Drug Design Physicochemical Properties in relation to biological

Drug Design (Physicochemical Properties in relation to biological activities) By Nohad A Atrushi 9/3/2017

Drug – Receptor Interactions forces The some binding forces involved as when simple molecules interact will be involved in interaction of drug with a functional or organized group of molecules , which may be called " biologic receptor site ".

# The forces involved in Drug-Receptor interaction are collected in table 2 -8 : • In most cases, it is desirable that the drug leaves the receptor site when the concentration decrease in the extracellular(EC) fluid. therefore , most useful drugs are held to their receptors by ionic or weaker bonds , when relatively long lasting or irreversible effects are desirable (ex. antibacterial, anticancer ) , Drugs that form covalent bonds are effective & useful.


• The alkylating agents, such as the nitrogen mustards (ex. mechlorethamine ) used in cancer chemotherapy given an example of drugs that act by formation of covalent bond. • • These are believed to form the reactive immonium ion intermediate, which alkylate & thereby link together proteins nucleic acids , preventing their normal participation in cell division.
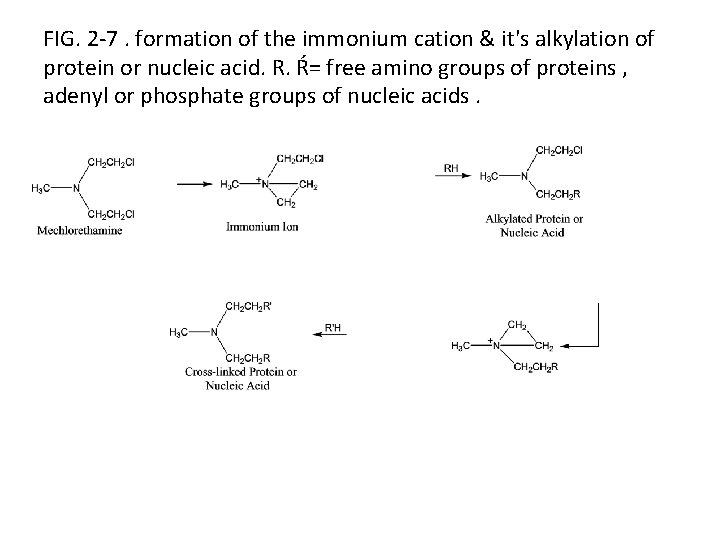
FIG. 2 -7. formation of the immonium cation & it's alkylation of protein or nucleic acid. R. Ŕ= free amino groups of proteins , adenyl or phosphate groups of nucleic acids.

• Covalent bond formation between drug and receptor is the basis of "Baker's concept" of active site directed irreversible inhibition. Enzymes inhibitors has supported experimental this concept. • Compounds studied posses appropriate structural features for reversible & highly selective association with an enzyme.
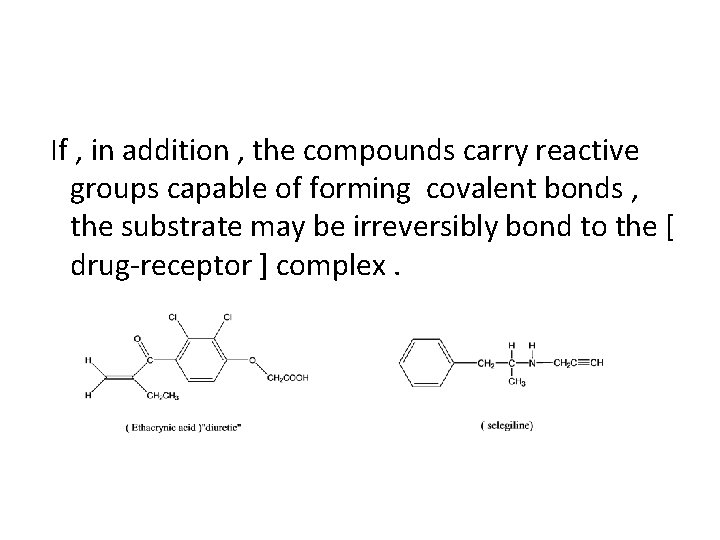
If , in addition , the compounds carry reactive groups capable of forming covalent bonds , the substrate may be irreversibly bond to the [ drug-receptor ] complex.

The diuretic drug , ethacrynic acid is an α , β unsaturated ketone , thought to act by covalent bond formation with sulfhydryl groups (thiol group) of ion transport systems in the renal tubules. Another examples of drugs which covalently binds to the receptor is selegiline which is an inhibitor of monoamino oxidase B (MAOB inhibitor).

Other examples of covalent bond formation between drug & biologic receptor site include the reaction of arsenicals & mercurials with cysteine thiol groups (- SH ), also the acylation of bacterial cell wall(C. W) constituents by penicillin & the phosphorylation of the serine hydroxyl moiety at the active site of cholinesterase by the organic phosphate. The most important to patient is to know that : - is desirable that most drug effects be reversible. for this to occur , relatively weak forces must be involved in the [drug – receptor]complex, at the same aim be strong enough that other binding site will not competitively deplete the site of action.

