Drug Regulatory Systems Strengthening RSS WHO Technical Briefing

Drug Regulatory Systems Strengthening (RSS) WHO Technical Briefing Seminar on Essential Medicines and Health Products 31 October 2013 WHO Headquarters, Geneva, Salle M 505 Dr Claudia P Alfonso

Outline l Global overview of vaccine production and immunization l The WHO Regulatory Systems Strengthening (RSS) Programme and the five-step capacity building model l Assessments of national regulatory authorities (NRA) and harmonized tool development l NRA Institutional Development Plans (IDP) | l Link between WHO Prequalification and NRA assessment 2 |

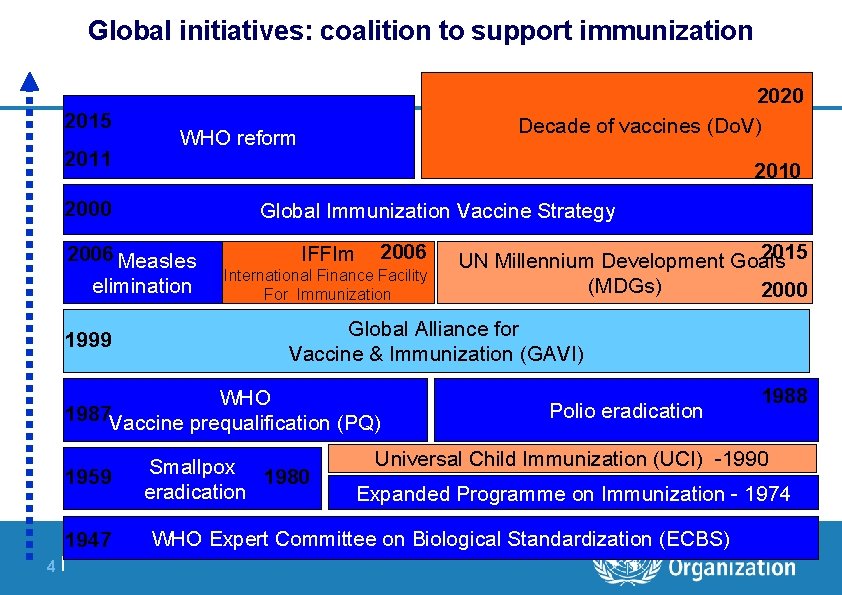
Global initiatives: coalition to support immunization 2015 2011 WHO reform 2010 2000 Global Immunization Vaccine Strategy 2006 Measles elimination 1999 2020 Decade of vaccines (Do. V) IFFIm 2006 International Finance Facility For Immunization Global Alliance for Vaccine & Immunization (GAVI) | WHO 1987 Vaccine prequalification (PQ) 1959 1947 4 | 2015 UN Millennium Development Goals (MDGs) 2000 Smallpox 1980 eradication Polio eradication 1988 Universal Child Immunization (UCI) -1990 Expanded Programme on Immunization - 1974 WHO Expert Committee on Biological Standardization (ECBS)

Global challenges 1. Demand for more vaccines 2. Raise of public awareness about vaccines 3. New standards promoted by WHO 4. Anticipate new needs for regulation vaccines 5. Quality control systems established in countries need increased investment to be sustained | 6. Domestic production not consistent in volume and quality has difficulty to meet c. GMP standards 7. Shift in vaccine market and increase segmentation 5 | 1. New initiatives and global coalition efforts: GAVI, Do. V, influenza, polio eradication, measles immunization, introduction of new vaccines, others 2. Public and media expectations increased through media, internet and social networks 3. Vaccine science requires new standards 4. Vaccines needs thorough regulations to address specificity 5. QC is more complex and expensive 6. Volume increasing and c. GMP standards raising too 7. EPI vaccines are traditionally produced by developing countries while new vaccines are produced by developed countries.

