DYNAMIC EQUILIBRIUM OF BODY FLUID I NJOMAN WIDAJADNJA

DYNAMIC EQUILIBRIUM OF BODY FLUID I NJOMAN WIDAJADNJA (FKIK-UNTAD) / DENNY AGUSTININGSIH (FK-UGM)

BODY FLUIDS ● ● Main Functions: ü Solvent ü Transport Other Functions: – Give shape and form to the cells – Regulate body temp. – Joint lubricant – Cushion body organs – Maintain peak performance

Body composition �In average young adult male: Body composition % of body weight Protein, & related substances 18% Fat 15% Mineral 7% Water 60%

Factors affecting body fluids �Water intake & output �Age: - infant: 73% - elderly: 45% �Sex: - adult male: 60% - adult female: 40 -50% �Body temperature healthy adult can sweat 1 liter/hr for 2 hrs (5% of body wt without problem) � perspire (1000 cc wet sheet) � Every degree F = 75 ml increase/day in fluid needs � �Obesity �Climate �Habits �Level of physical activity
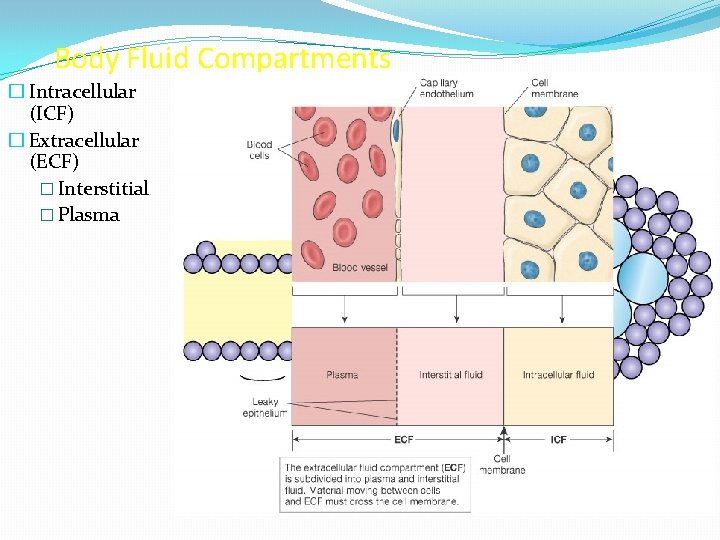
Body Fluid Compartments � Intracellular (ICF) � Extracellular (ECF) � Interstitial � Plasma Figure 5 -13: Body fluid compartments

Total Body Fluid by Compartment Total Body Water

COMPOSITION OF BODY FLUIDS

Electrolytes in body fluids �Ions form when electrolytes dissolve and dissociate � 4 general functions �Control osmosis of water between body fluid compartments �Help maintain the acid-base balance �Carry electrical current �Serve as cofactors
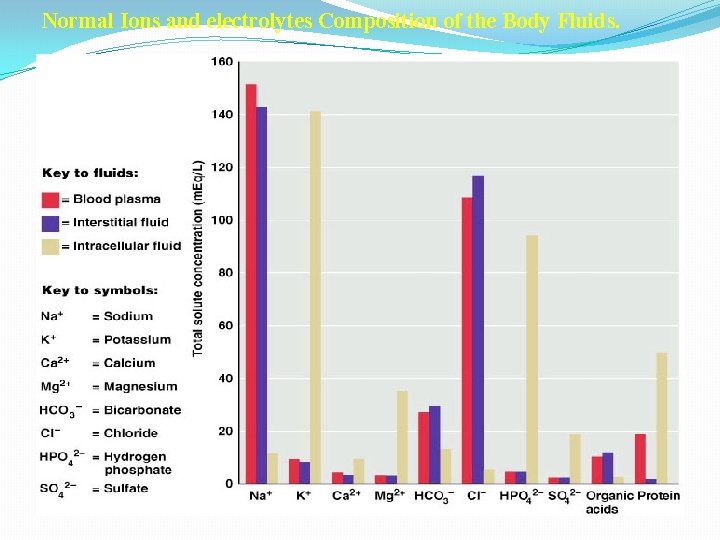
Normal Ions and electrolytes Composition of the Body Fluids. sodium pump ~ Na, K-ATPase: Na K capillary wall plasma membrane

