Drugs for Metabolic Disorders Diabetes mellitus BIMM 118
Drugs for Metabolic Disorders • Diabetes mellitus BIMM 118 • Hyperlipidemia
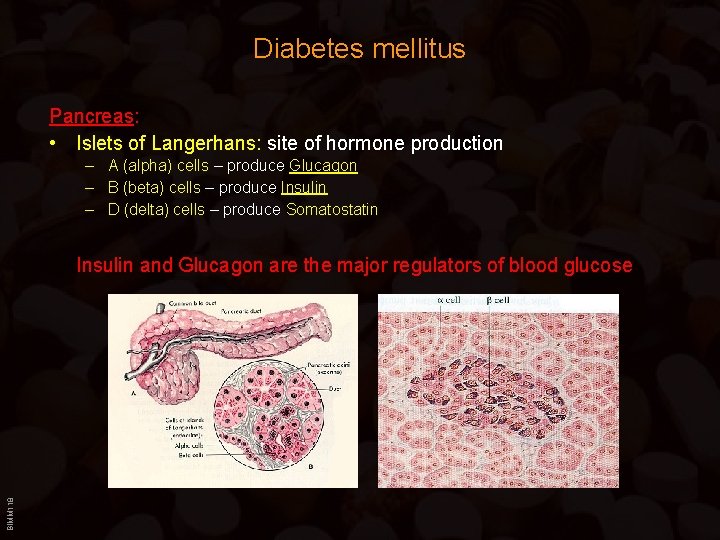
Diabetes mellitus Pancreas: • Islets of Langerhans: site of hormone production – A (alpha) cells – produce Glucagon – B (beta) cells – produce Insulin – D (delta) cells – produce Somatostatin BIMM 118 Insulin and Glucagon are the major regulators of blood glucose
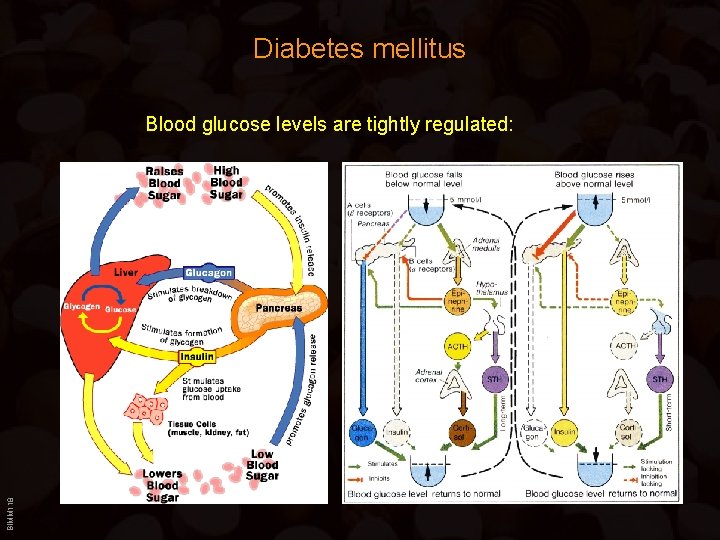
Diabetes mellitus BIMM 118 Blood glucose levels are tightly regulated:
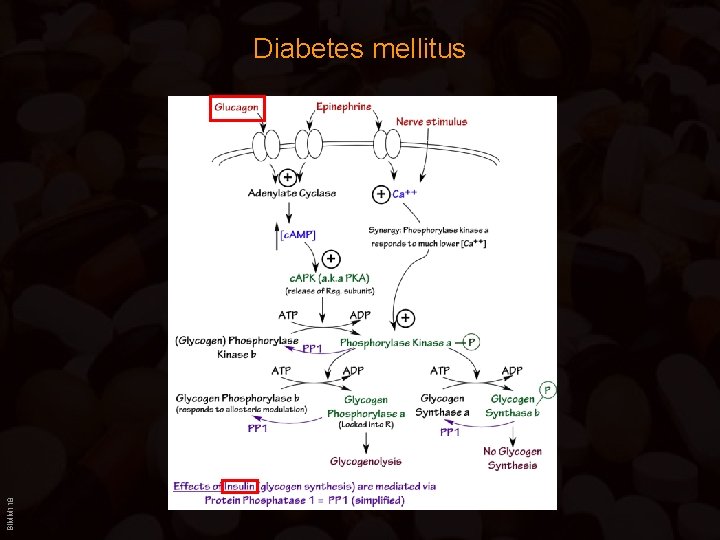
BIMM 118 Diabetes mellitus

Diabetes mellitus Insulin: • • First protein whose sequence was identified (1955) 51 amino acids; synthesized as proinsulin (84 aa) 6 -10 mg stored in the pancreas ~ 2 mg released per day • Liver, brain and red blood cells do not require Insulin for glucose uptake (only muscle and fat cells depend on insulin) Main release stimulus: elevated blood sugar Main effect: promote storage of glucose (increase in glucose uptake (GLUT 4) and glycogen synthesis) Also inhibits lipolysis, and promotes lipogenesis and amino acid uptake • • • BIMM 118 Glucagon: • • 29 amino acids Main release stimulus: hunger (= low blood sugar)

Diabetes mellitus Diabetis mellitus: • Group of metabolic diseases characterized by high blood sugar • Elevated levels of blood glucose (hyperglycemia) lead to spillage of glucose into the urine (diabetes mellitus means “sweet urine”) Two distinct clinical forms: • Type I (= insulin-dependent diabetes = juvenile onset diabetes) – Caused by destruction of the B cells – Generally appears in childhood – Absolutely dependent on insulin replacement • Type II (= insulin-independent diabetes = adult onset diabetes) – Caused by target cell resistance to insulin (Ins. R decreased, signaling defect) – Mostly obese patients (likely genetic predisposition) BIMM 118 – Obesity appears to reduce the number of insulin receptors – Can be treated with oral hypoglycemic drugs
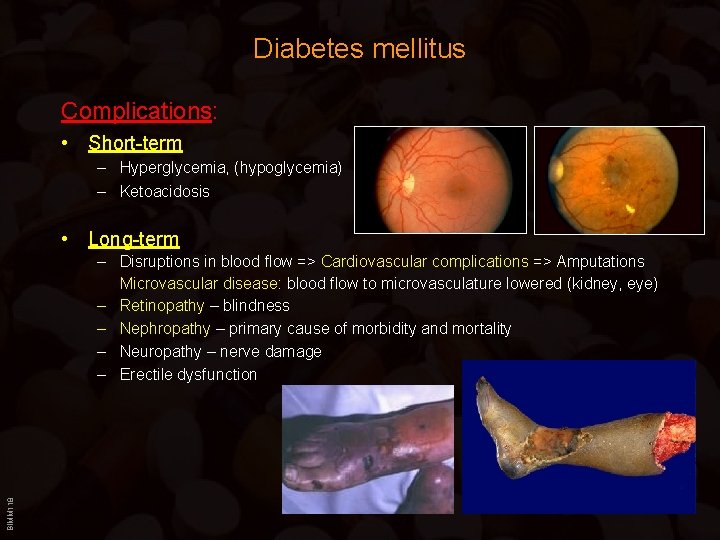
