Ductal Carcinoma InSitu DCIS Questions What are the

























- Slides: 32

Ductal Carcinoma In-Situ (DCIS)

Questions What are the histological differences between atypical ductal hyperplasia (ADH), ductal carcinoma in-situ (DCIS), atypical lobular hyperplasia (ALH) and lobular carcinoma in-situ (LCIS)? How do they each behave? What are the management options for DCIS and what is the evidence? What surveillance is recommended for DCIS?

Benign Breast Disease Nonproliferative Proliferative without atypia Proliferative with atypia ADH ALH LCIS Considered “high risk” as they are associated with an increase in the patient’s future risk of developing breast cancer Not “premalignant”

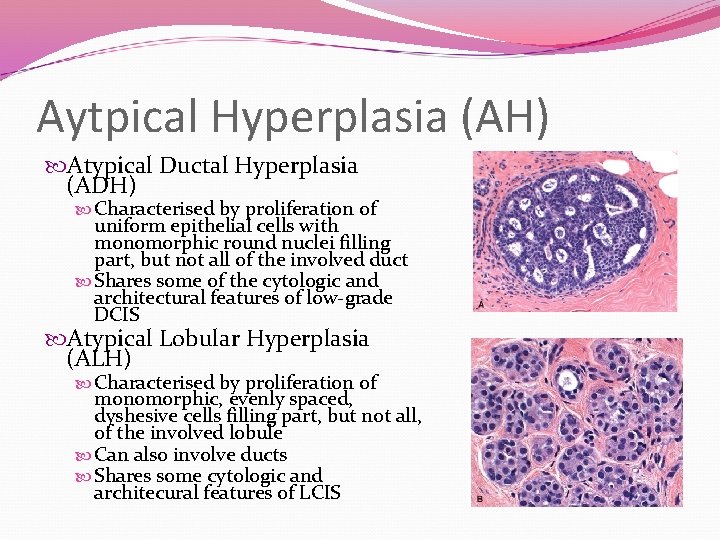
Aytpical Hyperplasia (AH) Atypical Ductal Hyperplasia (ADH) Characterised by proliferation of uniform epithelial cells with monomorphic round nuclei filling part, but not all of the involved duct Shares some of the cytologic and architectural features of low-grade DCIS Atypical Lobular Hyperplasia (ALH) Characterised by proliferation of monomorphic, evenly spaced, dyshesive cells filling part, but not all, of the involved lobule Can also involve ducts Shares some cytologic and architecural features of LCIS

ADH DCIS and invasive breast cancer is identified in 3387% of subsequent excision biopsies reported as ADH in the core biopsy

Risk of Cancer After AH AH (especially multifocal lesions) Increased risk (RR 3. 7 -5. 3) in ipsilateral and contralateral breast (higher in ipsilateral) Conflicting data to suggest that the risk is higher with ALH than ADH Relative risk increases when ADH occurs in women with a family history of breast cancer in a 1 st degree relative to 10 times that of the general population with no family history

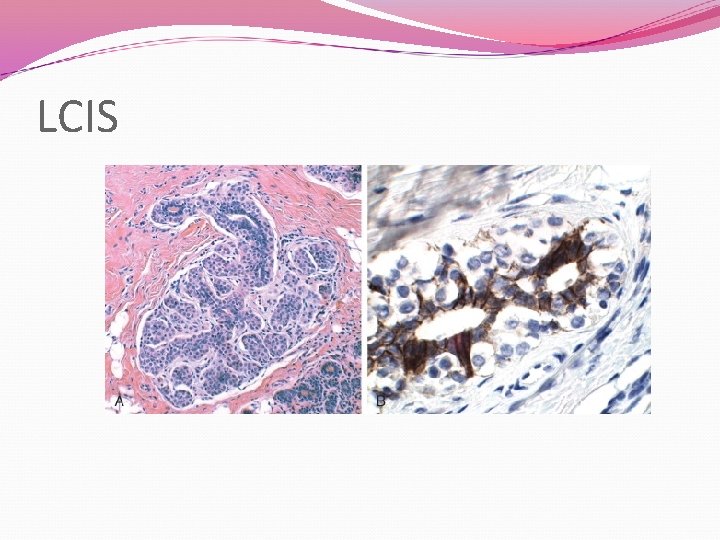
Lobular Neoplasia Spectrum of proliferative changes within the breast lobule that includes both atypical lobular hyperplasia (ALH) and LCIS Both associated with increased risk of invasive breast cancer LCIS is non-invasive, multicentric proliferation of the epithelial cells in the lobules and terminal ducts of the breast

