DRUGS USED TO TREAT Anemias Presented by Dr

DRUGS USED TO TREAT Anemias Presented by Dr. Sasan Zaeri Parm. D, Ph. D Fall, 2015

Agents Used in Anemias • Hematopoiesis: – the production from undifferentiated stem cells of circulating erythrocytes, platelets, and leukocytes • Essential factors: – Iron, vitamin B 12, folic acid and hematopoietic growth factors • Inadequate supply results in: – Anemia, thrombocytopenia and neutropenia 2

AGENTS USED IN ANEMIAS: IRON • Iron deficiency – the most common cause of chronic anemia – leads to pallor, fatigue, dizziness, exertional dyspnea, etc. – small erythrocytes with insufficient hemoglobin are formed >>> microcytic hypochromic anemia 3

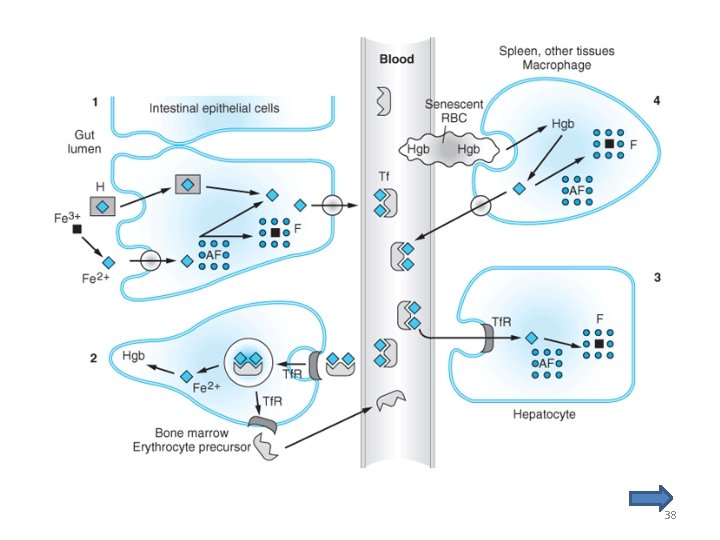
Pharmacokinetics • Elaborate system for regulating iron (figure) • Iron sources to support hematopoiesis: – catalysis of the hemoglobin in senescent or damaged erythrocytes – dietary iron from a wide variety of foods • iron requirements can exceed normal dietary supplies in – growing children and pregnant women (increased iron requirements) – menstruating women (increased losses of iron) 4

Pharmacokinetics • ABSORPTION – 0. 5 -1 mg/d iron from food by a normal individual – 1 -2 mg/d in normal menstruating women – 3 -4 mg/d in pregnant women • The iron in meat (heme iron) can be efficiently absorbed • Nonheme iron in foods must be reduced to ferrous iron (Fe 2+) before it can be absorbed 5

Pharmacokinetics • TRANSPORT – Iron is transported in the plasma bound to transferrin – The transferrin-iron complex enters maturing erythroid cells by receptor-mediated endocytosis – iron deficiency anemia is associated with an increased concentration of serum transferrin 6

Pharmacokinetics • STORAGE – primarily as ferritin in intestinal mucosal cells, macrophages in the liver, spleen, and bone and in parenchymal liver cells – the serum ferritin level can be used to estimate total body iron stores – Apoferritin (precursor of ferritin) levels is regulated by the levels of free iron • ↓ free iron → ↓ apoferritin → ↑ iron binding to transferrin • ↑ free iron → ↑ apoferritin → protection of organs from the iron toxic effects 7

Pharmacokinetics • ELIMINATION – no mechanism for excretion of iron: • regulation of iron balance must be achieved by changing intestinal absorption and storage of iron, in response to the body's needs 8

Clinical Pharmacology • The only clinical indication: – treatment or prevention of iron deficiency anemia • Patients with increased iron requirements: – – infants, especially premature infants children during rapid growth periods pregnant and lactating women patients with chronic kidney disease • Loss of erythrocytes at a relatively high rate during hemodialysis • Erythrocyte production at a high rate as a result of treatment with erythropoietin • Patients with iron malabsorption: – Patients after gastrectomy – patients with severe small bowel disease that results in generalized malabsorption 9

