Drugs Used in Coagulation Disorders Presented by Dr

Drugs Used in Coagulation Disorders Presented by Dr. Sasan Zaeri Pharm. D, Ph. D
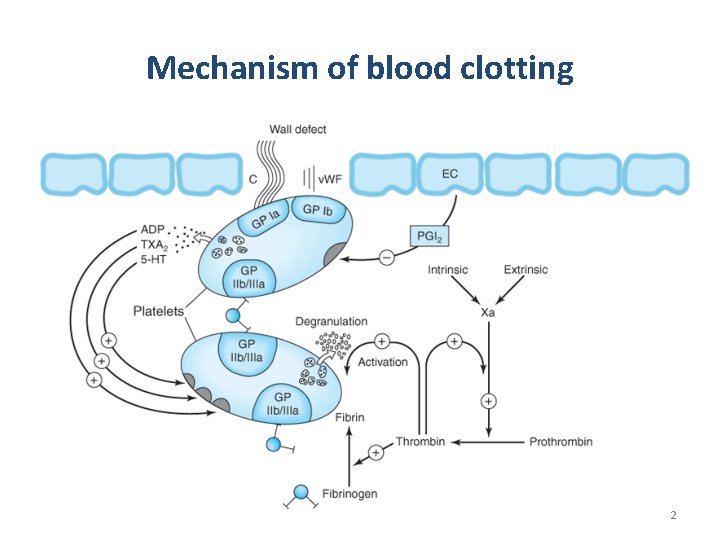
Mechanism of blood clotting 2
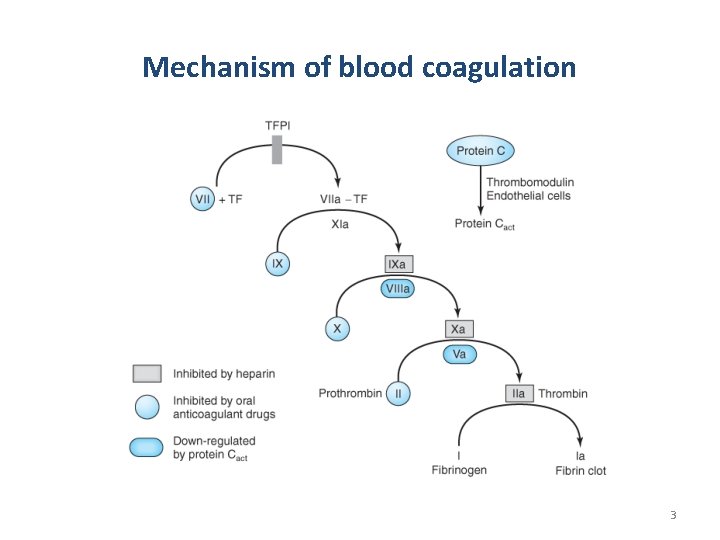
Mechanism of blood coagulation 3
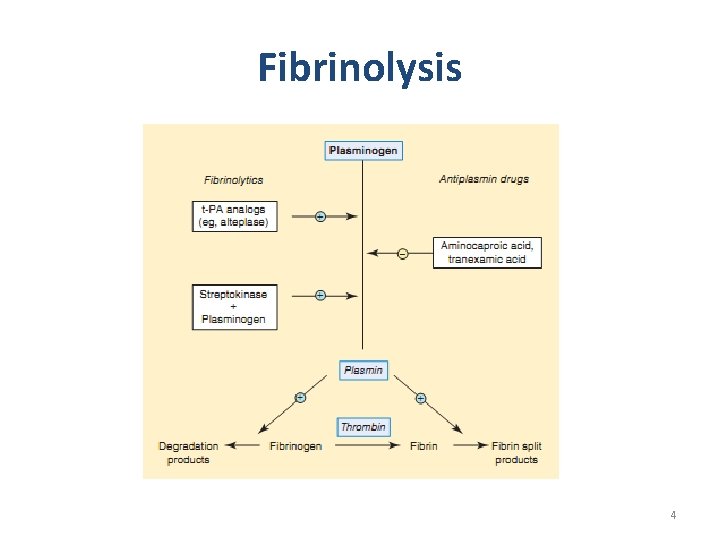
Fibrinolysis 4
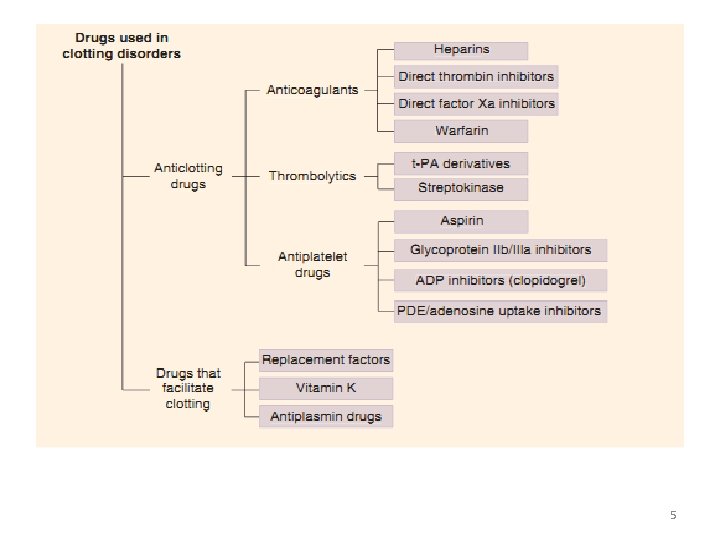
5

