Drill Draw name 5 isomers of C 5









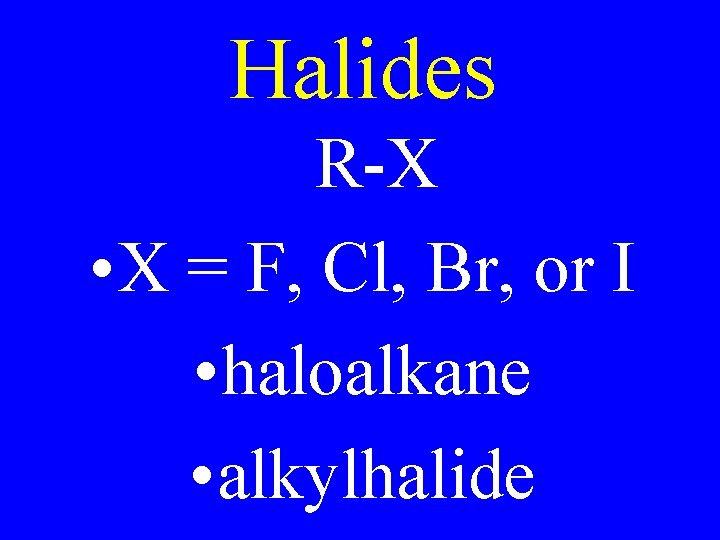


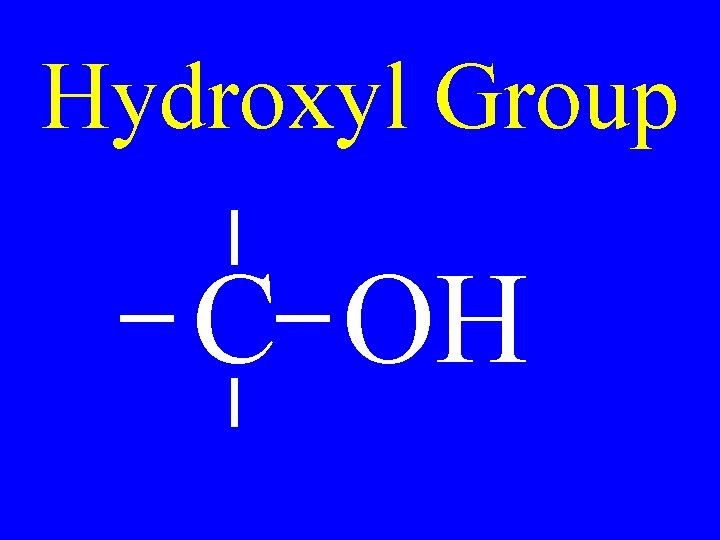






















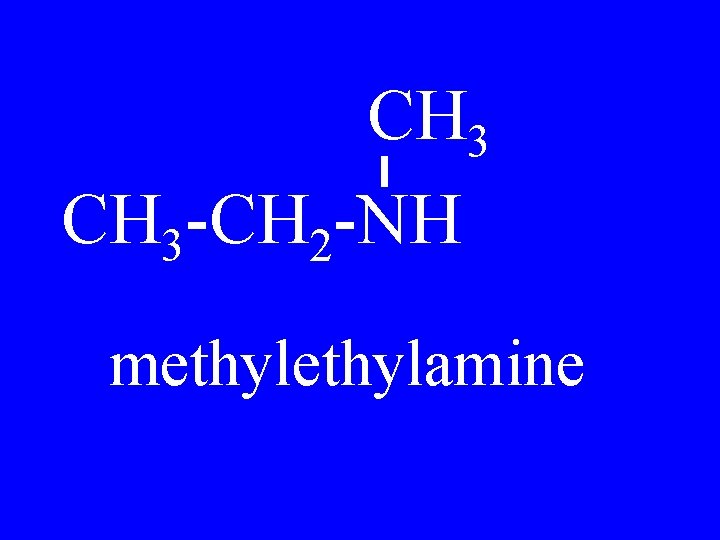




















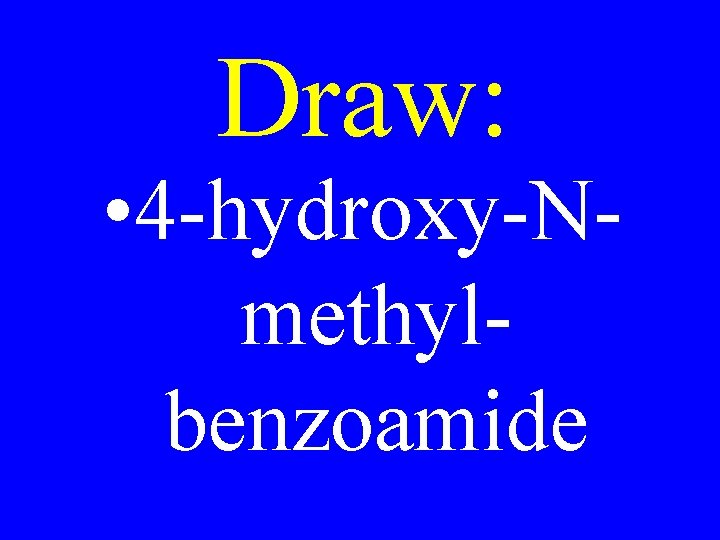


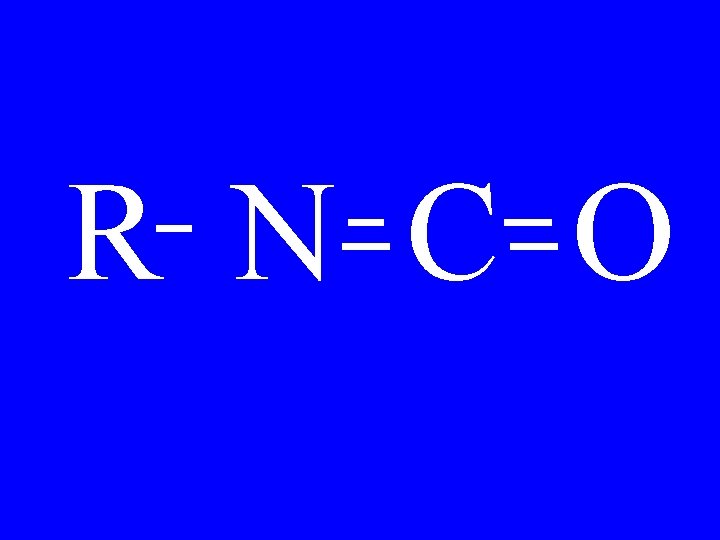














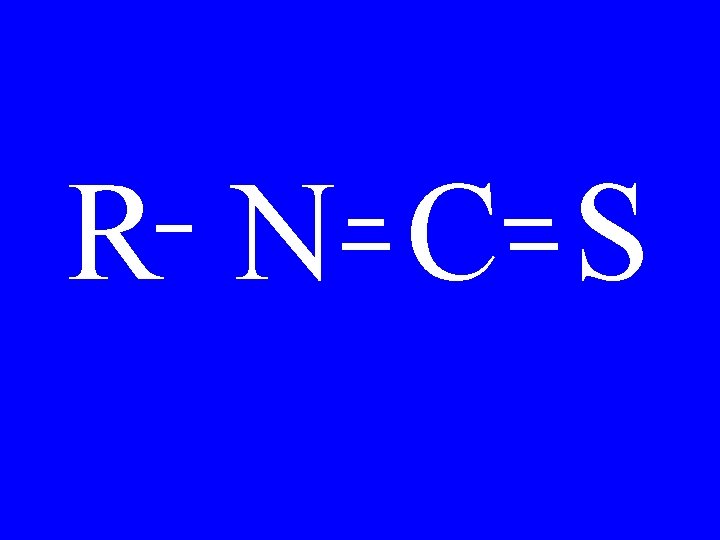