Diabetes mellitus Complications: • Short-term – Hyperglycemia, (hypoglycemia) – Ketoacidosis • Long-term BIMM 118 – Disruptions in blood flow => Cardiovascular complications => Amputations Microvascular disease: blood flow to microvasculature lowered (kidney, eye) – Retinopathy – blindness – Nephropathy – primary cause of morbidity and mortality – Neuropathy – nerve damage – Erectile dysfunction

Diabetes mellitus Insulin: Therapeutic insulin used to be purified from porcine or bovine pancreas => functionally active, but many patients developed an immune response Today, human insulin is produced by recombinant DNA technology Main side effect: Hypoglycemia (requires immediate attention!) “Natural” insulin and four modified insulins are used clinically: • Regular (Natural) Insulin – – BIMM 118 • Unmodified human insulin rapid acting with short duration (half-life 9 min) Only one that can be given IV (infusions, since injections are too brief acting) Useful for emergencies (hyperglycemic coma) Insulin Lispro (Humalog®) – – reversal of the order of the 28 th and 29 th amino acids of the Beta-chain Mutation prevents dimer formation more rapid acting – effects 5 -15 minutes Usually given right before meals
Diabetes mellitus Insulin: Main problem with using natural and rapid acting insulin: wide fluctuations in concentration => Longer lasting formulations: • Insulin Lente – mixed with zinc => forms micro-precipitates => takes longer to absorb => longer acting – Only for s. c. administration – Ultra-lente: longest acting • NPH Insulin BIMM 118 – regular insulin mixed with protamine (large positively charged protein) => delayed absorption – NPH = neutral protamine Hagedorn – Long acting

Diabetes mellitus Insulin: • Insulin Glargine (Lantus®) BIMM 118 – amino acid asparagine at position A 21 is replaced by glycine and two arginines are added to the C-terminus of the B-chain – low aqueous solubility at neutral p. H, but it is completely soluble at p. H 4 (as in the LANTUS injection solution). After injection into the subcutaneous tissue, the acidic solution is neutralized, leading to formation of microprecipitates from which small amounts of insulin glargine are slowly released, resulting in a relatively constant concentration/time profile over 24 hours with no pronounced peak.
