DRUGENZYME INTERACTION Course Drug Design Course code 0510412
















![Inhibitors [I] are substances that combine with Enzymes [E] and decrease its activity. Presence Inhibitors [I] are substances that combine with Enzymes [E] and decrease its activity. Presence](https://slidetodoc.com/presentation_image_h2/45e6a579eaeb69be0f4a81f30de28b09/image-28.jpg)





- Slides: 39

DRUG-ENZYME INTERACTION Course: Drug Design Course code: 0510412 Dr. Balakumar Chandrasekaran Dr. Bilal Al-Jaidi Assistant Professors, Faculty of Pharmacy, Philadelphia University-Jordan

Learning Outcomes At the end of this lesson students will be able to Ø Explain the enzyme, active-site of the enzyme, enzyme catalysis, and co-enzymes. Ø Define and describe the binding specificity. Ø Explain various enzymatic reactions, rate acceleration, and enzyme turn over. Ø Describe the enzyme inhibition mechanism and drug synergism.

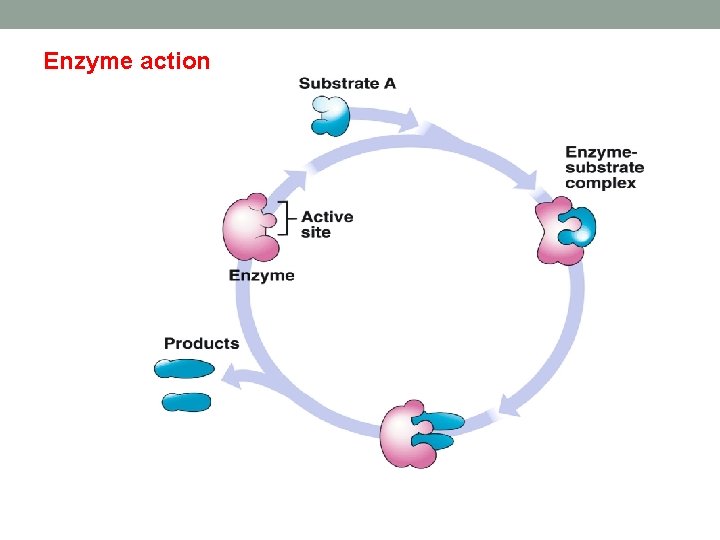
Introduction q. Enzymes are soluble proteins, floating in interstitial or extrastitial fluids. For example, in cell cytosol and in blood. q. Enzyme catalyses a chemical reaction on the substrate structure. q. Enzymes are produced by different cells of humans, animals, plants and microorganisms, which accelerate the rate of chemical reactions in living organisms. q. Substrate will enter enzyme active site then converted into metabolite. q. Ligand binds to receptor to activate, then leave without any change in its structure.

Enzyme activates reaction by: • Lowering transition state and intermediate energy level. • Raising the ground state energy of substrate. • This means substrate will be energetically unstable.

Enzyme action

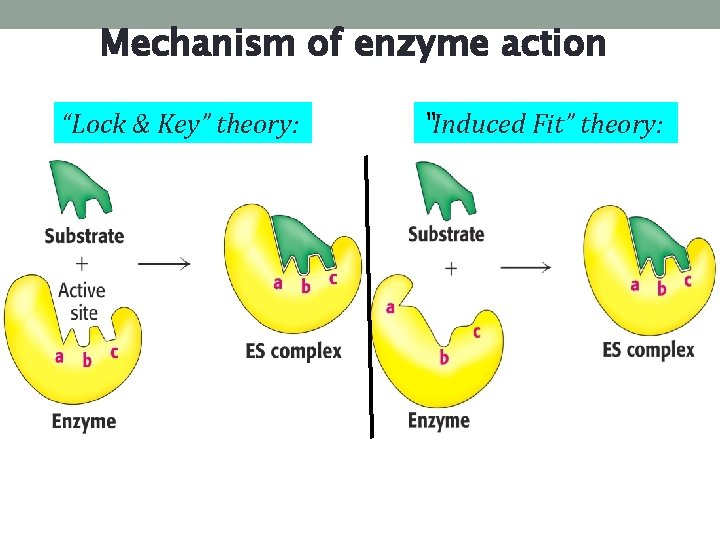
Mechanism of enzyme action “Lock & Key” theory: “Induced Fit” theory:

