Drugcoated Balloons and Stents in the Periphery Is

Drug-coated Balloons and Stents in the Periphery: Is There a Future? Ron Waksman, MD Professor of Medicine (Cardiology) Georgetown University, Associate Director Division of Cardiology Washington Hospital Center, Washington DC

Presenter Disclosure n n n Consultant : Biotronik, Medtronic, Boston Scientific. Abbott Speaker Biotronik BSC, Medtronic, Abbott Vascular, Astra Zeneca Research Grants: Biotronik, Medtronic, Boston Scientific Abbott Vascular, Lilly Dahaichi.

Scope of the Problem The Main Issues for intervention in the periphery is restenosis: for balloon recoil and intimal hyperplasia for stents stent fracture and intimal hyperplasia BARE STENT PATENCY Intra. Coil: 86% @ 12 Months; 85% @ 18 Months 7 -4. 5 cm lesion length Wallstent: 29 -68% @ 12 Months Symphony: 76% @ 3 Years 3. 2 cm lesion length Smart: 76% @ 12 Months; 66% @ 18 Months; 60% @ 24 Months. 12. 2 cm lesion length; 57% > 10 cm; f/u 302 days
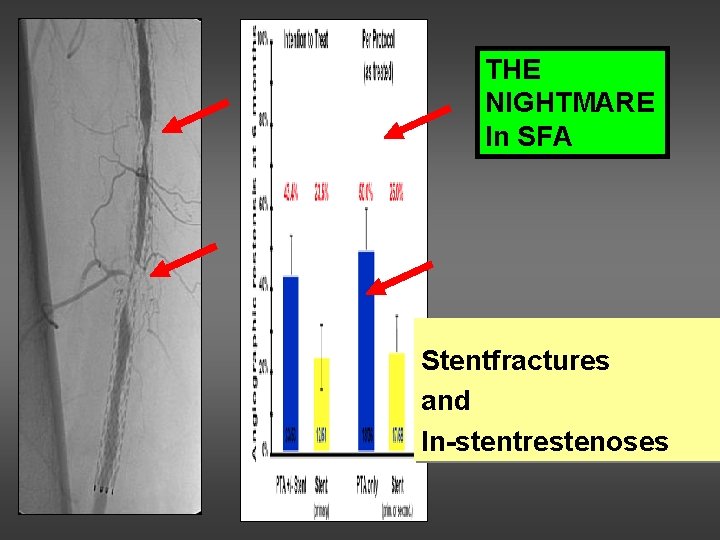
THE NIGHTMARE In SFA Stentfractures and In-stentrestenoses
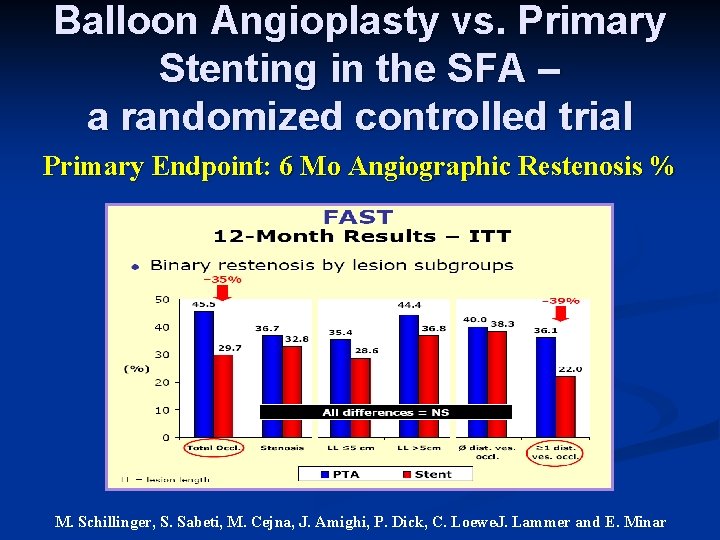
Balloon Angioplasty vs. Primary Stenting in the SFA – a randomized controlled trial Primary Endpoint: 6 Mo Angiographic Restenosis % M. Schillinger, S. Sabeti, M. Cejna, J. Amighi, P. Dick, C. Loewe. J. Lammer and E. Minar
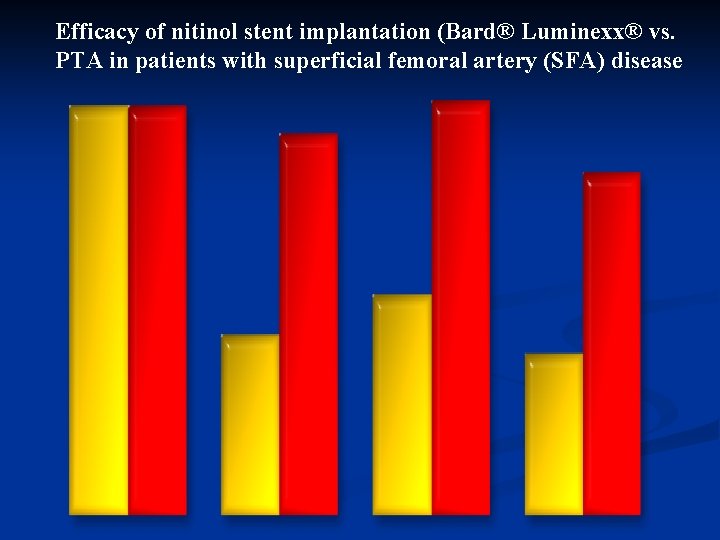
Efficacy of nitinol stent implantation (Bard® Luminexx® vs. PTA in patients with superficial femoral artery (SFA) disease
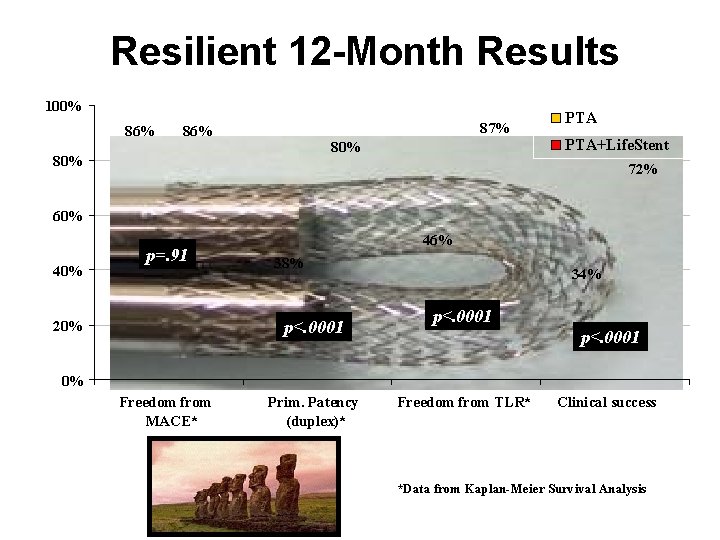
Resilient 12 -Month Results 100% 86% 87% 86% 80% PTA+Life. Stent 72% 60% 40% p=. 91 46% 38% p<. 0001 20% 34% p<. 0001 0% Freedom from MACE* Prim. Patency (duplex)* Freedom from TLR* Clinical success *Data from Kaplan-Meier Survival Analysis