Histology LCIS diagnosed when all of the following occur: Cellular proliferation is characterised by round, cuboidal or polyganal cells that are regularly arranged and evenly spaced Cell nuclei are predominantly round, monotonous and hyperchromatic Proliferation involves, distends and distorts at least half the acini in the terminal duct-lobular unit and fills involved lobular spaces, resulting in the absence of central lumia ALH is diagnosed with a lesion fails to meet at least one of the diagnostic criteria for LCIS in over 50% of acini within a lobular unit

LCIS

Cancer Risk ALH RR 3 -4 (compared with general population) Greater risk in ipsilateral breast LCIS RR 7 -9 Appears to be as common in the contralateral breast as in the ipsilateral breast Pleomorphic variant appears to have an aggressive biologic profile

Management and Follow-Up Core Bx of ADH excision biopsy due to association with DCIS or invasive breast cancer No strong evidence about whether to perform excision biopsy on women with LCIS or ALH on core Bx Surveillance appears to be best the best management option for women who have been diagnosed with ADH, LCIS or ALH as the only abnormality (annual clinical examination, annual bilateral mammography for at least 15 years following diagnosis)

Management and Follow-Up No role for CLE or mastectomy in the management of ADH, LCIS or ALH Insufficient evidence to recommend the use of tamoxifen for prevention of invasive breast cancer following a diagnosis of ADH, LCIS or ALH (Australian Guidelines)

DCIS Definition Abnormal proliferative condition of epithelial cells in the mammary ducts Cells display cytological features of malignancy but unlike invasive cancer, DCIS is confined within the ducts Why is it clinically significant? Historically data suggests that 20 -30% of untreated DCIS progresses to invasive cancer

No reliable predictors for probability to progression to invasive carcinoma Risk may be greater when DCIS displays features such as comedo necrosis or high nuclear grade Natural history is largely unknown

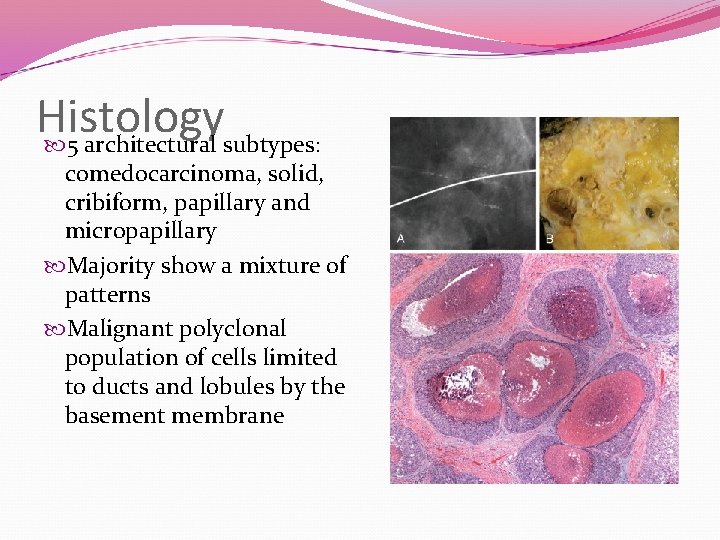
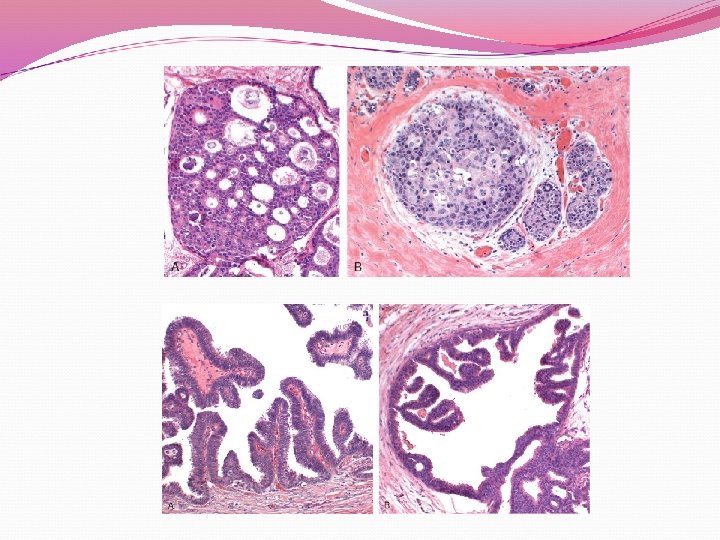
Histology 5 architectural subtypes: comedocarcinoma, solid, cribiform, papillary and micropapillary Majority show a mixture of patterns Malignant polyclonal population of cells limited to ducts and lobules by the basement membrane


