E 1 Basic Electrostatics General Physics 1 Electrostatics

E 1 Basic Electrostatics General Physics 1

Electrostatics • • • Complete the tutorial on electrostatics (both sides of handout) Tape activity www. sos. siena. edu/~rfinn/phys 140 s 09/ tape. pdf

Electrostatics • • electric charges that are not moving Definition of new symbols • • e- = electron p = proton n = neutron e+ = positron

Nature of charge • • Amber is a natural substance that can be naturally charged Few naturally occurring objects can be electrically charged Static electricity is easily shown with plastic, rubber, and synthetic fibers Electrical charges behave like positive and negative numbers

Electric Force • Similar form as gravitational force (weird, huh? ), except • • positive and negative charges Like charges repel; opposite charge attract

A proton is located just north of an electron. The net force that the electron feels due to the proton is in which direction? 1. 2. 3. 4. North South West East

Coulomb’s Law - magnitude • k = 9. 0 x 109 N m 2/C 2 • • q 1, q 2 = charges • • • Coulomb’s contant measured in Coulombs (C) 1 C is a lot of charge! r = distance between q 1 and q 2

Charge Carriers • The electron • 1 e- = -1. 602 x 10 -19 C • • • The proton • 1 p = 1. 602 x 10 -19 C • • smallest unit of charge, also referred to as e negatively charged Charge of 1 e- is -1 e mass = 9. 11 x 10 -31 kg positively charged Charge of 1 p is +1 e mass = 1. 67 x 10 -27 kg Charge is quantized!

Coulomb’s Law with different constants ε 0 =permittivity of free space (more relevant to future chapters)

Illustrations of Coulomb’s Law • • Comb & Paper Balloon on wall
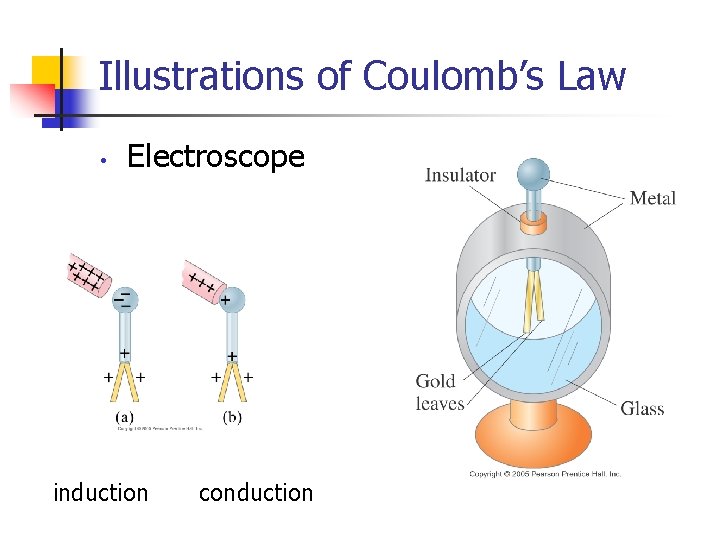
Illustrations of Coulomb’s Law • Electroscope induction conduction

Charge Conservation • The total charge of an isolated system is conserved. • • Rubbing a balloon on hair Triboelectric series • Decreasing tendency to lose electrons

