Dual Nature of Matter and Radiation By K

Dual Nature of Matter and Radiation By K. N. Rao

Free Electron in metal: In a Metal the electrons in the outer shells of the atoms are loosely bound. They are quite free to movie easily within the metal surface but can not leave the metal surface. “Such Loosely held electrons in the metal are called Free electrons”.

Work Function: (φ0) Work function of a metal is the minimum energy required by an electron to just escape from the metal surface so as to overcome the restraining forces at the surface. Ø Measured in e. V which is the unit commonly used in atomic and nuclear physics. Ø 1 e. V= J

Electron emission: The Phenomenon of emission of electrons from the surface of a metal surface is called emission 1) Thermionic Emission: It is the phenomenon of emission of electrons from the metal surface when heated suitably and the emitted electrons are called thermal electrons or thermions 2) Photoelectric Emission: It is the phenomenon of emission of electrons from the metal surface when heated suitably and the emitted electrons are called Photo electrons. 3) Field emission or cold cathode emission: It is the phenomenon of emission of electron from the metal surface under the application of strong electric field. (~108 Vm-1) 4) Secondary emission: It is the phenomenon of emission of electrons from the metal surfa in large number when fast moving electrons (primary electrons ) strike the metal surface. Th ejected electrons are called secondary electrons

Photoelectric effect: It is the phenomenon of emission of electrons from the surface of metals , when radiation of suitable frequency falls on it. The ejected electrons are called Photoelectrons and the current so produced is called photoelectric current

Hertz, Hallwachs and Lenard’s observation on Photoelectric effect: Hertz Observations: (1887), studying of electromagnetic waves, spark discharge , high voltage sparks across the detector plate when the emitter plate was illuminated by ultraviolet light from an arc lamp. Conclusion: when the metal surface is illuminated by radiation of suitable frequency the electrons are escaping out of the surface.
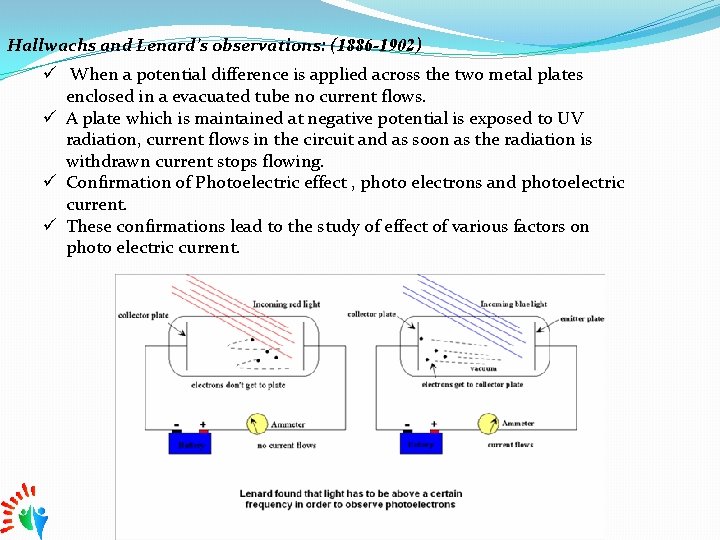
Hallwachs and Lenard’s observations: (1886 -1902) ü When a potential difference is applied across the two metal plates enclosed in a evacuated tube no current flows. ü A plate which is maintained at negative potential is exposed to UV radiation, current flows in the circuit and as soon as the radiation is withdrawn current stops flowing. ü Confirmation of Photoelectric effect , photo electrons and photoelectric current. ü These confirmations lead to the study of effect of various factors on photo electric current.