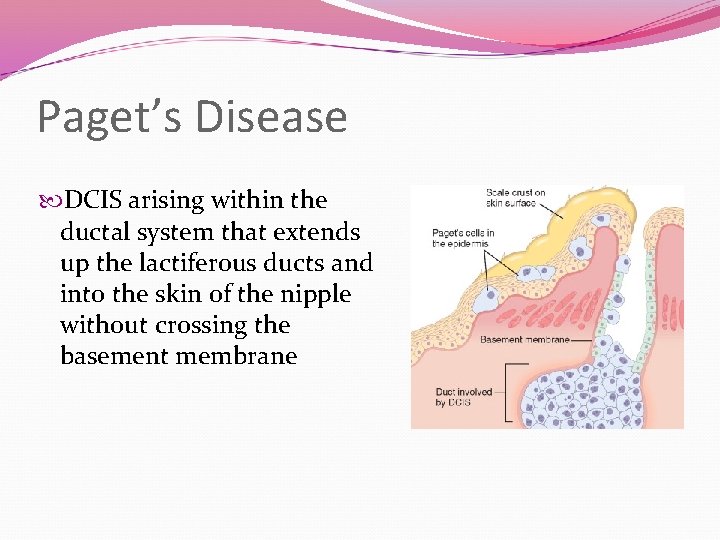
Paget’s Disease DCIS arising within the ductal system that extends up the lactiferous ducts and into the skin of the nipple without crossing the basement membrane

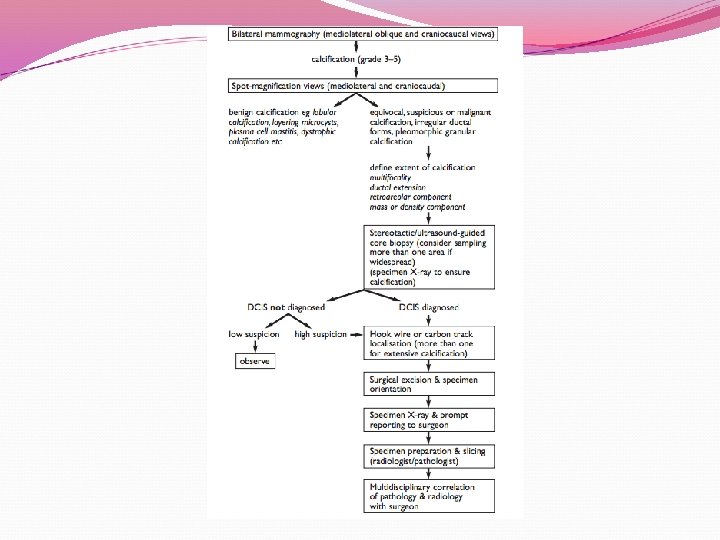
Diagnosis Most commonly detected as mammographic caclification BI-RADS Classification 0. Incomplete 1. Negative 2. Benign 3. Probably benign 4. Suspicious 5. Highly suggestive of malignancy 6. Known biopsy-proven malignant


General Principles of Management Small, mammographically detected lesions should be treated with complete local excision If disease is extensive, total mastectomy may be a more reliable treatment RCTs demonstrate a reduction in DCIS recurrence and invasive breast cancer if radiotherapy is performed after CLE ALND is not indicated Chemotherapy has never been investigated Tamoxifen in the adjuvant setting reduces risk of DCIS recurrence and invasive breast cancer MDT

Management Options in DCIS • Surgery (breast and axilla) • Radiotherapy • Chemoprophylaxis

Surgery Aim is to ensure complete excision with best possible cosmetic result Pre-operative localisation is essential for a mammographically detected impalpable lesion Optimal margins No reliable definition Must be completely excised “Clear margins” (no DCIS at section edge) provides acceptable local control when combined with radiotherapy <1 mm considered inadequate

Surgery Axillary Dissection No place for ALND What about SLNB? High suspicion for invasive cancer Large size Aggressive histologic features Palpable mass Surgery that will compromise ability to perform SLNB in the future

