Drug Therapy of Diabetes Michael Veltman MBBS FANZCA

Drug Therapy of Diabetes Michael Veltman MBBS FANZCA FASE Director of Anaesthesia Joondalup Health Campus

Overview Definition and Incidence Definition, incidence, classification Physiology & Pathophysiology Medical Managment Pharmacology

Definition and Incidence

Diabetes Mellitus History Mellitus versus Insipidus Insulin treatments Definition Plasma glucose > 7 m. M, Fasting Plasma glucose > 11. 1 m. M, 2 hours post GTT

Incidence Common Disease in the western world 6% of the world’s adult population (285 million) Rising rapidly - was only 30 million people in 1985

Aetiology & Classification

Classification Type I (5%) Autoimmune Other (rare) - Pancreatitis, toxins Type II (90 -95%) Insulin Resistance Gestational

Aetiology -Type I Due to a loss of insulin secreting cells β Islet cell produce insulin Multifactorial Genetic (30 -50%) Autoimmune

Aetiology - Type II Due to insulin resistance. Compensatory hypersecretion Ultimately leading to islet cell failure

Physiology
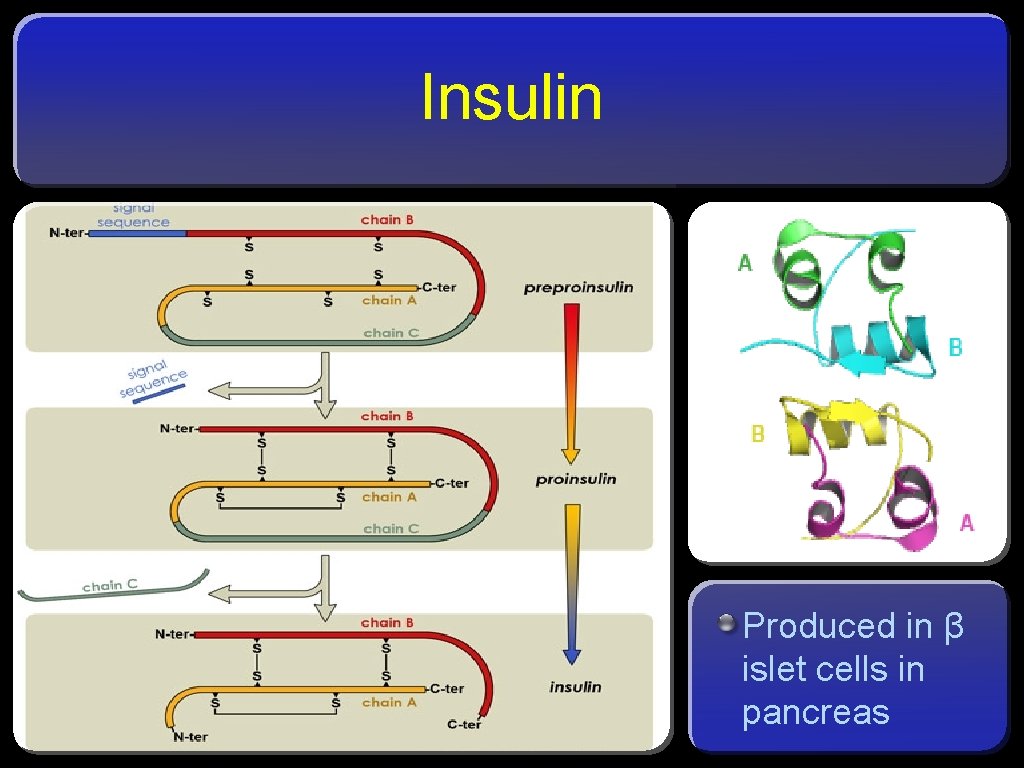
Insulin Produced in β islet cells in pancreas
Insulin Hormone produced by pancreas. Regulates metabolism: Carbohydrate Fat Insulin is needed for uptake of glucose