ICF differs considerably from ECF � ECF most abundant cation is Na+, anion is Cl- � Sodium � Impulse transmission, muscle contraction, fluid and electrolyte balance � Chloride � Regulating osmotic pressure, forming HCl in gastric acid � Controlled indirectly by ADH and processes that affect renal reabsorption of sodium � ICF most abundant cation is K+, anion are proteins and phosphates (HPO 42 -) � Potassium � Resting membrane potential , action potentials of nerves and muscles � Maintain intracellular volume � Regulation of p. H � Controlled by aldosterone � Na+ /K+ pumps play major role in keeping K+ high inside cells and Na+ high outside cell

HOMEOSTASIS �Claude Bernard (1878): animals have 2 environtments �- milieu exteriur : physically surrounds the whole organism �- milieu interieur : surrounds the tissue and cells of the organism � organic liquid � homeostasis
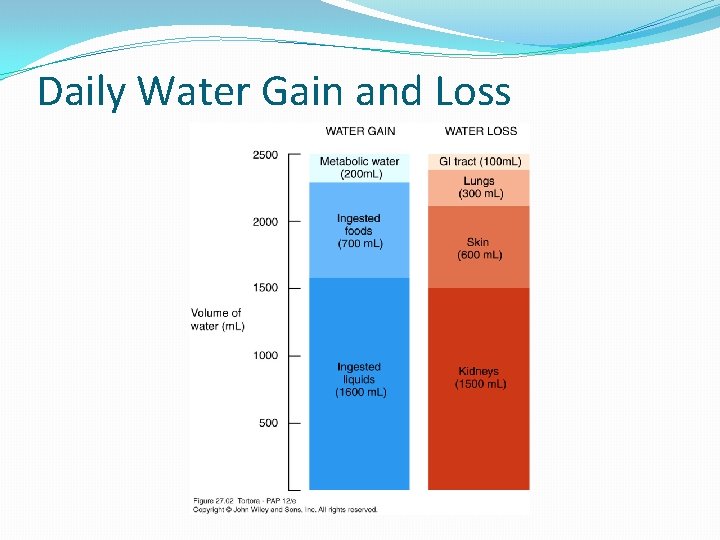
Daily Water Gain and Loss

TRANSPORT OF BODY WATER AND SOLUTE �Osmotic pressure (concentrations of all solutes in a fluid compartment) is equivalent between ECF and ICF compartments � the solutes move from the lower op to the higher op
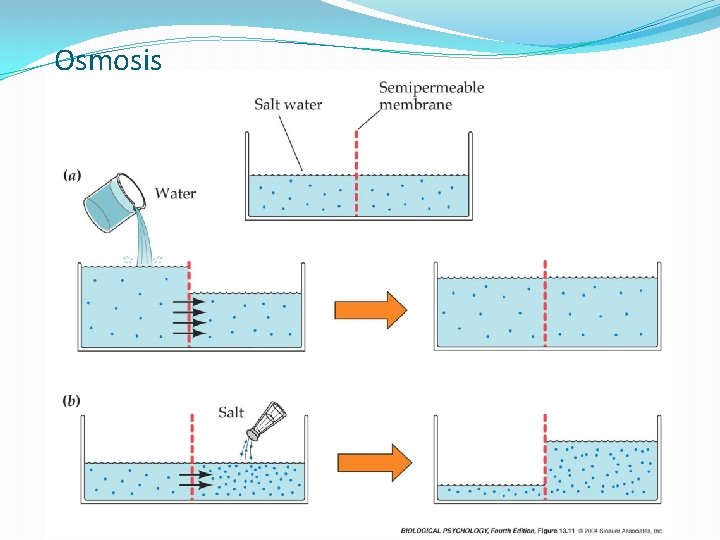
Osmosis
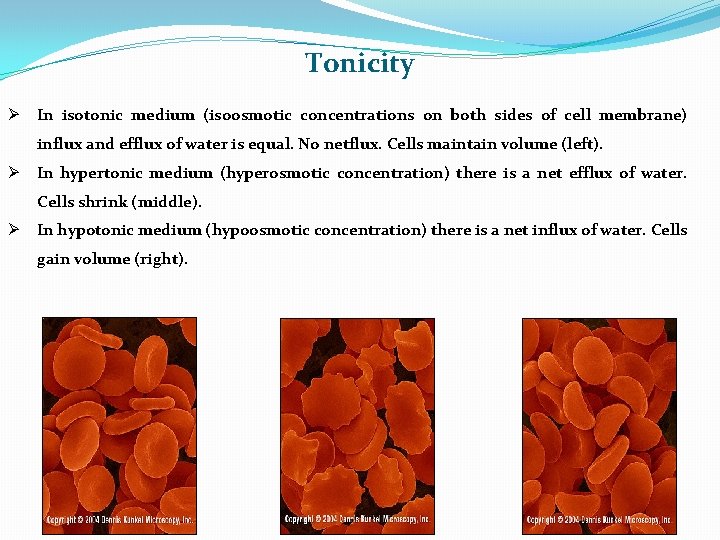
Tonicity Ø In isotonic medium (isoosmotic concentrations on both sides of cell membrane) influx and efflux of water is equal. No netflux. Cells maintain volume (left). Ø In hypertonic medium (hyperosmotic concentration) there is a net efflux of water. Cells shrink (middle). Ø In hypotonic medium (hypoosmotic concentration) there is a net influx of water. Cells gain volume (right).