- Slides: 188

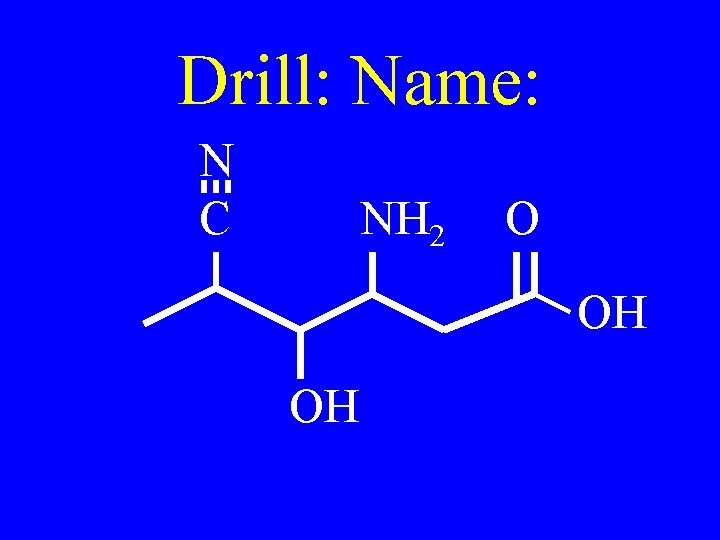
Drill: Draw & name 5 isomers of C 5 H 8

Functional Groups

Draw & Name Isomers of: C 4 H 6

Functional Groups • Chemically active part of an organic compound

Functional Groups • Anything other than singularly bonded carbon & hydrogen in an organic compound