Diabetes mellitus Insulin administration: BIMM 118 • • Subcutaneously (oral application impossible due to degradation) Only Regular Insulin can be given IV if needed Jet injectors Pen injectors Implantable insulin pumps Intranasal insulin - mucosal atrophy (abandoned) Pulmonary insulin (inhalation) - in clinical trial

Diabetes mellitus Oral hypoglycemic agents: Useful only in Type II diabetes! Sulfonylureas Stimulate insulin release (increase sensitivity of B cell towards glucose: block ATPgated K+ channel => membrane depolarization => Ca++ increase => insulin secretion), reduce serum glucagon levels, increase insulin binding on target cells First generation sulfonylureas: • • Tolbutamide (t 1/2= 6 -12 h) Chlorpropamide (not used anymore) Tolazamide Acetohexamide Second generation sulfonylureas: • Glimepiride (t 1/2= 18 -24 h), 100 x more BIMM 118 potent than Tolbutamide • • Glipizide Glyburide

Diabetes mellitus Oral hypoglycemic agents: -glitazones (Thiozolidinediones) Increase insulin sensitivity of target cells: function as PPARg agonists => promote transcription of insulin receptor signaling components and of glucose transporters Main side effect: hypoglycemia • Troglitazone – First of its class – Hepatotoxic! – No longer in use • • Rosiglitazone Pioglitazone BIMM 118 – Half-life ~ 7 hrs – Half-life of active metabolites up to 150 hrs !