Osmolarity vs. tonicity �related, but different concepts. �they both compare the solute concentrations of two solutions separated by a membrane. �osmolarity takes into account the total concentration of penetrating solutes and non-penetrating solutes, =jumlah partikel yg aktif secara osmotik(ion/molekul utuh) �tonicity takes into account the total concentration of only non-penetrating solutes. = `sifat larutan` dan bagaimana larutan tsb mempengaruhi volume dlm sel dlm hal capaian ekuilibrium (tampak hanya air yg bergeser-geser )

Osmolarity vs. tonicity �Penetrating solutes can diffuse through the cell membrane, causing momentary changes in cell volume as the solutes "pull" water molecules with them. �Non-penetrating solutes cannot cross the cell membrane, and therefore osmosis of water must occur for the solutions to reach equilibrium.

Osmolarity vs. tonicity �A solution can be both hyperosmotic and isotonic. �For example, the intracellular fluid and extracellular can be hyperosmotic, but isotonic – � if the total concentration of solutes in one compartment is different from that of the other, but one of the ions can cross the membrane, drawing water with it and thus causing no net change in solution volume.

Membandingkan Osmolaritas Larutan A = Glukosa 1 Osm Larutan B = Glukosa 2 Osm Larutan C = Na Cl 1 Osm A hiposmotik terhadap B B hiperosmotik terhadap A C isosmotik terhadap A A isosmotik terhadap C B hiperosmotik terhadap C C hiperosmotik terhadap B

Tonisitas Larutan Perilaku Sel di Dalam Larutan Penjelasan Sifat Larutan Terhadap Sel A Sel membengkak Larutan A hipotonik B Sel tidak berubah ukuran Larutan B isotonik C Sel mengkerut Larutan C hipertonik
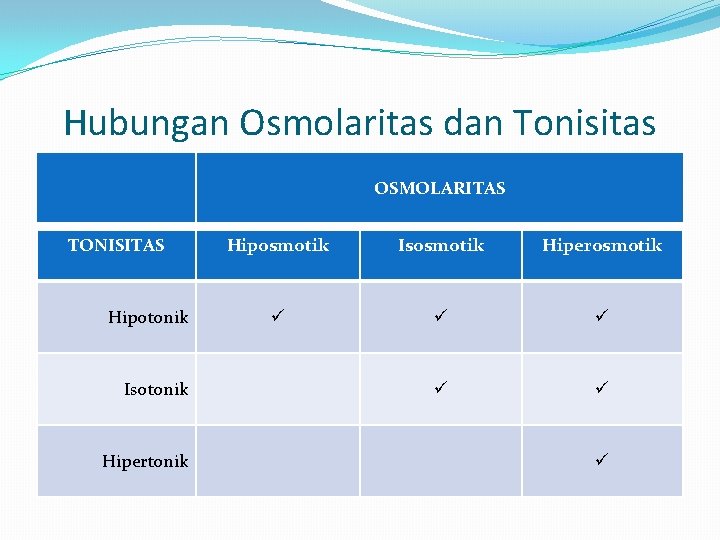
Hubungan Osmolaritas dan Tonisitas OSMOLARITAS TONISITAS Hipotonik Isotonik Hipertonik Hiposmotik Isosmotik Hiperosmotik ü ü ü