Functional Groups • The name of any organic compound ends with the suffix for the most important functional group

Terminology • R- represents the part of the organic compound you are not referring to

Terminology • R-OH • -OH is the functional group • R- anything else

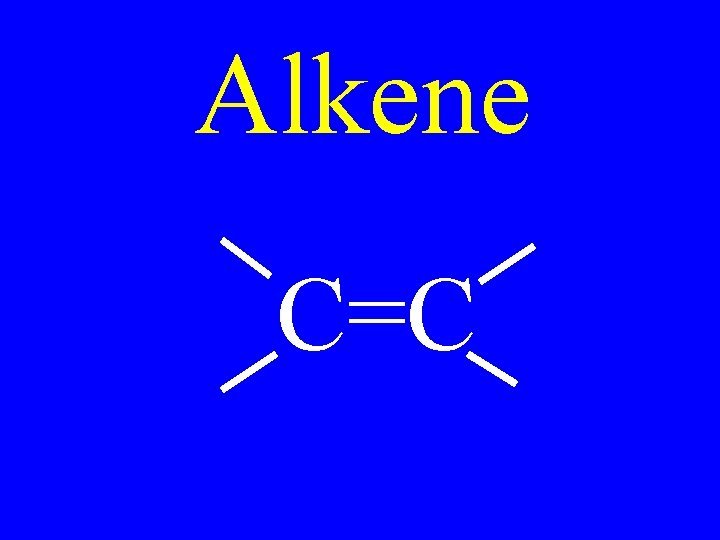
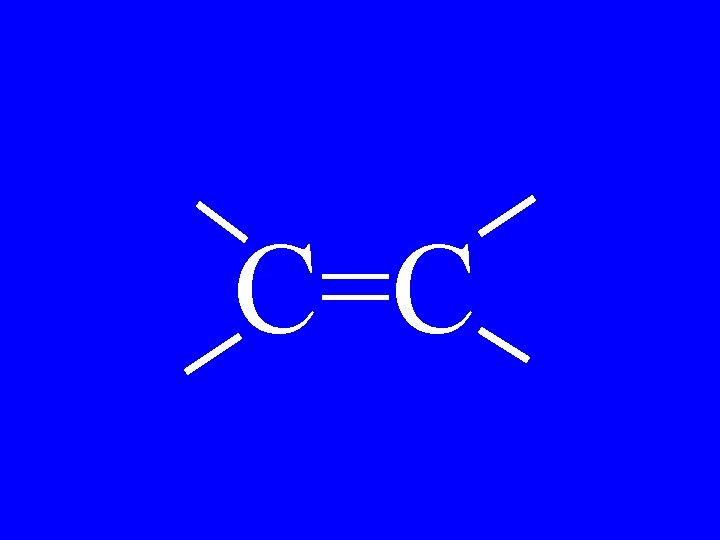
Alkene C=C