Enzyme Active site • Substrate binds to small area in enzyme structure known as active site: • Other names: Binding site, Binding domain, Active pocket

Enzyme Active site • Two types of amino acids available inside enzyme active- site: • Catalytic amino acids: Directly or indirectly participate in enzyme-substrate interactions. • Non-catalytic amino acids: • Complete the construction of active site pocket • Help shaping tunnels and opening to active site, especially when the pocket is deep inside. • Might play role in binding (anchoring) substrate to bring it close to catalytic amino acids

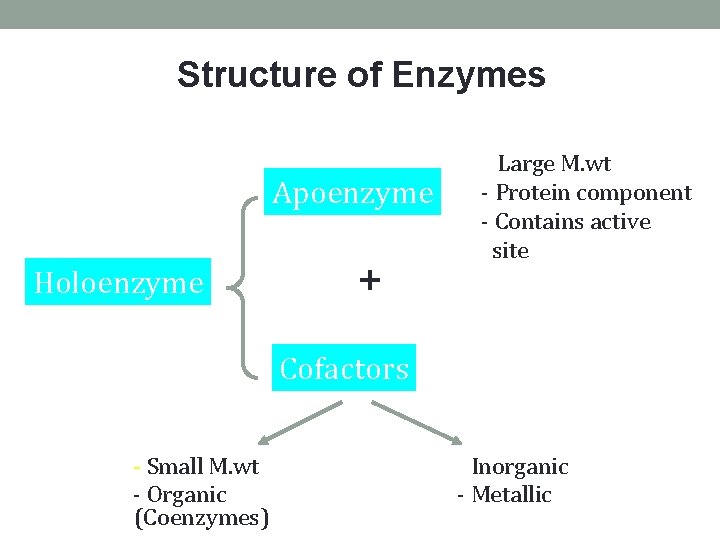
Structure of Enzymes Apoenzyme Holoenzyme + - Large M. wt - Protein component - Contains active site Cofactors - Small M. wt - Inorganic - Organic (Coenzymes) - Metallic

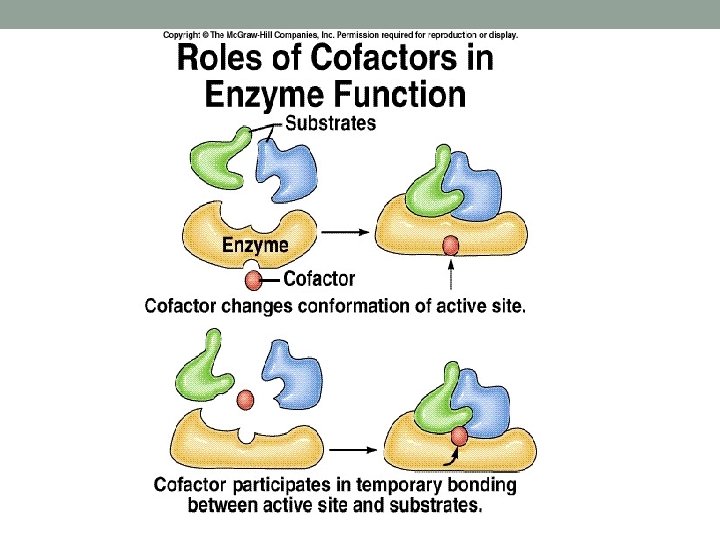
Enzyme Catalysis • Is characterized by: • Specificity • Rate acceleration • Reaction specificity: can be classified as: • Reaction specificity • Substrate specificity • Rate acceleration is due to the presence of co-enzymes in most of the cases.