Vaccine demand increases while number of producing countries decreases Vaccine producing 1990: 63 countries 1997: 52 countries 2012: 44 countries 6 |

Major vaccine producing countries 1997 -2012 146 vaccine manufacturers, 95% global production in 16 countries Denmark Russia Belgium Bulgaria France Italy China Canada USA Cuba Senegal India Japan Korea Indonesia Brazil Developing countries 7 |
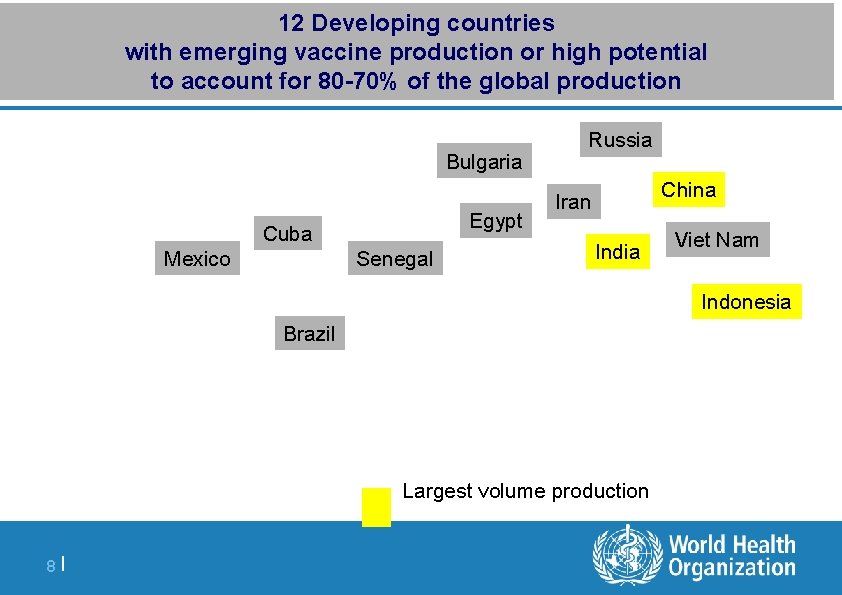
12 Developing countries with emerging vaccine production or high potential to account for 80 -70% of the global production Bulgaria Egypt Cuba Mexico Senegal Russia China Iran India Viet Nam Indonesia Brazil | Largest volume production 8 |
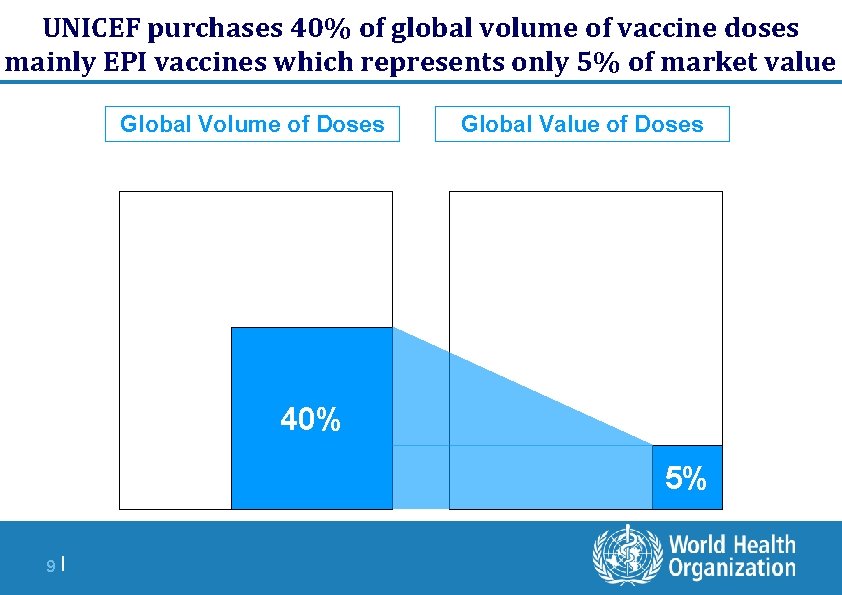
UNICEF purchases 40% of global volume of vaccine doses mainly EPI vaccines which represents only 5% of market value Global Volume of Doses Global Value of Doses 40% 5% 9 |