DES in the SFA SIROCCO I & II Trials • blinded, randomized, BMS versus DES (Sirolimus) • 36 patients (Sirocco I) + 57 patients (Sirocco II) • primary endpoint: Restenosis in stent after 6 months Duda et al. : Circulation. 2002; 106: 1505 -1509 Tepe et al: J Cardiovasc Surg. 2005; 46: 249 -259 Duda et al: J Endovasc Ther. 2006; 13: 701 -710
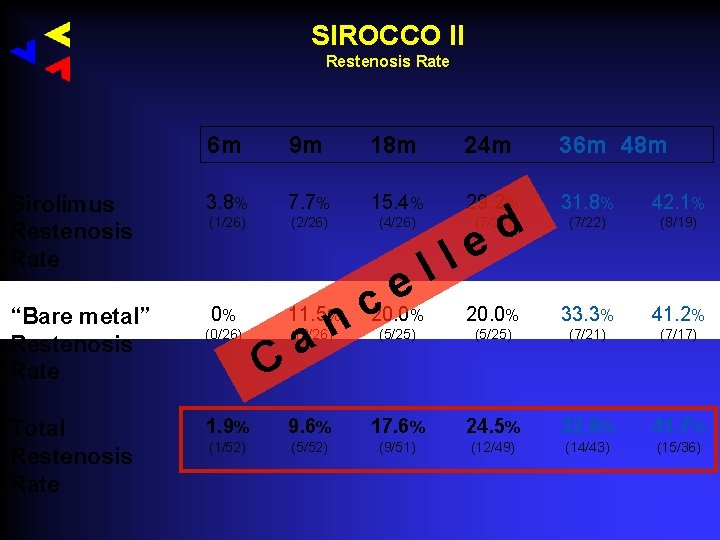
SIROCCO II Restenosis Rate 6 m 9 m 18 m 24 m 36 m 48 m Sirolimus Restenosis Rate 3. 8% 7. 7% 15. 4% 29. 2% 31. 8% 42. 1% (1/26) (2/26) (4/26) (7/24) (7/22) (8/19) “Bare metal” Restenosis Rate 0% 20. 0% 33. 3% 41. 2% (5/25) (7/21) (7/17) Total Restenosis Rate (0/26) c 20. 0 n 11. 5% a C l l e (3/26) % d e 1. 9% 9. 6% 17. 6% 24. 5% 32. 6% 41. 7% (1/52) (5/52) (9/51) (12/49) (14/43) (15/36)

