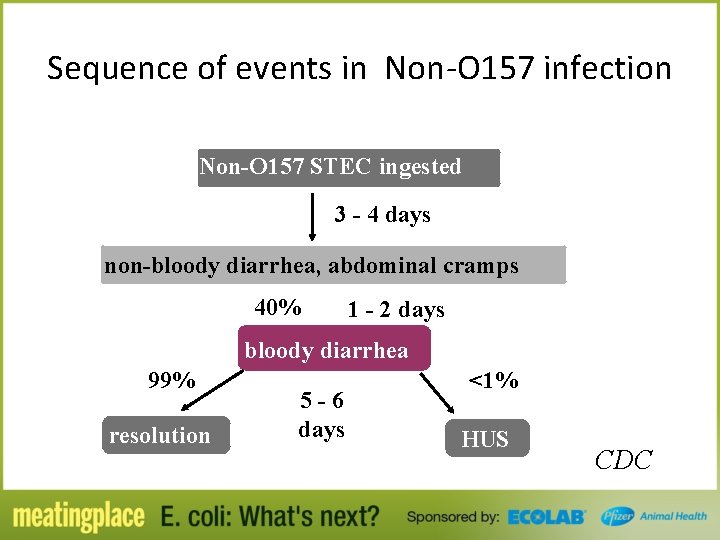
Sequence of events in Non-O 157 infection Non-O 157 STEC ingested 3 - 4 days non-bloody diarrhea, abdominal cramps 40% 1 - 2 days bloody diarrhea 99% resolution 5 -6 days <1% HUS CDC

Food commodities implicated outbreaks of non-O 157 STEC infections with known food commodity, 19902007 Commodity Fruit-nuts Number outbreaks 3 Food items Juice, apple cider, berries Dairy 2 Leafy vegetables 1 Cheese, margarine Lettuce No outbreaks due to beef

But it all changed in 2010 with the recall of ground beef because of human illness associated with O 26 contamination

Will Non-O 157 STECs Get Adulterant Designation? • It is a matter of when and not if any more • Less than 12 months

Detection

Testing for STEC • E. coli O 157 – Unusual feature: does not ferment sorbitol • Non-O 157 STEC – Lack unusual features – A group of organisms

USDA Approach • • Meat sample Enrich Screen for eae + stx If negative – STOP If positive, PCR to determine if it is a top 6 If one of the top 6 – Sample is POSITIVE If not a top 6 – Sample is NEGATIVE

Non-O 157 Prevalence

Prevalence of Non-O 157 STEC • Commercial fed cattle processing plants as a function of the season of the year • Commercial cow/bull processing plants • Commercial lamb processing plants • Imported raw ground beef material (trim) • National ground beef supply We are very appreciative of the U. S. meat industry for allowing us to use their facilities as our laboratory.
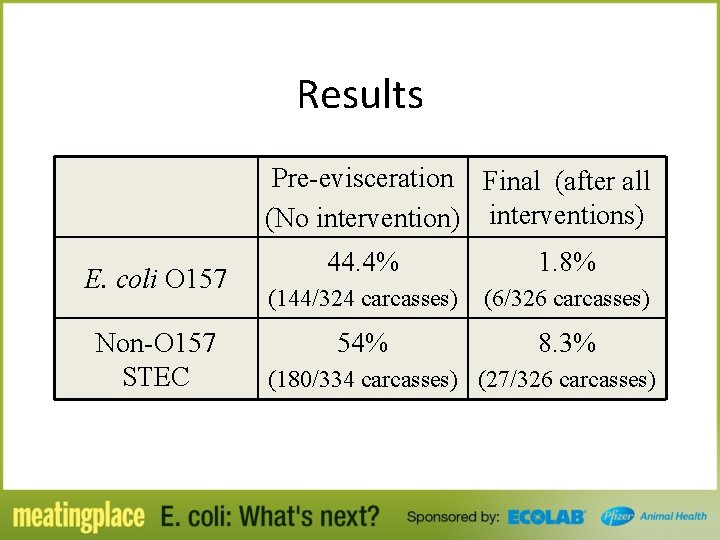
Results Pre-evisceration Final (after all (No intervention) interventions) E. coli O 157 Non-O 157 STEC 44. 4% 1. 8% (144/324 carcasses) (6/326 carcasses) 54% 8. 3% (180/334 carcasses) (27/326 carcasses)

