DTI Acquisition Guide Donald Brien February 2016 Overview

DTI Acquisition Guide Donald Brien February 2016

Overview • The basic sequence: Once or twice refocused SE EPI • DTI specific parameters: b-value and directions • Standard MRI parameters: TE / TR / Resolution / FOV / Parallel Imaging / Filters / Fat Sat. • Orientation and Encoding Direction(s) – Pros and cons • Top up and the general problems with EPI imaging (acquisition vs post-processing) • Scenarios and Example Images

General MRI Acquisition Considerations • There is theory, and then there is practice. Always pilot your protocol and run pilot analysis. • Optimizing your study requires: – Thinking ahead. What do you need from the data? – What are the limitations? Time, participant issues – What can be mitigated through parameter setup? – What can be mitigated by post-processing? Are other scans necessary to allow this?
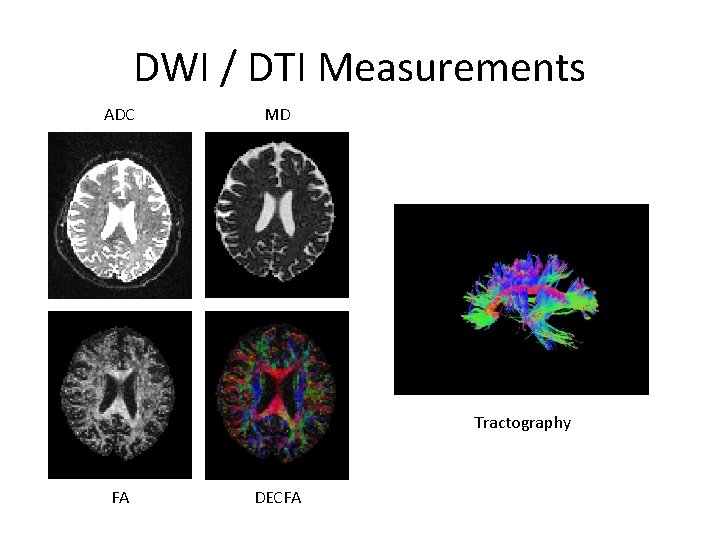
DWI / DTI Measurements ADC MD Tractography FA DECFA
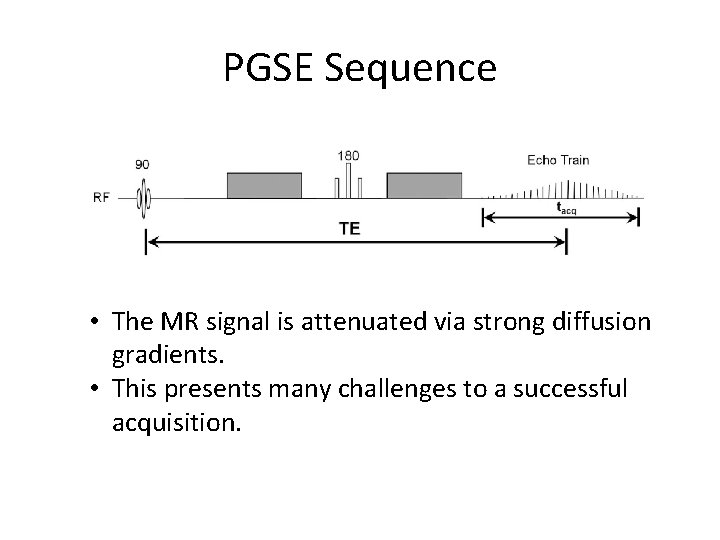
PGSE Sequence • The MR signal is attenuated via strong diffusion gradients. • This presents many challenges to a successful acquisition.
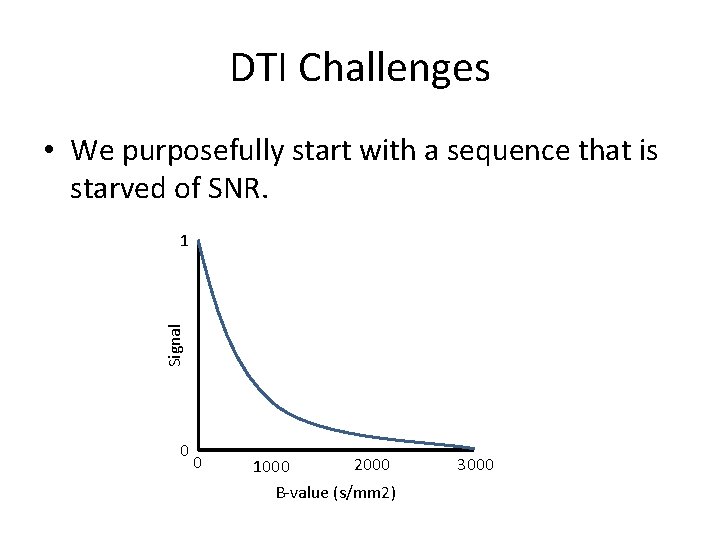
DTI Challenges • We purposefully start with a sequence that is starved of SNR. Signal 1 0 0 2000 1000 B-value (s/mm 2) 3000