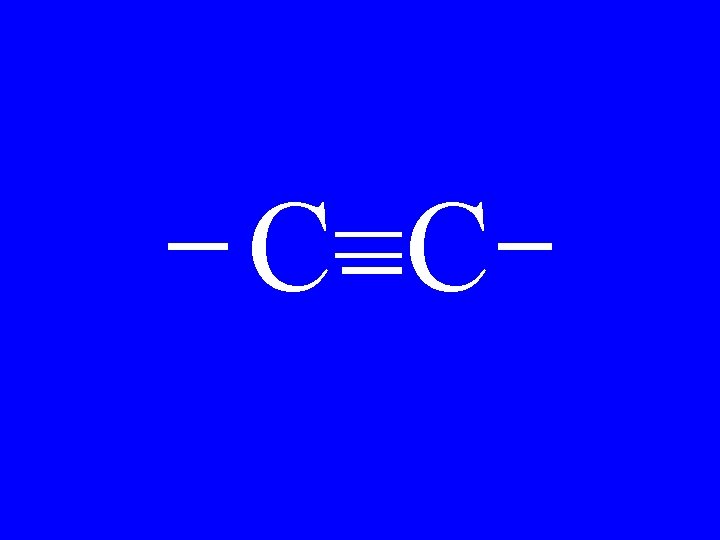
Alkyne C=C
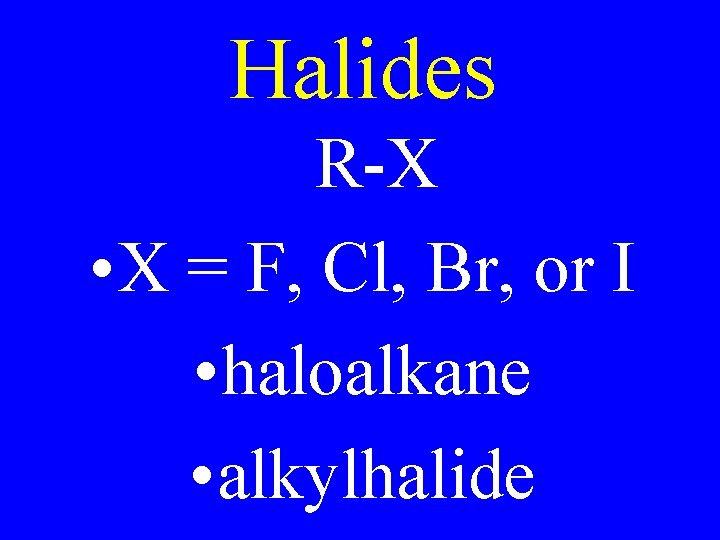
Halides R-X • X = F, Cl, Br, or I • haloalkane • alkylhalide

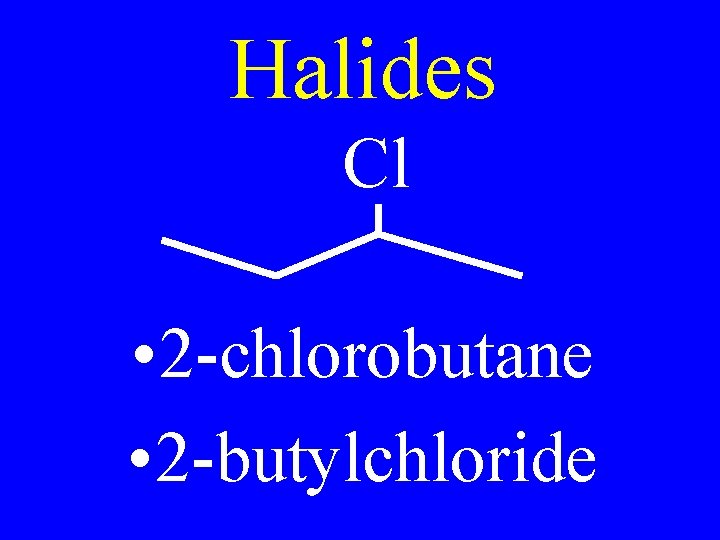
Halides Cl • 2 -chlorobutane • 2 -butylchloride