Vaccines are different from drugs ü Biological products (Variation) ü Heat sensitive (Cold chain) ü Sensitive to light and freezing ü Limited shelf life ü Mandated by public health programme (EPI or NIP) ü Targeted to healthy children and mothers ü Given| for prevention - recipient cannot judge treatment ü Pennies per dose, new vaccines are more expensive ü Limited number of products and manufacturers ü Given once or only a few times 10 |

WHO Prequalification Principals l Reliance on NRAs l General understanding of the product – presentations offered – production process – quality control methods – quality system in place | – clinical data relevant to the target population l Assurance of production consistency through compliance with GMP 11 |

WHO DEFINITION OF VACCINES OF ASSURED QUALITY l The National Regulatory Authority (NRA) is able to regulate independently vaccines against standards of quality, safety and efficacy (QSE) in accordance with the WHO recommended functions defined by WHO, and l There are no unresolved confirmed reports of quality-related problems. | 12 |

Ensuring vaccine quality for use in national immunization programmes: concept evolution Training Benchmarking Assessment Technical Monitoring Impact support Progress 1996 1997 1998 2004 2011 Regulatory Systems Strengthening 60 functional NRAs, 80% of all producing countries, 97% vaccines of assured quality
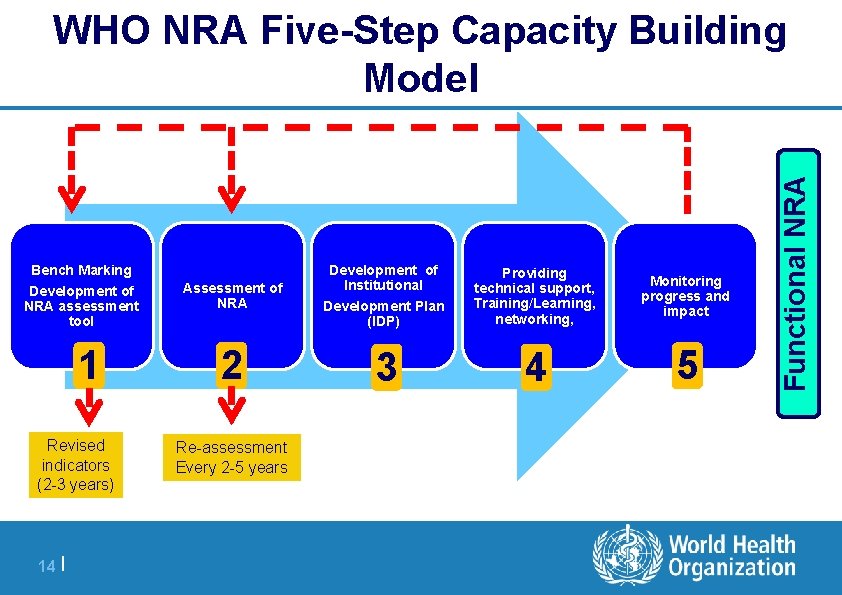
Bench Marking Development of NRA assessment tool 1 Revised indicators (2 -3 years) 14 | Assessment of NRA Development of Institutional Development Plan (IDP) Providing technical support, Training/Learning, networking, Monitoring progress and impact 2 3 4 5 | Re-assessment Every 2 -5 years Functional NRA WHO NRA Five-Step Capacity Building Model
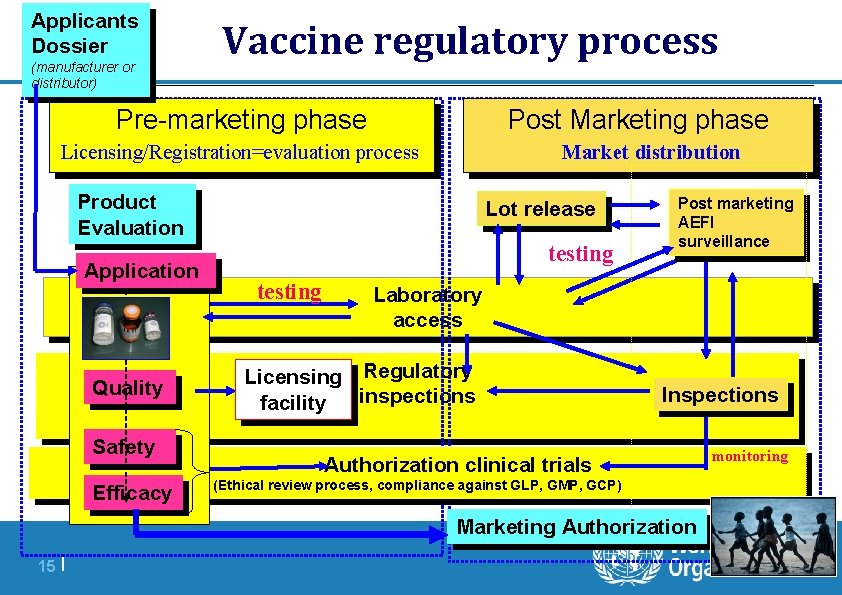
Applicants Dossier (manufacturer or distributor) Vaccine regulatory process Pre-marketing phase Post Marketing phase Market distribution Licensing/Registration=evaluation process Product Evaluation Application Quality Safety Efficacy Lot release testing Post marketing AEFI surveillance Laboratory access Licensing Regulatory inspections facility Inspections Authorization clinical trials (Ethical review process, compliance against GLP, GMP, GCP) Marketing Authorization 15 | monitoring
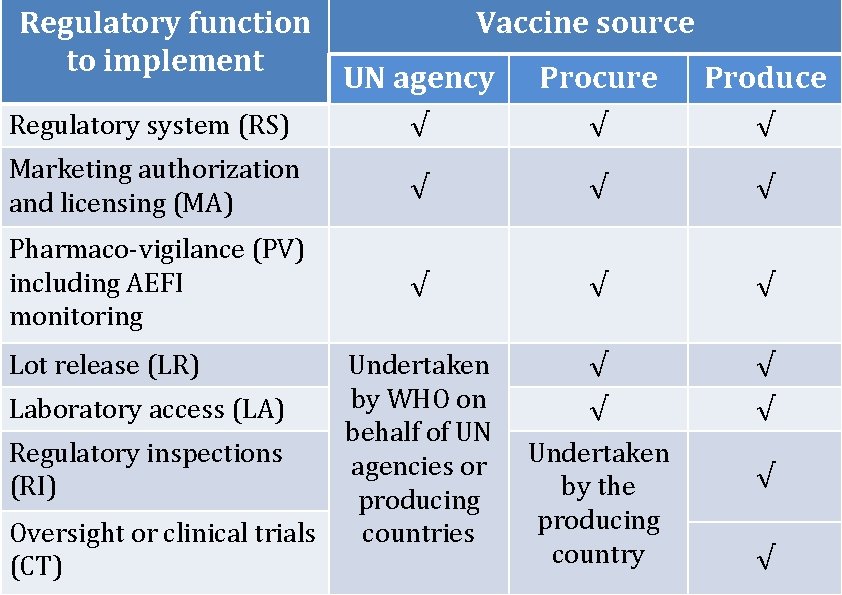
Regulatory function to implement Vaccine source UN agency Procure Produce Regulatory system (RS) √ √ √ Marketing authorization and licensing (MA) √ √ √ Pharmaco-vigilance (PV) including AEFI monitoring √ √ √ Undertaken by WHO on behalf of UN agencies or producing countries √ √ Lot release (LR) Laboratory access (LA) Regulatory inspections (RI) Oversight or clinical trials (CT) Undertaken by the producing country √ √