More Drug Eluting Stent Trials Strides Trial(Abbott Vascular) a. Everolimus coating b. Study: 104 patients Clinical 30 d 6 mo Duplex Ultrasound Angiography 12 mo 18 mo 2 yr 3 yr 4 yr 5 yr
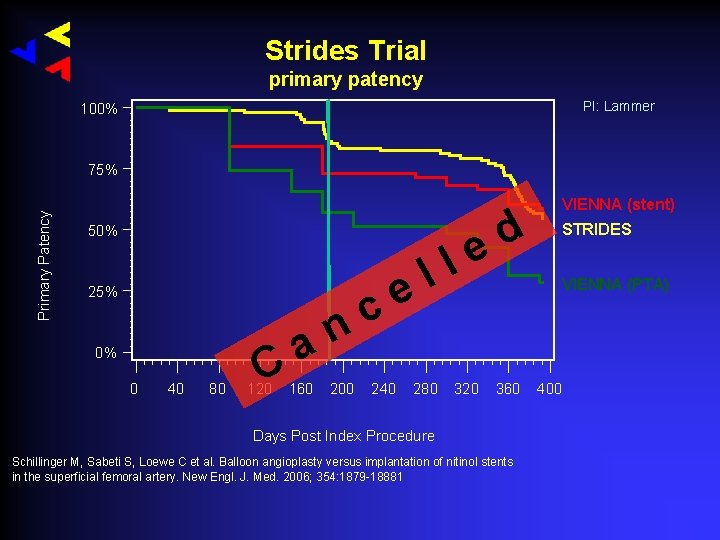
Strides Trial primary patency PI: Lammer 100% Primary Patency 75% 50% 25% 0% 0 40 80 a C 120 160 c n 200 240 l l e 280 d e 320 VIENNA (stent) STRIDES VIENNA (PTA) 360 Days Post Index Procedure Schillinger M, Sabeti S, Loewe C et al. Balloon angioplasty versus implantation of nitinol stents in the superficial femoral artery. New Engl. J. Med. 2006; 354: 1879 -18881 400

Zilver PTX® Drug-Eluting Stent • Designed for the SFA • CE Marked – Investigational in the US and Japan • Dual therapy stent – Mechanical support: Zilver Flex® Stent Platform – Drug coating: Paclitaxel only • No polymer or binder • 3 µg/mm 2 dose density PTX Coated • Sponsor: Cook Medical Uncoated

Zilver PTX® Randomized Trial • 12 -month event-free survival – Primary safety endpoint – Per patient freedom from death, amputation, target lesion revascularization, or worsening Rutherford score (by 2 classes or to class 5 or 6) • 12 -month primary patency – Primary effectiveness endpoint – Per lesion patency by duplex ultrasonography, patent = PSVR < 2. 0 (or angiography if available, patent = diameter stenosis < 50%) – One lesion per limb, bilateral treatment allowed • 5 year ongoing follow-up – 2, 3, 4, and 5 year patency evaluations for all stent patients and a randomly selected subset of patients with acutely successful PTA – 3 and 5 year stent radiographs