Experimental study of Photoelectric effect: Emitter plate(cathode) Collector plate (anode)
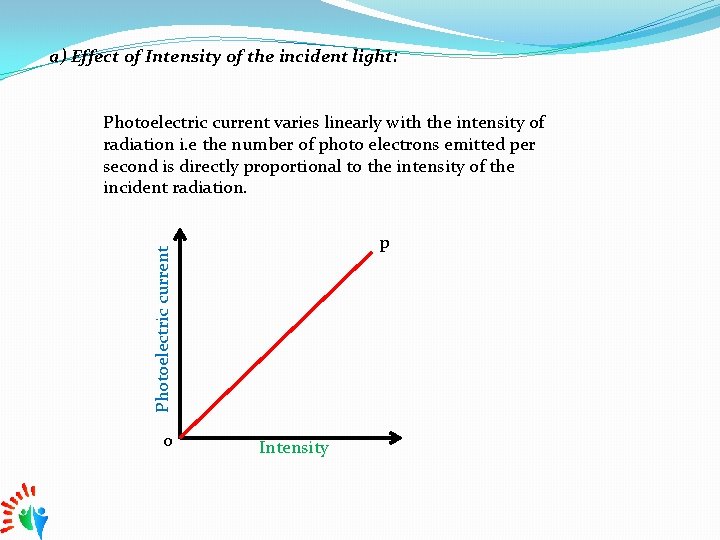
a) Effect of Intensity of the incident light: Photoelectric current varies linearly with the intensity of radiation i. e the number of photo electrons emitted per second is directly proportional to the intensity of the incident radiation. Photoelectric current p o Intensity
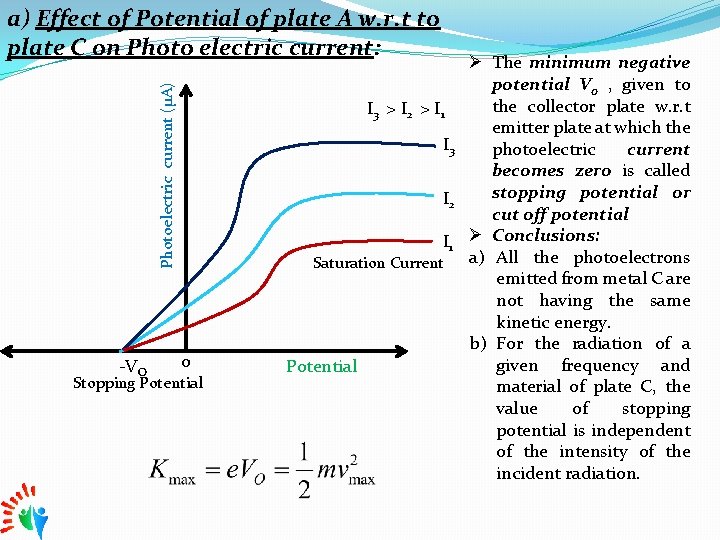
Photoelectric current (μA) a) Effect of Potential of plate A w. r. t to plate C on Photo electric current: -VO I 3 > I 2 > I 1 I 3 I 2 I 1 Saturation Current o Stopping Potential Ø The minimum negative potential Vo , given to the collector plate w. r. t emitter plate at which the photoelectric current becomes zero is called stopping potential or cut off potential Ø Conclusions: a) All the photoelectrons emitted from metal C are not having the same kinetic energy. b) For the radiation of a given frequency and material of plate C, the value of stopping potential is independent of the intensity of the incident radiation.
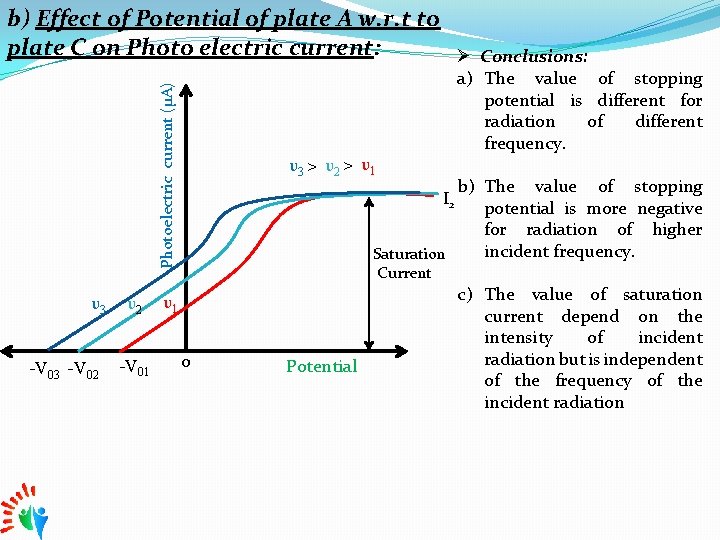