STEC Prevalence in Imported and Domestic Boneless Beef Trim Used for Ground Beef

STEC Frequency of STEC isolation in boneless beef trim by country of origin

Serotypes of STEC isolated by country Underlined serotypes have been associated with human illness. Bolded serotypes have been associated with Hemolytic Uremic Syndrome (HUS).

A National Survey of the Prevalence of non-O 157 Shiga toxin-producing E. coli in Ground Beef

BIFSCo Database Microbiological Regions 1 2 4 5 8 6 3 7

Ground Beef Non-O 157 STEC 4133 samples Supplies by ground beef manufacturers over a 24 months period 24. 3% (1006/4133) of the samples were positive for Shiga toxin (stx 1 or stx 2). • In all, 0. 2% of the samples (10/4133) could be considered a food safety threat. • •

Control Hide is the principal source of most pathogens. An effective control in order of significance is: 1 st - Dressing practices 2 nd - Effective interventions • Effective – scientific support • Works in your hands – validation • Must make contact the potential pathogens 3 rd - Effective test and hold

Summary and Conclusions • STEC are a natural part of the animal microflora. • Some Non-O 157 STEC can cause severe disease in humans. • Non-O 157 STEC is found at high frequency in preharvest samples (feces and hides). • The prevalence on carcass depends on the adequacy of dressing practices.

Summary and Conclusions • A thoughtful and comprehensive approach should be used to develop a national policy with respect to the control of non-O 157 STEC. • This approach should be such that remove the most virulent E. coli from the food supply not just some and not just any. • In this way we are truly reducing the burden to public health of pathogenic E. coli without undue economic hardship.

Mike Mullen Corporate Account Manager For more information contact: melissa. meinke@ecolab. com www. ecolab. com or 800 -392 -3392

The Sinful Six Interventions to Minimize the Risk for Beef Processors: A Research and Application Update Harshavardhan Thippareddi, Ph. D. Associate Professor and Extension Food Safety Microbiologist Dept. of Food Science and Technology University of Nebraska, Lincoln, NE 68583

Outline • • • Introduction Prevalence of STECs Antimicrobial Interventions for STECs Non-O 157 STEC Challenges for the processor Non-intact Beef – STECs Summary

Innovative Interventions Developed and Applied by the Beef Industry • 1993 E. coli O 157: H 7 Pacific Northwest – Knife trimming and water washing • 2009 – Multiple meat processing interventions – Sequential – Hurdle Technologies – Primary and Secondary – At all phases of meat processing

Outline • • • Introduction Prevalence of STECs Antimicrobial Interventions for STECs Non-O 157 STEC Challenges for the processor Non-intact Beef – STECs Summary

Non-O 157 Shiga toxin producing E. coli – From FSIS Public Meeting, 2007 • Cattle STEC prevalence data varies: – 0 -19%, dairy cattle 1, 2, 3, 4 – 19. 4 – 56. 3% 5, beef cattle feces/hides • Food prevalence data very limited: – Pre-evisceration beef carcasses >50%5, 6 – Retail ground beef, 2. 3%7 1 Wachsmuth et al. , 1991 2 Wells et al. , 1991 3 Cray et al. , 1996 4 Thran et al. , 2001 5 Barkocy-Gallagher, et al. , 2003 6 Arthur et al. , 2002 7 Samadpour et al. , 2006 Dr. Hagen, 2007

Outline • • • Introduction Prevalence of STECs Antimicrobial Interventions for STECs Non-O 157 STEC Challenges for the processor Non-intact Beef – STECs Summary