H-bond : many drugs posses groups , such as carbonyl , Hydroxyl , amine & imino which the first structure capabilities of acting as acceptor or donors in the formation of Hbond. * Vander waal’s forces: - are attractive forces created by the polarizability of molecules & are exerted when any two uncharged atoms approach each other very closely. * Hydrophobic bond : - is a concept used to explain attraction between nonpolar regions of the receptor & the drug.

Physical Properties (water and lipid solubility) • Partition coefficient – lipophilic vs. hydrophilic character of drug – determines water solubility of drug substances – affects drug distribution – confers target-drug binding interactions 12

Water Solubility 13

Water Solubility Ø Given that we are ~75% water, the solubility of a drug in water directly affects the route of administration, distribution, and elimination (ADME). Ø The most important two key factors that influence this are: § Hydrogen bonding: more H-bonds => solubility § Ionisation: dissociable ions => solubility 14

Predicting Water Solubility Ø Empirical Approach Ø Analytical Approach 15

Predicting Water Solubility Empirical Approach Ø Lemke has developed an approach to predicting water solubility based upon the “solubilising potential” of various functional groups, versus the number of carbons Ø Given that most drugs are polyfunctional, the second column is most relevant 16




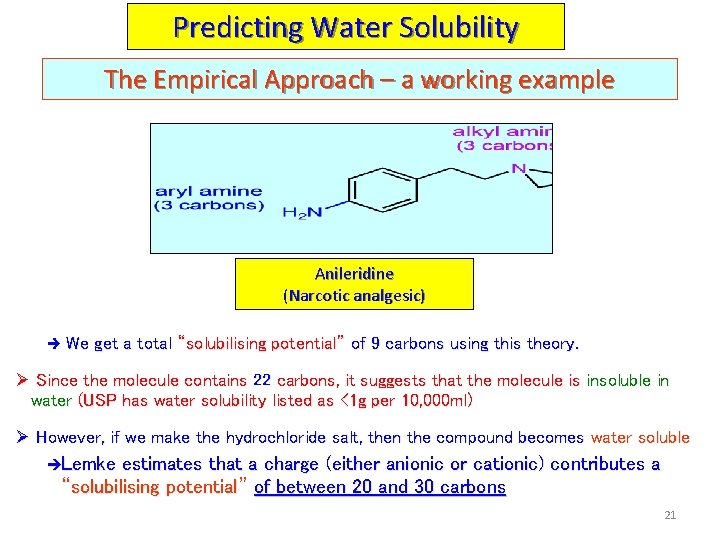
Predicting Water Solubility The Empirical Approach – a working example Anileridine (Narcotic analgesic) è We get a total “solubilising potential” of 9 carbons using this theory. Ø Since the molecule contains 22 carbons, it suggests that the molecule is insoluble in water (USP has water solubility listed as <1 g per 10, 000 ml) Ø However, if we make the hydrochloride salt, then the compound becomes water soluble èLemke estimates that a charge (either anionic or cationic) contributes a “solubilising potential” of between 20 and 30 carbons 21

Predicting Water Solubility Analytical Approach Ø The alternative approach for predicting water solubility utilises the “log. P” of molecules Ø Essentially, log. P is a measure of lipophilicity (hydrophobic) properties of a molecule Ø It is determined by measuring the “partition coefficient” between water and octanol for a given molecule (i. e. the solubility of the compound in octanol versus the solubility of the compound in water) Ø Octanol is used as a mimic of the characteristics of a lipid membrane (polar at one end, long hydrocarbon chain at the other) ØLog. P is calculated by adding the contributions from each functional group in the molecule Ø A hydrophobic substituent constant π has been assigned to most organic 22 functional groups, such that Log. P = ∑ π (fragments)







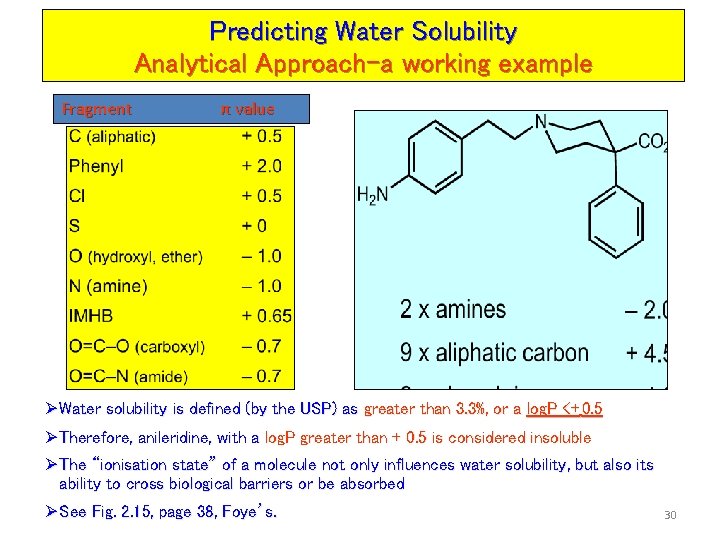
Predicting Water Solubility Analytical Approach-a working example Fragment π value Ø Water solubility is defined (by the USP) as greater than 3. 3%, or a log. P <+ 0. 5 Ø Therefore, anileridine, with a log. P greater than + 0. 5 is considered insoluble Ø The “ionisation state” of a molecule not only influences water solubility, but also its ability to cross biological barriers or be absorbed Ø See Fig. 2. 15, page 38, Foye’s. 30