Photoelectric current (μA) b) Effect of Potential of plate A w. r. t to plate C on Photo electric current: υ3 -V 02 υ2 -V 01 Ø Conclusions: a) The value of stopping potential is different for radiation of different frequency. υ3 > υ2 > υ1 b) The value of stopping potential is more negative for radiation of higher incident frequency. Saturation I 2 Current υ1 o Potential c) The value of saturation current depend on the intensity of incident radiation but is independent of the frequency of the incident radiation
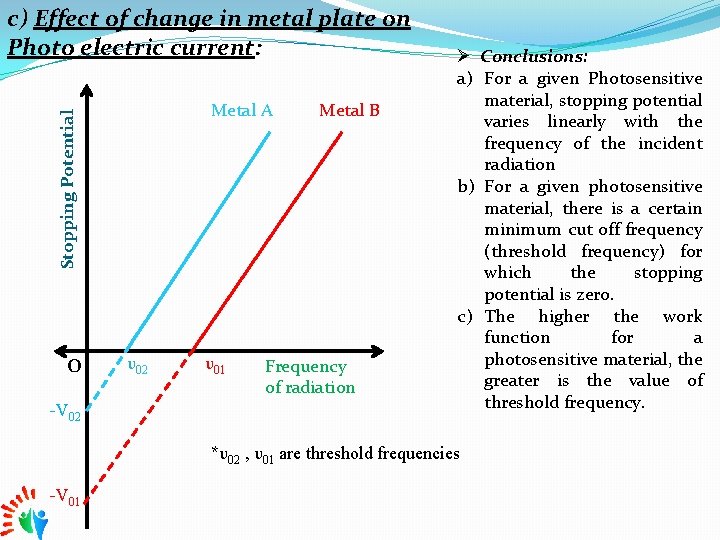
c) Effect of change in metal plate on Photo electric current: Stopping Potential Metal A o -V 02 υ01 Metal B Frequency of radiation Ø Conclusions: a) For a given Photosensitive material, stopping potential varies linearly with the frequency of the incident radiation b) For a given photosensitive material, there is a certain minimum cut off frequency (threshold frequency) for which the stopping potential is zero. c) The higher the work function for a photosensitive material, the greater is the value of threshold frequency. *υ02 , υ01 are threshold frequencies -V 01