What cellular tools are available to counteract swelling or shrinking? �Rapid: opening of channels, activation of transporters �Slower: accumulation or disposition of intracellular organic osmolytes
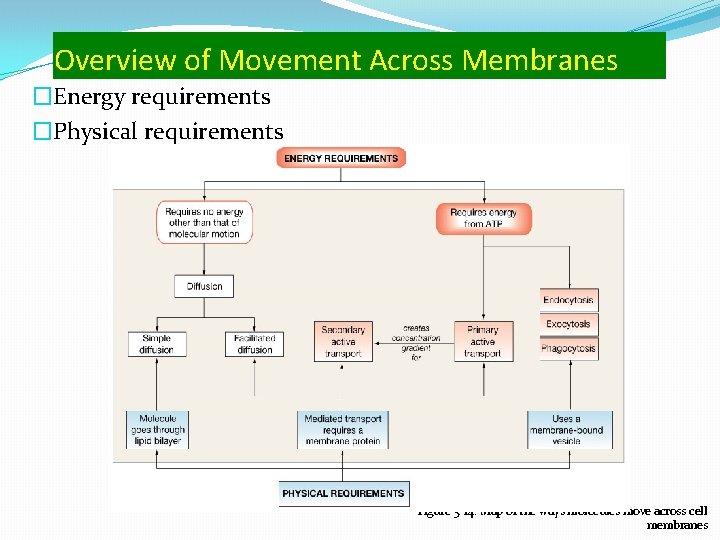
Overview of Movement Across Membranes �Energy requirements �Physical requirements Figure 5 -14: Map of the ways molecules move across cell membranes
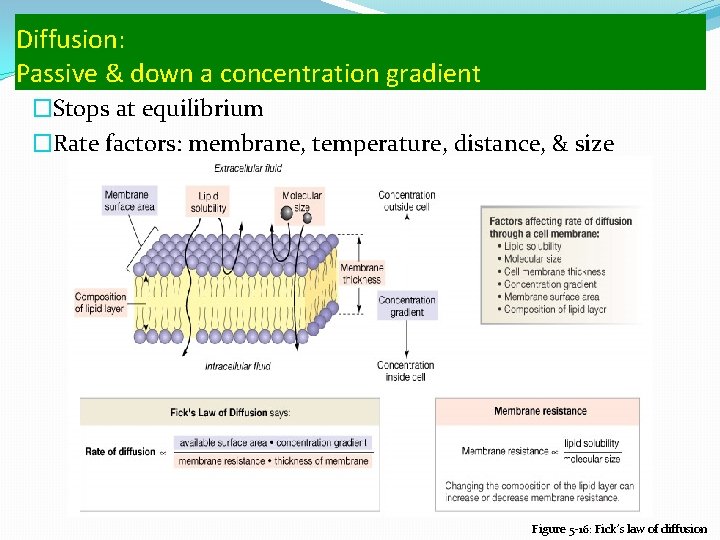
Diffusion: Passive & down a concentration gradient �Stops at equilibrium �Rate factors: membrane, temperature, distance, & size Figure 5 -16: Fick’s law of diffusion

ICF ECF ICF Hypotonic Hypertonic ICF Hypotonic ECF Volume Expansion Isotonic ECF ICF ECF Volume Contraction Isotonic Hypertonic ECF Osmolality PATHOPHYSIOLOGICAL CHANGES IN EXTRACELLULAR FLUID VOLUME ICF From H. Valtin
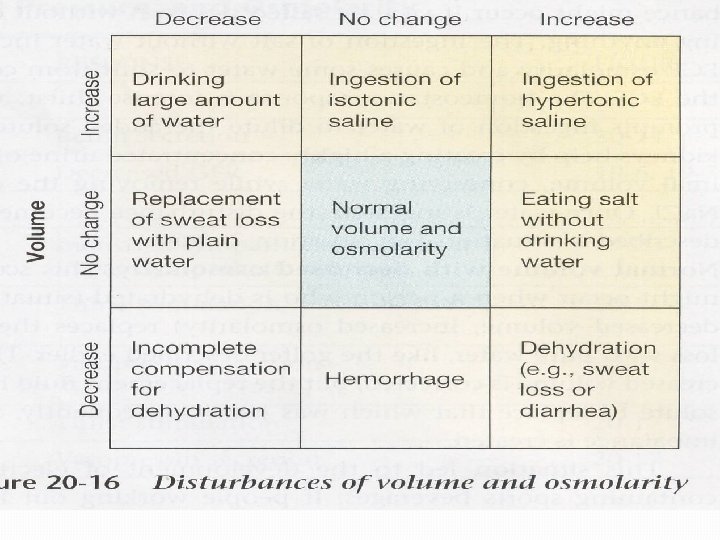