DTI Challenges • Bulk motion during gradients creates unpredictable apparent diffusion. • Echo-Planar Imaging (EPI) – Acquire entire k-space in one echo – Bulk motion is minimized and more consistent – Many volumes can now be acquired in a feasible time

DTI Challenges • EPI Readout • We read out the entire k-space in one shot • But we pay a price: distortion in the phaseencoding direction
DTI Challenges
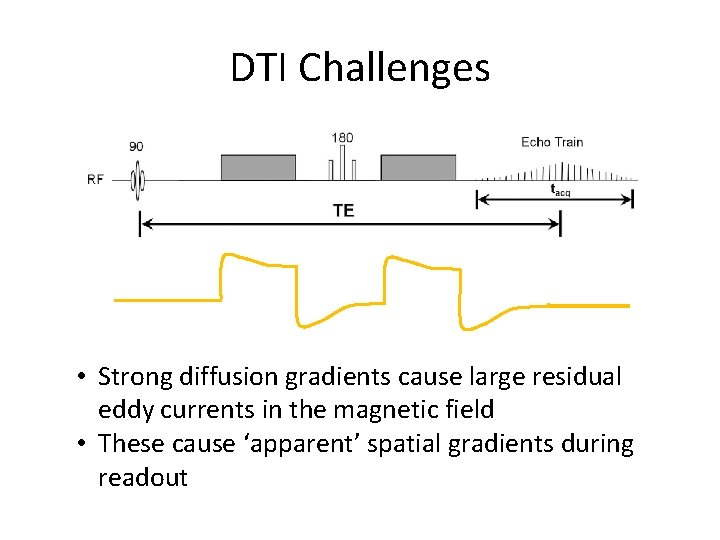
DTI Challenges • Strong diffusion gradients cause large residual eddy currents in the magnetic field • These cause ‘apparent’ spatial gradients during readout
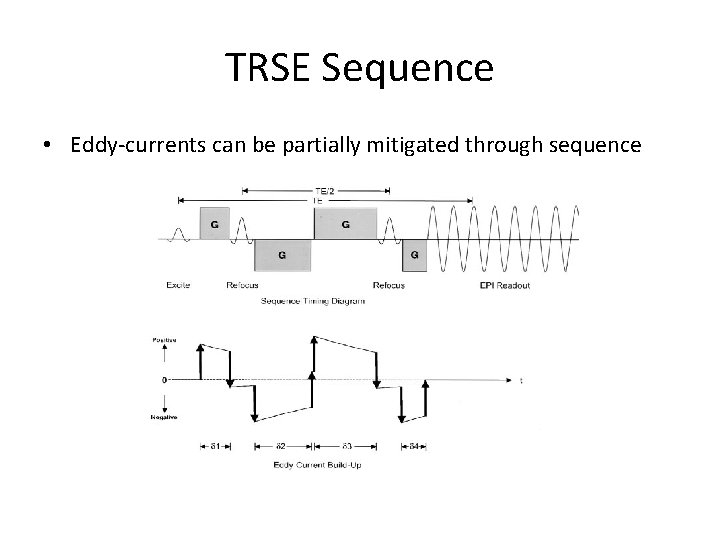
TRSE Sequence • Eddy-currents can be partially mitigated through sequence