Insulin Effects
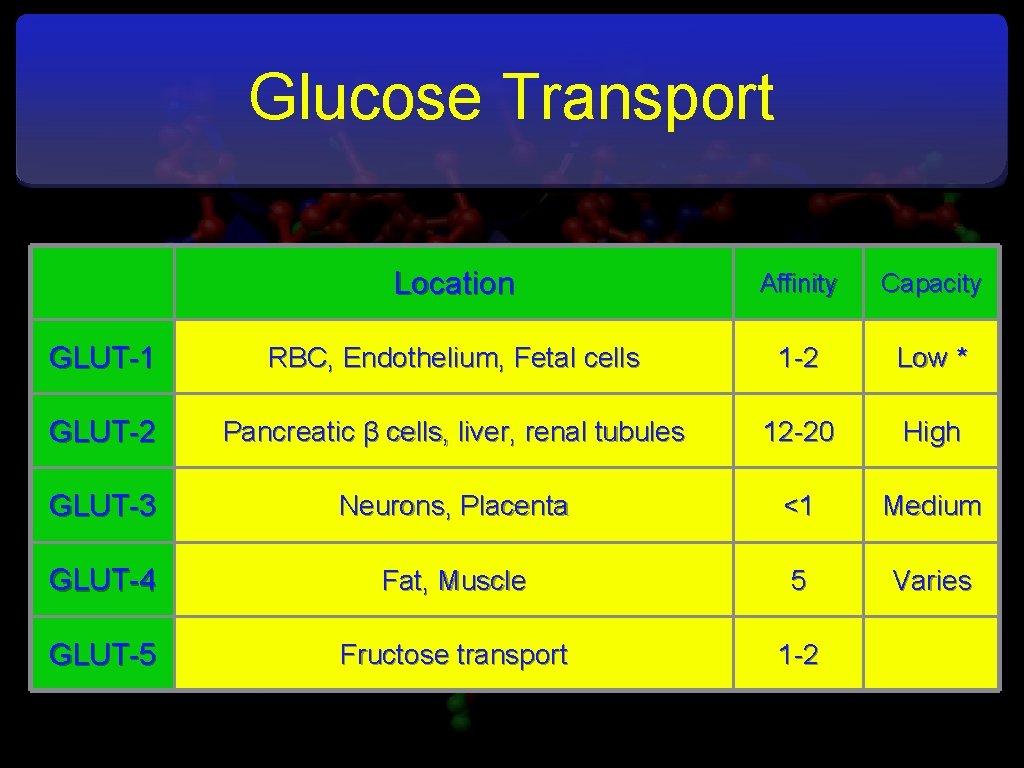
Glucose Transport Location Affinity Capacity GLUT-1 RBC, Endothelium, Fetal cells 1 -2 Low * GLUT-2 Pancreatic β cells, liver, renal tubules 12 -20 High GLUT-3 Neurons, Placenta <1 Medium GLUT-4 Fat, Muscle 5 Varies GLUT-5 Fructose transport 1 -2

Cellular physiology Binds to the insulin receptor α subunit Also binds to IGF-I Causes activation of Tyrosine Kinase Phosphorylation of of cytoplasmic protiens

Insulin - Rapid Effects Rapid increases in transport of Glucose Amino Acids Potassium

Insulin - Intermediate Effects Increased protein synthesis Decreased protein degredation Glycolysis Inhibition of gluconeogenesis

Insulin - Delayed Effects Increased lipogenesis Mediated through increased production of enzymes

Pathophysiology

Diabetic Complications Acute Hyperglycaemia Polyuria. (+ loss of electrolytes) Impaired immune function Impaired injury response Myocytes Neurons Diabetic ketoacidosis

Hyperglycaemia is specifically known to be bad in certain clinical states Acute Myocardial Infarction Neurological Injury Critically unwell patients with sepsis

Diabetic Ketoacidosis Medical Emergency Acidosis is the key feature Excess production of Aceto-acetate, β Hydroxy butyrate. Fall in p. H Acidic Urine Loss of Na+ and some K+ in urine
Chronic Diabetes More a disease of fat metabolism Elevated free fatty acids Altered metabolism of Acetyl-Co. A Ketone formation (source of energy)

Diabetic Complications Chronic Accelerated Vascular Disease Peripherally Coronary Cerebral Retinopathy Neuropathy Nephropathy

Diabetes versus Hyperglycaemia The “diagnosis” of Type 2 diabetes is late Delayed diagnosis: 10 -12 years Undiagnosed illness is common This affects interpretation of many studies

Medical Management Diabetic Ketoacidosis Hypoglycaemia Hyperglycaemia Chronic management Fasting diabetics Other options