Safety Event-free Survival
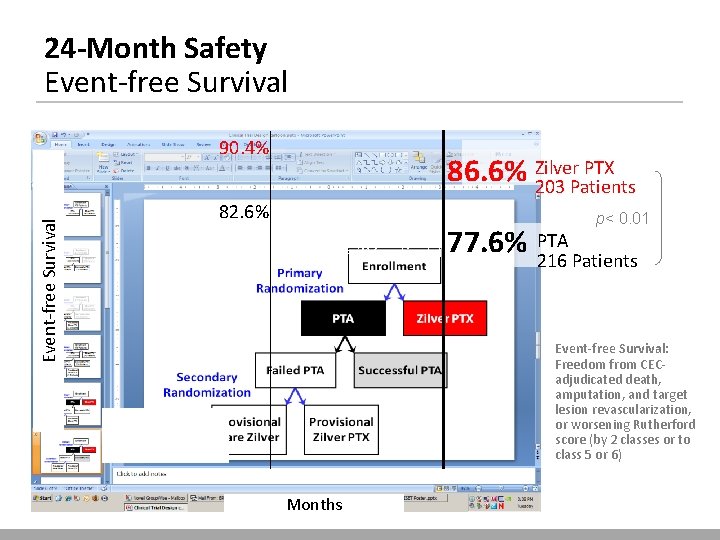
24 -Month Safety Event-free Survival 90. 4% PTX 86. 6% Zilver 203 Patients 82. 6% 77. 6% (n = 216 patients) p< 0. 01 77. 6% PTA 216 Patients Event-free Survival: Freedom from CECadjudicated death, amputation, and target lesion revascularization, or worsening Rutherford score (by 2 classes or to class 5 or 6) Months

Low Stent Fracture Rate • 546 stents implanted – 453 Zilver PTX (average of 1. 5 stents per patient) – 93 Zilver BMS • X-ray core laboratory analysis of 457 stents at 12 months • Four stent fractures – No associated adverse events 0. 9% stent fracture rate through 12 months (next evaluations at 3 and 5 years)
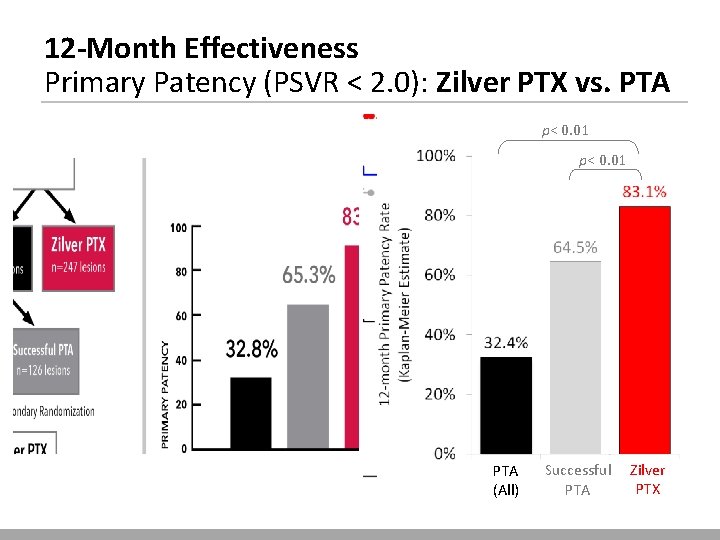
12 -Month Effectiveness Primary Patency (PSVR < 2. 0): Zilver PTX vs. PTA p< 0. 01 PTA (All) Zilver Successful Zilver PTX PTA
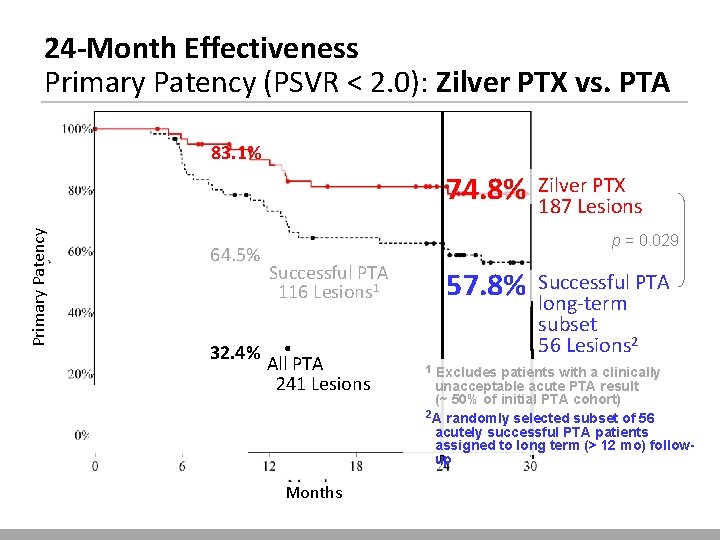
24 -Month Effectiveness Primary Patency (PSVR < 2. 0): Zilver PTX vs. PTA 83. 1% Primary Patency 74. 8% 64. 5% 32. 4% Zilver PTX 187 Lesions p = 0. 029 Successful PTA 116 Lesions 1 All PTA 241 Lesions Months 57. 8% 1 Successful PTA long-term subset 56 Lesions 2 Excludes patients with a clinically unacceptable acute PTA result (~ 50% of initial PTA cohort) 2 A randomly selected subset of 56 acutely successful PTA patients assigned to long term (> 12 mo) followup
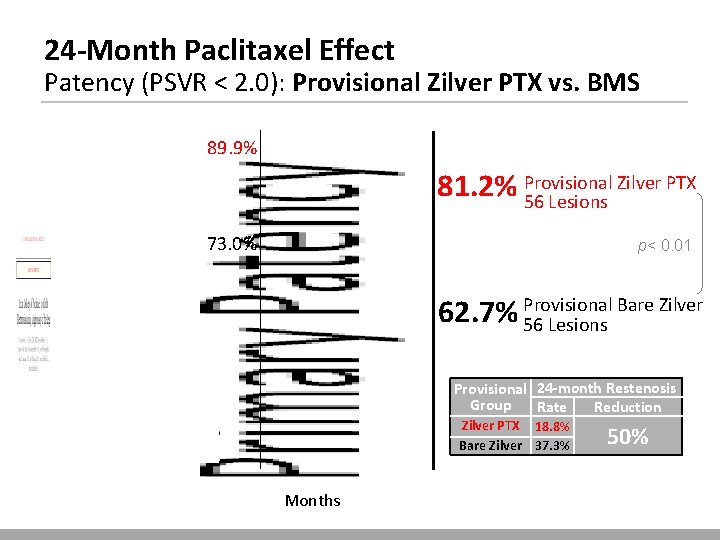
24 -Month Paclitaxel Effect Patency (PSVR < 2. 0): Provisional Zilver PTX vs. BMS 89. 9% Zilver PTX 81. 2% Provisional 56 Lesions 73. 0% p< 0. 01 Bare Zilver 62. 7% Provisional 56 Lesions Provisional 24 -month Restenosis Group Rate Reduction Zilver PTX 18. 8% Bare Zilver 37. 3% Months 50%