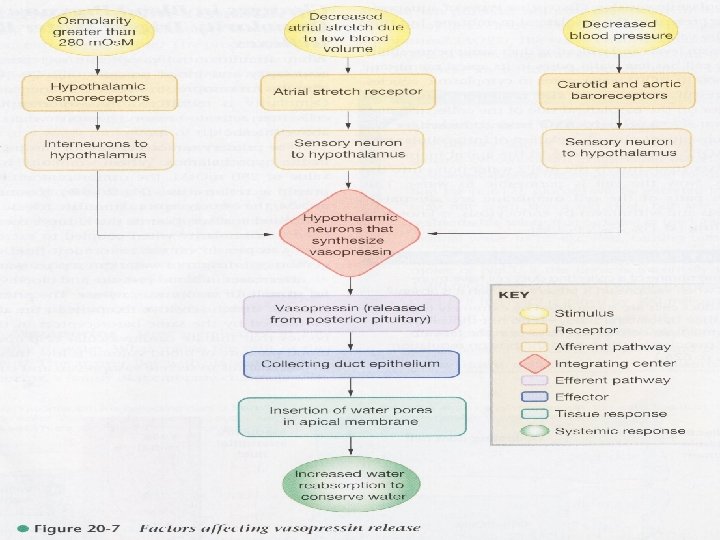
CONTROL OF WATER INTAKE Primarily by THIRST Stimuli: 1) Increased Osmolality 2) Decreased Arterial Pressure 3) Decreased Blood Volume 4) Angiotensin II
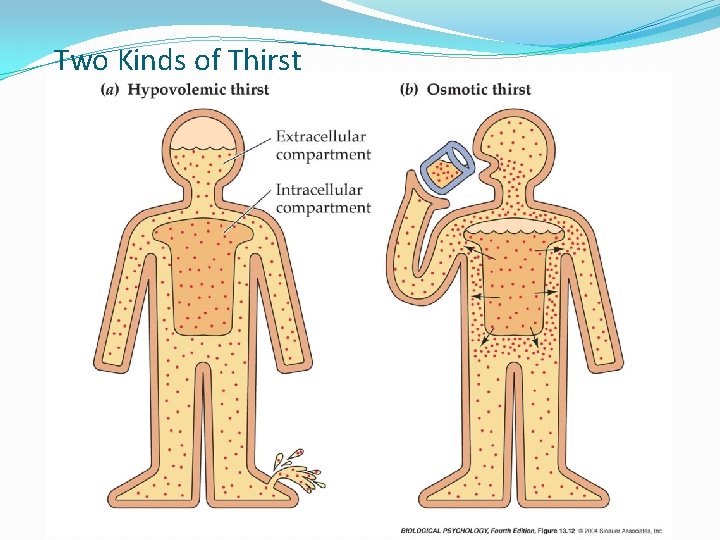
Two Kinds of Thirst
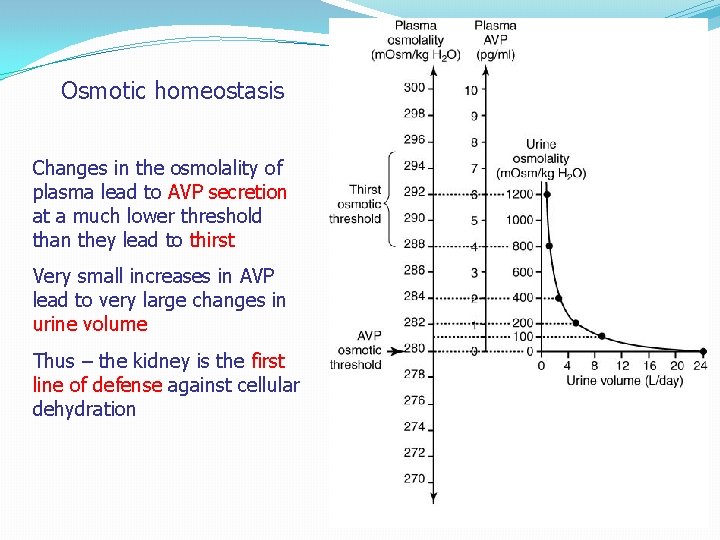
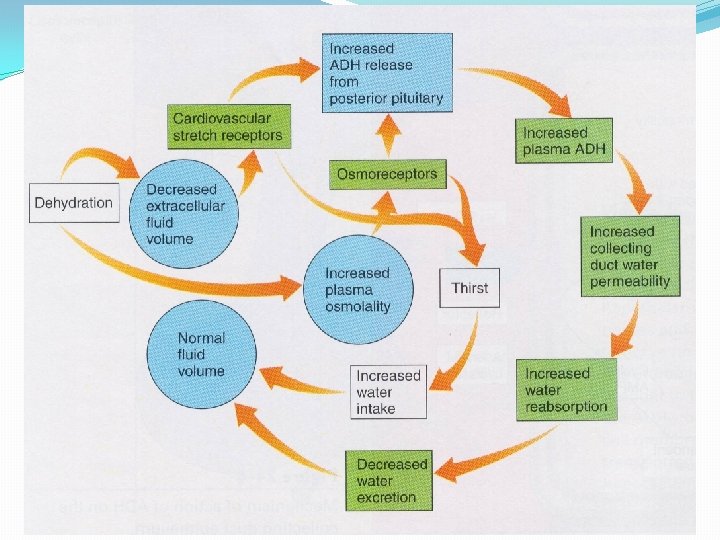
