DrNahlaa Khalifa King AbdulAziz University Faculty of Applied
Dr-Nahlaa Khalifa

King Abdul-Aziz University Faculty of Applied Medical Science Clinical Nutrition Department – Third Year FOOD ANALYSIS Moisture and Total Solids Analysis

Moisture and Total Solids Analysis 1. Moisture is an important analytical procedure that can performed on a food product, is an assay the amount of moisture 1. The dry matter that remain after moisture removal is total solids.

Moisture and Total Solids Analysis 1. 2. 3. 4. 5. Introduction Oven Drying Methods. Distillation Procedures. Chemical Methods. Physical Methods

Introduction A. Importance of Moisture Assay B. Forms of Water in Foods C. Sample Collection and Handling

A. Importance of Moisture Assay (1) Moisture is a quality factor in the preservation of some products, such as dehydrated vegetables and fruits, dried milks, (2) Moisture is used as a quality factor for such as jams, jellies, sugar some food product, syrups… , to prevent sugar crystallization. (3) Reduced moisture is used for convenience in packaging ping of concentrated milk, fruit juice… (4) Moisture (or solids) content is often specified in compositional standards, such as Cheddar cheese must be < 39% moisture, enriched flour must be < 15% moisture.

B. Forms of Water in Foods (1) Free water (2) ADSORBED WATER (3) Water of hydration

1. Free water WATER CAN ACT AS A SOLVENT FOR MOLECULAR DISPERSION. WHEN WATER EXISTS AS A SOLVENT, IT CAN BE THOUGHT OF AS FREE WATER, NOT BOUND TO ANY THING AND THEREFORE AVAILABLE FOR CHEMICAL REACTIONS, MICROBIAL GROWTH, AND PHYSICAL CHANGES.

2. Adsorbed � WATER ADSORBED (CHEMICALLY BOUND…. NOT ABSORBED LIKE A water SPONGE DOES) CAN EXIST IN MONO OR POLY MOLECULAR LAYERS ON THE INTERNAL OR EXTERNAL SURFACES OF MOLECULES IN THE FOOD (IN THE CELL o WALLS). Proteins and starches have strong tendencies forming such interactions with water (held tightly to protein). o Because fat is hydrophobic, it does not usually interact with the water phase in food, however, there are phospholipids and lipoproteins that associate at lipidwater interfaces and create emulsion.

3. Water of hydration o Sometimes, water can be chemically bound to certain compounds like lactose (forming a stable monohydrate), also some salts such as Na So 4. 10 H 2 O , proteins and polysaccharides (hydrogen bonding). o This “water of hydration” may vary from 0. 5 to 30% of the total water present. This water is particularly difficult to remove for analytical purposes. 2

C. Sample Collection and Handling o How to minimize inadvertent moisture losses or gains during sample collection and handling? Precautions must be taken to minimize inadvertent moisture during sample collection and handling: (1) Any exposure of a sample to the open atmosphere should be as short as possible (2) Any heating of a sample by friction during grinding should be minimized. (3) Headspace in the sample storage container should be minimal minimized

Moisture content in food ■ Cucumber 96% ■ Whole milk 87% ■ Yogurt 89% ■ Potato 80% ■ Raisin 18% ■ Roasted peanut < 2% ■ Vegetable oil ~0%

WHAT WAS PROXIMATE ANALYSIS? n n Ash, Protein, Fat, Carbohydrate Sum of other Proximate + Moisture = ~100%

Oven Drying Methods 1. General Information. 2. Forced Draft Oven. 3. Vacuum Oven. 4. Microwave Analyzer. 5. Infrared Drying.