Surgery Mastectomy Widespread contiguous or multi-focal DCIS Widespread microcalcification in the presence of proven DCIS Recurrence of DCIS following initial treatmen when either of the above indiciations is present Woman’s choice Other relevant risk factors for breast cancer

Radiotherapy Lower recurrence rate demonstrated (4 RCTs) when DCIS is treated with complete local excision (CLE) and adjuvant radiotherapy compared with CLE alone regardless of grade and pathological subgroup Insufficient statistical power to detect small differences in survival 2009 Meta Analysis of RTx compared to no further treatment following excision showed RTx resulting in reduction in risk of all ipsilateral breast events (HR 0. 49; 95% CI 0. 41 -0. 58)

Risk of Recurrence Relatively low for DCIS with good prognostic pathological features Clear margins Low-grade No necrosis Small extent (<10 mm) High grade DCIS with necrosis, close margins and larger size should have radiotherapy

Systemic Treatment Chemotherapy Never investigated or used in treatment with DCIS

Systemic Treatment Tamoxifen Must have ER + disease In women who have breast conservation treatment, postoperative tamoxifen is more effective than placebo in reducing the risk of invasive breast cancer (NSABP B-17 and B-24) (8. 5 vs 10% Meta-analysis (2009): Tamoxifen reduces recurrence risk of ipsilateral breast (HR 0. 75, 95%CI 0. 60 -. 092), trend to reduction in risk of invasive carcinoma (HR 0. 79, 95%CI 0. 60 -1. 01), lower risk for contralateral carcinoma (HR 0. 57, 95%CI 0. 39 -0. 83) No apparent survival benefit Risks: endometrial cancer, VTE

Systemic Treatment NCCN 2013 Guidelines Consider tamoxifen for 5 years: Patients treated with breast conservation, especially if ER + Patients treated with surgery alone and ER + Australian Guidelines 2003 Further research required, tamoxifen may reduce risk of subsequent local invasive breast cancer in women who have had breast conservation treatment No data for aromatase inhibitors Limited data for HER 2 -directed therapy

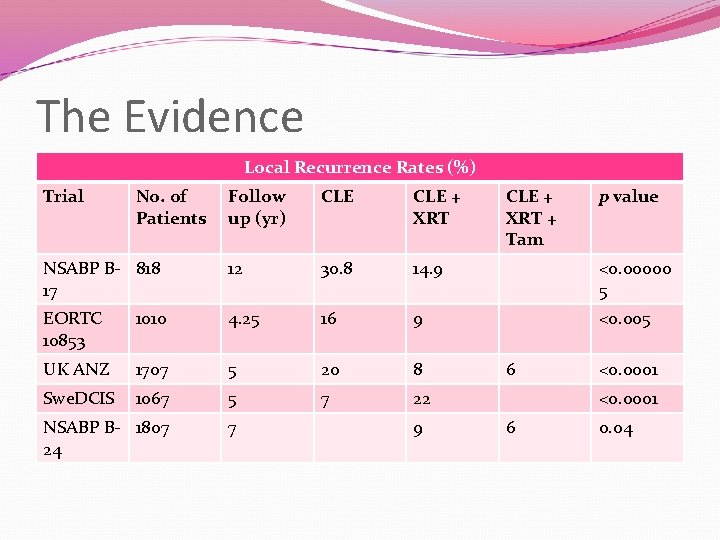
The Evidence Local Recurrence Rates (%) Trial No. of Patients Follow up (yr) CLE + XRT NSABP B- 818 17 12 30. 8 14. 9 <0. 00000 5 EORTC 10853 1010 4. 25 16 9 <0. 005 UK ANZ 1707 5 20 8 Swe. DCIS 1067 5 7 22 NSABP B- 1807 24 7 9 CLE + XRT + Tam 6 p value <0. 0001 6 0. 04

Post Treatment Surveillance Overall survival for women >67 is similar to those without breast cancer Women >67 are more likely to die of cardiovascular disease than breast cancer Australian Guidelines No evidence defining optimum follow-up protocol Consensus recommendation is for annual review with examination and mammography indefinitely

Post Treatment Surveillance NCCN Guidelines Interval history and physical examination every 6 -12 months for 5 years then anually Mammogram annually and 6 -12 months post radiation therapy if breast conserved Breast awareness If treated with tamoxifen Annual gynae assessment Opthalmology exam Manage symptoms as per guidelines