Stereochemistry and Biological Activity 31


Stereochemistry and Biological Activity Ø The physicochemical properties of a drug are not only influenced by which functional groups are present, but also by the spatial arrangement of groups. Ø The spatial arrangement of groups is especially important when dealing with biological systems, since receptors are susceptible to the shape of a molecule. Ø Stereoisomers contain the same number and kinds of atoms, the same arrangement of bonds, but a different spatial arrangement of atoms. Ø A carbon atom with four different substituents is an asymmetric molecules. Ø Stereochemistry is primary: – Optical isomerism (Enantiomers, Diastereomers) – Geometric isomerism – Conformational isomerism 33

Designation of stereoisomerism Ø Cahn, Ingold & Prelog (1956) devised a system of nomenclature for stereoisomer Ø Prioritise atoms around a chiral centre, based upon the atomic weight of the atom Ø Once you have assigned priority from 1 (= highest) to 4 (= lowest), then “look from the chiral centre towards the lowest priority and count from 1 to 3 è If you count clockwise it is “R” è If you count anticlockwise it is “S” 34
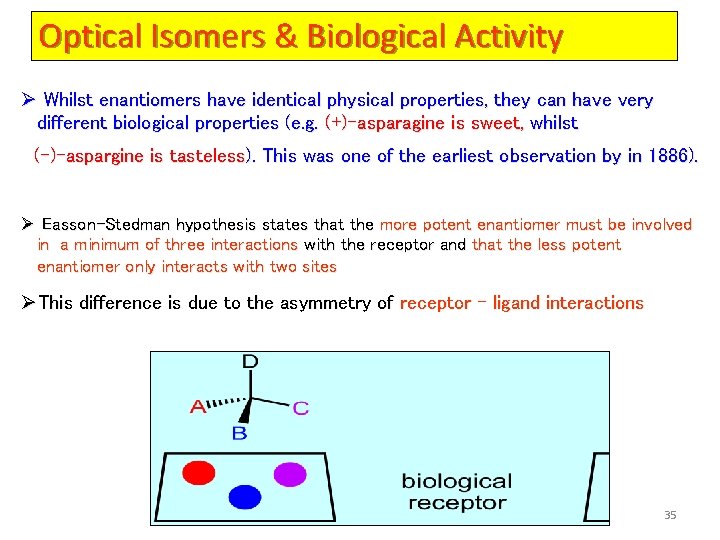
Optical Isomers & Biological Activity Ø Whilst enantiomers have identical physical properties, they can have very different biological properties (e. g. (+)-asparagine is sweet, whilst (–)-aspargine is tasteless). This was one of the earliest observation by in 1886). Ø Easson-Stedman hypothesis states that the more potent enantiomer must be involved in a minimum of three interactions with the receptor and that the less potent enantiomer only interacts with two sites ØThis difference is due to the asymmetry of receptor – ligand interactions 35
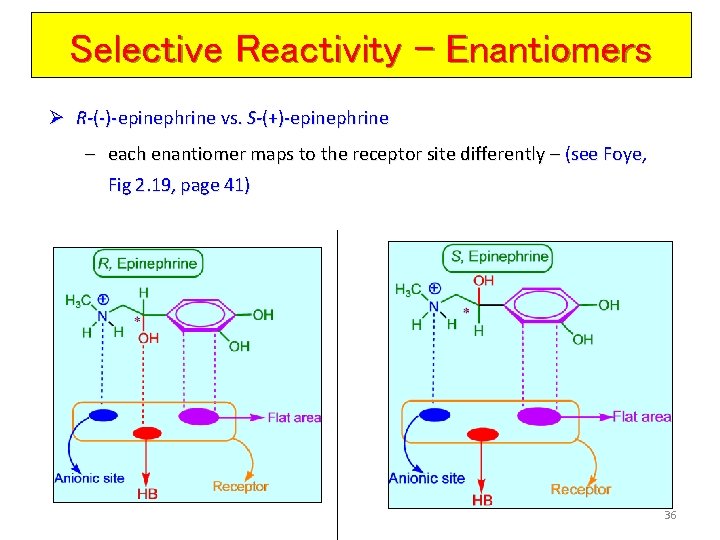
Selective Reactivity - Enantiomers Ø R-(-)-epinephrine vs. S-(+)-epinephrine – each enantiomer maps to the receptor site differently – (see Foye, Fig 2. 19, page 41) 36