Antimicrobial Interventions for Slaughter, Fabrication and Grinding

Antimicrobial Agents: Classification n n Direct Food Additives ► Sod. or Pot. Lactates, Buffered sodium citrate, sod. Diacetate and Lactoferrin, Irradiation ► Considered ingredients, need to be labeled as such Secondary Direct Food Additives ► Peroxy acids, ASC, Ozone ► No labeling requirement

Antimicrobial Interventions n Slaughter: – Chemical dehairing – Hot water rinses – Steam pasteurization – Steam vacuum – Chemical rinses – Lactoferrin

Antimicrobial Interventions n Fabrication: – Organic acid rinses – Sanova – Ozone – Per-acetic acid – Lauric Arginate – Lactoferrin

Antimicrobial Interventions n Trim for Grinding: n Ground Beef: – Organic acid rinses – Ozone – Per-acetic acid – ASC – Multiple hurdle technology – High Pressure Processing – Irradiation

Chemical Rinses n n n Chlorine, Organic acids most commonly used – Organic acids – lactic, acetic, citric Improves microbiological quality of carcasses Other chemicals include Per-oxy acetic acid, Acidified sodium chlorite, CPC
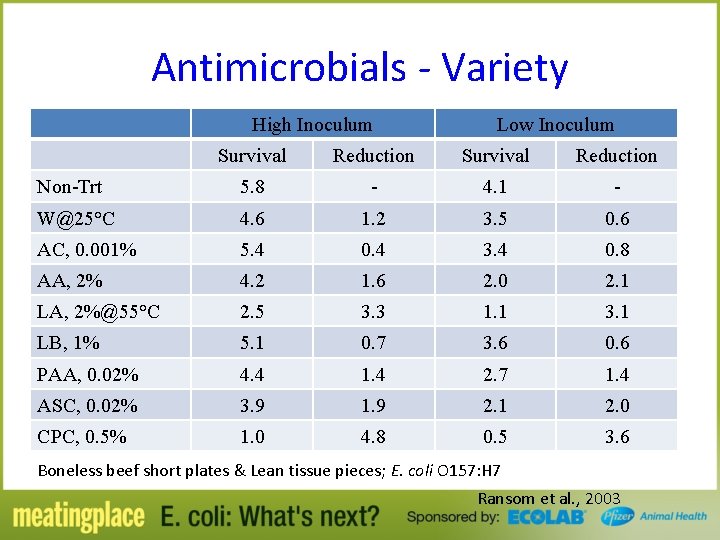
Antimicrobials - Variety High Inoculum Low Inoculum Survival Reduction Non-Trt 5. 8 - 4. 1 - W@25°C 4. 6 1. 2 3. 5 0. 6 AC, 0. 001% 5. 4 0. 4 3. 4 0. 8 AA, 2% 4. 2 1. 6 2. 0 2. 1 LA, 2%@55°C 2. 5 3. 3 1. 1 3. 1 LB, 1% 5. 1 0. 7 3. 6 0. 6 PAA, 0. 02% 4. 4 1. 4 2. 7 1. 4 ASC, 0. 02% 3. 9 1. 9 2. 1 2. 0 CPC, 0. 5% 1. 0 4. 8 0. 5 3. 6 Boneless beef short plates & Lean tissue pieces; E. coli O 157: H 7 Ransom et al. , 2003
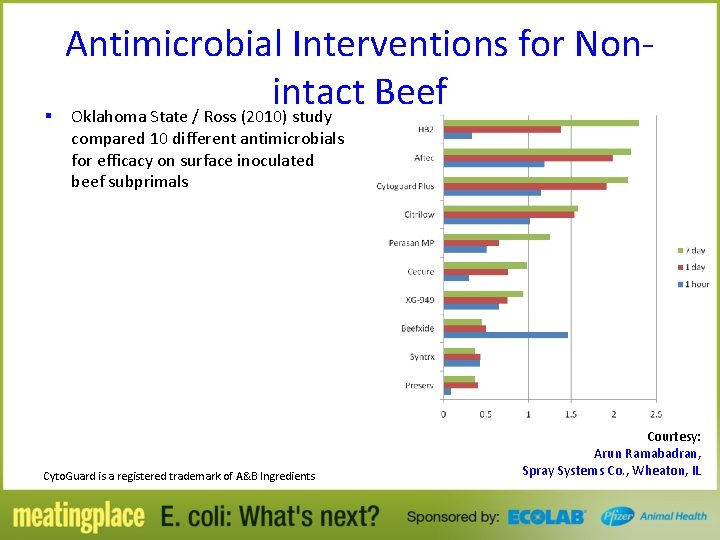
Antimicrobial Interventions for Nonintact Beef § Oklahoma State / Ross (2010) study compared 10 different antimicrobials for efficacy on surface inoculated beef subprimals Cyto. Guard is a registered trademark of A&B Ingredients Courtesy: Arun Ramabadran, Spray Systems Co. , Wheaton, IL