Clinical Pharmacology • The most common cause of iron deficiency in adults: blood loss – Menstruating women lose about 30 mg of iron with each menstrual period • many premenopausal women have low iron stores or even iron deficiency – In men and postmenopausal women, the most common site of blood loss is the gastrointestinal tract 10

Clinical Pharmacology • TREATMENT – with oral or parenteral iron preparations • Oral iron corrects the anemia just as rapidly and completely as parenteral iron in most cases if iron absorption from the gastrointestinal tract is normal • for patients with advanced chronic kidney disease who are undergoing hemodialysis and treatment with erythropoietin, parenteral iron administration is preferred 11

Clinical Pharmacology • Oral iron therapy – Only ferrous salts should be used • Ferrous sulfate, ferrous gluconate and ferrous fumarate – Different iron salts provide different amounts of elemental iron • Ferrous fumarate> Ferrous sulfate>ferrous gluconate – 200 -400 mg/d of elemental iron corrects iron deficiency most rapidly 12

Clinical Pharmacology • Oral iron therapy – lower daily doses can be given: • slower but still complete correction of iron deficiency – Treatment should be continued for 3 -6 months after correction of the cause of the iron loss • This corrects the anemia and replenishes iron stores 13

Clinical Pharmacology • Oral iron therapy • Common adverse effects: – nausea, epigastric discomfort, abdominal cramps, constipation, and diarrhea • Adverse effects can often be overcome by – lowering the daily dose of iron – taking the tablets immediately after or with meals – Changing from one iron salt to another • Patients taking oral iron develop black stools – This may obscure the diagnosis of continued gastrointestinal blood loss 14

Clinical Pharmacology • Parenteral iron therapy • Parenteral therapy should be reserved for – patients unable to tolerate or absorb oral iron • • patients with various postgastrectomy conditions patients with previous small bowel resection inflammatory bowel disease malabsorption syndromes – patients with extensive chronic blood loss who cannot be maintained with oral iron alone: • advanced chronic renal disease including hemodialysis and treatment with erythropoietin 15

Clinical Pharmacology • Parenteral iron therapy • Iron-dextran – deep IM injection or IV infusion – the IV route is used most commonly – Adverse effects of IV route: • headache, light-headedness, fever, arthralgias, nausea and vomiting, back pain, flushing, urticaria, bronchospasm, and, rarely, anaphylaxis and death – a small test dose should always be given before full IM or IV doses are given 16

Clinical Pharmacology • Parenteral iron therapy • Alternative preparations to iron-dextran: Ironsucrose and iron-gluconate – Can be given only by IM route – Less likely than iron dextran to cause hypersensitivity reactions 17

Clinical Toxicity • ACUTE IRON TOXICITY – Seen almost exclusively in young children who accidentally ingest iron tablets • Even 10 tablets can be lethal in young children • Adult patients should be instructed to store tablets out of the reach of children – Manifestations: • • • necrotizing gastroenteritis Vomiting abdominal pain bloody diarrhea Severe metabolic acidosis Coma and death 18

Clinical Toxicity • ACUTE IRON TOXICITY – Urgent treatment is necessary – Whole bowel irrigation should be performed – Deferoxamine, a potent iron-chelating compound, can be given systemically to bind iron that has already been absorbed and to promote its excretion in urine and feces – Activated charcoal does not bind iron and thus is ineffective – Appropriate supportive therapy for gastrointestinal bleeding, metabolic acidosis, and shock must also be provided 19

Clinical Toxicity • CHRONIC IRON TOXICITY – Also known as iron overload or hemochromatosis – excess iron is deposited in the heart, liver, pancreas, and other organs – It can lead to organ failure and death – occurs in • patients with inherited hemochromatosis (a disorder characterized by excessive iron absorption) • patients who receive many red cell transfusions over a long period of time (e. g. patients with thalassemia major) 20

Clinical Toxicity • CHRONIC IRON TOXICITY – In the absence of anemia is treated by intermittent phlebotomy – parenteral deferoxamine is much less efficient • deferoxamine can be the only option for iron overload in patients with thalassemia major – deferasirox (oral iron chelator) has been approved for treatment of iron overload • Deferasirox appears to be as effective as deferoxamine at reducing liver iron concentrations and is much more convenient 21