1. General (1) REMOVALInformation OF MOISTURE-- TWO-STAGE PROCESS n Liquid products: predried over a steam bath before drying in an oven dried. n Solid product: Breads and grain are often air dried, then ground and oven dried (2) Decomposition of other food constituents Carbohydrates decompose at 100 C according to the reaction: p C 6 H 12 O 6 6 C + 6 H 2 O n Sucrose hydrolysis can result in utilization of moisture. n Loss of volatile constituents: acetic, propionic, and butyric acids;alcohols, esters, aldehyde… n Oxidation of unsaturated fatty acids n

1. General Information (3) TEMPERATURE CONTROL (4) Types of pans n n The AOAC international moisture pan is about 5. 5 cm in diameter with an insert cover Disposable aluminum foil pans and glass fiber discs for cover, which prevent spattering and allow the surface to breath (5) Handling and preparation of pans n n Use only tongs to handle any pan. Pans must be vacuum oven dried for 3 hr before use. Store dry moisture pans in a functioning desiccators. The glass fiber covers should be dried for one hour before use (6) Calculations

2. Forced Draft Oven WEIGHED INTO A ●THE SAMPLE IS RAPIDLY PREDRIED MOISTURE PAN COVERED AND PLACED IN THE OVEN FOR A SELECTED TIME IF o A STANDARDIZED forced draft oven. METHOD is used with or without a NO EXITS. steam bath predrying treatment to determine the solids content of fluid milks (AOAC method 990. 19, 990. 20) o Weight and reweight the dried sample and pan until two successive weighing taken 30 min apart agree within a specified limit, for example, 0. 1 -02 mg for a 5 g sample. o Be aware of sample transformation Browning reaction n Lipid oxidation n

q CONVENTIONAL OVEN 3. Temperature *THE GREATEST TEMPERATURE VARIATION EXISTS. control *HOT AIR SLOWLY CIRCULATES WITHOUT THE AID OF A FAN. *AIR MOVEMENT IS OBSTRUCTED FURTHER BY PANS PLACED IN THE OVEN. *A 10 CO TEMPERATURE DIFFERENTIAL o Forced draft Oven ACROSS A CONVENTIONAL OVEN IS NOT n Have the least temperature UNUSUAL. differential across the interior of all ovens, usually not o Vacuum greater. Oven than 1 o C. n Glass panel in the door: the glass is a heat sink. n Air inlet and discharge design: mounted up and bottom. n To minimize cold spots and to

4. Microwave Analyzer with microprocessor o Microwave analyzer is specified in the AOAC International procedures for total solids analysis of processed tomato products, and moisture analysis of meat and poultry products (AOAC Method 985. 14). o Temperature used depends on the product, such as 70 C for fruits and other high-sugar products. o The sample must be uniform, appropriate size. o The sample must be centrally located and evenly distributed. o Microwave drying provides a fast, accurate method to analyze many foods for moisture content.

5. Infrared Drying o Infrared drying involves penetration of heat into the sample being dried, which can significantly shorten the required drying time , to 10 -25 min (compared with conventional oven). o Equipped with forced ventilation to remove moisture air and an analytical balance to read moisture content directly. o Factors must be controlled include distance of the infrared source from the dried material and thickness of the sample. o Suitable for qualitative in-process use.

6. Calculations
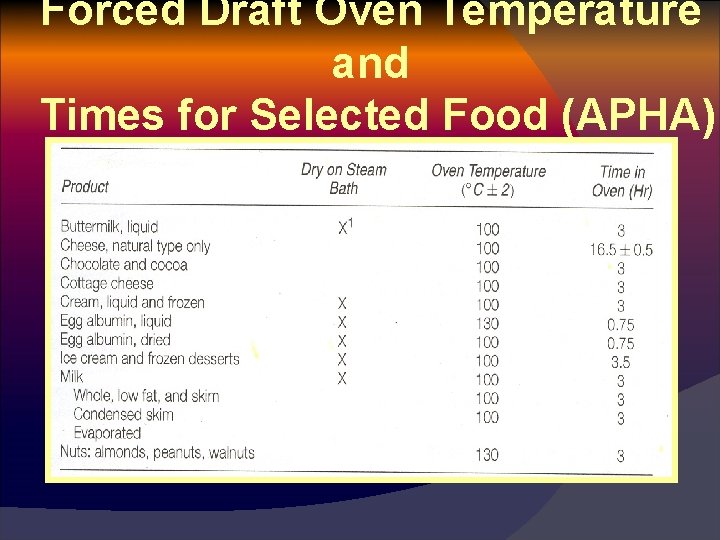
Forced Draft Oven Temperature and Times for Selected Food (APHA)

o Rapid Heating: Temperature distribution problems. o Good accuracy: Programmable temperature profile. o Expensive: Limited sample numbers

q Rapid drying, q Weights by difference, q Wide temperature range, q Programmable temperature profile, q Expensive, getting cheaper, q Limited sample

