Drill n Determine the name of the following

























- Slides: 27

Drill n Determine the name of the following Molecules? 1. CO 2 2. CO

Objective n The student will be able to: n Complete practice problems in order to name Acids and Bases.

SAT/HSA Enrichment n There are … Carbon atoms represented by a. b. c. d. 3 Mg(HCO 3)2 2 3 5 6

Quiz n Naming Molecular Compounds!

Acid Demonstration n Demo I n Pour approximately 50 m. L of vinegar into 100 m. L beaker. n Take approximately 2 grams of sodium bicarbonate (baking soda) and add it to the vinegar.

Acid Demonstration n Demo II n Place 2 grams of mossy zinc in a large test tube. n Add approximately 10 m. L of 3 M hydrochloric acid to the test tube. n Immediately loosely stopper the test tube. n Light a wooden splint. n Remove the stopper and place wooden splint near the mouth of the test tube.

Acid Demonstration n Demo III n Pour 250 m. L of 0. 1 Na. OH into a 1000 m. L Erlenmeyer flask. n Add a few drops of phenolphthalein indicator to the solution. n Place solution on a magnetic stirrer. n While stirring, add 50 m. L of 3 M HCl solution.

Acid Demonstration n All the reactions involve acids and we will be studying many reactions that involve acids. n Therefore, you should know how to name and write the formulas of acids.

Naming and Writing Formulas for Acids and Bases How are they named?

Naming and Writing Formulas for Acids and Bases n Some ants can give painful stings when threatened or disturbed. Certain ant species called formicines have poison glands that produce venom containing formic acid. You will learn the names and formulas of some important acids such as formic acid.

Naming Acids n An acid is a compound that contains one or more hydrogen atoms and produces hydrogen ions (H+) when dissolved in water. Acids have various uses.

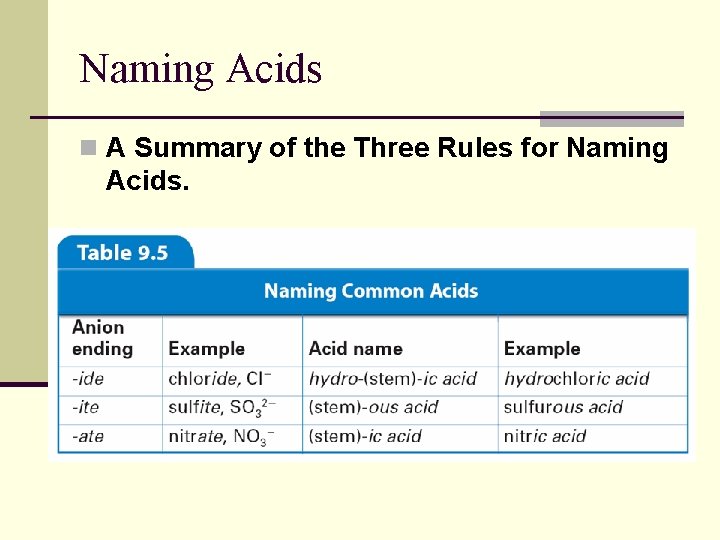
Naming Acids n Three rules can help you name an acid with the general formula Hn. X. n When the name of the anion (X) ends in -ide, the acid name begins with the prefix hydro-. n The stem of the anion has the suffix -ic and is followed by the word acid.

9. 4 Naming Acids n When the anion name ends in -ite, the acid name is the stem of the anion with the suffix ous, followed by the word acid.

9. 4 Naming Acids n When the anion name ends in -ate, the acid name is the stem of the anion with the suffix ic followed by the word acid.

Naming Acids n A Summary of the Three Rules for Naming Acids.

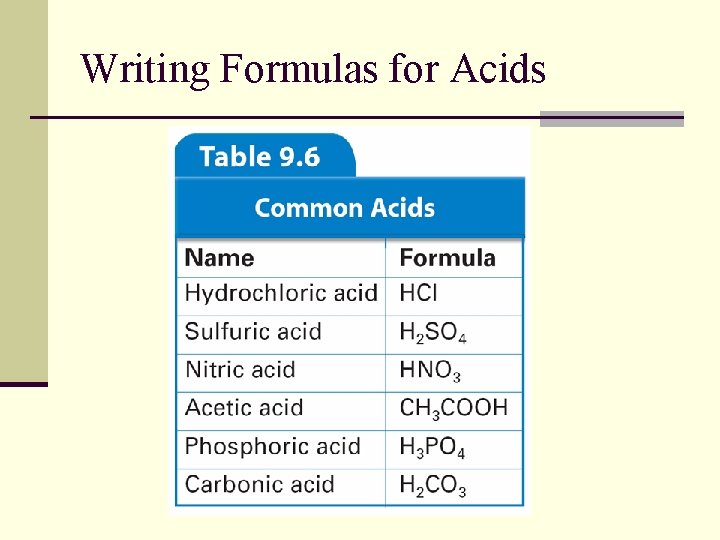
Writing Formulas for Acids n Use the rules for writing the names of acids in reverse to write the formulas for acids. n What is the formula for hydrobromic acid? Following Rule 1, hydrobromic acid (hydroprefix and -ic suffix) must be a combination of hydrogen ion (H+) and bromide ion (Br–). The formula of hydrobromic acid is HBr.

Writing Formulas for Acids

Names and Formulas for Bases n Bases are named in the same way as other ionic compounds—the name of the cation is followed by the name of the anion. n For example, aluminum hydroxide consists of the aluminum cation (Al 3+) and the hydroxide anion (OH–). The formula for aluminum hydroxide is Al(OH)3.

9. 4 Names and Formulas for Bases n Sodium hydroxide (Na. OH) is a base that is used to make paper.

9. 4 Names and Formulas for Bases n Cleaners and soap contain sodium hydroxide.

Section Quiz 1. The name for H 2 S(aq) is a. b. c. d. sulfuric acid. hydrosulfuric acid. sulfurous acid. hydrosulfurous acid.

Section Quiz 2. The chemical formula for chlorous acid is a. b. c. d. HCl. O 2. HCl. O 3. HCl. O 4. HCl.

Section Quiz 3. The correct chemical name for NH 4 OH is a. b. c. d. nitrogen tetrahydrogen hydroxide. nitrogen pentahydrogen oxide. ammonium oxyhydride. ammonium hydroxide.

Acids Organizer n Complete an Acid organizer

Acid Nomenclature n Complete Acid Nomenclature worksheet.

Summary Questions 1. What is the difference b/w the formulas of 2. 3. 4. 5. acids and bases? What is the difference b/w naming an acid and base? What do acids and bases have in common? What type of elements are the same in both? When the anion ends in -ite the acid suffix is? When the anion ends in –ate the acid suffix is?

Homework n Naming Acids worksheet n Exit ticket: n Naming Acids and Bases