Conclusions: Zilver PTX Stent 9 0 0 2 k y r r a o s m i v – d concerns E Ø Interim results show no safety with the Zilver a l d e PTX stent C A e n nd D a F e p e h Ø Effectiveness results appear favorable att 24 months m n i m • High rate of freedom from TLR even for restenotic l o a c lesions v e r o r • Clinical outcomes are significantly improved at 24 S p U months for both de novo and restenotic lesions, p a Ø Durability of the Zilver® Flex® stent platform is excellent in the SFA, even for lesions with previously implanted stents • 12 -month fracture rate 1. 1% in previously stented lesions ® ® including previously stentedlesions • PTX coating reduces 24 -month restenosis rates by 50%
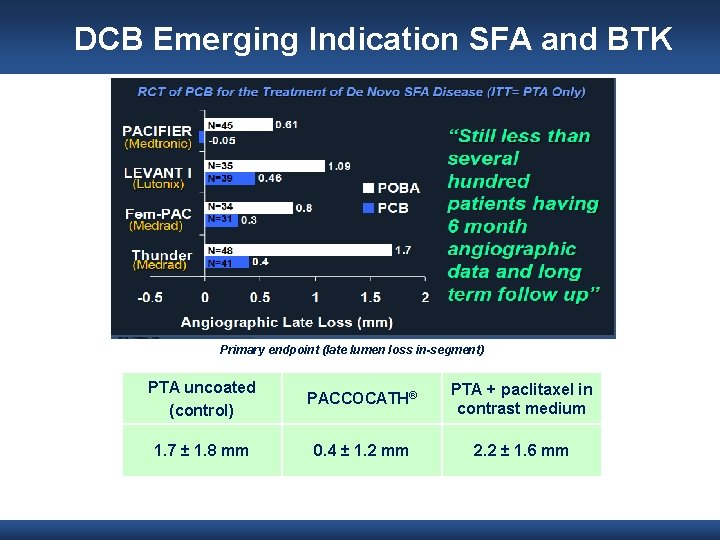
DCB Emerging Indication SFA and BTK Primary endpoint (late lumen loss in-segment) PTA uncoated (control) PACCOCATH® PTA + paclitaxel in contrast medium 1. 7 ± 1. 8 mm 0. 4 ± 1. 2 mm 2. 2 ± 1. 6 mm Tepe, N Engl J Med 2008; 358: 689 -99