Diabetic Ketoacidosis

Critical Care Setting Aiming to prevent high (>8. 0 m. Mol) levels of glucose Intravenous insulin is the drug of choice Benefits: Critically unwell Myocardial injury Neurological injury (? )

Diabetic Ketoacidosis Aim to restore deficits in: Insulin Potassium and Sodium Water Life threatening condition 5% mortality

Critical Care Setting Insulin can be used to lower potassium Usually given with glucose Doesn’t shift potassium out of the body

Hypoglycaemia

Hypoglycaemia Medical emergency BSL < 4. 0 Treatment Glucose - Up to 50 ml of 50% Glucagon - 1 mg

Hyperglycaemia

Hyperglycaemia Aim to keep glucose < 8. 0 m. Mol most of the time. More aggressive control has uncertain benefit in most studies

DIGAMI Hyperglycaemia with AMI 620 patients follow up 3. 4 (1. 6 -5. 6) yrs. 11% reduction in mortality One life was saved for nine treated patients.
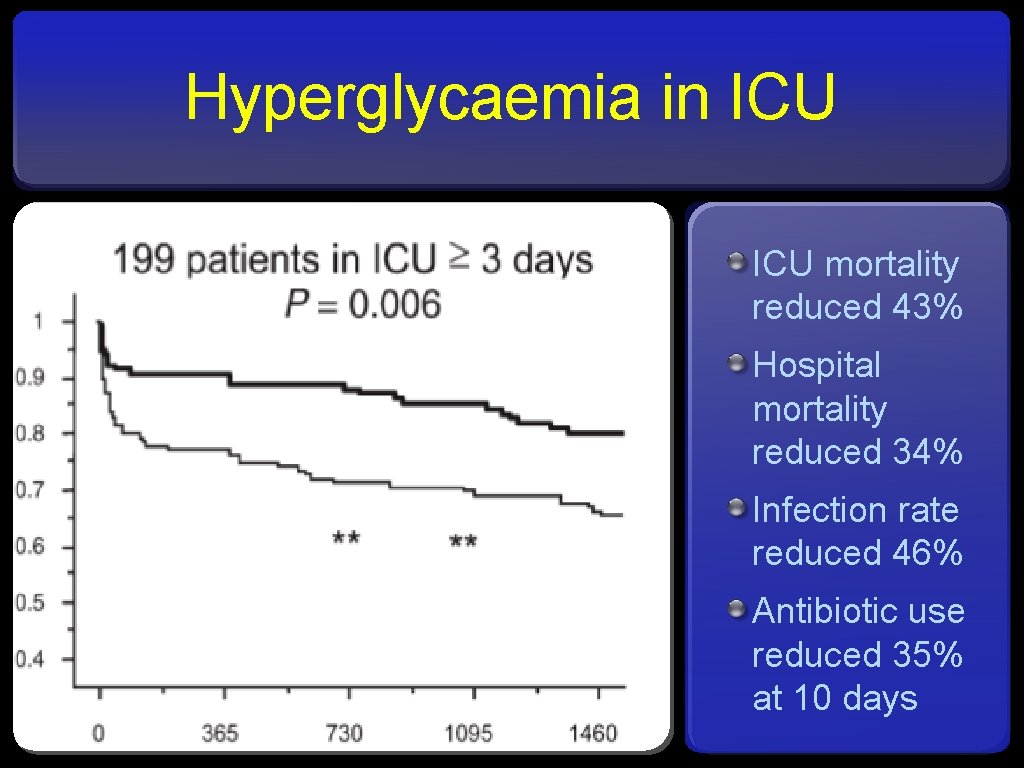
Hyperglycaemia in ICU mortality reduced 43% Hospital mortality reduced 34% Infection rate reduced 46% Antibiotic use reduced 35% at 10 days

Chronic management

Aim in the community Minimise the longer term impact of elevated glucose Day to day fluctuation is not that important Glycosylated Haemaglobin is very good measure Avoid hypoglycaemic events Reduce cardiovascular risk