Diastereomers – Asymmetric Centres Diastereomers: Ø Stereoisomers with the same number and kinds of atoms, but in a different spatial arrangement (any stereoisomers compound that is not an enantiomer) Ø These compounds have different physical and chemical properties Ø These arise from compounds possessing two or more asymmetric centres Ø Consider isomethadol • • • 2 asymmetric carbons 4 isomers (2 pairs of enantiomers) only the (3 S, 5 S)-isomer has analgesic activity. 37
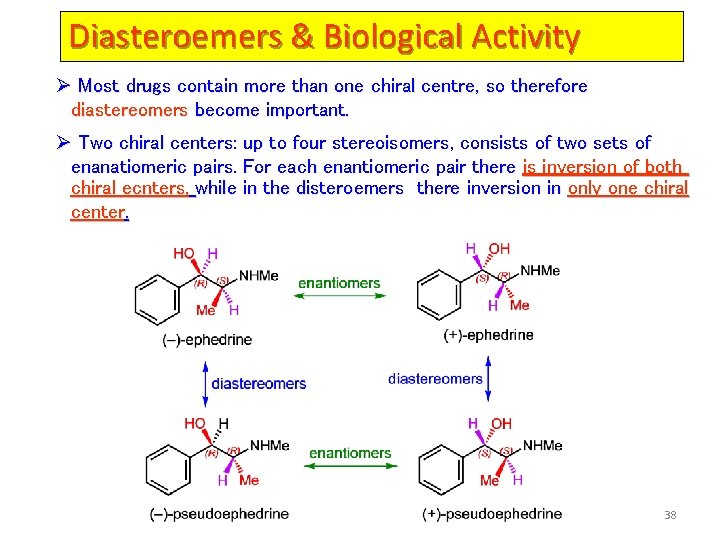
Diasteroemers & Biological Activity Ø Most drugs contain more than one chiral centre, so therefore diastereomers become important. Ø Two chiral centers: up to four stereoisomers, consists of two sets of enanatiomeric pairs. For each enantiomeric pair there is inversion of both chiral ecnters, while in the disteroemers there inversion in only one chiral center. 38

Enantiomeric Pair Differences • Some examples – Isomethadol (cf methadone) - analgesic – Acetylisomethadol - transformation induced – Etomidate - nonbarbiturate hypnotic – Ibuprofen - NSAID/Analgesic – Naproxen - NSAID/Analgesic – Verapamil - Ca channel blocker – Warfarin - anticoagulant 39
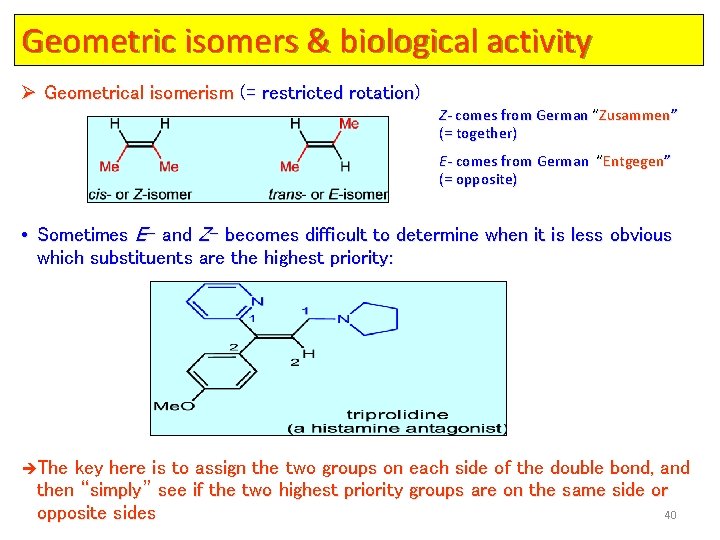
Geometric isomers & biological activity Ø Geometrical isomerism (= restricted rotation) Z- comes from German “Zusammen” (= together) E- comes from German “Entgegen” (= opposite) • Sometimes E- and Z- becomes difficult to determine when it is less obvious which substituents are the highest priority: èThe key here is to assign the two groups on each side of the double bond, and then “simply” see if the two highest priority groups are on the same side or 40 opposite sides