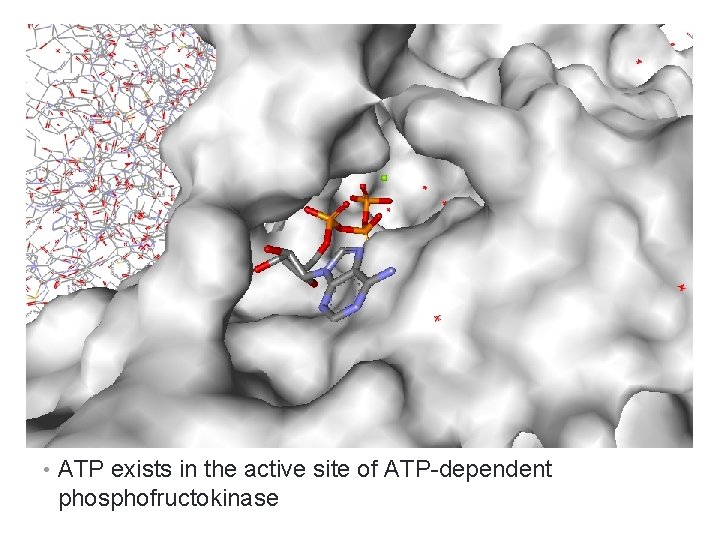
Co-Enzymes • Are any organic molecules or metal ions that are essential for catalytic action of enzyme. • Examples: Ø ATP Ø Coenzyme A Ø Glutathione Ø Ascorbic acid Ø Lipoic acid Ø Zn, Co, Fe, Mg cations


• ATP exists in the active site of ATP-dependent phosphofructokinase

Substrate specificity • Substrate specificity can be categorized into: • Very specific: one type of substrate can fit the active site Examples: carboxyesterase, COMT, Acetylcholinesterase • Broad: more than one substrate can bind and converted into metabolite. Cytochrome P 450 is an example • Enzymes are substrate specific because: • They have distinguished active site compared to others • Different amino acids in active site play a major role in specificity.

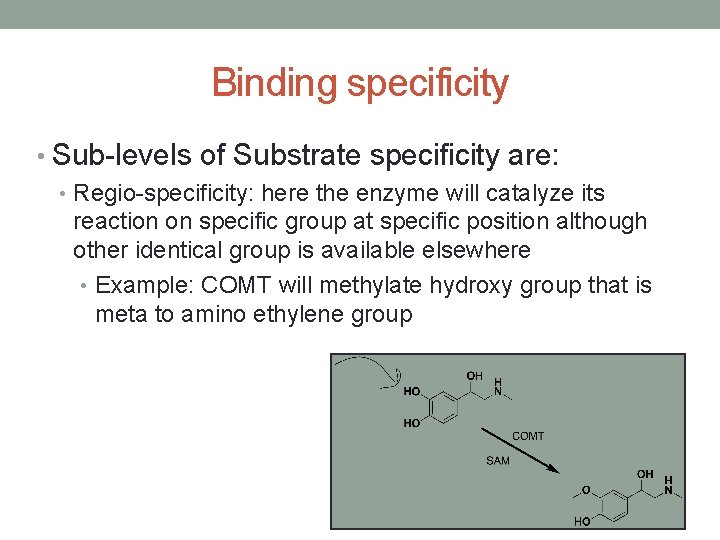
Binding specificity • Sub-levels of Substrate specificity are: • Regio-specificity: here the enzyme will catalyze its reaction on specific group at specific position although other identical group is available elsewhere • Example: COMT will methylate hydroxy group that is meta to amino ethylene group