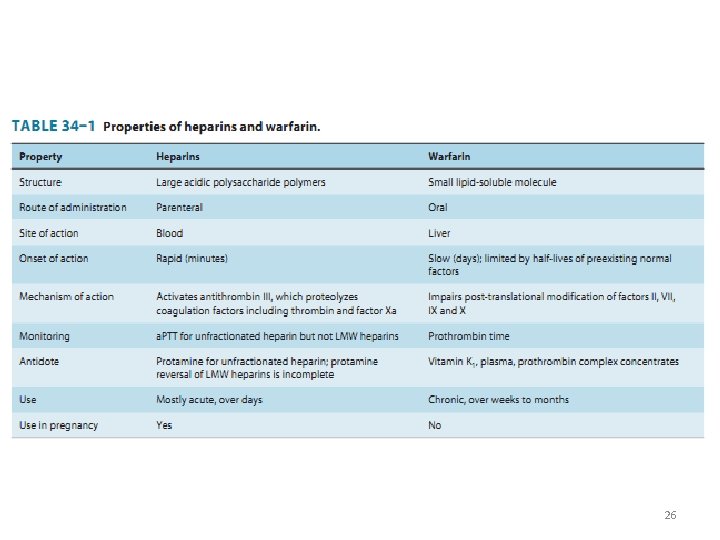
ANTICOAGULANTS Classification • Three major types of anticoagulants: – Heparin and related products • must be used parenterally – Direct thrombin and factor Xa inhibitors • used parenterally or orally – Orally active coumarin derivatives (e. g. warfarin) 6

ANTICOAGULANTS Heparin • A large sulfated polysaccharide polymer obtained from animal sources • Highly acidic and can be neutralized by basic molecules – Protamine sulfate (heparin antidote) • Given IV or SC to avoid the risk of hematoma associated with IM injection 7

ANTICOAGULANTS Heparin • Low-molecular-weight (LMW) heparin – Enoxaparin, Dalteparin, Tinzaparin – Greater bioavailability (SC) – Longer durations of action • Administered once or twice a day • Fondaparinux – A small synthetic drug that contains the biologically active pentasaccharide – Administered SC once daily 8