Geometric isomers & biological activity Ø cis/trans isomers have different physical properties distribution in biologic system varies – generally leads to distinct biological activity • But … difficult to correlate activity differences with stereochemistry alone – eg different p. Kas of isomers => different levels of ionisation and hence => differing penetration or absorption 41
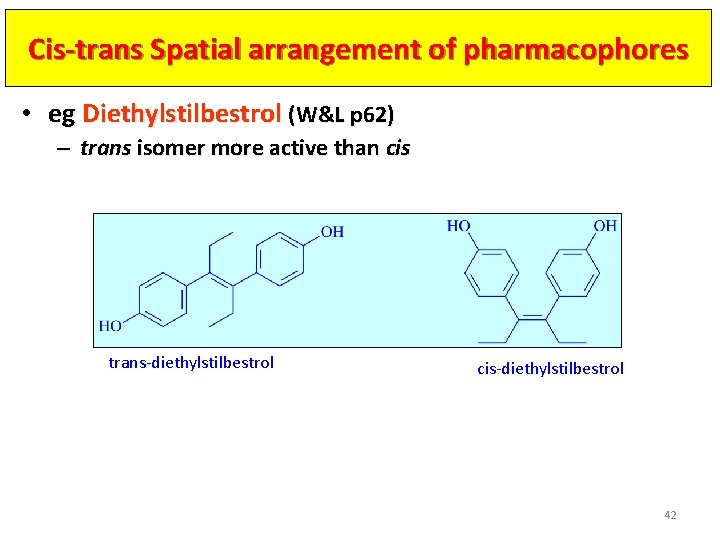
Cis-trans Spatial arrangement of pharmacophores • eg Diethylstilbestrol (W&L p 62) – trans isomer more active than cis trans-diethylstilbestrol cis-diethylstilbestrol 42
Conformational Isomers Ø Conformational isomerism - Eliel’s definition Ø “. . . the no identical spatial arrangement of atoms in a molecule, resulting from rotation about one or more single bonds. ” Ø Involves both acyclic and cyclic drug molecules – acyclic - flexible - Newman and sawhorse models – cyclic - rigid - chair/boat model of conformers Ø cyclic molecules of more interest medicinally 43
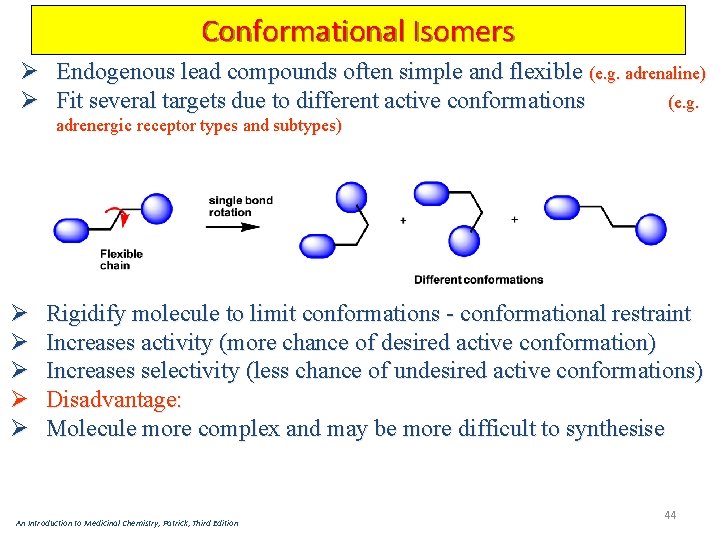