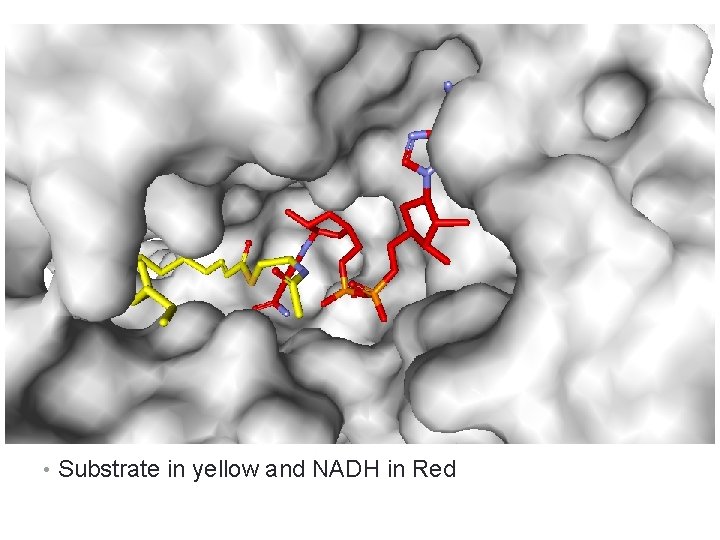
Binding specificity • Determinant factors that make enzymes Regio- specific: • Which group is the closest to the catalytic amino acids • Which group is the nearest to the site where Co- enzyme lies: • ATP for phosphorylation • NADH/NAD+ for redox reaction • SAM for methylation

• Substrate in yellow and NADH in Red

Binding specificity • Sub-levels of Substrate specificity are: • Stereo-specificity: here the enzyme will catalyze its reaction on one of substrate isomers or enantiomer more than the other • This is why some drug will be active only when they are in its pure S or R isomer form • Examples: • R isomer of Adrenaline is much more active than S • R isomer of Salbutamol is much more active than S • S-methacoline is more active than R-methacoline • S-ibuprofen is more active than R-ibuprofen


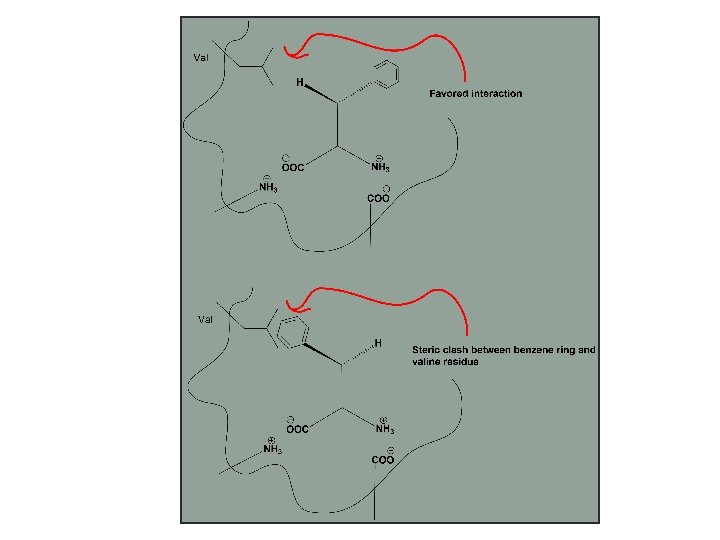
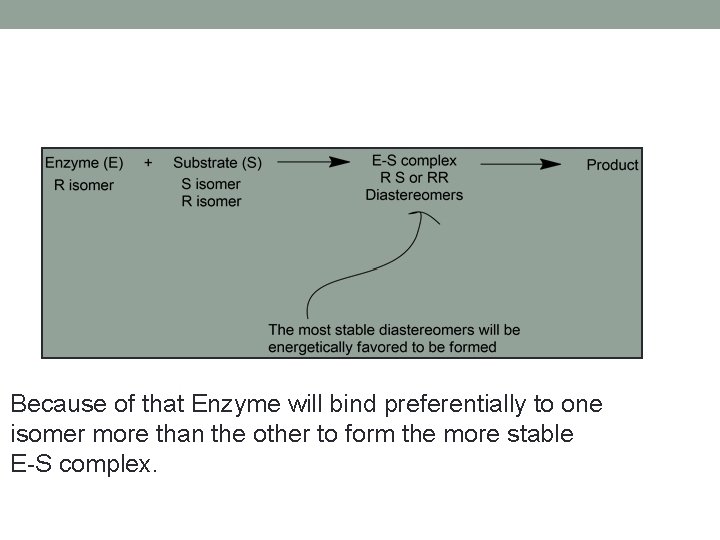
Why is Enzyme specific? • Because enzyme is chiral molecule, only L- amino acids are available and amino acids are all chiral except Glycine (Why) • Chiral compounds will bind to form diastereomers (E-S complex), that have different physicochemical properties…. The most stable diastereomer will be energetically favored to form. • Also amino acid residues available in active site might play a role in this specificity

Because of that Enzyme will bind preferentially to one isomer more than the other to form the more stable E-S complex.