Heparin Mechanism and effects • Heparin binds to antithrombin III (ATIII): – irreversible inactivation of thrombin and factor Xa • 1000 -fold faster than ATIII alone • Heparin provides anticoagulation immediately after administration • Heparin monitoring – Activated partial thromboplastin time (a. PTT) 9
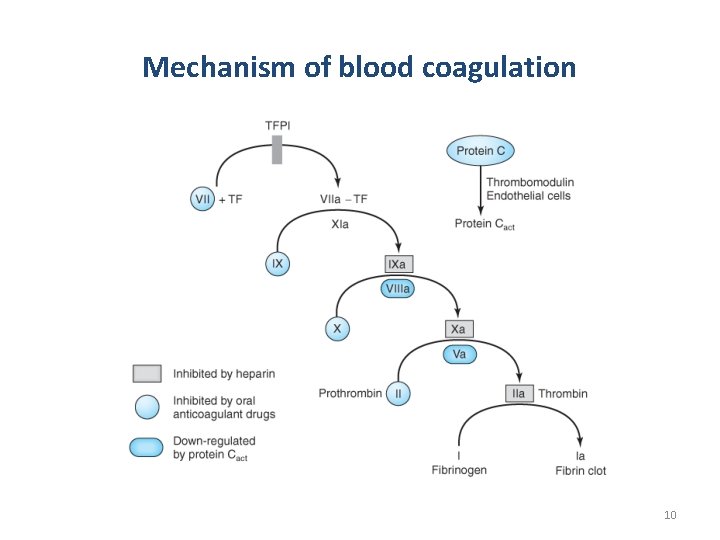
Mechanism of blood coagulation 10

Mechanism and effects • LMW heparins and fondaparinux – bind ATIII – same inhibitory effect on factor Xa as heparin– ATIII – they fail to affect thrombin • a more selective action – a. PTT not required • potential problem in renal failure due to decreased clearance 11

Clinical uses • When anticoagulation is needed immediately e. g. when starting therapy • Common uses: – DVT – Pulmonary embolism – acute myocardial infarction • in combination with thrombolytics for revascularization • in combination with glycoprotein IIb/IIIa inhibitors during angioplasty and placement of coronary stents • The drug of choice in pregnancy 12

Toxicity • Increased bleeding (most common) – may result in hemorrhagic stroke – Protamine as antidote • Not effective for LMW heparins and fondaparinux • Heparin-induced thrombocytopenia (HIT) • • • Due to antibody against complex of heparin and platelet factor 4 May yield venous thrombosis less likely with LMW heparins and fondaparinux • Osteoporosis – Due to prolonged use of unfractionated heparin 13

Direct Thrombin Inhibitors • Lepirudin – Recombinant form hirudin (Hirudo medicinalis) • Desirudin and Bivalirudin – Modified forms of hirudin • Argatroban – A small molecule with a short half-life • Dabigatran – Orally active 14

Mechanism and effects • These drugs inhibit both soluble thrombin and the thrombin enmeshed within developing clots • Bivalirudin – also inhibits platelet activation 15

Clinical uses • Alternatives to heparin – primarily in patients with HIT • Coronary angioplasty – Bivalirudin in combination with aspirin v. Monitoring using a. PTT requiured 16

Toxicity • Bleeding – No reversal agents exist • Anaphylactic reactions – Prolonged infusion of lepirudin induces antibodies that form a complex with lepirudin and prolong its action 17

Direct Oral Factor Xa inhibitors • Rivaroxaban and Apixaban – Rapid onset of action – Shorter half-lives than warfarin – Given as fixed oral doses and do not require monitoring 18

Rivaroxaban and Apixaban • Bind to both free factor Xa and factor Xa bound in the clotting complex • Rivaroxaban is approved for: – Prevention of venous thromboembolism following hip or knee surgery – Prevention of stroke in patients with atrial fibrillation • Toxicity – Bleeding • No reversal agents exist 19