Conformational Isomers Ø Endogenous lead compounds often simple and flexible (e. g. adrenaline) Ø Fit several targets due to different active conformations (e. g. adrenergic receptor types and subtypes) Ø Ø Ø Rigidify molecule to limit conformations - conformational restraint Increases activity (more chance of desired active conformation) Increases selectivity (less chance of undesired active conformations) Disadvantage: Molecule more complex and may be more difficult to synthesise An Introduction to Medicinal Chemistry, Patrick, Third Edition 44
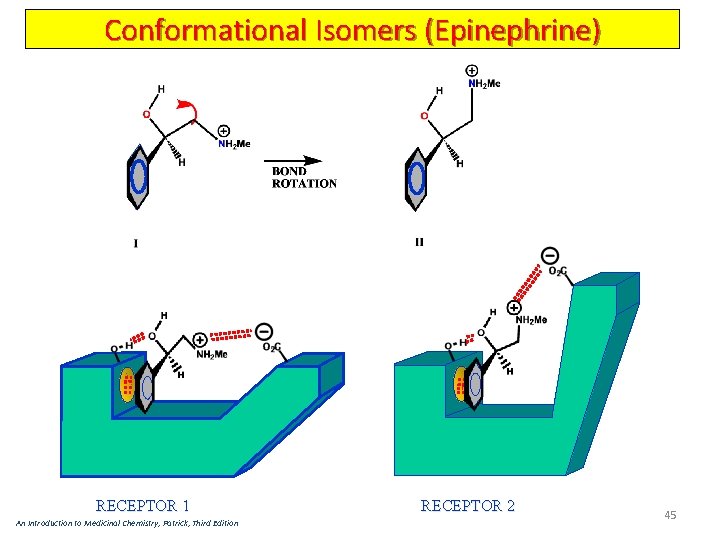
Conformational Isomers (Epinephrine) RECEPTOR 1 An Introduction to Medicinal Chemistry, Patrick, Third Edition RECEPTOR 2 45
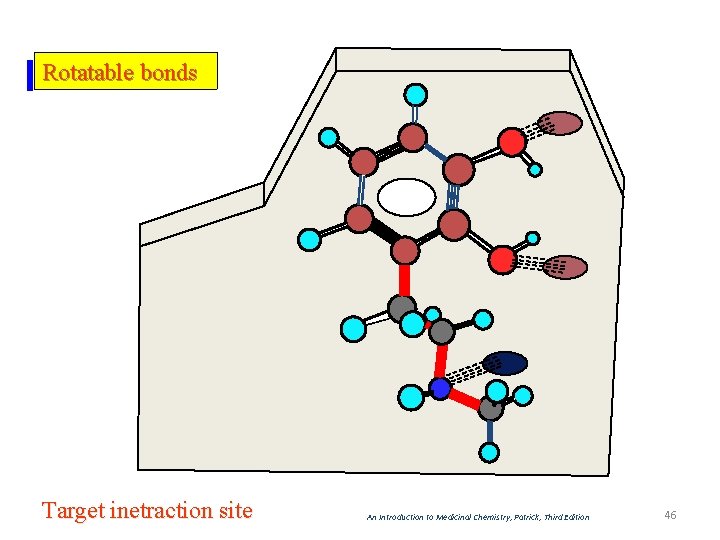
Rotatable bonds Target inetraction site An Introduction to Medicinal Chemistry, Patrick, Third Edition 46
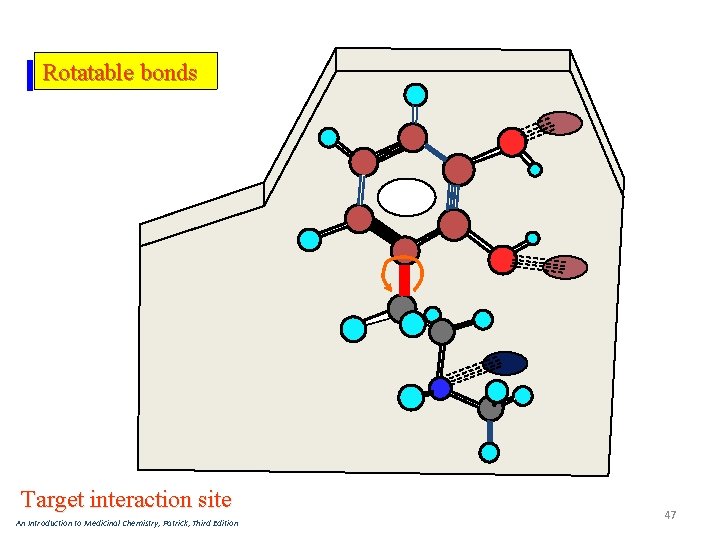
Rotatable bonds Target interaction site An Introduction to Medicinal Chemistry, Patrick, Third Edition 47
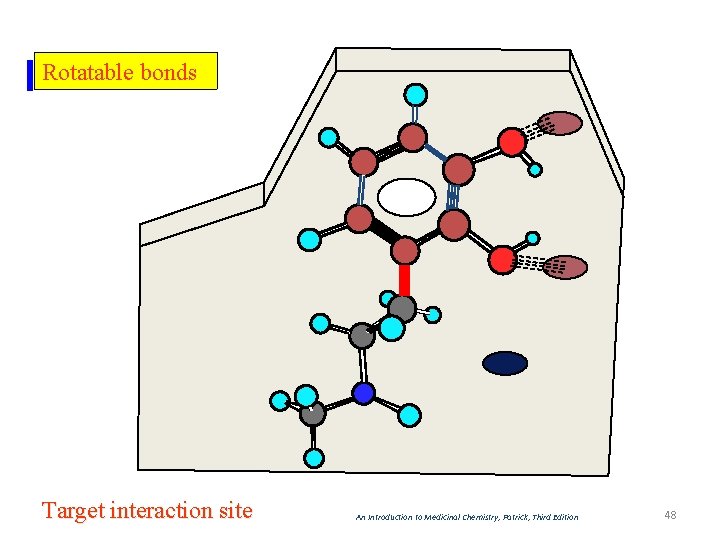
Rotatable bonds Target interaction site An Introduction to Medicinal Chemistry, Patrick, Third Edition 48