Hb. A 1 c Haemoglobin has a 120 day lifespan Glycosylation happens spontaneously Glycosylation is proportional to glucose level Normal level is 4. 0 -5. 9 % Level above 6. 5% suggests diabetes Target in diabetes is < 7. 0%
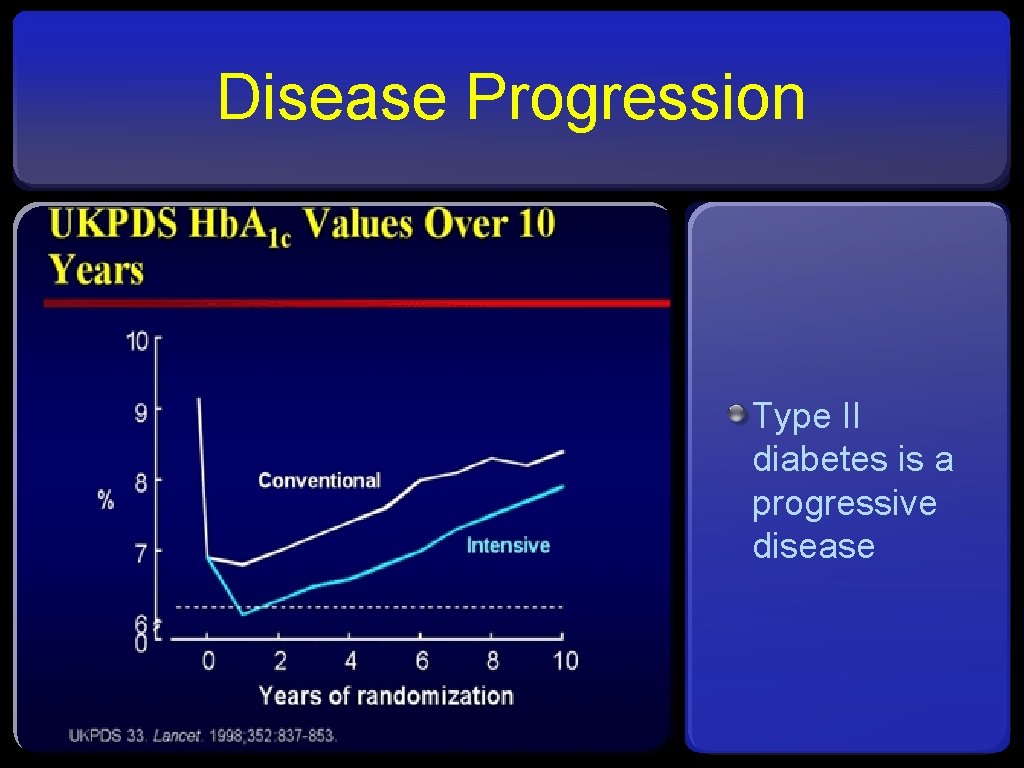
Disease Progression Type II diabetes is a progressive disease

Diet control Multifactorial approach Avoid high glycaemic index agents Weight loss Exercise

Oral Agents Aim for good glycaemic control Ideally normalise BSL within 2 -4 hours of a meal Many choices of agent Metformin Sulphonylureas

Insulin Necessary with type I diabetics from onset of disease Can be combined with oral agents in type II diabetes Bedtime insulin Can be sole agent