Warfarin • Small lipid-soluble molecule – readily absorbed after oral administration • Highly bound to plasma proteins (>99%) • Its elimination depends on metabolism by cytochrome P 450 enzymes 20
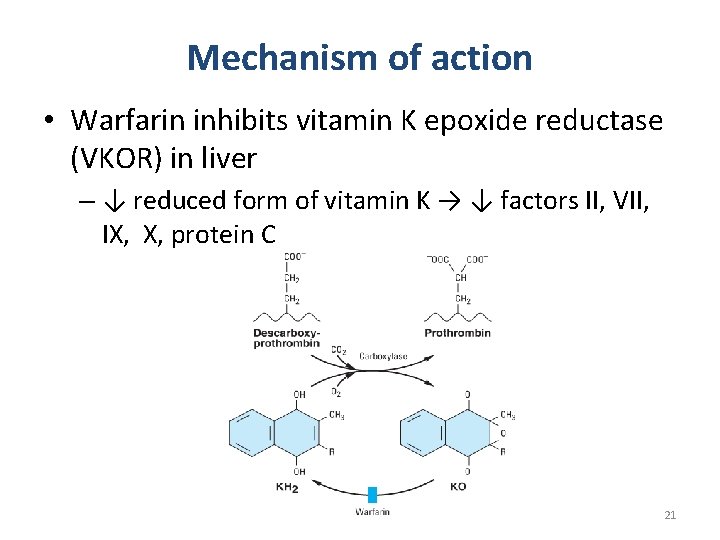
Mechanism of action • Warfarin inhibits vitamin K epoxide reductase (VKOR) in liver – ↓ reduced form of vitamin K → ↓ factors II, VII, IX, X, protein C 21

• Anticoagulant effect is observed within 8 -12 h • The action of warfarin can be reversed by: – Vitamin K 1 (slowly within 6 -24 h) – Transfusion with fresh or frozen plasma (more rapid reversal) • Warfarin monitoring: – Prothrombin time (PT) expressed by INR – INR: 2 -3 22

Clinical uses • Chronic anticoagulation in all of the clinical situations described for heparin – Exception: anticoagulation in pregnant women • In DVT 1. Heparin + warfarin (5 -7 days) 2. Warfarin (3 -6 months) 23
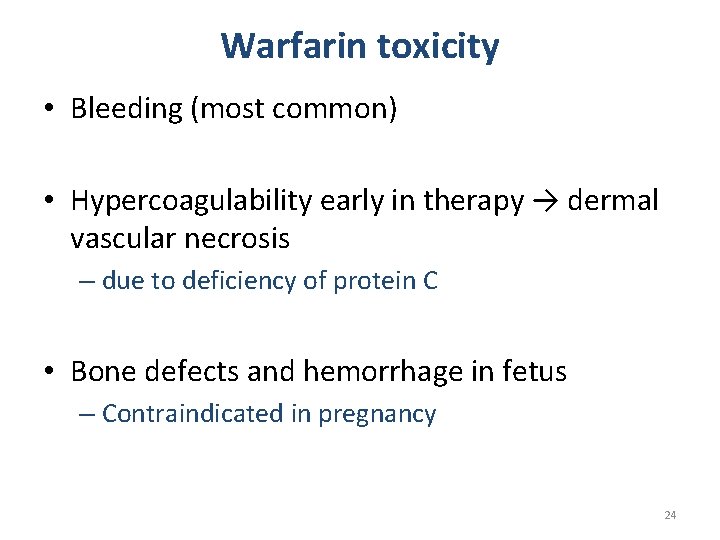
Warfarin toxicity • Bleeding (most common) • Hypercoagulability early in therapy → dermal vascular necrosis – due to deficiency of protein C • Bone defects and hemorrhage in fetus – Contraindicated in pregnancy 24

Warfarin toxicity • Drug interactions – Cytochrome P 450 inducers • carbamazepine, phenytoin, rifampin, barbiturates – Cytochrome P 450 inhibitors • amiodarone, selective serotonin reuptake inhibitors, cimetidine • Cytochrome P 450 2 C 9 and VKOR gene polymorphism – Dose tailoring based on genetic profile (!) 25
26