AGENTS USED IN ANEMIAS: Vitamin B 12 • Vitamin B 12 serves as a cofactor for several essential biochemical reactions in humans • Deficiency of vitamin B 12 leads to – anemia – gastrointestinal symptoms – neurologic abnormalities • Most common cause of B 12 deficiency: – inadequate absorption of dietary vitamin B 12 especially in older adults • Active forms of the vitamin in humans: – Deoxyadenosylcobalamin – Methylcobalamin 22

VITAMIN B 12 • Inactive forms of the vitamin: – Cyanocobalamin (available for therapeutic use) – Hydroxocobalamin (available for therapeutic use) – Other cobalamins found in food sources • The chief dietary source of vitamin B 12 : – Meat, liver, eggs and dairy products • Vitamin B 12 is sometimes called extrinsic factor to differentiate it from intrinsic factor 23

Pharmacokinetics • Vitamin B 12 is avidly stored in the liver – with an average total storage pool of 3000 -5000 mcg (Lasting for 5 years) • Vitamin B 12 is absorbed only after it complexes with intrinsic factor secreted by the parietal cells of the gastric mucosa – intrinsic factor-vitamin B 12 complex is subsequently absorbed in the distal ileum • Vitamin B 12 deficiency results from malabsorption of vitamin B 12 due to – lack of intrinsic factor – loss or malfunction of the specific absorptive mechanism in the distal ileum • Vitamin B 12 is transported to the various cells of the body bound to a plasma glycoprotein, transcobalamin II 24

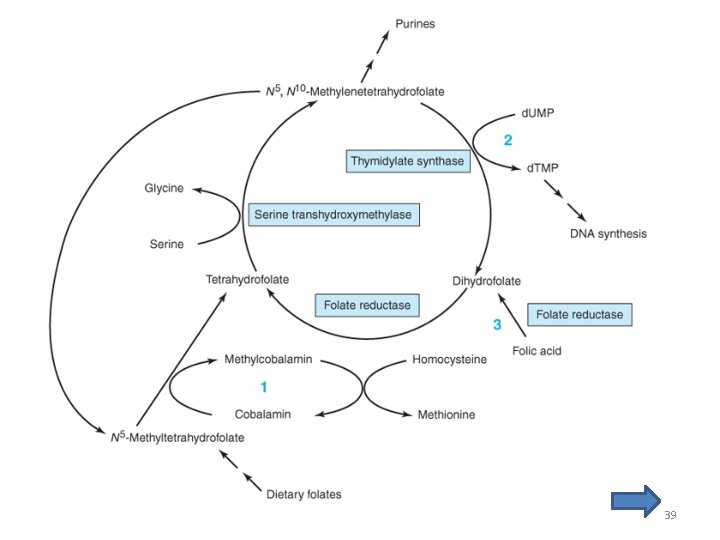
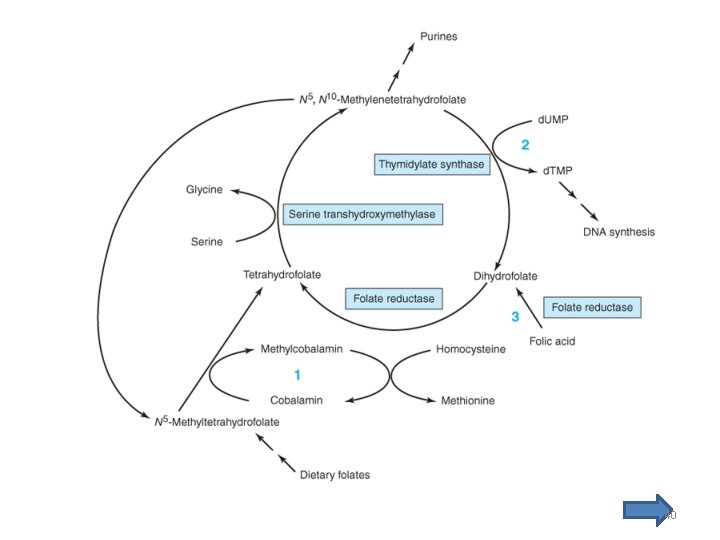
Pharmacodynamics • Methylcobalamin converts N 5 methyltetrahydrofolate to tetrahydrofolate (figure) – N 5 -methyltetrahydrofolate: major dietary and storage folate – Tetrahydrofolate: precursor of folate cofactors • ↓ Vitamin B 12 → ↓ Tetrahydrofolate → ↓folate cofactors → ↓d. TMP and purines and DNA in rapidly dividing cells 25