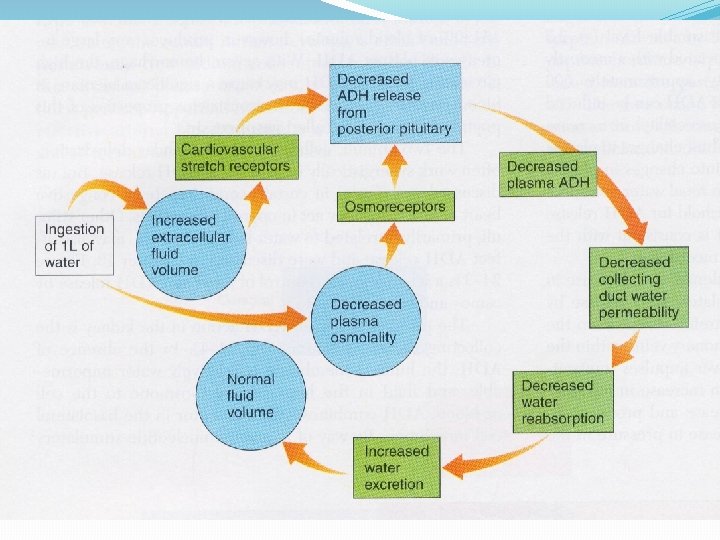
Osmotic homeostasis Changes in the osmolality of plasma lead to AVP secretion at a much lower threshold than they lead to thirst Very small increases in AVP lead to very large changes in urine volume Thus – the kidney is the first line of defense against cellular dehydration