THROMBOLYTIC AGENTS • Streptokinase – synthesized by streptococci • Urokinase – Human enzyme produced by kidneys • Anistreplase – complex of purified human plasminogen and bacterial streptokinase • Alteplase, Tenecteplase and Reteplase – Recombinant forms of t-PA 27
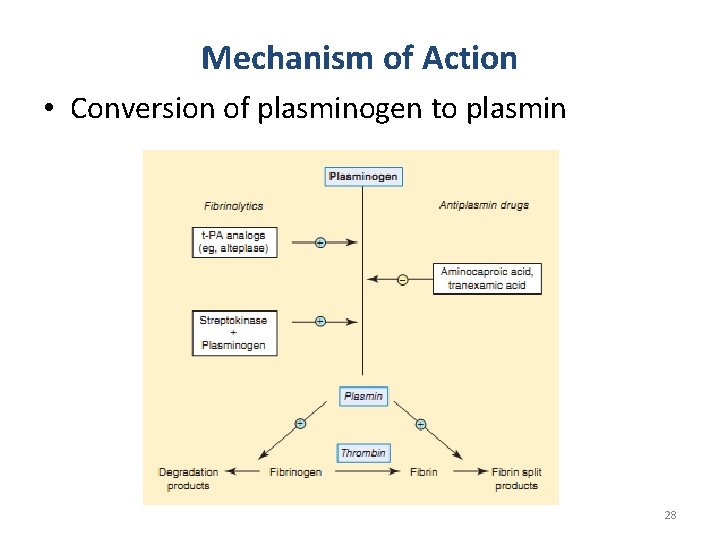
Mechanism of Action • Conversion of plasminogen to plasmin 28

t-PA • Fibrin selectivity – In theory, it should result in less danger of widespread bleeding – In fact, t-PA’s selectivity appears to be quite limited • Reteplase – slightly faster onset of action – longer half-life • Tenecteplase – longer half-life 29

Clinical Uses • Alternative to coronary angioplasty – Best result in ST-elevated MI and bundle branch block – Prompt recanalization if used within 6 h • Ischemic stroke – Better clinical outcome if used within 3 h – Cerebral hemorrhage must be ruled out before such use • Severe pulmonary embolism 30

Toxicity • Bleeding – Same frequency with all thrombolytics – Cerebral hemorrhage (most serious manifestation) • Allergic reactions (streptokinase) – Even at first dose (streptococcal infection history) – Loss of drug efficacy – Not observed with recombinant forms of t-PA • BUT, t-PA is more expensive and not much more effective 31
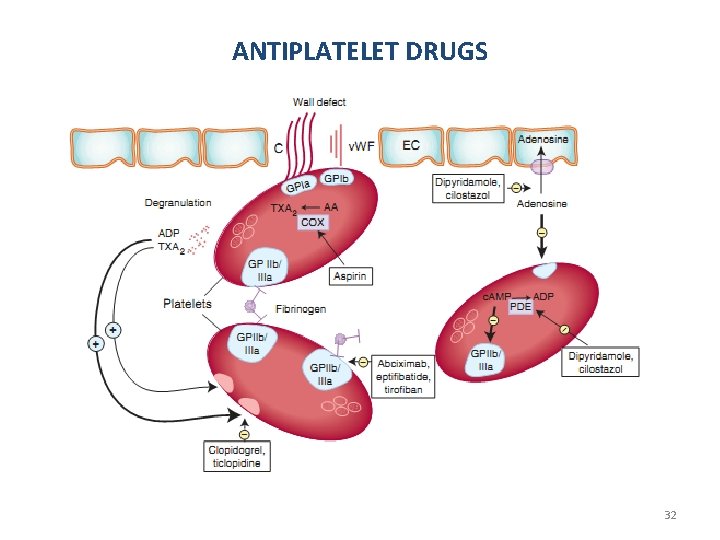
ANTIPLATELET DRUGS 32

ANTIPLATELET DRUGS • Aspirin acts on COX irreversibly – several-day effect • Other NSAIDs not used as antiplatelet drug – May interfere with aspirin antiplatelet effect • Abciximab (monoclonal antibody), eptifibatide and tirofiban – reversibly inhibit glycoprotein IIb/IIIa • Clopidogrel, prasugrel and ticlopidine (prodrugs) – irreversibly inhibit the platelet ADP receptor 33

ANTIPLATELET DRUGS • Dipyridamole and cilostazol – Inhibit phosphodiesterase enzymes → ↑ c. AMP – Inhibit reuptake of adenosine by endothelial cells and RBCs • Adenosine acts through platelet adenosine A 2 receptors to increase platelet c. AMP 34