Chronology of indicators in the WHO NRA assessment tool for vaccines 17 |

Harmonized tool: regulatory functions 1. NATIONAL REGULATORY SYSTEM 2. REGISTRATION AND MARKETING AUTHORIZATION 3. LICENSING ACTIVITIES 4. POST-MARKETING SURVEILLANCE (+ LOT RELEASE FUNCTION) 5. OVERSIGHT OF CLINICAL TRIALS 6. INSPECTIONS AND ENFORCEMENT ACTIVITIES 7. LABORATORY ACCESS AND TESTING 8. VIGILANCE AND RISK MANAGEMENT 9. CONTROL OF PROMOTION AND ADVERTISING 10. CONTROL OF NARCOTICS, PSYCHOTROPIC SUBSTANCES AND PRECURSORS 11. PHARMACEUTICAL PERSONNEL National Regulatory System, 18 | Status of vaccine indicators
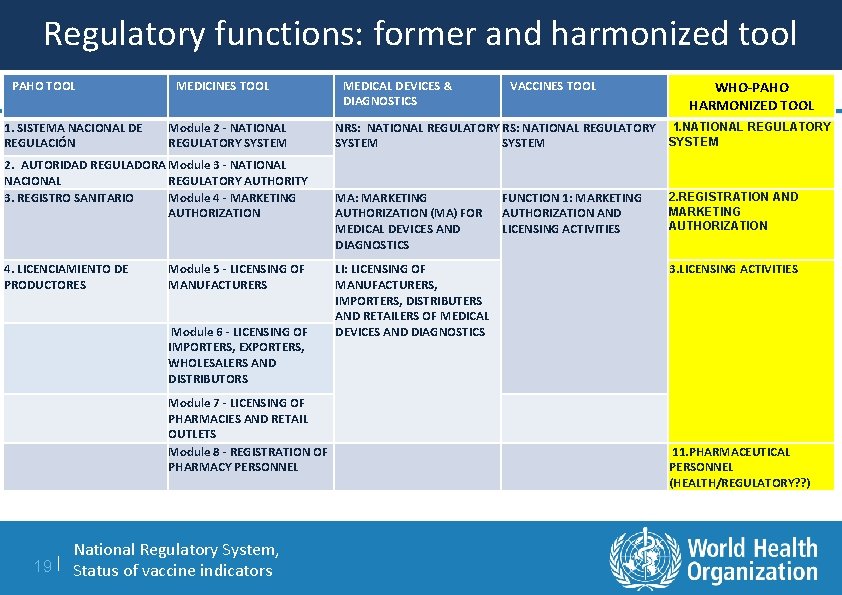
Regulatory functions: former and harmonized tool PAHO TOOL 1. SISTEMA NACIONAL DE REGULACIÓN MEDICINES TOOL Module 2 - NATIONAL REGULATORY SYSTEM 2. AUTORIDAD REGULADORA Module 3 - NATIONAL NACIONAL REGULATORY AUTHORITY 3. REGISTRO SANITARIO Module 4 - MARKETING AUTHORIZATION MEDICAL DEVICES & DIAGNOSTICS MA: MARKETING AUTHORIZATION (MA) FOR MEDICAL DEVICES AND DIAGNOSTICS Module 5 - LICENSING OF MANUFACTURERS Module 6 - LICENSING OF IMPORTERS, EXPORTERS, WHOLESALERS AND DISTRIBUTORS Module 7 - LICENSING OF PHARMACIES AND RETAIL OUTLETS Module 8 - REGISTRATION OF PHARMACY PERSONNEL National Regulatory System, 19 | Status of vaccine indicators WHO-PAHO HARMONIZED TOOL NRS: NATIONAL REGULATORY RS: NATIONAL REGULATORY 1. NATIONAL REGULATORY SYSTEM 4. LICENCIAMIENTO DE PRODUCTORES VACCINES TOOL LI: LICENSING OF MANUFACTURERS, IMPORTERS, DISTRIBUTERS AND RETAILERS OF MEDICAL DEVICES AND DIAGNOSTICS FUNCTION 1: MARKETING AUTHORIZATION AND LICENSING ACTIVITIES 2. REGISTRATION AND MARKETING AUTHORIZATION 3. LICENSING ACTIVITIES 11. PHARMACEUTICAL PERSONNEL (HEALTH/REGULATORY? ? )