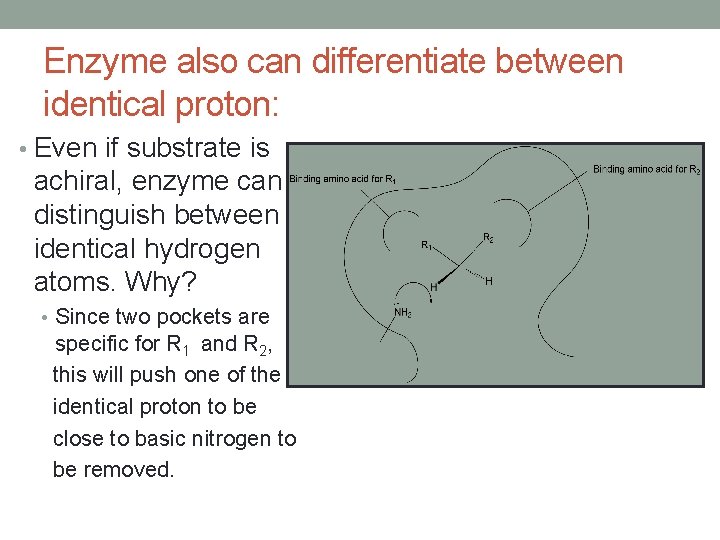
Enzyme also can differentiate between identical proton: • Even if substrate is achiral, enzyme can distinguish between identical hydrogen atoms. Why? • Since two pockets are specific for R 1 and R 2, this will push one of the identical proton to be close to basic nitrogen to be removed.

Rate acceleration • Enzyme accelerate reactions by: • Stabilizing the reaction transition state…. This will lower the activation energy. • By destabilizing drug molecule. • Bring drug close to the cofactor that normally carries reactive species such as phosphate, hydride, oxygen, … • Reaction rate could reach 1010 – 1014 the rate of non- enzymatic reactions.

Enzyme turnover • Is the number of molecules of substrate converted to product per unit time per molecule of enzyme active site. • Some enzyme have multiple subunits and each subunit might have more than one active site. • As the number of subunits increased, turnover number increased.

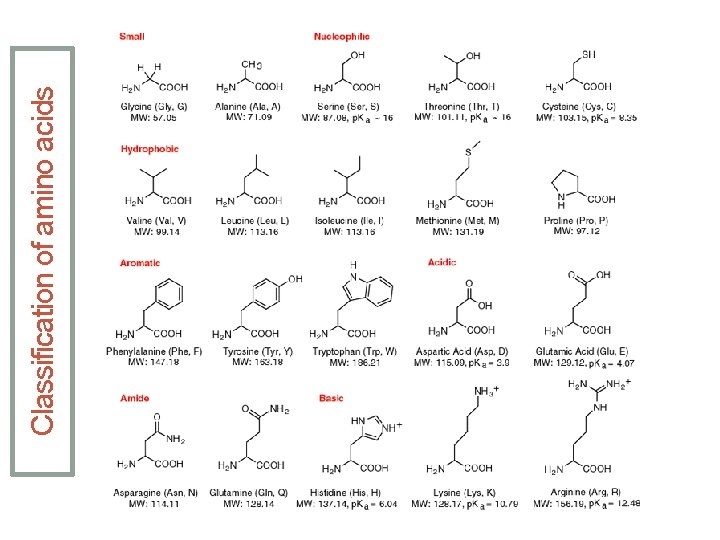
Classification of amino acids

Enzyme inhibition • Enzyme inhibitor: will slow down or block enzyme catalysis • Mainly irreversible ……. Enzyme inactivation • The consequences of enzyme inhibition depends on the function of the enzyme • Examples: • GABA aminotransferase deactivates GABA…. Inhibition of this enzyme will accumulate GABA …. Anticonvulsant action • Xanthine will be oxidized into uric acid…. Inhibition of this enzyme will help in Gout patients