Clinical Uses • Aspirin – To prevent first or further MI – To prevent transient ischemic attacks, ischemic stroke, and other thrombotic events 35

Clinical Uses • Glycoprotein IIb/IIIa inhibitors – To prevent restenosis after coronary angioplasty – In acute coronary syndromes (unstable angina and non-Qwave acute MI) • Clopidogrel and ticlopidine – To prevent transient ischemic attacks and ischemic strokes • especially in patients who cannot tolerate aspirin – To prevent thrombosis in patients with coronary artery stent (clopidogrel) 36

Clinical Use • Dipyridamole – To prevent thrombosis in those with cardiac valve replacement (adjunct to warfarin) – For secondary prevention of ischemic stroke (in combination with aspirin) • Cilostazol – To treat intermittent claudication (a manifestation of peripheral arterial disease) 37

Toxicity • Aspirin causes GI and CNS effects • All antiplatelet drugs significantly enhance the effects of other anticlotting agents • Major toxicities of the glycoprotein IIb/IIIa inhibitors: – Bleeding – Thrombocytopenia (in chronic use) 38

Toxicity • Ticlopidine – Bleeding in up to 5% of patients – Severe neutropenia in about 1% – Thrombotic thrombocytopenic purpura (TTP) • a syndrome characterized by the disseminated formation of small thrombi, platelet consumption and thrombocytopenia 39

Toxicity • Clopidogrel is less hematotoxic • Dipyridamole and cilostazol – headaches and palpitations (most common) – Cilostazol; contraindicated in patients with CHF (↓survival) 40

DRUGS USED IN BLEEDING DISORDERS • Causes of Inadequate blood clotting: – Vitamin K deficiency – Genetic defects in clotting factor synthesis (hemophilia) – A variety of drug-induced conditions – Thrombocytopenia 41

Vitamin K • Deficiency of vitamin K in – Older persons with abnormalities of fat absorption (most common) – Newborns – Hospitalized patients • Treatment – Oral or parenteral phytonadione (vitamin K 1) • Caution: dyspnea in fast infusion 42

Clotting Factors • Treatment of hemophilia – Fresh plasma – Factor VIII (for hemophilia A) and factor IX (for hemophilia B) • Purified products • Recombinant products 43

Desmopressin • Vasopressin V 2 receptor agonist – Increases activity of von Willebrand factor VIII • Used to prepare patients with mild hemophilia A or von Willebrand disease for surgery 44
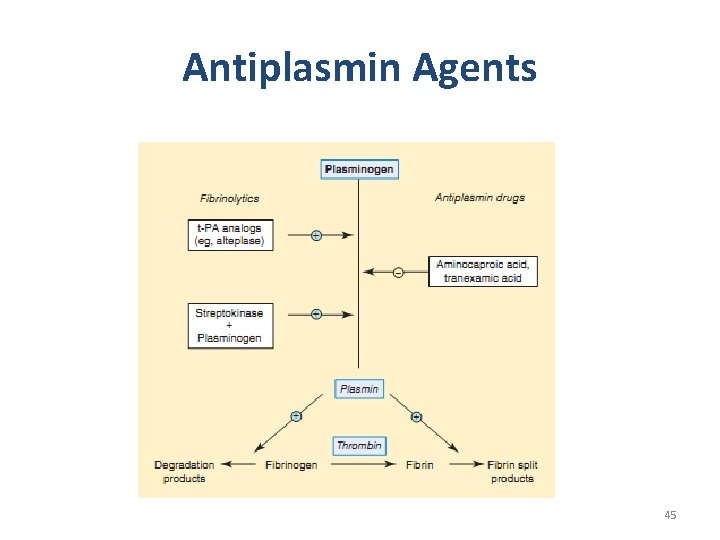
Antiplasmin Agents 45

Antiplasmin Agents • Aminocaproic acid and tranexamic acid – To prevent or manage acute bleeding episodes in patients with hemophilia and others bleeding disorders 46
- Slides: 46