Outline • • • Introduction Prevalence of STECs Antimicrobial Interventions for STECs Non-O 157 STEC Challenges for the processor Non-intact Beef – STECs Summary

Non-O 157 STEC Challenges for the Beef Processor • Biology and characteristics of the organisms are unknown • Characteristics of Significance – Thermal resistance – Acid resistance – Dehydration – Aw – Irradiation – Behavior to antimicrobial interventions traditionally used in beef processing

Characteristics of Significance in Meat Processing – Growth @ 10°C Strain O 26: H 11 survived, but did not grow LPD - 11. 3 h to 39. 4 h Growth rate – 0. 032 to 0. 045 log CFU/g/h O 145 and O 157 had shorter LPD compared to O 45, O 103, and O 121 (O 111 had intermediate LPD) • After end of LPD, no difference in growth rate • • (Chatzikyriakidiu et al. , 2011)
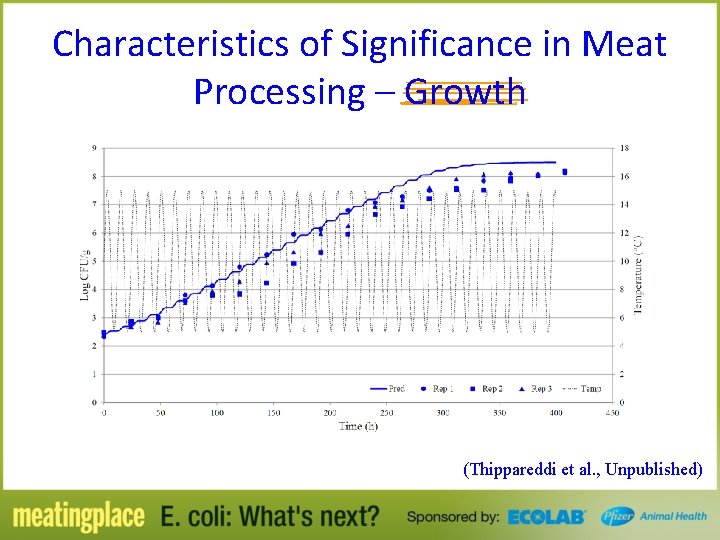
Characteristics of Significance in Meat Processing – Growth (Thippareddi et al. , Unpublished)

Characteristics of Significance in Meat Processing – Thermal Resistance L. monocytogenes Salmonella spp. Lean Fatty 125°F 81. 3 71. 1 61 -62 54. 3 135°F 2. 6 5. 8 3. 8 -4. 2 5. 43 145°F 0. 6 1. 2 0. 6 -0. 7 0. 54 Z Value 9. 3 11. 4 10 10 Broth Ground Beef STECs 130°F (54. 4°C) 9. 39 -31. 15 140°F (60°C) 0. 63 -1. 28 150°F (65. 5°C) 0. 08 -0. 21 1. 16 -0. 76 (Fain et al. , 2009; Leong et al. , 2011, Vasan et al. , 2011)