Pharmacodynamics • Methylfolate trap: – The accumulation of folate as N 5 methyltetrahydrofolate and the associated depletion of tetrahydrofolate cofactors in vitamin B 12 deficiency • Folic acid can be reduced to dihydrofolate and tetrahydrofolate by the enzyme dihydrofolate reductase (figure) – this explains why the megaloblastic anemia of vitamin B 12 deficiency can be partially corrected by ingestion of relatively large amounts of folic acid 26

Pharmacodynamics • Other enzymatic reaction that requires vitamin B 12 : – isomerization of methylmalonyl-Co. A to succinyl-Co. A • Vitamin B 12 deficiency causes neurologic damage via: – ↑ methylmalonyl-Co. A or ↓ methionine • Administration of folic acid in the setting of vitamin B 12 deficiency will not prevent neurologic manifestations 27

Clinical Pharmacology • Vitamin B 12 deficiency manifestations: – megaloblastic anemia (most characteristic clinical manifestation) – The neurologic syndrome • paresthesias, weakness , ataxia, other central nervous system dysfunctions • Correction of vitamin B 12 deficiency arrests the progression of neurologic disease, but it may not fully reverse neurologic symptoms that have been present for several months 28

Clinical Pharmacology • Upon diagnosis of megaloblastic anemia: – it must be determined whether vitamin B 12 or folic acid deficiency is the cause • This can usually be accomplished by measuring serum levels of the vitamins 29

Clinical Pharmacology • The most common causes of vitamin B 12 deficiency: – pernicious anemia – partial or total gastrectomy – conditions that affect the distal ileum • Pernicious anemia results from defective secretion of intrinsic factor by the gastric mucosal cells 30

Clinical Pharmacology • Treatment of vitamin B 12 deficiency – Parenteral injections of vitamin B 12 are required for therapy – Vitamin B 12 for parenteral injection is available as cyanocobalamin or hydroxocobalamin • Hydroxocobalamin is preferred because it is more highly protein-bound – Initial therapy: 100 -1000 mcg of vitamin B 12 IM daily or every other day for 1 -2 weeks to replenish body stores – Maintenance therapy: 100 -1000 mcg IM once a month for life 31

AGENTS USED IN ANEMIAS: FOLIC ACID • Reduced forms of folic acid are required for synthesis of amino acids, purines, and DNA • The consequences of folate deficiency: – Anemia – congenital malformations in newborns 32

Pharmacokinetics • The richest sources of folic acid: – yeast, liver, kidney and green vegetables • Body stores of folates are relatively low and daily requirements high – folic acid deficiency and megaloblastic anemia can develop within 1 -6 months after the intake of folic acid stops 33

Clinical Pharmacology • Folate deficiency results in a megaloblastic anemia that is indistinguishable from the anemia caused by vitamin B 12 deficiency – folate deficiency does not cause the characteristic neurologic syndrome seen in vitamin B 12 deficiency 34

Clinical Pharmacology • Causes of folic acid deficiency – inadequate dietary intake of folates – alcohol dependence – liver diseases (diminished hepatic storage of folates) – Pregnancy • maternal folic acid deficiency may cause fetal neural tube defects e. g. spina bifida – hemolytic anemia 35

Clinical Pharmacology • Causes of folic acid deficiency – renal dialysis (folate loss during dialysis) – Drugs • Methotrexate, trimethoprim and pyrimethamine – Leading to megaloblastic anemia • Long-term therapy with phenytoin – Rarely leading to megaloblastic anemia 36

Clinical Pharmacology • Treatment of folic acid deficiency: – 1 mg/d folic acid orally • reverses megaloblastic anemia • restore normal serum folate levels • replenishes body stores of folates – Therapy should be continued until the underlying cause of the deficiency is removed or corrected • Folic acid supplementation to prevent folic acid deficiency should be considered in high-risk patients: – pregnant women, patients with alcohol dependence, hemolytic anemia, liver disease, or certain skin diseases, and patients on renal dialysis 37
38
39
40
- Slides: 40