Diabetes mellitus Oral hypoglycemic agents: Biguanides • Metformine BIMM 118 – – – – Only drug in this class in use Increase glucose uptake and inhibit gluconeogenesis in the liver Mechanism unclear (AMPK? ) Also lowers LDL and VLDL Adverse side effects: Diarrhea, nausea Benefitial side effect: appetite suppressant! Does not cause hypoglycemia Not for patients with liver or kidney disease (predisposition to lactic acidosis)

Diabetes mellitus Novel concepts: Alpha-Glucosidases: – – Intestinal enzymes in the small intestine Break down complex carbohydrates (Starch, Glygogen) Alpha-Glucosidase Inhibitors: – – • Acarbose – – • Also inhibits alpha-amylases No significant absorption => no systemic side effects Used to prevent postprandial hyperglycemia Side effects: Diarrhea, flatulence (intestinal bacteria digest the carbohydrates!) Miglitol – – BIMM 118 Inhibit carbohydrate breakdown => less monosaccharides available for absorption Saccharides that act as competitive enzyme inhibitors DO NOT increase insulin levels !! Maybe useful in Type I diabetes as well? Systemically absorbed No effect on alpha-amylases
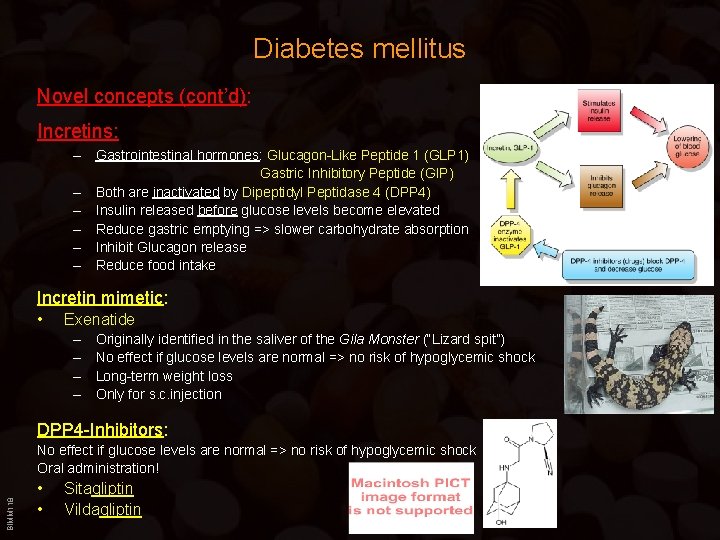
Diabetes mellitus Novel concepts (cont’d): Incretins: – Gastrointestinal hormones: Glucagon-Like Peptide 1 (GLP 1) Gastric Inhibitory Peptide (GIP) – Both are inactivated by Dipeptidyl Peptidase 4 (DPP 4) – Insulin released before glucose levels become elevated – Reduce gastric emptying => slower carbohydrate absorption – Inhibit Glucagon release – Reduce food intake Incretin mimetic: • Exenatide – – Originally identified in the saliver of the Gila Monster (“Lizard spit”) No effect if glucose levels are normal => no risk of hypoglycemic shock Long-term weight loss Only for s. c. injection DPP 4 -Inhibitors: BIMM 118 No effect if glucose levels are normal => no risk of hypoglycemic shock Oral administration! • • Sitagliptin Vildagliptin