Enzyme inhibition • Enzyme inhibitors in contrast to receptor antagonists, are closely similar inn structure to enzyme natural substrate……. It should be strongly bind to enzyme active site…. inhibiting the binding of substrate. • Ideal Enzyme inhibitor should: • be specific for one target enzyme • target essential enzymes in essential metabolic pathway • Selectively inhibit essential enzymes in non human cells that are unique, does not exist in human cells.
![Inhibitors I are substances that combine with Enzymes E and decrease its activity Presence Inhibitors [I] are substances that combine with Enzymes [E] and decrease its activity. Presence](https://slidetodoc.com/presentation_image_h2/45e6a579eaeb69be0f4a81f30de28b09/image-28.jpg)
Inhibitors [I] are substances that combine with Enzymes [E] and decrease its activity. Presence of [I] decreases the rate of Enzyme catalyzed reaction. Enzymes are subject to reversible or irreversible inhibition. Irreversible inhibitors usually cause stable, covalent alterations in the enzyme. The consequence of irreversible inhibition is a decrease in the concentration of active enzyme.

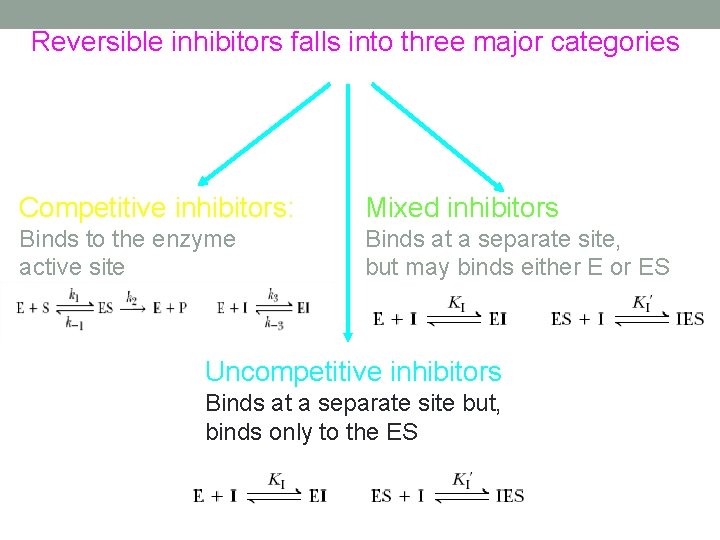
Reversible inhibitors falls into three major categories Competitive inhibitors: Mixed inhibitors Binds to the enzyme active site Binds at a separate site, but may binds either E or ES Uncompetitive inhibitors Binds at a separate site but, binds only to the ES

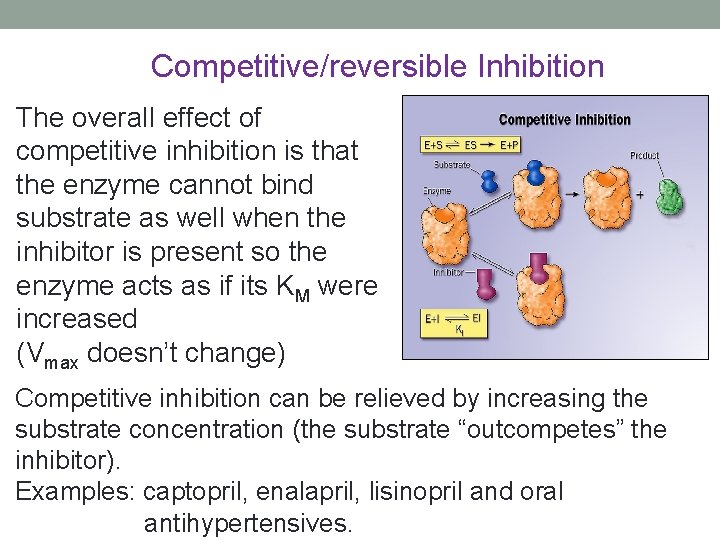
Competitive/reversible Inhibition The overall effect of competitive inhibition is that the enzyme cannot bind substrate as well when the inhibitor is present so the enzyme acts as if its KM were increased (Vmax doesn’t change) Competitive inhibition can be relieved by increasing the substrate concentration (the substrate “outcompetes” the inhibitor). Examples: captopril, enalapril, lisinopril and oral antihypertensives.