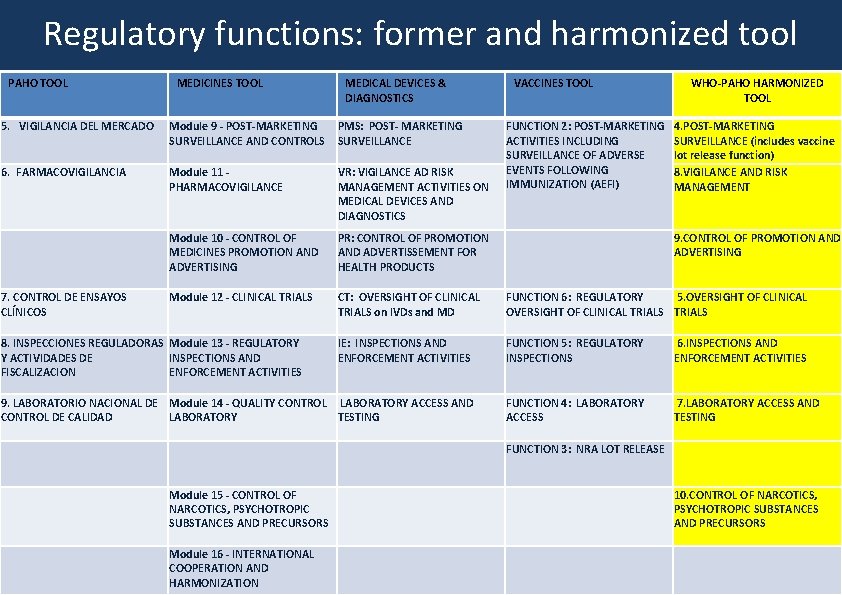
Regulatory functions: former and harmonized tool PAHO TOOL MEDICINES TOOL MEDICAL DEVICES & DIAGNOSTICS VACCINES TOOL 5. VIGILANCIA DEL MERCADO Module 9 - POST-MARKETING SURVEILLANCE AND CONTROLS PMS: POST- MARKETING SURVEILLANCE 6. FARMACOVIGILANCIA Module 11 - PHARMACOVIGILANCE VR: VIGILANCE AD RISK MANAGEMENT ACTIVITIES ON MEDICAL DEVICES AND DIAGNOSTICS Module 10 - CONTROL OF MEDICINES PROMOTION AND ADVERTISING PR: CONTROL OF PROMOTION AND ADVERTISSEMENT FOR HEALTH PRODUCTS 7. CONTROL DE ENSAYOS CLÍNICOS Module 12 - CLINICAL TRIALS CT: OVERSIGHT OF CLINICAL TRIALS on IVDs and MD FUNCTION 6: REGULATORY 5. OVERSIGHT OF CLINICAL TRIALS IE: INSPECTIONS AND ENFORCEMENT ACTIVITIES FUNCTION 5: REGULATORY INSPECTIONS 6. INSPECTIONS AND ENFORCEMENT ACTIVITIES 9. LABORATORIO NACIONAL DE Module 14 - QUALITY CONTROL LABORATORY ACCESS AND CONTROL DE CALIDAD LABORATORY TESTING FUNCTION 4: LABORATORY ACCESS 7. LABORATORY ACCESS AND TESTING FUNCTION 3: NRA LOT RELEASE Module 15 - CONTROL OF NARCOTICS, PSYCHOTROPIC SUBSTANCES AND PRECURSORS 8. INSPECCIONES REGULADORAS Module 13 - REGULATORY Y ACTIVIDADES DE INSPECTIONS AND FISCALIZACION ENFORCEMENT ACTIVITIES National Regulatory System, Module 16 - INTERNATIONAL | 20 Status of vaccine indicators COOPERATION AND HARMONIZATION FUNCTION 2: POST-MARKETING ACTIVITIES INCLUDING SURVEILLANCE OF ADVERSE EVENTS FOLLOWING IMMUNIZATION (AEFI) WHO-PAHO HARMONIZED TOOL 4. POST-MARKETING SURVEILLANCE (includes vaccine lot release function) 8. VIGILANCE AND RISK MANAGEMENT 9. CONTROL OF PROMOTION AND ADVERTISING 10. CONTROL OF NARCOTICS, PSYCHOTROPIC SUBSTANCES AND PRECURSORS