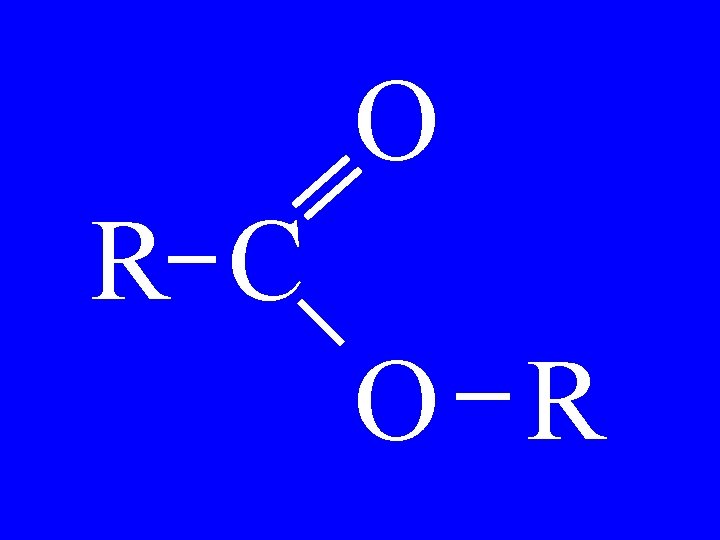
Ethers R 1 -O-R 2 • alkylether • alkoxyalkane

Ethers CH 3 CH 2 -O-CH 3 • Methylether • methoxyethane
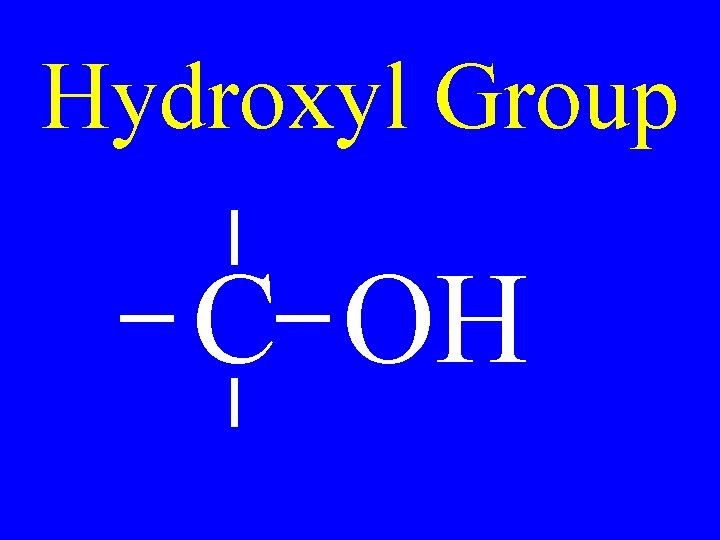
Hydroxyl Group C OH