Diabetes mellitus Novel concepts (cont’d): Amylin: – – Pancreatic hormone (also from b-Islet cells) Reduces gastric emptying Inhibit Glucagon release Promotes satiety (=> decreased food intake) Amylin mimetics: • Pramlintide BIMM 118 – Only drug other than insulin approved for Type I Diabetes !! – Used in combination with insulin
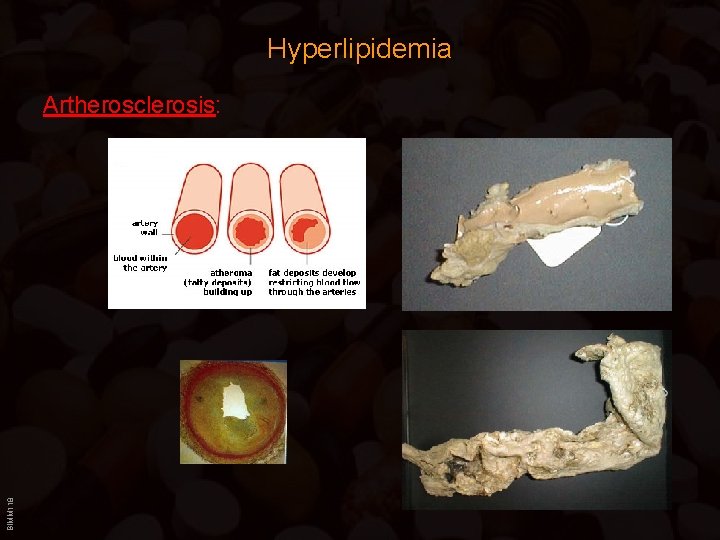
Hyperlipidemia BIMM 118 Artherosclerosis:
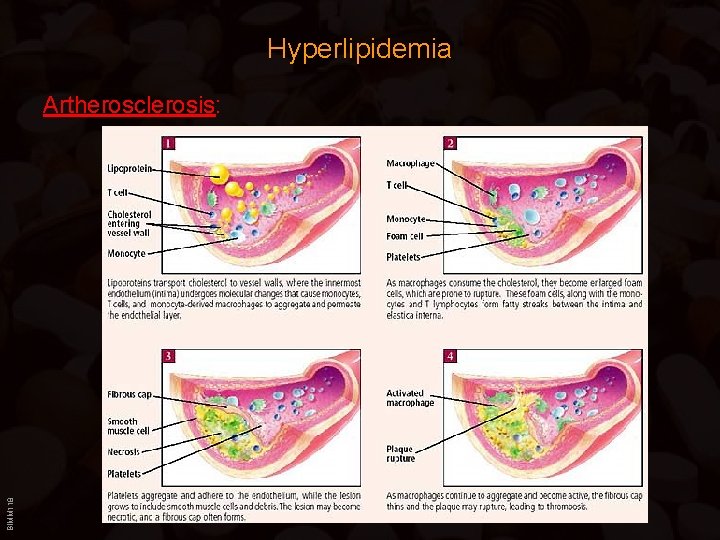
Hyperlipidemia BIMM 118 Artherosclerosis:
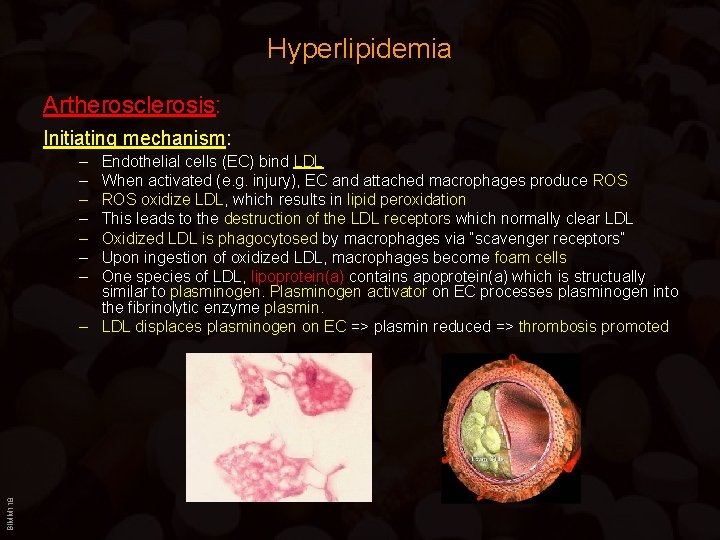
Hyperlipidemia Artherosclerosis: Initiating mechanism: – – – – BIMM 118 Endothelial cells (EC) bind LDL When activated (e. g. injury), EC and attached macrophages produce ROS oxidize LDL, which results in lipid peroxidation This leads to the destruction of the LDL receptors which normally clear LDL Oxidized LDL is phagocytosed by macrophages via “scavenger receptors” Upon ingestion of oxidized LDL, macrophages become foam cells One species of LDL, lipoprotein(a) contains apoprotein(a) which is structually similar to plasminogen. Plasminogen activator on EC processes plasminogen into the fibrinolytic enzyme plasmin. – LDL displaces plasminogen on EC => plasmin reduced => thrombosis promoted
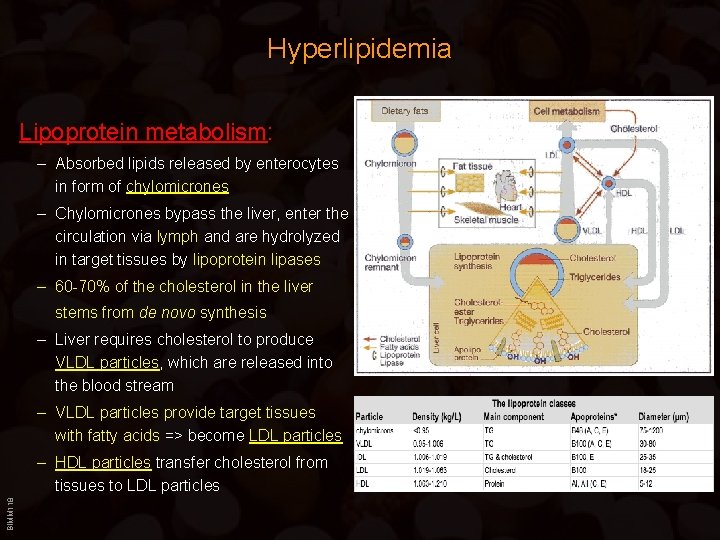
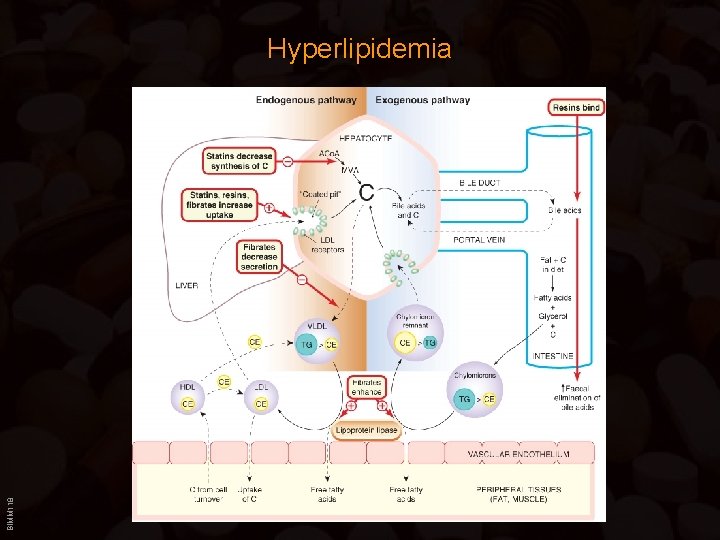