DTI Specific Parameters • b-values – 1000 s/mm 2 is the standard because most sensitive to changes in diffusion in the brain – Also good choice because most data is acquired at this bvalue, which will allow your results to be comparable to normative datasets • 9. 1: 1 DWI: b 0 to minimize variance in tensor model • # of directions – 20 directions minimum for accurate anisotropy measurements – Benefit to tensors flattens out around 30 directions – diminishing returns
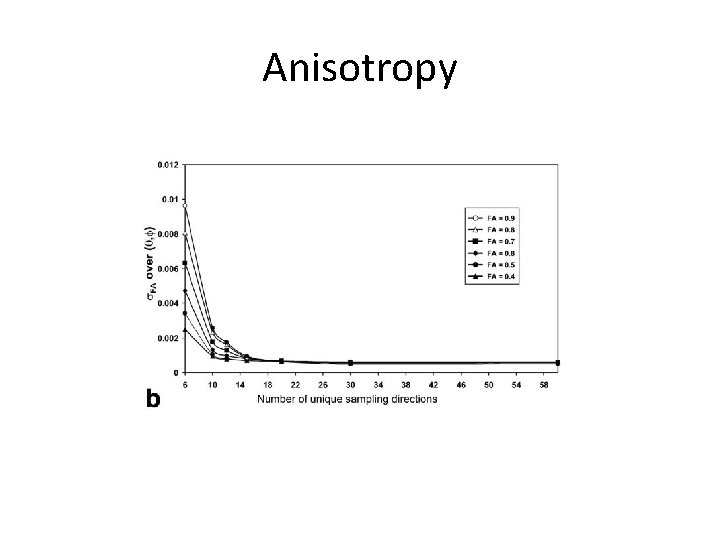
Anisotropy
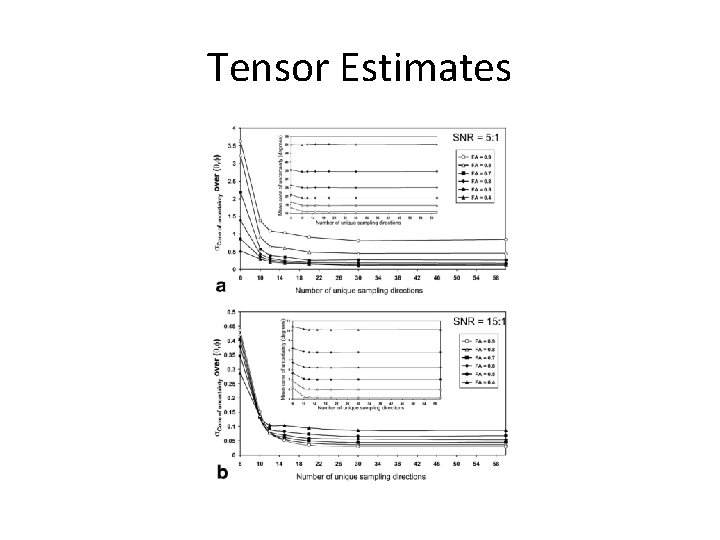
Tensor Estimates

Common MRI Parameters • Fat. Sat. on because lipid signal interferes (off resonance chemical shift) with water diffusion signal. • TE short as possible to maximize SNR and same for all measurements • TR must be > 3 x T 1 of tissue (5 s or more), but viable (10 mins or less total typically) • Resolution tends to be lower to compensate for lower SNR and permit a quick readout