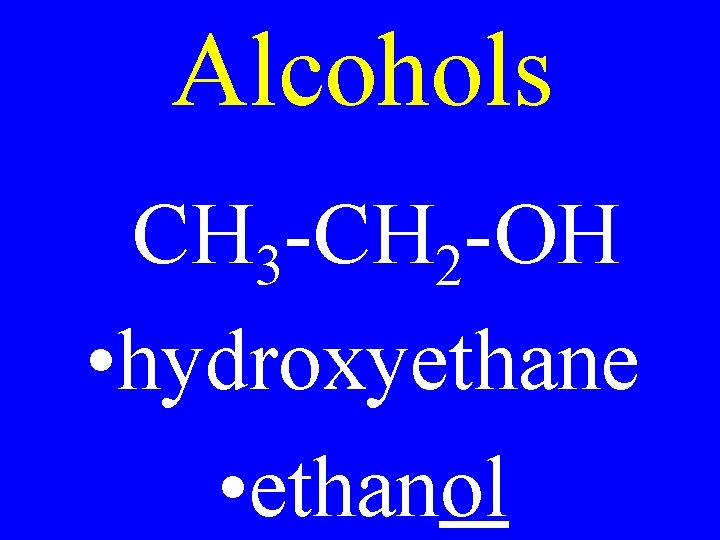
Alcohols R-OH • hydroxyalkane • alkanol

Alcohols CH 3 -CH 2 -OH • hydroxyethane • ethanol

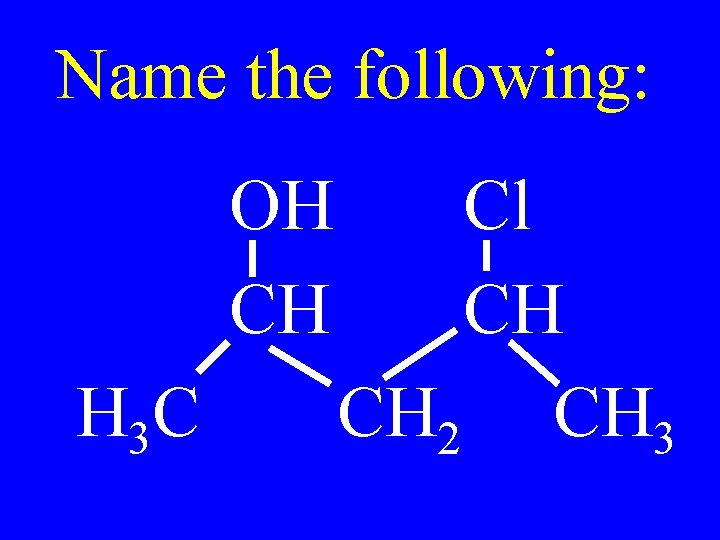
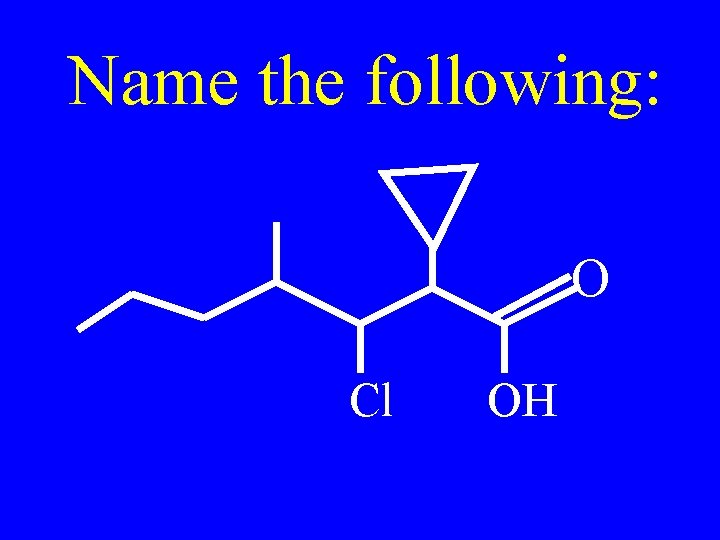
Name the following: OH CH H 3 C Cl CH CH 2 CH 3

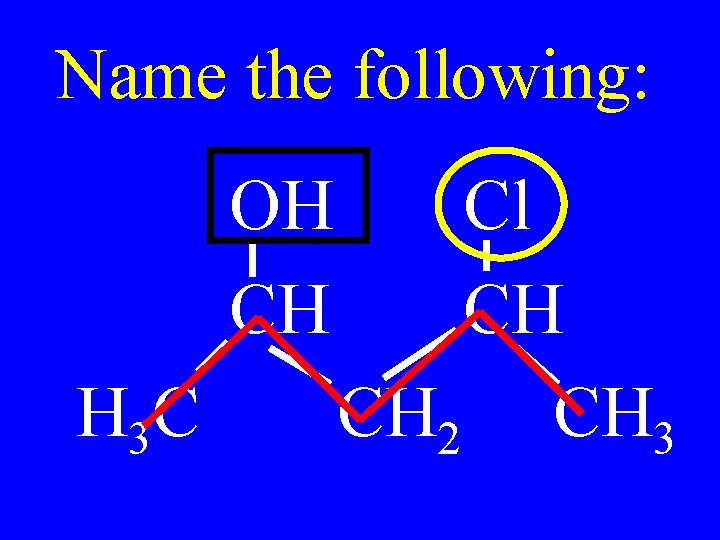
Name the following: OH CH H 3 C Cl CH CH 2 CH 3