Retinopathy Starts within a few years of diagnosis (5 -10) Leading cause of blindness in first world Generally preventable Good glucose control Laser therapy. C-peptide (? )
Fundoscopy Normal retina Left eye
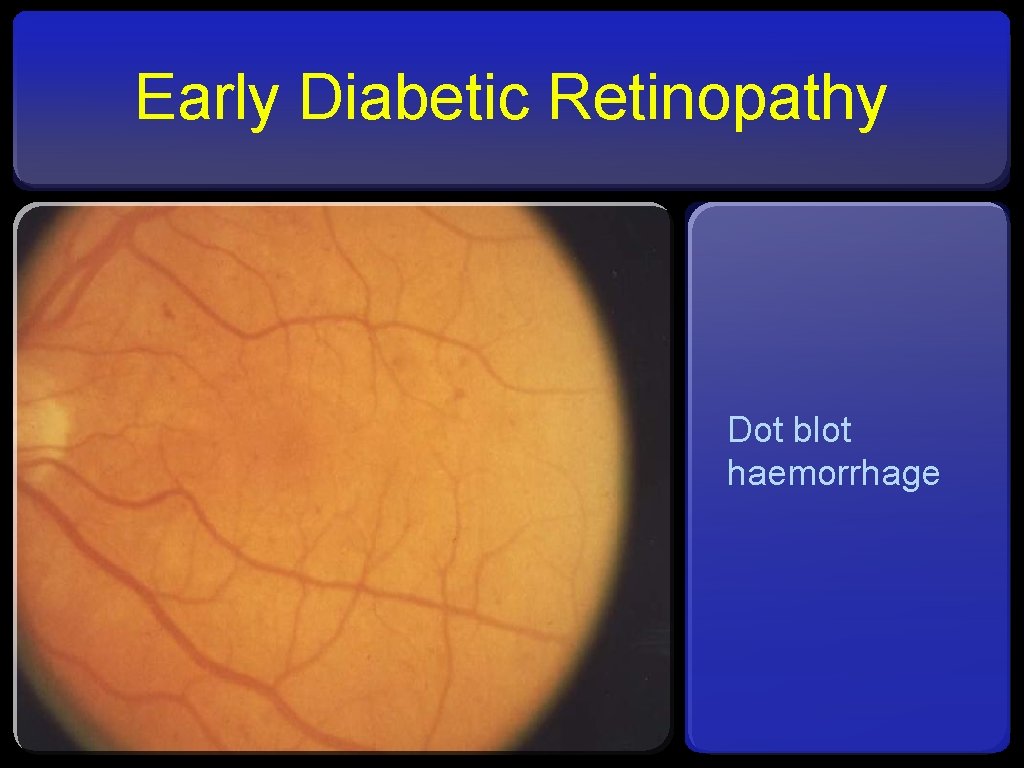
Early Diabetic Retinopathy Dot blot haemorrhage
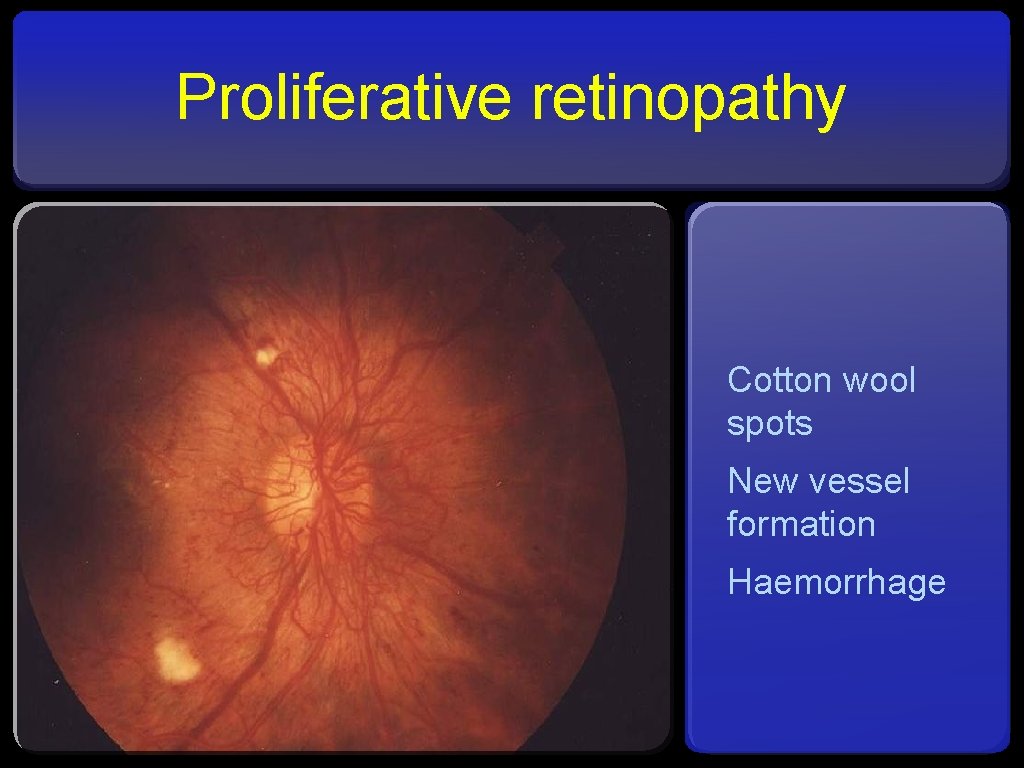
Proliferative retinopathy Cotton wool spots New vessel formation Haemorrhage
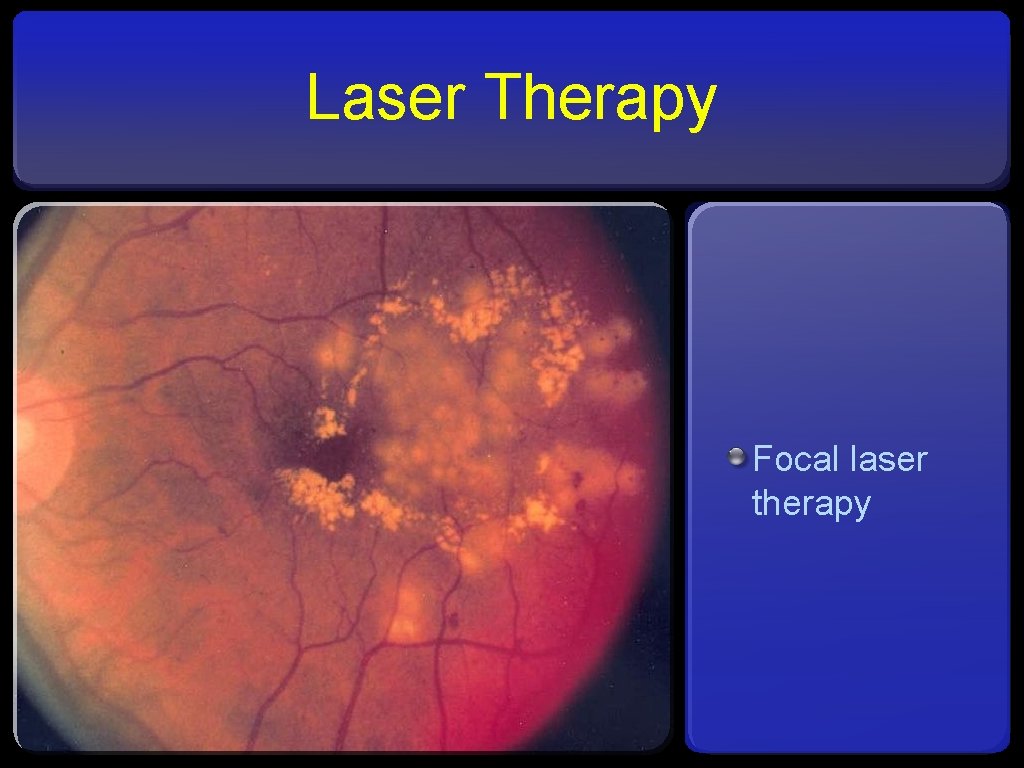
Laser Therapy Focal laser therapy