Other Parameters • FOV: ~24 cm to cover brain and prevent aliasing • Slice Gap: 0% - use interleaved ordering • Bandwidth: Generally higher for SNR and fast imaging, but testing important • Parallel Imaging: GRAPPA for faster k-space sampling – reduce distortion vs lower SNR • Partial Fourier: May introduce artifacts, but does reduce TE.
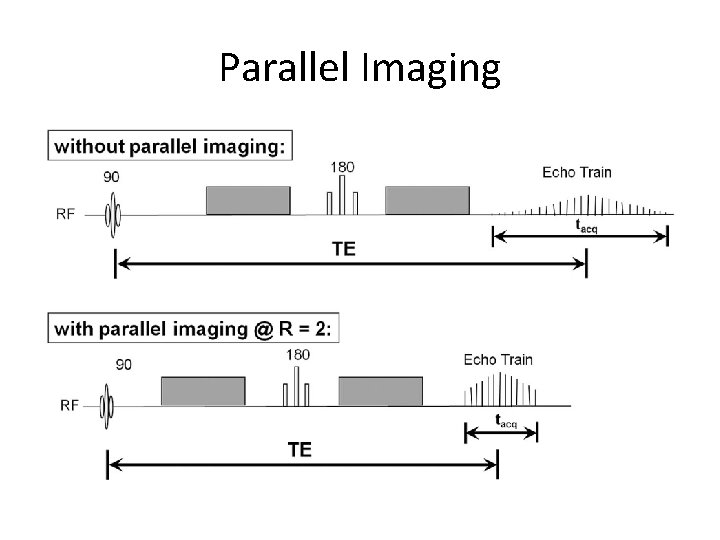
Parallel Imaging

Orientation and Encoding Directions • A>>P is preferred for single scan studies – Minimizes asymmetry biases (does not eliminate) • Axial or axial oblique? – Axial may reduce ghosting – Measures should be rotationally invariant (if 7+ measurements) either way – Be consistent and nearly axial (ACPC is common)
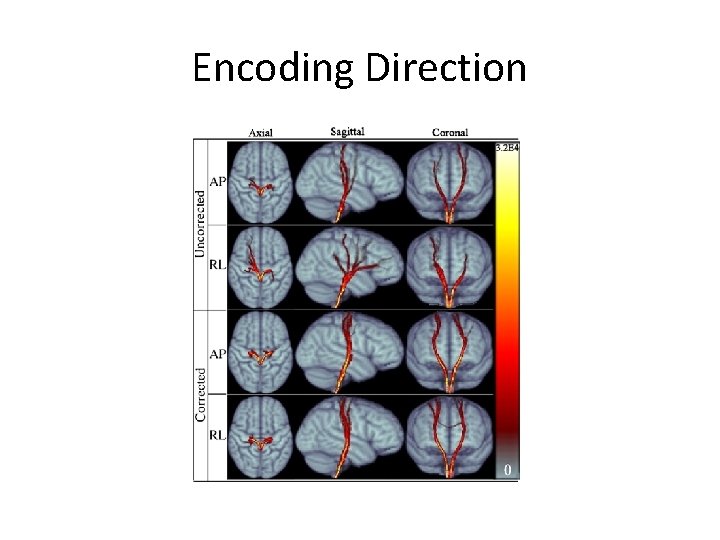
Encoding Direction
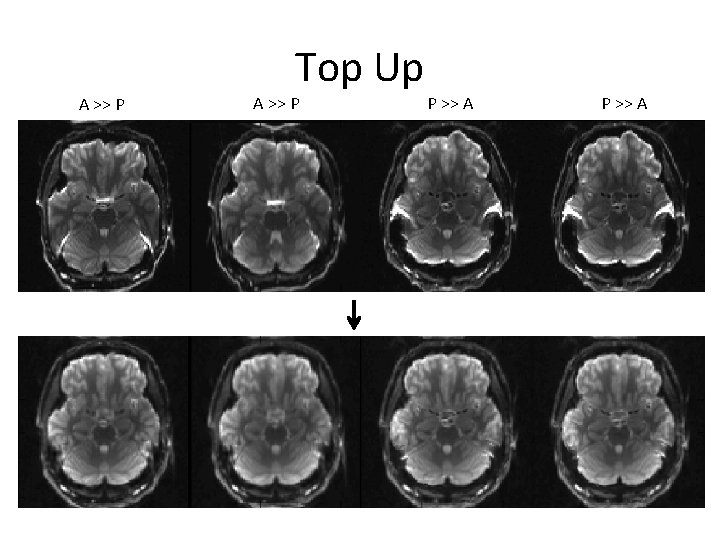
A >> P Top Up A >> P P >> A