Rigidification Methods - Introduce rings ØBonds within ring systems are locked and cannot rotate freely An Introduction to Medicinal Chemistry, Patrick, Third Edition 49

Isosterism and Bioisosterism 50

Isosterism and Bioisosterism Ø A poor “drug profile” includes issues such as bioavailability, unwanted side effects, inability to cross biological barriers, poor pharmacokinetics. ØThese undesirable features could be due to specific functional groups in the molecule. Ø Modify this molecule to reduce these undesirable features WITHOUT losing the desired biological activity with other groups having similar properties is known as ISOSTERIC or BIOISOSTERIC replacement. ØIn 1919 Langmuir first developed the concept of isosterism to describe the similarities in physical properties among atoms (same number of valence electrons O and S). Ø In 1925 Grimm developed his hydride displacement law (illustration of similar physical properties among closely related functional groups) ØThus, NH 2 is considered to be isosteric to OH, SH, CH 3) 51

Grimm’s isosteres - 1925 Ø Descending diagonally from left to right in the table H atoms are added to maintain the same number of valence electrons for each group of atoms within a column. Ø Each member of a vertical group is isoelectronic 52
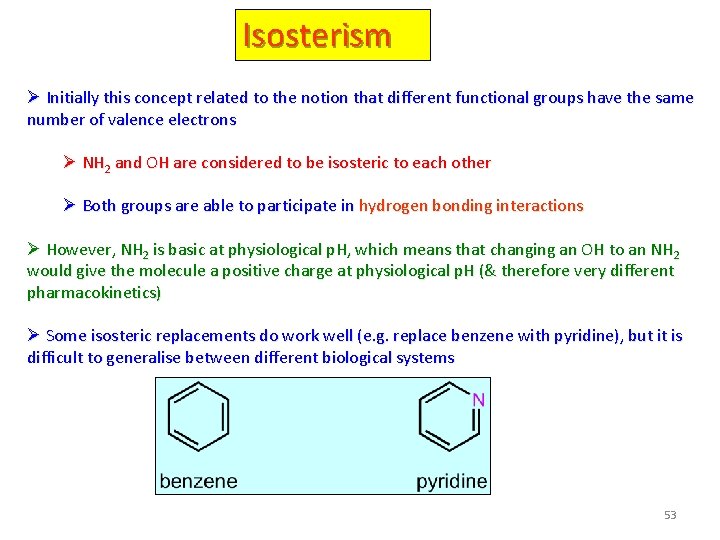
Isosterism Ø Initially this concept related to the notion that different functional groups have the same number of valence electrons Ø NH 2 and OH are considered to be isosteric to each other Ø Both groups are able to participate in hydrogen bonding interactions Ø However, NH 2 is basic at physiological p. H, which means that changing an OH to an NH 2 would give the molecule a positive charge at physiological p. H (& therefore very different pharmacokinetics) Ø Some isosteric replacements do work well (e. g. replace benzene with pyridine), but it is difficult to generalise between different biological systems 53
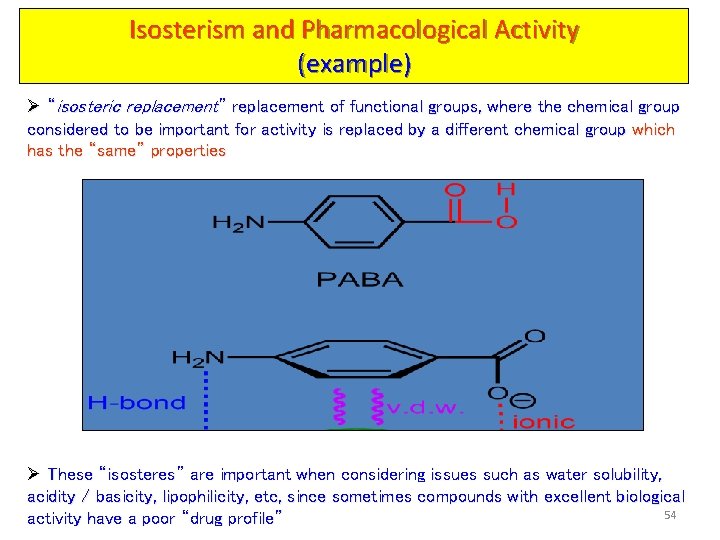
Isosterism and Pharmacological Activity (example) Ø “isosteric replacement” replacement of functional groups, where the chemical group considered to be important for activity is replaced by a different chemical group which has the “same” properties Ø These “isosteres” are important when considering issues such as water solubility, acidity / basicity, lipophilicity, etc, since sometimes compounds with excellent biological 54 activity have a poor “drug profile”