Characteristics of Significance in Meat Processing – Acid Resistance • E. coli O 157: H 7 reductions of >7. 70 log CFU/ml was observed in lemon and lime juices* within 72 h at 22°C • STECs (six serotypes) reduction of >6 log within 72 h at 22°C in lemon and lime juices *Lemon Juice: 6. 8° Brix; 2. 62 p. H; 4. 62 TA *Lime Juice: 7. 4° Brix; 2. 54 p. H; 4. 82 TA (Enache et al. , 2009; Kataoka et al. , 2011)
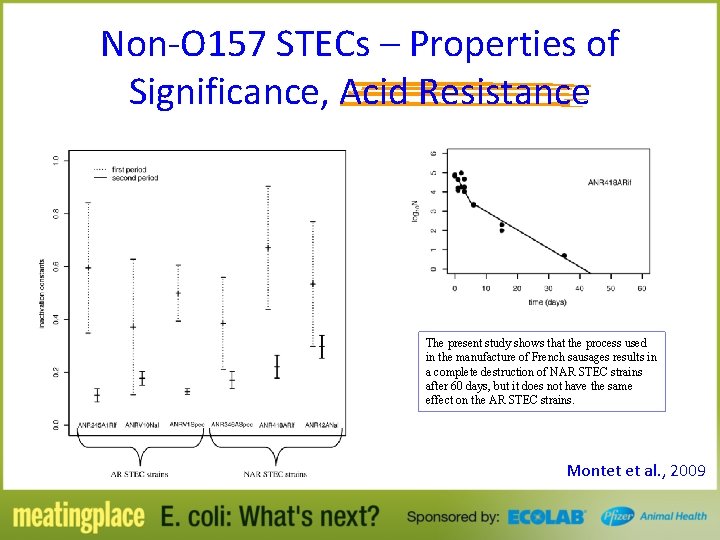
Non-O 157 STECs – Properties of Significance, Acid Resistance The present study shows that the process used in the manufacture of French sausages results in a complete destruction of NAR STEC strains after 60 days, but it does not have the same effect on the AR STEC strains. Montet et al. , 2009

Outline • • • Introduction Prevalence of STECs Antimicrobial Interventions for STECs Non-O 157 STEC Challenges for the processor Non-intact Beef – STECs Summary

Translocation and Thermal Inactivation of Shiga Toxin. Producing E. coli in Blade and Chemically Tenderized Beef 1. 2. 3. 4. Luchansky, J. B. , J. E. Call, R. K. Phebus, and H. Thippareddi. 2008. Translocation of surface inoculated Escherichia coli O 157: H 7 into beef subprimals following blade tenderization. J. Food Prot. 71: 2190 -2197. Luchansky, J. B. , A. C. S. Porto-Fett, B. Shoyer, R. K. Phebus, H. Thippareddi, and J. E. Call. 2009. Thermal inactivation of Escherichia coli O 157: H 7 in blade tenderized beef steaks cooked on a commercial openflame gas grill. J. Food Prot. 72: 1404 -1411. Luchansky, J. B. , A. C. S. Porto-Fett, B. A. Shoyer, J. E. Call, W. Schlosser, W. Shaw, N. Bauer, and H. Latimer. 2011. Inactivation of Shiga toxin-producing O 157: H 7 and non-O 157: H 7 Shiga toxin-producing Escherichia coli in brine-injected, gas-grilled steaks. J. Food Prot. 74: 1054 -1064. Luchansky, J. B. , A. C. S. Porto-Fett, B. A. Shoyer, J. E. Call, W. Schlosser, W. Shaw, N. Bauer, and H. Latimer. 2011. Fate of Shiga toxin-producing O 157: H 7 and non-O 157: H 7 Shiga toxin-producing Escherichia coli in blade tenderized beef steaks cooked on a commercial open-flame gas grill. J. Food Prot. , Submitted 06 -02 -11.