DCB Peripheral Market / Data Overview Why the Enthusiasm Est Peripheral DCB Market Size and Growth • If proven successful, DCB could represent largest new market in peripheral space • Early studies demonstrate reduction in restenosis without need for permanent implant but these are in short lesions • Applicability may be as a stand alone therapy in type A and B lesions or as adjunctive to BMS in type C and D lesions Six month Late Loss Twenty four month TVR (Thunder)
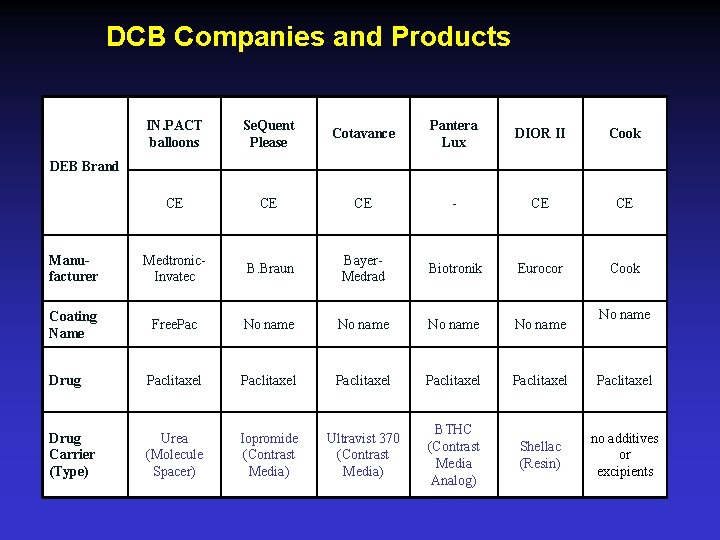
DCB Companies and Products IN. PACT balloons Se. Quent Please Cotavance Pantera Lux DIOR II Cook CE CE CE - CE CE Manufacturer Medtronic. Invatec B. Braun Bayer. Medrad Biotronik Eurocor Cook Coating Name Free. Pac No name Drug Paclitaxel Paclitaxel Drug Carrier (Type) Urea (Molecule Spacer) Iopromide (Contrast Media) Ultravist 370 (Contrast Media) BTHC (Contrast Media Analog) Shellac (Resin) no additives or excipients DEB Brand No name
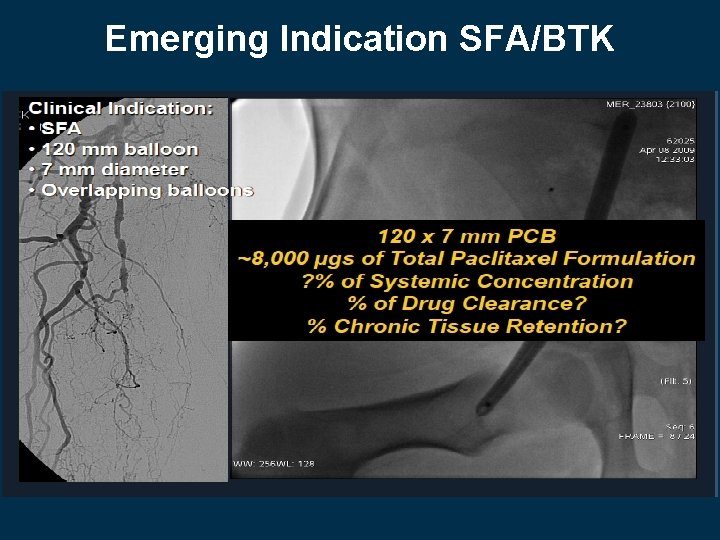
Emerging Indication SFA/BTK

Systemic Release of Paclitaxel

Majority of PTA procedures ended with dissection and a stent may be needed

Particulate Formation Drug Loss, Dissolution and Tissue Effects

DEB REGULATROY CHALLEMGES • DCB: Class. III combinationproduct • Balloon CDRH, Drug CDER • Similarchallenges and requirements to DES • Animalstudies up to 180 days time course of healing • Large randomizedclinicaltrials • Exipientparticlesreleaseduponinflationmicrovascularobstuction • Potential for large doses of druglossduringpassagein vivo • Consistncyand uniformity of thedrugovertheballoon? • Combinationswithstentscouldbeharmfulthrombosis • Large vesselslonglesions: Requires large dose of drugtoxicity? • Multiple balloonsforlonglesions • Can the DCB be used for multiple inflations? • Consider affects of bail out stenting.