Nephropathy Looking for proteinuria Microalbuminuria is first sign Definitive Dx requires kidney biopsy (not needed)

Management Nephropathy ACE inhibition or ARB’s (not both) Diet (Protien, Potassium) Avoid NSAID’s including COX-2 inhibitors New treatments C-Peptide

Neuropathy Affects all nerve fibers, including motor, sensorty and autonomic fibres Multifactorial causes Microvascular disease Glycosylation end products Protein Kinase C activation Sorbitol / Polyol pathways
Effects Sensorimotor Loss of sensory pathways (esp vibration) Chronic pain Autonomic Diarrhoea, orthostatic hypotension, gastropariesis Cranial nerve palsy (3 rd nerve)

Treatment Pain control TCA’s (Amitriptyline, Nortriptyline) SNRI’s (Duloxitine, Venlafaxine) Gabapentinoids (Pregabalin, Gabapentin)

Other Treatments α-lipoic acid Methylcobalmin (Vit B 12 subform) C-Peptide (experimental) Photo therapies Glucose control

Vascular Disease Both peripheral and coronary vascular disease risk Control other risk factors Smoking, hypertension, cholesterol

Fasting in Diabetes

Fasting Type I diabetes Needs insulin Reduce dosage Type II diabetes Often needs no oral agents

Other Treatment Options
Other treatments Bariatric surgery Pancreatic transplantation Implantable pumps

Medical Management Diabetic Ketoacidosis Hypoglycaemia Hyperglycaemia Chronic management Fasting diabetics Other options

Oral Agents

Types of oral agents Sensitisers Biguanides (Metformin, Phenyformin) Thiazolidines (Rosiglitazone, Pioglitazone) Secretagogues Sulphonylureas (glipizide, glyburide, gliclazide) Meglitinides (repaglinide, nateglinide) α glucosidase inhibitors Peptide analogs