Laws of photoelectric emission: 1) For a given metal and frequency of incident radiation , the number of photoelectrons ejected per second is directly proportional to the intensity of incident light. 2) For a given metal, there exist certain minimum frequency of the radiation below which no emission of photo electrons takes place. This frequency is called threshold frequency. 3) Above threshold frequency , the maximum kinetic energy of the emitted electrons is independent of the intensity of the incident light but depends only upon the frequency of the incident light. 4) The photoelectric emission is an instantaneous process.

Einstein’s Photoelectric equation: v Light=collection of tiny packets of energy called quanta v One quanta=One Photon v One Photon energy , E=hυ=hc/λ Assumptions made by Einstein: 1. A part of the energy of the photon is used in liberating the electron from the metal surface which is equal to the work function φ0. 2. The rest of the energy of the photon is used in imparting the maximum kinetic energy Kmax to the emitted electron. 1 Eq. 1 is called Einstein’s Photoelectric Equation Note: The immediate consequence of Photoelectric emission is that particle nature of light has come in to play.

Relation between Cut of potential, frequency of the incident photon and threshold frequency: 1 If V 0 is the cut potential or stopping potential and ‘e’ is the charge of the electron then 2 and if is the threshold frequency, work function is given by 3 From Eq’s 1, 2 & 3 we have We have 4 From Eq. 4 Comparing the above equation with
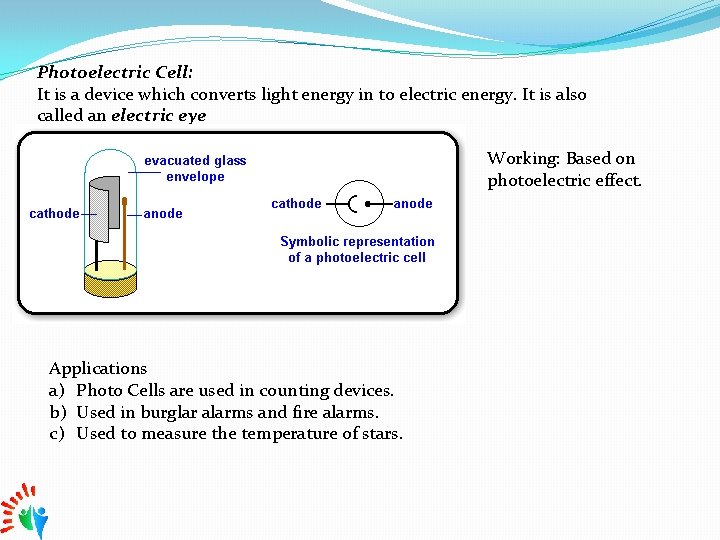
Photoelectric Cell: It is a device which converts light energy in to electric energy. It is also called an electric eye Working: Based on photoelectric effect. Applications a) Photo Cells are used in counting devices. b) Used in burglar alarms and fire alarms. c) Used to measure the temperature of stars.

Dual Nature of Radiation: Interference Diffraction Wave Nature of light Dual Nature of Light Polarization Photoelectric Effect Particle Nature of light

De Broglie Dualistic Hypothesis: According to this a moving material particle sometimes acts as a wave and some times acts as a particle. The wave associated with the moving particle is called matter wave or De-Broglie wave whose wavelength is called de-Broglie wavelength. Derivation: De-Broglie Wavelength

De-Broglie wavelength of an electron: Let m and e be the mass and charge of electron Work done in driving the charge between the electrodes Hence, Implies, Hence de-Broglie wavelength is,
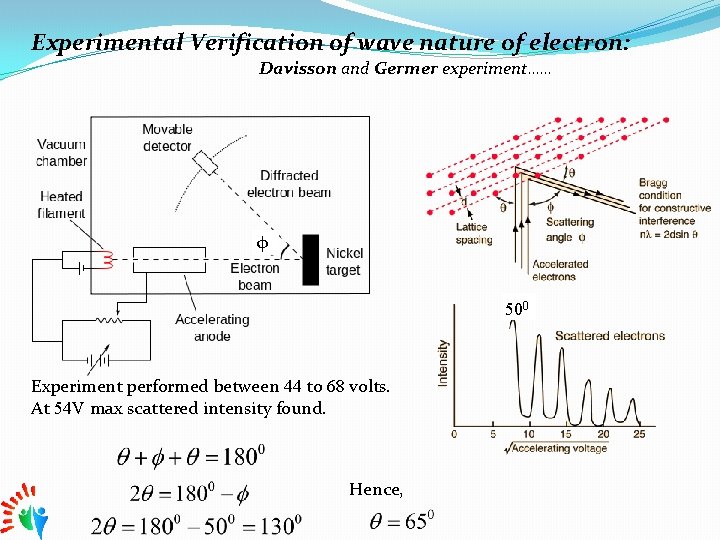
Experimental Verification of wave nature of electron: Davisson and Germer experiment…… φ 500 Experiment performed between 44 to 68 volts. At 54 V max scattered intensity found. Hence,

Acc. Braggs’s Law 2 First order diffraction (n=1) Lattice spacing for Nickel Crystal=0. 91 A 0 Hence, 1 Acc. To de-Broglie, From eq. 1 and 2 it is obvious that the experimental result of davisson Germer experiment is matching well with the de-Broglie wavelength. Hence we an conclude that for material particles wave nature exists

n a h T k u o y
- Slides: 22