Drug Utilization Review Drug Utilization Evaluation An Overview














- Slides: 15

Drug Utilization Review & Drug Utilization Evaluation: An Overview Presentation Developed for the Academy of Managed Care Pharmacy Updated: February 2016

Learning Objectives • Differentiate between the terms Drug Utilization Review (DUR) and Drug Utilization Evaluation (DUE) • Explain the role of a pharmacist in a DUR program • Provide an example for each of the three types of DUR’s: prospective, concurrent, and retrospective • Describe the key stakeholders vested in a successful DUR/DUE program

Definition of DUR and DUE • Drug Utilization Review (DUR): An authorized, structured, ongoing review of health care provider prescribing, pharmacist dispensing, and patient use of medication. • Drug Utilization Evaluation (DUE): A qualitative evaluation of drug use, prescribing, and member fill patterns to determine the appropriateness of drug therapy. AMCP. Glossary of Managed Care Terms. http: //www. amcp. org/Managed. Care. Terms/

Goals of DUR and DUE • Improve quality of care and overall drug effectiveness • Prevent adverse drug reactions • Encourage the practice of evidence-based, clinically appropriate, cost-effective drug use • Reduce drug misuse and abuse • Reduce costs related to inappropriate drug use

Pharmacist Role in DUR & DUE • Identifies opportunities for quality improvement • Participates in efforts to improve: – Patient outcomes – Quality of programs • Promotes appropriate drug use to reduce overall health care costs and improve access to care • Carries out ethical and professional responsibility

A Model DUR Program • Access to member drug utilization data • Qualified pharmacists with authority to review • Knowledge of population served and delivery system • Availability of established standards for comparison • Measurement of utilization review outcomes

Prospective DUR • A screening method by which a health care provider reviews the necessity of drug therapy before it is dispensed or administered Electronic DUE programs at retail pharmacies Prior authorization (PA) programs Drug-drug and drug-disease interactions Dosing appropriateness Drug-patient precautions (due to age, allergies, gender, pregnancy, etc. ) – Medication directions – Formulary substitutions (e. g. , therapeutic interchange, generic substitution) – Inappropriate duration of drug treatment – – – AMCP. Glossary of Managed Care Terms. http: //www. amcp. org/Managed. Care. Terms/

Concurrent DUR • A screening method by which a health care provider reviews the necessity of drug therapy at the time of dispensing or during treatment – – – – – Case management Review of patient records Research projects that follow patients in randomized, controlled trials Real-time system edits at the point of service Over or underutilization of medication Excessive or insufficient dosing Drug-drug interactions Drug-disease interactions Drug dosage modifications AMCP. Glossary of Managed Care Terms. http: //www. amcp. org/Managed. Care. Terms/

Retrospective DUR • A screening method by which a health care provider reviews the necessity of drug therapy after it has been dispensed or treatment has started – Review of medical charts, electronic medical records and/or claims data to assess appropriate drug use – Review provider prescribing patterns – Quality assurance analyses – Developing standard guidelines to achieve target outcomes at a population level – Appropriate generic use – Use of formulary medications whenever appropriate – Therapeutic appropriateness and/or duplication AMCP. Glossary of Managed Care Terms. http: //www. amcp. org/Managed. Care. Terms/

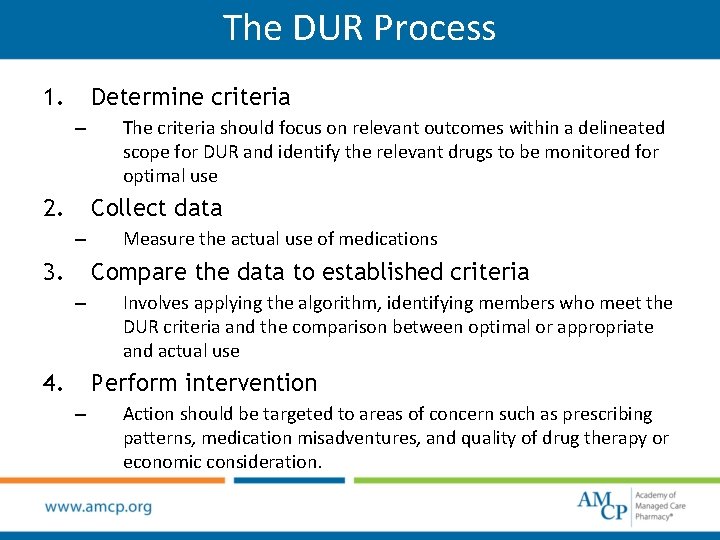
The DUR Process 1. Determine criteria – 2. The criteria should focus on relevant outcomes within a delineated scope for DUR and identify the relevant drugs to be monitored for optimal use Collect data – 3. Measure the actual use of medications Compare the data to established criteria – 4. Involves applying the algorithm, identifying members who meet the DUR criteria and the comparison between optimal or appropriate and actual use Perform intervention – Action should be targeted to areas of concern such as prescribing patterns, medication misadventures, and quality of drug therapy or economic consideration.

The DUR Process 5. Analyze results – 6. Document DUR – 7. Evaluate the outcomes and document reasons for positive and negative results Report the findings to the appropriate team within the organization (e. g. , the pharmacy & therapeutics committee) and/or individual prescribers when appropriate Re-evaluate the program (on-going)

Who Benefits from DUE/DUR? • Accrediting bodies/Government • National Committee for Quality Assurance • Joint Commission on Accreditation of Healthcare Organizations • Omnibus Budget Reconciliation Act 1990 • • Plan member Health care provider Pharmacist Health care system

Example "This asthma is really slowing me down. This prescription isn't helping much. " • Example Scenario: Tim's asthma is not well controlled, and he uses his inhaler multiple times a day. Tim's therapy should most likely be increased to prevent further medical complications. • Pharmacist Interaction: A pharmacist conducted concurrent DUR at the health plan and noticed that Tim was only prescribed an as-needed inhaler. With the pharmacist's recommendation to the prescriber, derived from evidence-based guidelines, Tim was prescribed a maintenance asthma medication. • Benefit: Although another medication was added, the patient and the health plan have an overall cost savings. The added prescription vastly decreases Tim's likelihood of a costly emergency room visit for a severe asthma attack and enhances Tim's quality of life.

Conclusion • A pharmacist performs DUR/DUE to improve overall access and quality of care, and to reduce costs • Each type of DUR represents an important step in ensuring that the member receives the most appropriate, cost-effective medication • A successful DUR/DUE program benefits all health care players, including the member

Thank you to AMCP member Alvah Stahlnecker for updating this presentation for 2016.