Multishot Imaging

Scenarios 1 • Grey matter diffusion (DWI) – 3 orthogonal DWIs and a b 0 would be sufficient as grey matter is mostly isotropic • White matter diffusion (DWI) – 6 maximally spread DWIs and a b 0 for rotationally invariant measures of white matter MD • Whole Brain FA measurements (DTI) – 6 minimum, but 20 would be ideal.

Scenarios 2 • Pediatric DTI imaging – Motion susceptibility and time limit with small head • 20 directions, 1 AP scan, 1 extra b 0, maximize SNR, lower bvalue due to developing brain (~700 s/mm 2), SAR considerations • ALS / Parkinsons imaging – Motion susceptibility and time limit • 20 directions, 1 AP scan, 1 extra b 0 • Healthy Controls – Ideal candidates. Adult, good at staying still. • 30 directions, AP and PA, 3 extra b 0

Scenarios 3 • NHP or other animal or small structure imaging – Very small head. Probably very little motion issues due to anesthesia. • 20 -30 directions, but can use the increased time to our advantage. More averages, more directions, less acceleration. • Multishot scans. Separate k-space into many read outs to drastically decrease susceptibility problems. • Many extra b 0 • AP and PA – susceptibility problems likely exaggerated with small FOV, strong gradients, and anatomical differences (large eyes) • Investigate b-value considerations in animal