Osmotic homeostasis The initial response to cellular dehydration is release of arginine vasopressin (AVP) – the antidiuretic hormone AVP is synthesized in the supraoptic n. and paraventricular n. of the hypothalamus and transported along axons to the posterior pituitary. AVP is stored in secretory granules in posterior pituitary until an increase in osmolality of body fluids initiates its secretion into the blood AVP acts on V 2 receptors (c. AMP) in the kidney to increase water permeability by inserting aquaporin channels into cell membranes Water moves out of the distal convoluted tubule of the kidney by osmosis through these channels – decreasing osmolality There is also an increased water reabsorption by the kidney and decrease in urine flow
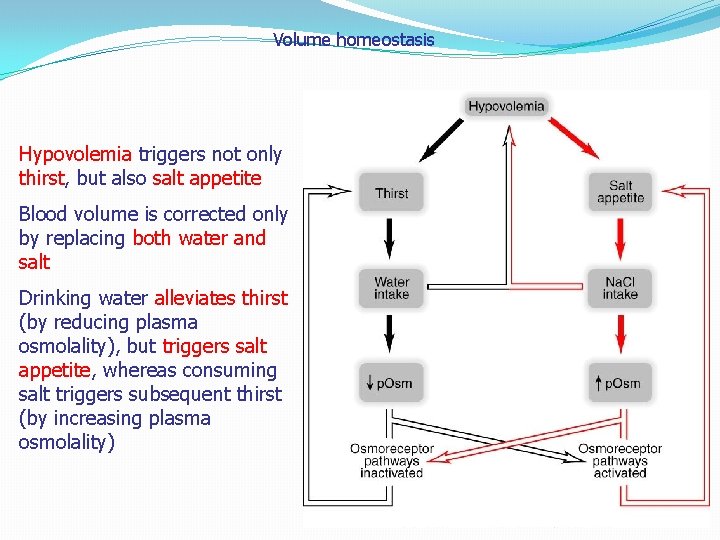
Volume homeostasis Hypovolemia triggers not only thirst, but also salt appetite Blood volume is corrected only by replacing both water and salt Drinking water alleviates thirst (by reducing plasma osmolality), but triggers salt appetite, whereas consuming salt triggers subsequent thirst (by increasing plasma osmolality)
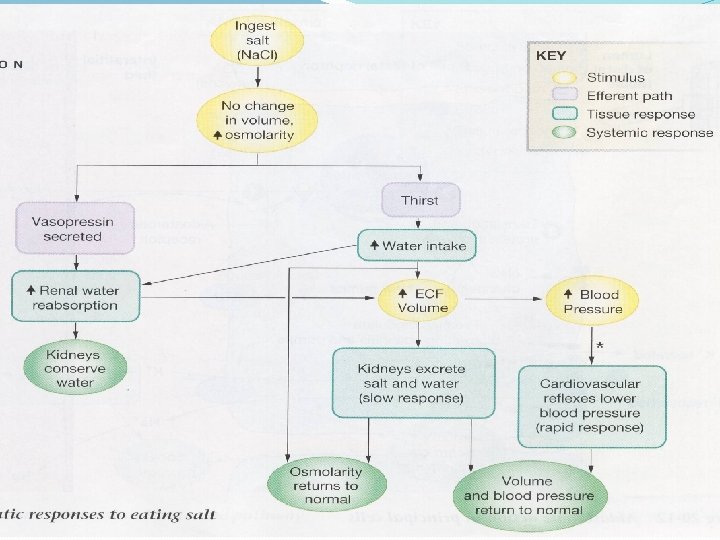
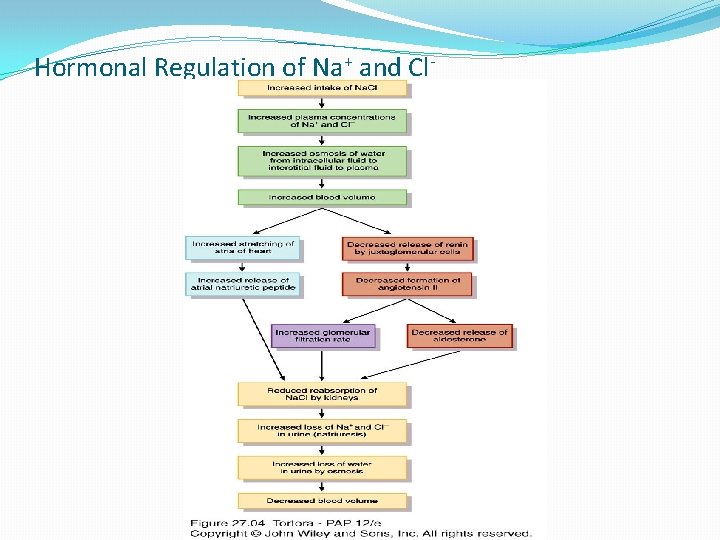
Hormonal Regulation of Na+ and Cl-
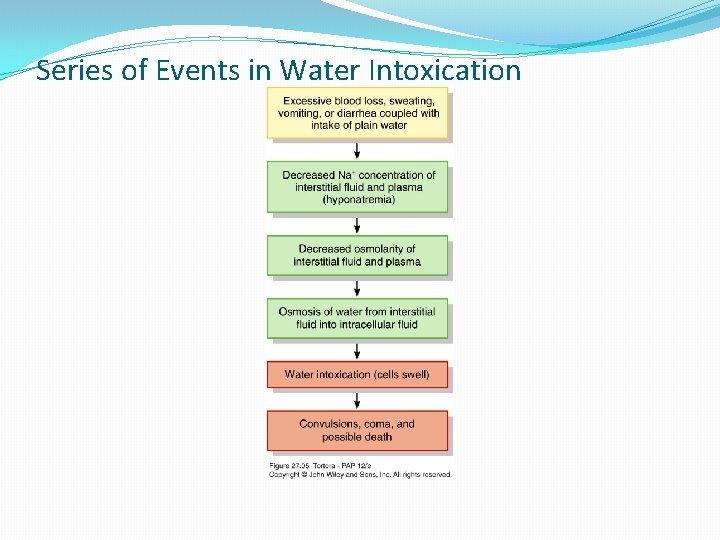
Series of Events in Water Intoxication