21 |
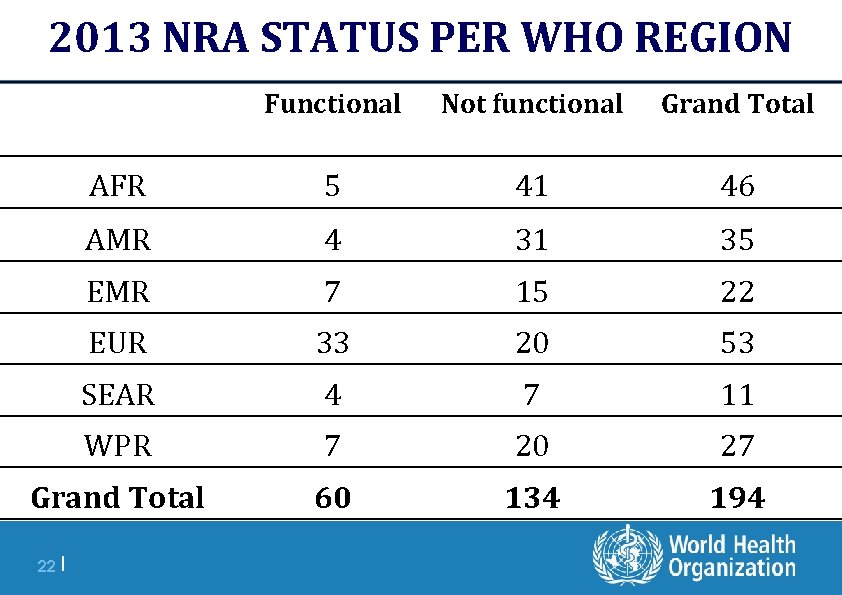
2013 NRA STATUS PER WHO REGION Functional Not functional Grand Total AFR 5 41 46 AMR 4 31 35 EMR 7 15 22 EUR 33 20 53 SEAR 4 7 11 WPR 7 20 27 Grand Total 60 134 194 22 |
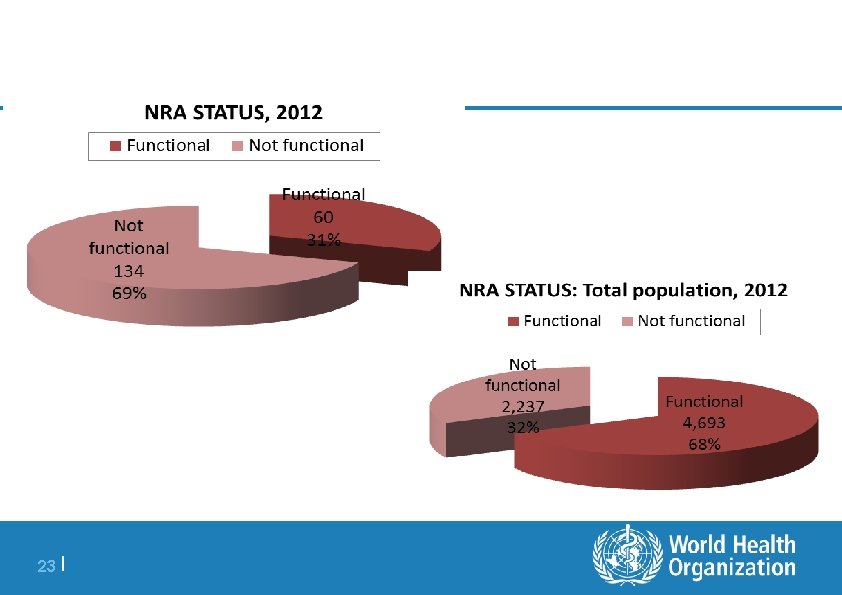

24 |

| 25 | GRACIAS MERCI
- Slides: 25