Hyperlipidemia Lipoprotein metabolism: – Absorbed lipids released by enterocytes in form of chylomicrones – Chylomicrones bypass the liver, enter the circulation via lymph and are hydrolyzed in target tissues by lipoprotein lipases – 60 -70% of the cholesterol in the liver stems from de novo synthesis – Liver requires cholesterol to produce VLDL particles, which are released into the blood stream – VLDL particles provide target tissues with fatty acids => become LDL particles BIMM 118 – HDL particles transfer cholesterol from tissues to LDL particles
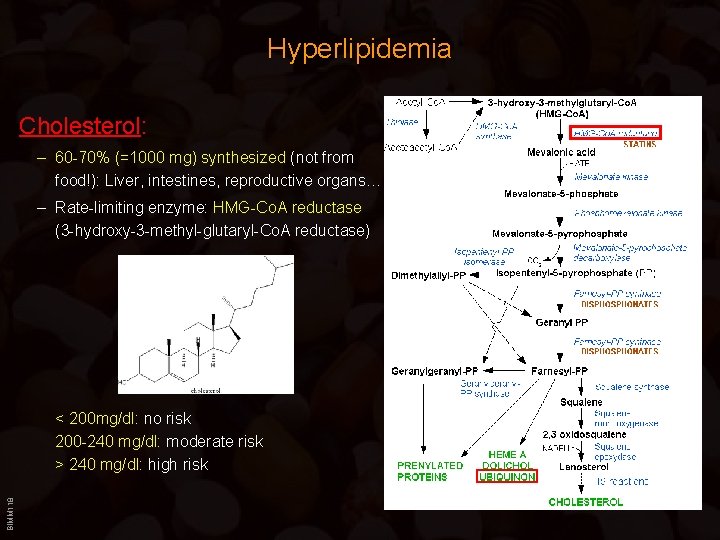
Hyperlipidemia Cholesterol: – 60 -70% (=1000 mg) synthesized (not from food!): Liver, intestines, reproductive organs… – Rate-limiting enzyme: HMG-Co. A reductase (3 -hydroxy-3 -methyl-glutaryl-Co. A reductase) BIMM 118 < 200 mg/dl: no risk 200 -240 mg/dl: moderate risk > 240 mg/dl: high risk
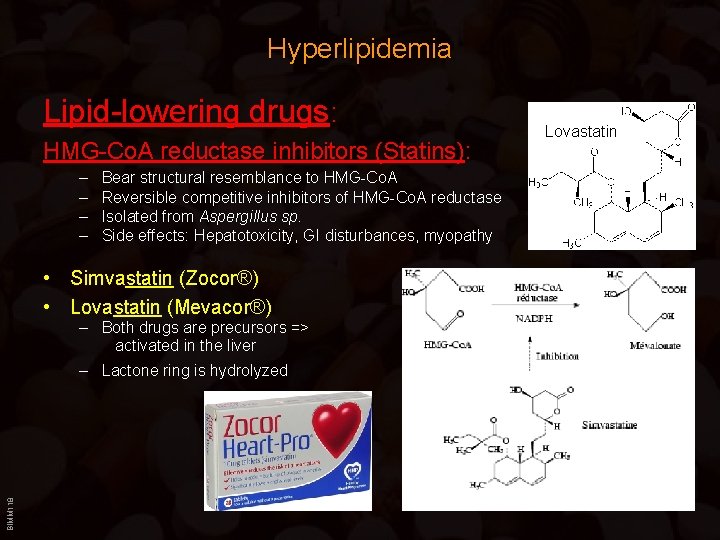
Hyperlipidemia Lipid-lowering drugs: HMG-Co. A reductase inhibitors (Statins): – – Bear structural resemblance to HMG-Co. A Reversible competitive inhibitors of HMG-Co. A reductase Isolated from Aspergillus sp. Side effects: Hepatotoxicity, GI disturbances, myopathy • Simvastatin (Zocor®) • Lovastatin (Mevacor®) – Both drugs are precursors => activated in the liver BIMM 118 – Lactone ring is hydrolyzed Lovastatin
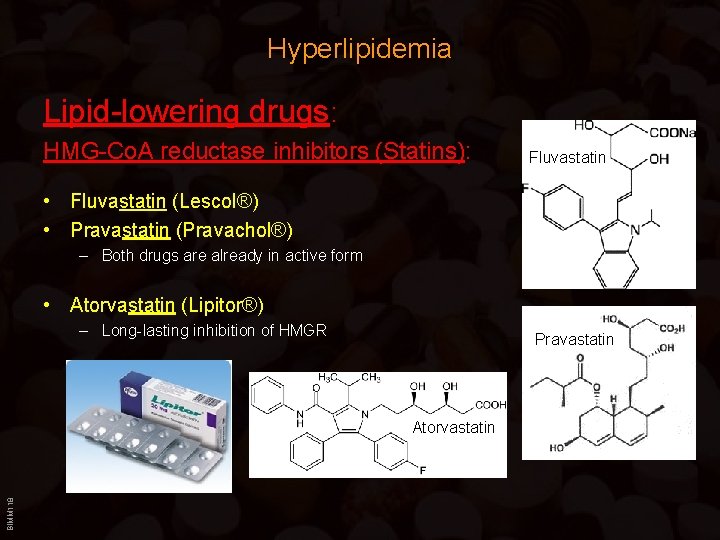
Hyperlipidemia Lipid-lowering drugs: HMG-Co. A reductase inhibitors (Statins): Fluvastatin • Fluvastatin (Lescol®) • Pravastatin (Pravachol®) – Both drugs are already in active form • Atorvastatin (Lipitor®) – Long-lasting inhibition of HMGR Pravastatin BIMM 118 Atorvastatin