Conductors and Insulators • Charges aren’t free to move around in an insulator • • Why they may be charged If charge is left on them due to rubbing another material, the charge can’t go anywhere Examples – amber, plastics, synthetic materials Charges are free to move very easily in materials called conductors • Examples – metals, salty water
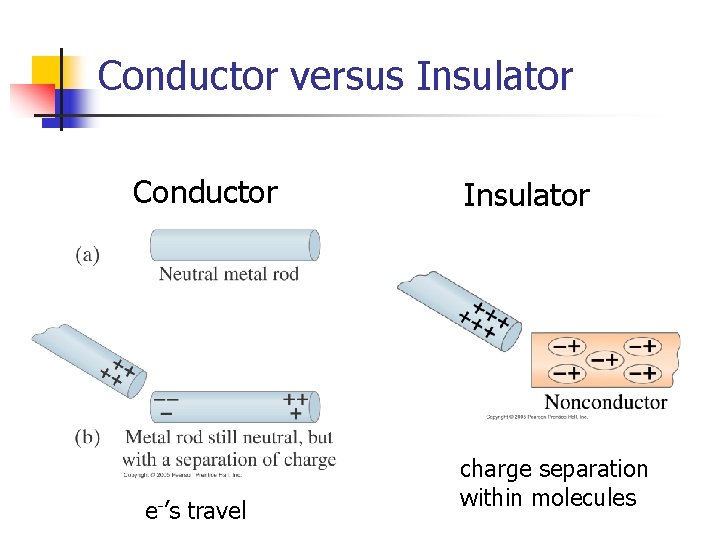
Conductor versus Insulator Conductor e-’s travel Insulator charge separation within molecules
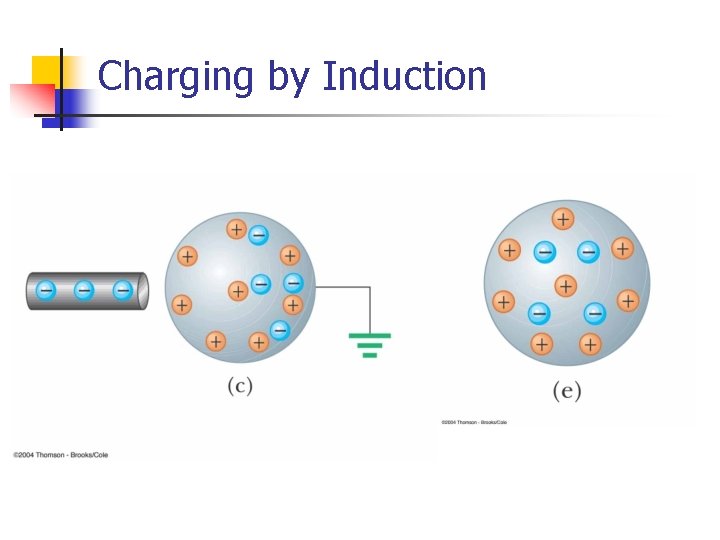
Charging by Induction

Net Force from Multiple Charges • • An electrostatic force exists between each pair of charges according to Coulomb’s law Add components of forces to get net force when adding multiple charges

Adding Vectors Magnitude and direction of Net Force given by:
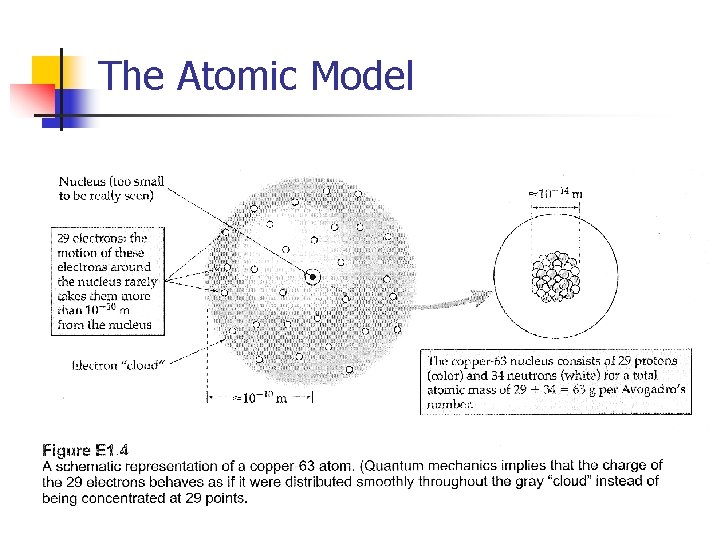
The Atomic Model

How objects become charged • • • A macroscopic object is composed of a huge number of tiny atoms Each atom has a tiny nucleus (includes protons and neutrons) surrounded by a cloud of electrons Equal number of electrons and protons Avogadro’s number of protons or neutrons have a mass of ~ 1 g. Total mass of electrons is negligible Most of the volume of an atom is occupied by the electron clouds

Coulomb’s Law – examples • • Review Example E 1. 2 • Determine direction of force Review Example E 1. 3 • Determine direction and magnitude of force

Van de Graff Generator • Demonstration • • In-class Pie plates Ion motor Extra Credit - see schedule and moodle

Group Problems • • • E 1 S. 3 E 1 S. 5 E 1 S. 10

Charges in a line • What is the net force on the 48 C charge?

Charges in a line - Solution +x Q 1 Q 2 Q 3

Charges in a line - Solution +x Q 1 Q 2 Do signs make sense? !! Q 3

Charges in a square Q 1 Q 2 Q 4 Q 3 +y +x Find net force on Q 1

Charges in a square Q 1 Q 2 Q 4 Q 3 The x and y components of the net force are given by: Do signs of forces make sense? Does trig make sense?

Charges in a square

Charges in a square
- Slides: 29