Acid-base balance �Major homeostatic challenge is keeping H+ concentration (p. H) of body fluids at appropriate level � 3 D shape of proteins sensitive to p. H �Diets with large amounts of proteins produce more acids than bases which acidifies blood �Several mechanisms help maintain p. H of arterial blood between 7. 35 and 7. 45 �Buffer systems, exhalation of CO 2, and kidney excretion of H+ / reabsorption of HCO 3 -

Buffer systems �Act to quickly temporarily bind H+ �Raise p. H but do not remove H+ �Most consist of weak acid and salt of that acid functioning as weak base �Major buffer systems: proteins, carbonic acid/bicarbonate, phosphates Copyright 2009, John Wiley & Sons, Inc.

Buffer systems �Protein buffer system �Most abundant buffer in ICF and blood plasma �Hemoglobin in RBCs �Albumin in blood plasma �Free carboxyl group acts like an acid by releasing H + �Free amino group acts as a base to combine with H+ �Side chain groups on 7 of 20 amino acids also can buffer H+

Buffer Systems �Carbonic acid / bicarbonate buffer system � Based on bicarbonate ion (HCO 3 -) acting as weak base and carbonic acid (H 2 CO 3) acting as weak acid � HCO 3 - is a significant anion in both ICF and ECF � Because CO 2 and H 2 O combine to form this buffer system cannot protect against p. H changes due to respiratory problems in which there is an excess or shortage of CO 2 �Phosphate buffer system � Dihydrogen phosphate (H 2 PO 4 -) and monohydrogen phosphate (HPO 42 -) � Phosphates are major anions in ICF and minor ones in ECF � Important regulator of p. H in cytosol

Exhalation of carbon dioxide �Increase in carbon dioxide in body fluids lowers p. H of body fluids �Because H 2 CO 3 can be eliminated by exhaling CO 2 it is called a volatile acid �Changes in the rate and depth of breathing can alter p. H of body fluids within minutes �Negative feedback loop
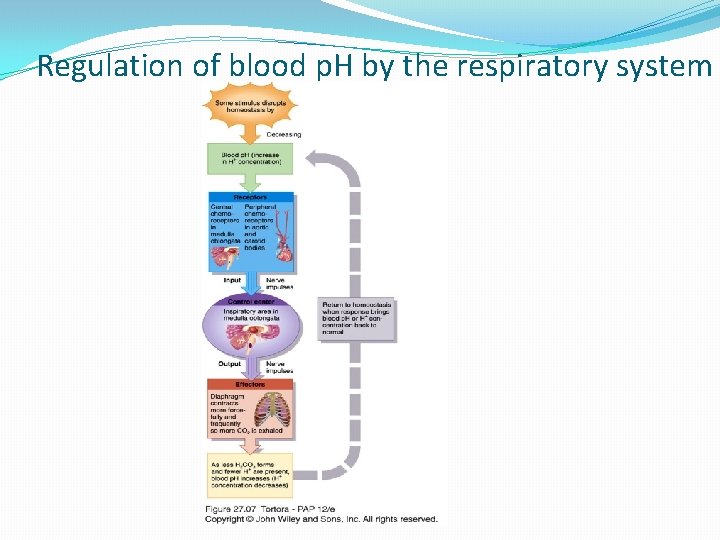
Regulation of blood p. H by the respiratory system

Acid-base imbalances �Normal p. H range of arterial blood 7. 35 -7. 45 � Acidosis – blood p. H below 7. 35 � Alkalosis – blood p. H above 7. 45 �Major physiological effect of � Acidosis – depression of synaptic transmission in CNS � Alkalosis – overexcitability of CNS and peripheral nerves Copyright 2009, John Wiley & Sons, Inc.

Changes in p. H �Acidosis – p. H below 7. 35 �CNS depression �Alkalosis – p. H above 7. 45 �CNS hyerexcitability Copyright 2009, John Wiley & Sons, Inc.

Some acid-base terminology �Acid: a proton donor �Base (alkali): a proton acceptor �Fixed (metabolic) acid: any acid except CO 2 �Metabolic acidosis/alkalosis – due to excess of any fixed acid or base �Respiratory acidosis/alkalosis – due to excess/deficit of CO 2

Cytoplasmic p. H is closely regulated �Generally, cells are more alkaline than would be expected if H+ was in electrochemical equilibrium (more than level 7 =7, 35 -7, 45) �The Na+/H+ exchanger is an acid extruder �The Cl-/HCO 3 - exchanger is an acid loader �Both of these processes operate at some level all the time
![�An increase in cytoplasmic [H+] due to addition of fixed acid (intracellular metabolic acidosis) �An increase in cytoplasmic [H+] due to addition of fixed acid (intracellular metabolic acidosis)](http://slidetodoc.com/presentation_image_h2/c766450482ca693e532ab96992ba7222/image-50.jpg)
�An increase in cytoplasmic [H+] due to addition of fixed acid (intracellular metabolic acidosis) stimulates acid extrusion and inhibits acid loading vice versa for intracellular metabolic alkalosis. Cells can usually defeat this kind of challenge, so long as there is not an extracellular p. H disturbance as well. �If there is an extracellular p. H disturbance, it will affect the responses of both acid loaders and acid extruders.
- Slides: 50