Distillation Procedures 1. Overview: Ø AOAC approved technique for moisture analysis of spices (AOAC method 986. 21), cheese (AOAC method 868. 19), and animal feeds (AOAC method 925. 04). Ø 2. Give good. Distillation accuracy and precision for nuts, Reflux with Immiscible oils, soaps, and waxes. Solvent: o Use a solvent less dense than water -- toluene or xylene. o Use a solvent more dense than water -tetrachlorethylene

Apparatus for reflux distillation of moisture from a food (Bidwell-Sterling o Observe a hazymoisture cloud risingtrap) in the distillation flask emulsion of water in toluene o Sources of error Formation of emulsions that will not break n Clinging of water droplets to dirty apparatus n Decomposition of the sample with production of water n

Chemical Methods (Karl Fischer Titration) o Determination of water in many low- moisture foods, such as dried fruits and vegetables (AOAC Method 967. 19 E-G), candies, chocolate (AOAC Method 977. 10), roasted coffee, oils and fats (AOAC Method 984. 20), or any lowmoisture food high in sugar or protein. o The method is quite rapid, accurate, and uses no heat.

Karl Fischer Titration 1. One sample at a time, 2. Good accuracy, 3. Specific toward water, 4. Endpoint subjective, 1. 2. Volumetric Coulometric

1) Recommended for foods that have low moisture or samples that give erratic results such as high sugar and fatty foods. Karl Fischer Titration 2) The chemistry of the reaction (iodine reduction) 2 H 2 O + SO 2 + I 2 --> H 2 SO 4 + 2 HI Iodine reduction in the presence of sulfite. 3) The reaction was modified to include methanol and pyridine to ensure dissolution of all components in the mixture. 4) Dissolve sample in methanol and titrate with KFR (iodine : sulfite: pyridine; 1: 3: 10). 5) Each ml of KFR titrated equals 3. 5 mg water.

Physical Methods For Moisture 1. Electrical Methods. a. Dielectric Methods, b. Conductivity Method 2. 3. 4. 5. 6. Hydrometry. Refractometry Infrared Spectroscopy. Freezing Point. Water Activity

Physical Methods For 1. Electrical Methods Moisture • Measured against a known moisture content, • Conductivity – measures the strength of an electrical current passed through a food. • The lower the moisture the higher the resistance. • Capacitance – measures a change in resistance to an electrical current. A. Dielectric Method 1. Measuring the change in capacitance or resistance to an electric current passed through a sample 2. Limited to food systems that contain no more than 30 -35% moisture 3. The moisture determination in dielectric-type meters is based on the fact that the dielectric constant of water (80. 37 at 20 C)

B. Conductivity Method 1. The electrical resistance of wheat with 13% moisture is seven times as great as that with 14% moisture and 50 times that with 15% moisture 2 HYDROME TRY 1. Hydrometers use several principles and 2. 3. 4. 5. instruments, (solids suspended in liquids are buoyed by a force equal to the weight of water displaced). Weight per unit volume is determined by using a calibrated weight. Measures sugar, salt, alcohol, … etc. Accurate. specific gravity ( density) measurements.
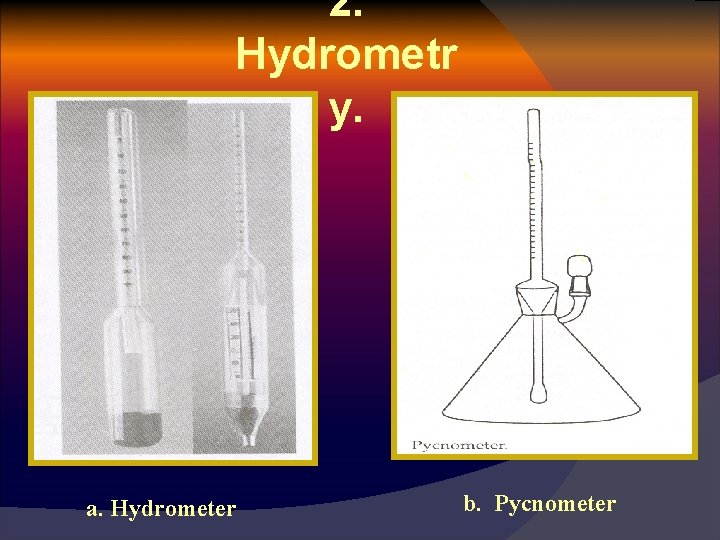
2. Hydrometr y. a. Hydrometer b. Pycnometer