Sulphonylureas

Sulphonylureas Glicizide, glipizide, glibenclamide Inhibit potassium channel Increase the amount of insulin secreted. Effective, inexpensive Hypoglycaemia & Weight Gain

Mechanism of action Bind to ATP-dependent K+ channel Hyperpolarises the β cell membrane Opens voltage gated Ca++ channels Fusion of insulin granulae with membrane

Sulphonylureas Short half life - require BD or TDS dosing Metabolised by P 450 enzymes

Interactions Increased risk of hypoglycaemia Aspirin, allopurinol, sulphonylamides, fibrates Worsening glucose tolerance Steroids, isoniazide, OCP, sympathomimetics, thyroid hormones

Complications Hypoglycaemia Weight Gain Overstimulation of β cells Possible risk of disease acceleration. Teratogenic Little to no survival benefit seen

Biguanides (Metformin)

Uses Type II diabetes Prediabetes (less benefit than lifestyle Δ) Polycystic ovarian disease Gestational diabetes (? foetal safety) Reduced pancreatic cancer risk Reduced weight gain from other agents.

Mechanism of Action Inhibits gluconeogenesis to 1/3 baseline Most Type II diabetics have 3 x increase Activates AMP-activated protein kinase AMPK activation increases SHP Inhibits gluconeogenesis genes

Kinetics Bioavailable 50 -60% Peak 1 -3 hours, 8 hours with SR Minimal plasma protein binding High volume of distribution (10 L/kg) Not metabolised Cleared by tubular secretion (T 1/2 6. 2 hrs)

Advantages Reduces diabetic complications Mortality reduction of 30% c. f. insulin or sulphonylureas Mortality reduction of 40% c. f. diet control Less weight gain Lower risk of hypoglycaemia

Contraindications Lactic acidosis risks Renal, Lung, Liver, Heart disease No actual evidence of harm however Cease before iodine contrast Actual risk is 9/100 000 person years

Adverse Effects GIT Diarrhoea, GI upset, Nausea, Vomiting Hypoglycaemia Alcohol

Glitazones

Glitazones Insulin sensitiser (Binds PPAR receptor) Makes cells more sensitive to insulin Third line drug Usually in combination with metformin

Adverse effects Increased risk of AMI(Rosiglitazone) Heart failure (Pioglitazone) Stroke (Rosiglitazone) Bone fractures Bladder Cancer (Pioglitazone) Macular oedema (? ) & Acute hepatitis (? )

Adverse Effects Most glitazones have been withdrawn from some market, somewhere for some safety concern.

Advantages Low risk of hypoglycaemia Caution with alcohol or other hypoglycaemic agents. May be a better alternative than going to insulin Pioglitazone may reduce rate of atheroma progression. Raises HDL, lowers TG and hs. CRP.

Insulin

Insulin Manufacture Done with recombinant DNA USE bacteria or yeast. Species variation exists Essentially all human insulin now. Lower reaction rates to human insulins

Insulin Administration Subcutaneous Intravenous Insulin Other routes Inhalational, transdermal, intranasal, oral
Insulin Pharmacology Types of insulin Mechanisms of action Dosage

Insulin Ultra fast acting Fast Acting Intermediate Acting Long Acting

Ultra Fast Acting Lispro (Lysine and proline swap on B chain) Aspart (Aspartic Acid for Proline on B chain) Both have high soluability Rapid uptake - Onset 15 minutes Peak effect at 45 -90 minutes Duration 3 -5 hours.

Fast Acting Insulin (“Actrapid”) Standard insulin Onset 30 minutes Peak 2 -4 hours Duration up to 6 hours

Intermediate Acting Mixtard (Insulin/protamine) Onset 2 hours Peak 4 -6 Duration 12 hours

Long Acting Used to use Zinc (Ultralente insulin) Mostly now use modified insulins Glargine Adds arginine to C end of B chain Low p. Ka leads to slow absorption Detemir Binds myristic acid to the Lysine at B 29 High affinity for Albumin

Long Acting Onset slow - ? 4 hours Duration 24 hours No discernable peak.

Exogenous Versus Endogenous Insulin has different effects depending on route of administration Ideal route is into the portal vein Next best is in peritoneal dialysis bag Intravenous is better in the acute setting Subcutaneous is the easiest for most.

Summary Definition and Incidence Definition, incidence, classification Physiology & Pathophysiology Medical Managment Pharmacology
- Slides: 92