Proof of Concept to Pivotal Trials 1. Safety and efficacyfirst in human trialsfordenovolesions (DCB vs bare balloon) Paccocath – Cotavance technology (Thunder, Fem. Pactrial) Lutonix – Levant 1 2. New indications a) SFA - In stent restenosis b) SFA - With stents c) SFA - with atherectomy d) BTK e) AV-Fistula 3. For US approval (DCB vs bare balloon)
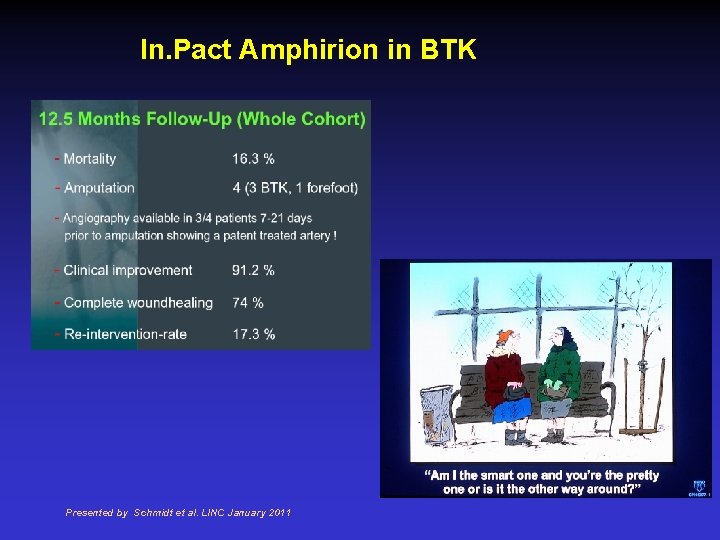
In. Pact Amphirion in BTK Presented by Schmidt et al. LINC January 2011

DCB Summary The data to support the use of DEB in the periphery for SFA and tibial disease is limited Long DCB balloons for the SFA may result with spill large doses of drug to the blood stream, could be toxic. The coating and particulates on the balloon are not stable and may clogged distal vessels Crossover to stent after balloon is risky and may associate with increase risk of stent thrombosis DCB and BMS associated with high rates of stent thrombosis, there are healing issues Regulatory requirements for combination therapy in the US are challenging

DCB is it disruptive technology? “Am I the smart one and you’re the pretty one or is it the other way around? ”

DES for Fempop Trials second look Zilver® PTx® N 1º patency 12 mo RESILIENT 1º DES PTA + provisional BMS Optimal PTA Provisional DES Provisional BMS PTA 247 188 126 63 62 134 72 83. 1% 67. 0% 65. 3% 89. 9% 73. 0% 81. 3% 36. 7% Duplex ultrasound, patent = PSVR , 2. 0 (or angiography if available, patent = diameter stenosis <50%) Combination of ultrasound confirmed patency and TLR “It is possible that in selected cases and due to the scaffolding effect, peripheral DES may be more effective than PCB…then if we are obliged to migrate into a combined approach (PCB + BMS), PCB use will be restricted to longer lesions and smaller vascular territories” Dake, TCT 2010 & Katzen ISET 2008

Future of DES DCB? • 2 out of 3 DES studies in the SFA failed (sirocco, stride) • 1 DES (Zilver. PTX) since September 2009 has CE-mark Ø DES for SFA is challenging combination device Ø It is imperative that the stent platform will be fracture free. Ø With higher Patentcy rates DES should replace BMS in the periphery Ø DCB technology emerged as a potential alternative to balloon in SFA and tibial indication k n a u o Y Th Ø The next wave of studies should be DCB vs DES Ø The future holds for both technologies if they will continue to provide supportive data and superiority in efficacy without compromising safety when compared to BMS and PTA alone
- Slides: 35