Bio-isosterism Ø This process attempts to overcome the limitations of isosteric replacement by considering not just the similarity in chemical structure between functional groups, but to also look at the biological effects • Friedman definition “bio-isosteres are functional groups or molecules that have chemical and physical similarities producing broadly similar biological properties ” • Burger definition: “bio-isosteres are compounds or groups that possess near equal molecular shape and volumes, and with exhibit similar physical properties such as hydrophobicity”. Ø The key point is that the same pharmacological target is influenced by bioisosteres as agonist or antagoinist. ØThere are two general types of “bio-isosteres” • Classical and non-classical 55
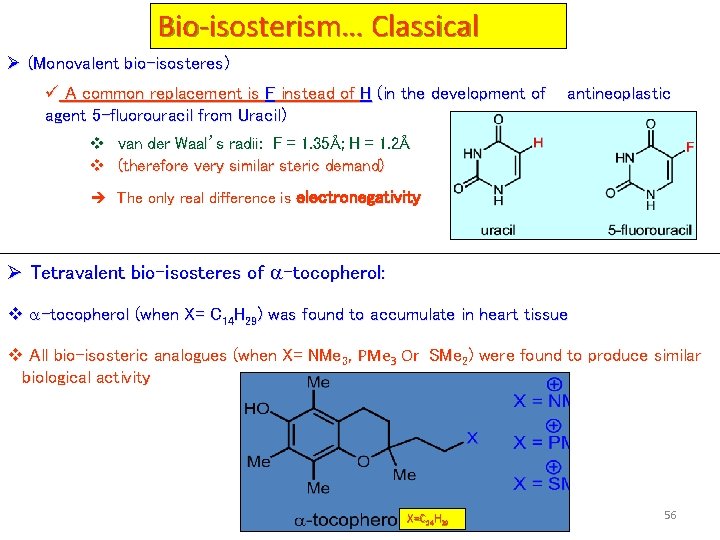
Bio-isosterism… Classical Ø (Monovalent bio-isosteres) ü A common replacement is F instead of H (in the development of agent 5 -fluorouracil from Uracil) antineoplastic v van der Waal’s radii: F = 1. 35Å; H = 1. 2Å v (therefore very similar steric demand) è The only real difference is electronegativity Ø Tetravalent bio-isosteres of -tocopherol: v -tocopherol (when X= C 14 H 29) was found to accumulate in heart tissue v All bio-isosteric analogues (when X= NMe 3, PMe 3 Or SMe 2) were found to produce similar biological activity X=C 14 H 29 56
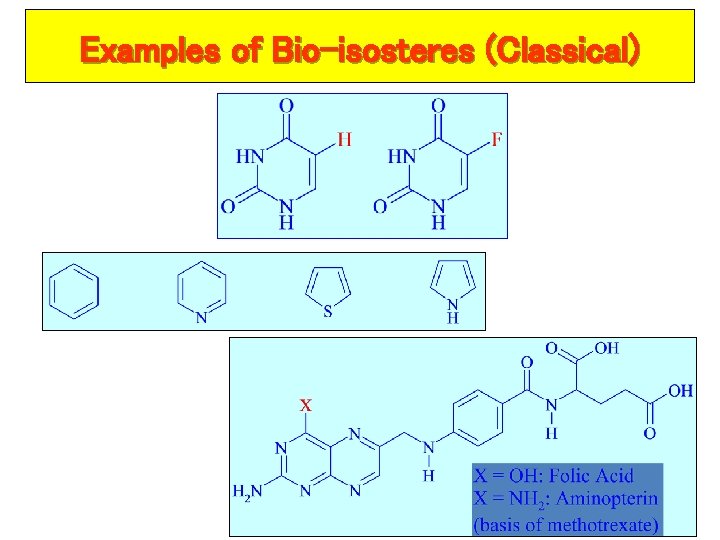
Examples of Bio-isosteres (Classical) 57
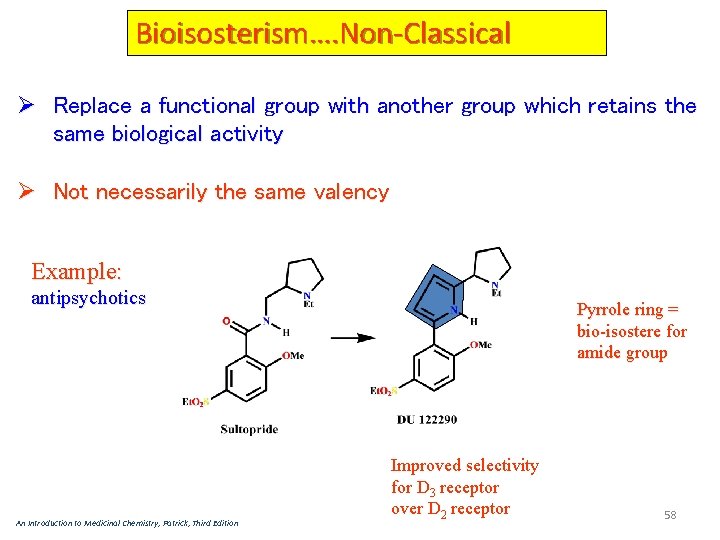
Bioisosterism…. Non-Classical Ø Replace a functional group with another group which retains the same biological activity Ø Not necessarily the same valency Example: antipsychotics An Introduction to Medicinal Chemistry, Patrick, Third Edition Pyrrole ring = bio-isostere for amide group Improved selectivity for D 3 receptor over D 2 receptor 58

Take Home? ? Ø Relationships of Functional Groups to Pharmacological Activity (SARs) Ø Physiochemical Properties of drug molecules § § § Acid - base properties of drug molecules p. H and p. Ka (Henderson-Hassalbach Equation) ionisation and absorption Water and lipid Solubility (hydrogen and ion bonds) Predicting water solubility • Empirical approach • Analytical Approach § Partition coefficient • absorption/distribution Ø Stereochemistry and pharmacological activity Ø Optical isomerism (enantiomers and distereomers) Ø Conformation isomers Ø Geometric isomers (cis and trans) Ø Isosterism and Bioisosterism Ø drug design 59

ANY QUESTIONs? 60
- Slides: 60