Hyperlipidemia Lipid-lowering drugs: HMG-Co. A reductase inhibitors (Statins): • Statins accumulate in the liver (usually an undesired drug effect) • Cholesterol synthesis is predominantly effected in the liver => hepatocytes must meet their cholesterol requirements through different mechanisms => • Hepatic upregulation of the LDL-receptors => increase in LDL BIMM 118 uptake => decrease in circulating LDL
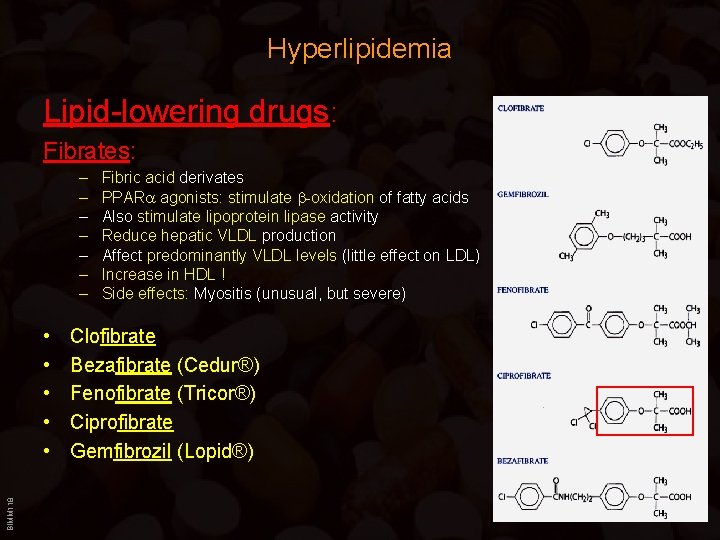
Hyperlipidemia Lipid-lowering drugs: Fibrates: – – – – BIMM 118 • • • Fibric acid derivates PPARa agonists: stimulate b-oxidation of fatty acids Also stimulate lipoprotein lipase activity Reduce hepatic VLDL production Affect predominantly VLDL levels (little effect on LDL) Increase in HDL ! Side effects: Myositis (unusual, but severe) Clofibrate Bezafibrate (Cedur®) Fenofibrate (Tricor®) Ciprofibrate Gemfibrozil (Lopid®)

Hyperlipidemia Lipid-lowering drugs: Bile acid binding resins: – Anion exchange resins – Prevent reabsorption and enterohepatic recirculation of bile acids => increase in hepatic LDL receptors => increase in hepatic LDL uptake => Reduced LDL in the plasma – Side effects: resins are not absorbed => no systemic side effects mostly bloating, constipation, diarrhea – Interfer with absorption of drugs (e. g. digoxin) and fat-soluble vitamins – Not particularly appetizing BIMM 118 • Cholestyramine • Colestipol
BIMM 118 Hyperlipidemia
- Slides: 28