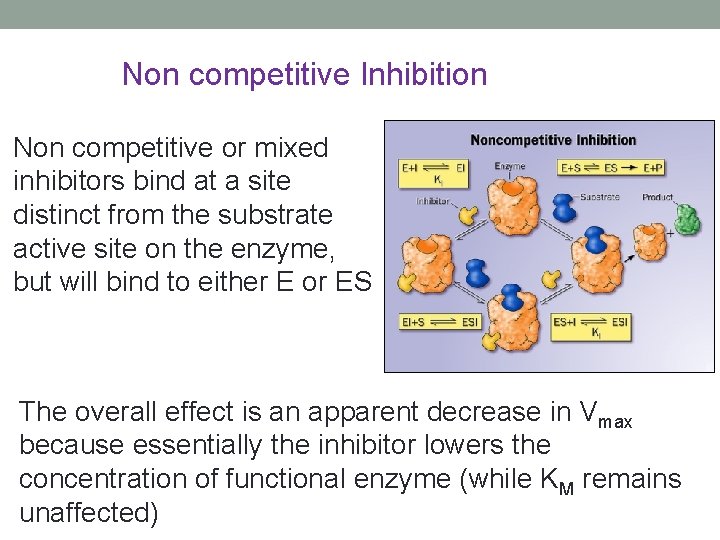
Non competitive Inhibition Non competitive or mixed inhibitors bind at a site distinct from the substrate active site on the enzyme, but will bind to either E or ES The overall effect is an apparent decrease in Vmax because essentially the inhibitor lowers the concentration of functional enzyme (while KM remains unaffected)

Uncompetitive inhibitors Binds at a sight distinct from the substrate but only in the ES complex (non competitive inhibitor binds to either free enzyme or the ES complex

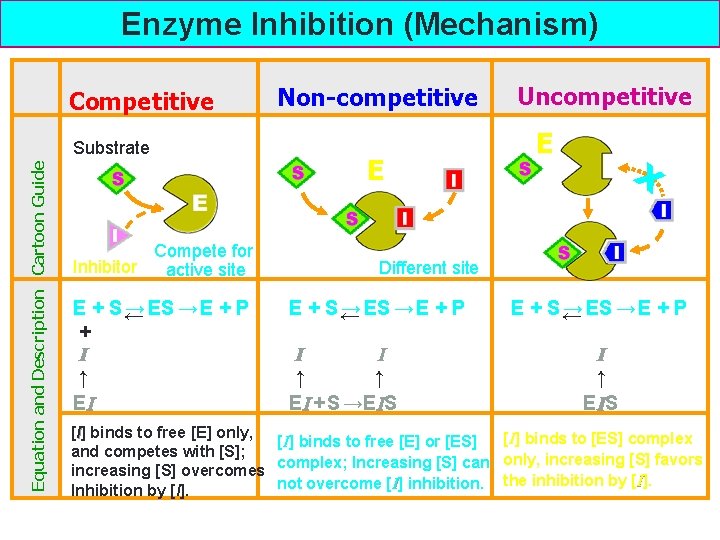
Enzyme Inhibition (Mechanism) Competitive Equation and Description Cartoon Guide Substrate Compete for Inhibitor active site E + S← → ES → E + P + I ↓↑ EI Non-competitive E Uncompetitive E X Different site E + S← → ES → E + P + + I I ↓↑ ↓↑ EI + S →EIS E + S← → ES → E + P + I ↓↑ E IS [I] binds to free [E] only, [I] binds to [ES] complex [I] binds to free [E] or [ES] and competes with [S]; complex; Increasing [S] can only, increasing [S] favors increasing [S] overcomes not overcome [I] inhibition. the inhibition by [I]. Inhibition by [I].