Scenarios 4 • Small structure imaging in awake participants – Imaging a smaller region of interest may require higher resolution and a higher # of directions, but motion and SNR an issue • Less slices, more focused scan to reduce TR • Train participants on staying still outside of the scanner / Pick participants carefully • Cardiac gaiting? • Multiband imaging – increase number of directions without increasing time, but preserving SNR. Not available commercially yet. Long reconstruction.
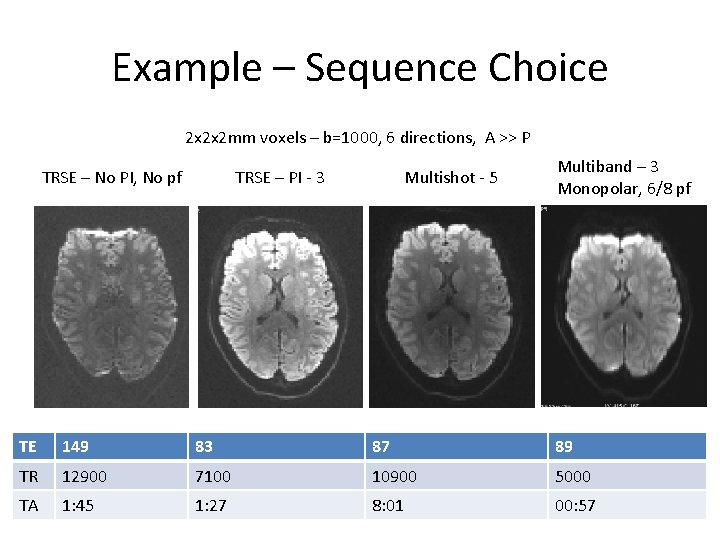
Example – Sequence Choice 2 x 2 x 2 mm voxels – b=1000, 6 directions, A >> P TRSE – No PI, No pf TRSE – PI - 3 Multishot - 5 Multiband – 3 Monopolar, 6/8 pf TE 149 83 87 89 TR 12900 7100 10900 5000 TA 1: 45 1: 27 8: 01 00: 57
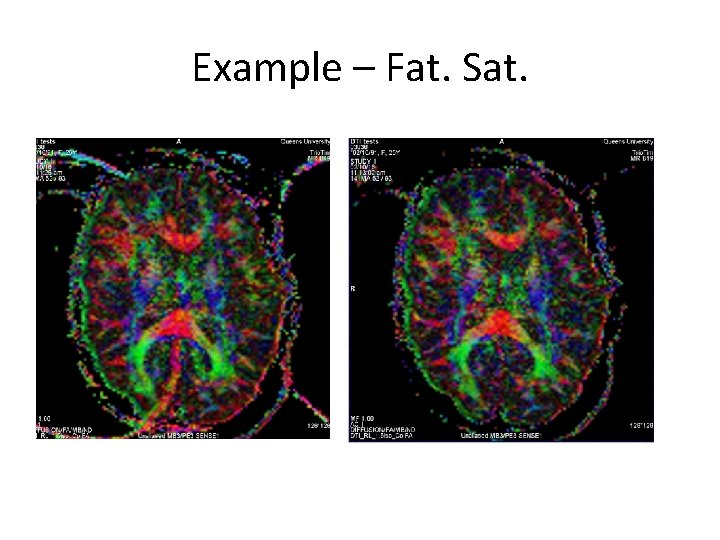
Example – Fat. Sat.
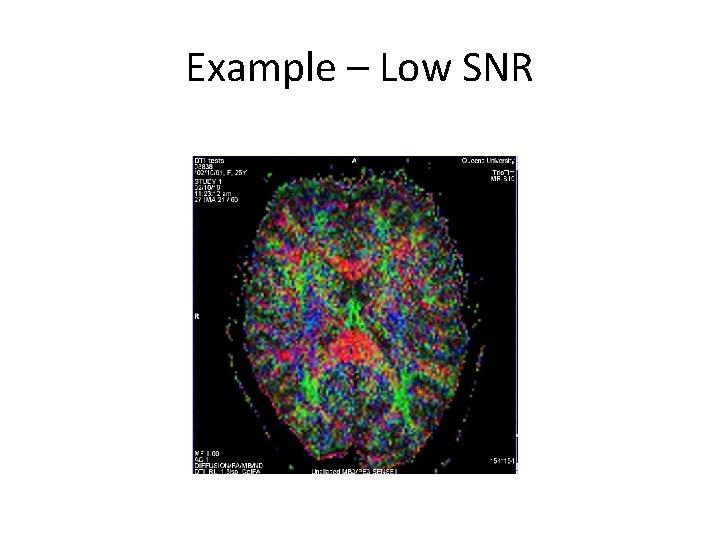
Example – Low SNR

Conclusions 1. 2. 3. What measures will you ultimately need? – What tools do you plan to use? – 5. 6. Resolution / SNR / directions / etc… What trade-offs are you willing to make given your requirements and limitations? – 4. MD, ADC, FA, Tractography? Time constraints, motion issues, resolution requirements What can be mitigated through parameters vs post-processing? – Parallel imaging, partial fourier, multi-shot, multiband What additional scans should you acquire? – Additional encoding, T 2 w anatomical, b 0 field? The interaction of DWI / DTI parameters is complex. – – Consult previous research before designing your acquisition. Even with careful theoretical preparation, careful pilot testing is fundamental to any MRI study.
- Slides: 29