STEC and ECOH behave similarly! • No discernible differences in translocation between ECOH and STEC following blade tenderization or chemical injection of beef sub-primals – Majority of cells to top-most 1 cm – More transfer via single pass and lean side inoculation and tenderization than double pass and fat side inoculation and tenderization (NCBA Study) Courtesy: Dr. John Luchansky & the ARS Non-Intact Team

STEC and ECOH behave similarly! • No discernible differences in thermal resistance between STEC and ECOH following cooking of blade tenderized or chemically -injected steaks – Higher temperatures generated greater lethality – No difference in lethality based on steak thickness (1. 0 or 1. 5 in. ) – Subtle differences in thermal resistance between steaks injected with lactate-containing brine compared to brine without lactate that were cooked at 140 F Courtesy: Dr. John Luchansky & the ARS Non-Intact Team

Outline • • • Introduction Prevalence of STECs Antimicrobial Interventions for STECs Non-O 157 STEC Challenges for the processor Non-intact Beef – STECs Summary

Summary • STECs seem to be similar in characteristics to E. coli O 157: H 7 based on current research • Very few interventions eliminate the E. coli O 157: H 7 and non-O 157 STEC risk • Novel technologies such as carcass irradiation and HPP should be optimized and validated to assure safety of intact as well as non-intact beef products

Together, we can reduce E. coli O 157. Dr. Brad Morgan, Food Safety Specialist Pfizer Animal Health • • • The beef industry has made significant strides in reducing risks and costs associated with foodborne pathogens. – However, food safety remains a significant concern due to increasing media and regulatory scrutiny. – E. coli O 157 is naturally occurring in animals and the environment, which makes it an ever-present challenge. – The beef industry has a good track record for safety, but there is always room to improve. Now there is a tool available to your suppliers that can help reduce E. coli O 157 before it enters the packing plant — in the cattle, at the feedlot — thereby helping reduce E. coli O 157 throughout the beef processing chain. – Escherichia Coli Bacterial Extract vaccine* with SRP® technology is the only conditionally licensed product that can help reduce E. coli O 157** carrier state prevalence and shedding at the source — in the cattle. – Reducing the amount of E. coli O 157 coming into the plant can give other interventions in place an opportunity to be as effective as possible. The goal for us at Pfizer Animal Health is to help ensure continued confidence in the industry to help keep demand for our products strong. * This product license is conditional. Efficacy and potency test studies are in progress. ** Escherichia coli O 157: H 7 bacterial strain 1 Woerner DR, Ransom JR, Sofos JN, Dewell GA, Smith GC, Salman MD, et al. Determining the Prevalence of Escherichia coli O 157 in Cattle and Beef from the Feedlot to the Cooler. J Food Prot 2006; 69: 2824 -2827.

E. COLI: Legal Issues and the Big Six Robert G. Hibbert K&L Gates LLP

E. coli 0157: H 7 Precedent – – – 1994 Policy Statement Texas Food Industry v. Espy (1994) Extension to other products – trim, non-intact Avoidance of rulemaking Integration with HACCP

E. coli Precedent (continued) – Commercial response – Technological responses – Exception proves the rule-labeling and product handling distinctions

Shiga–Toxin Producing E. Coli Serotypes – – Outbreaks Methodology issues Marler petition Pending policy statement

Legal Issues (Regulatory) – Adulterants under 1(m)(1) vs. 1 (m)(3) – Policy by Recall – Abandonment of rulemaking (listeria RTE precedent) – HACCP and inspection implementation

Legal Issues (Commercial) – Guarantees and requirements – Role of testing – Reliability of testing – Tort liability – Insurance concerns

QUESTIONS & ANSWERS

FOR MORE INFORMATION Mohammad Koohmaraie, Ph. D. : mk@iehinc. com Harshavardhan Thippareddi, Ph. D. : hthippareddi 2@unl. edu Robert G. Hibbert: robert. hibbert@klgates. com Mike Fielding: mfielding@meatingplace. com Bill Mc. Dowell: bmcdowell@meatingplace. com Ecolab: melissa. meinke@ecolab. com Pfizer Animal Health: Brad. morgan@pfizer. com Webinar recording and Power. Point presentation will be emailed to you within 48 hours. For more information: www. meatingplace. com/webinars
- Slides: 70