Irreversible inhibition Irreversible inhibitors/ enzyme inactivators: Covalently combine with or destroy a functional group on an enzyme that is essential for the enzymes activity, or that form a particularly stable non covalent association that does the same thing Most irreversible inhibitors are toxic substances, either natural (cyanide, penicillin) or synthetic (parathion – an insecticide).

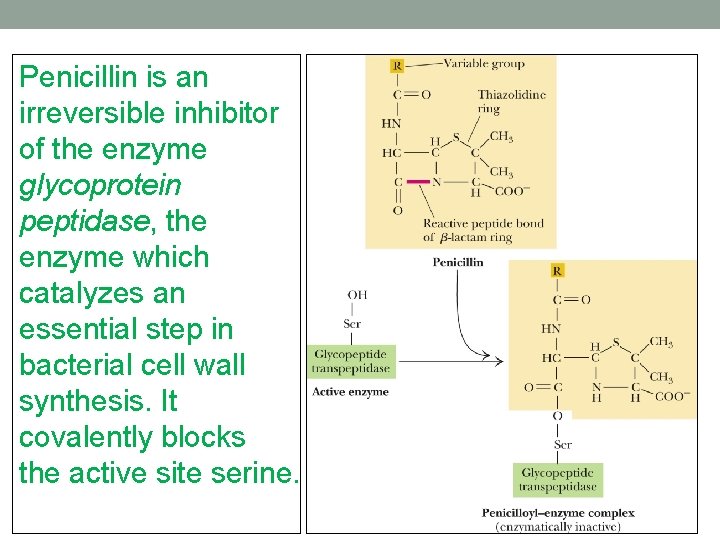
Penicillin is an irreversible inhibitor of the enzyme glycoprotein peptidase, the enzyme which catalyzes an essential step in bacterial cell wall synthesis. It covalently blocks the active site serine.

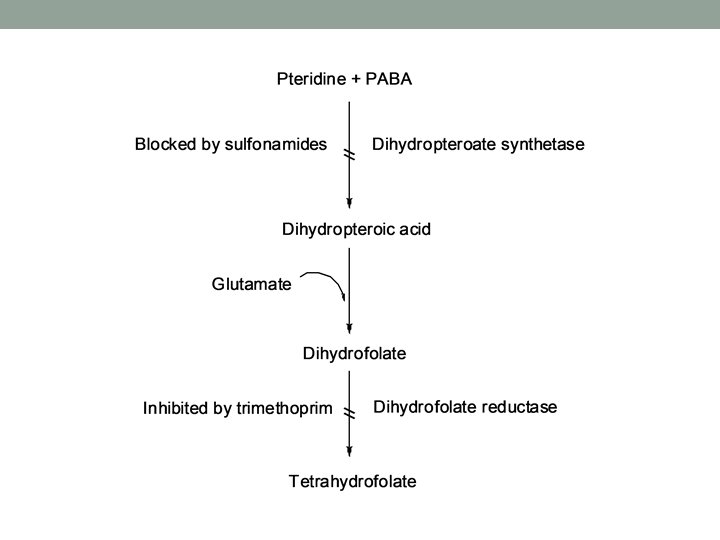
Drug synergism • When two drugs are given in combination, therapeutic effect of them is greater than the sum of their effects if they are given individually. • Mechanisms of enzyme synergism: • inhibition of drug destroying enzymes: Example: calvulanic acid with amoxicillin • Sequential blocking: • Inhibiting two or more consecutive steps or enzymes in the same metabolic pathway • Example: Trimethorprim with sulfamethoxazole


Recommended Books 1. The organic chemistry of drug design by Richard B. Silverman. Second edition, Elsevier, 2004. 2. An introduction to Medicinal Chemistry by Graham L. Patrick. Fourth edition, Oxford, 2009. 38