3. Refractometry Ø All chemical compounds have an index of refraction. Ø The refractive index (RI) of an oil, syrup, or other liquid is a dimensionless constant. Ø RI varies with concentration of the compound, temperature, and wavelength of light. Ø Measure solid in syrup (AOAC Method 9. 32. 14 C), soluble solids in fruits and fruit product (AOAC Method 932. 12 ; 976. 20; 983. 17), approximate sugar concentration in food.

4. Infrared Spectroscopy o The use of near-infrared (NIR) bands (1400 -1450, 1920 -1950 nm) are characteristic of -OH stretch of the water molecule and can be used to determine the moisture of a food o The use of mid-infrared milk analyzers to determine fat, protein, lactose and total solids in milk (AOAC Method 972. 16)

5. Freezing Point ü The freezing point of milk is its most constant physical property. ü The freezing point varies within narrow limits. ü Hortver (H) is the surname of the inventor of the first freezing point apparatus. ü % H 2 O added = (0. 517 -T)/0. 517 x 100%. (AOAC Method 961. 07). ü FDA will reject all milk with freezing points above -0. 503 C (-0. 525 H).

6. Water Activity q Aw =P/P 0, Aw = ERH/100 Where: P = partial pressure of water above the sample P 0 = partial pressure of pure water at the same T. q Equilibrating a sample in a chamber held at constant relative humidity by means of a saturated salt solution q Chilled mirror technology v The water vapor in the headspace condenses on the surface of a mirror that is cooled in a controlled manner v The dew point is determined by the temperature at which condensation takes place --- relative humidity.

Comparison of Methods 1. PRINCIPLE 2. Nature of Sample 3. Intended Purposes

Comparison of Methods OVEN DRYING METHODS INVOLVE THE REMOVAL OF MOISTURE FROM THE SAMPLE AND THEN A WEIGHT DETERMINATION OF THE 1. Principle SOLIDS REMAINING TO CALCULATE THE MOISTURE CONTENT , DISTILLATION PROCEDURES ALSO INVOLVE A SEPARATION OF THE MOISTURE FROM THE SOLIDS, AND THE MOISTURE IS QUANTITIES DIRECTLY BY VOLUME. KARL FISCHER TITRATION IS BASED ON CHEMICAL REACTIONS OF THE MOISTURE PRESENT, REFLECTED AS THE AMOUNT OF TITRANT USED.

Comparison of Methods 1. Principle o Hydrometric methods are based on the relationship between specific gravity and moisture content. o The RI method is based on how water in a sample affects the refraction of light. o Near-infrared analysis of water in foods is based on measuring the absorption a wavelengths characteristic of the molecule vibration in water o Freezing point is a physical property of milk that is changed by a change in solute concentration or water added

Comparison of Methods Most food will tolerate oven drying at high 2. o. Nature of Sample temperature. o Vacuum oven drying at reduced temperature may overcome some problem for some foods, such as loss of volatiles, undergo chemical reaction at high temperature. o Karl Fischer titration is used for foods with very low in moisture or high in fats and sugars. o The use of a pycnometer, hydrometer, and refractometer requires liquid samples

Comparison of Methods 3. Intended Purposes ►FOR QUICKLY, QUALITY CONTROL PUR ►FOR REGULATORY AND NUTRITION LABE

Thank you
- Slides: 43