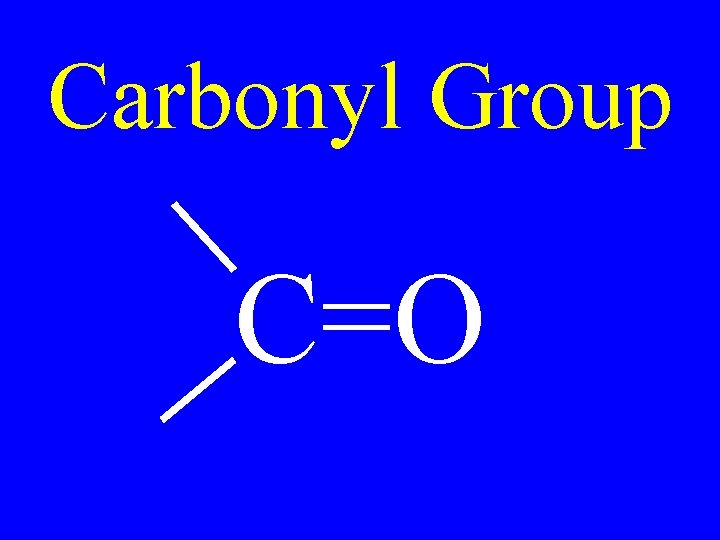
Carbonyl Group C=O

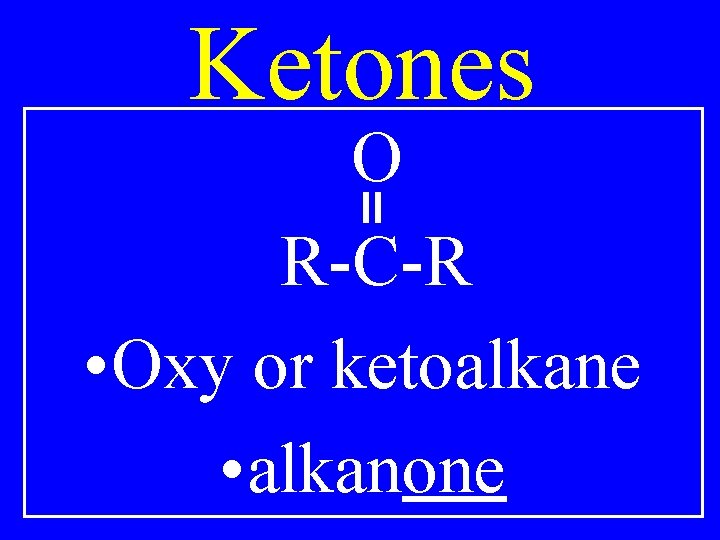
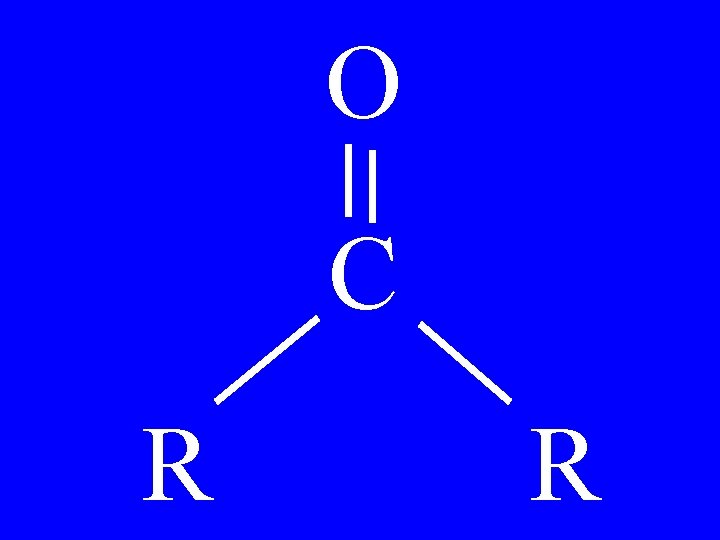
Ketones O R-C-R • Oxy or ketoalkane • alkanone

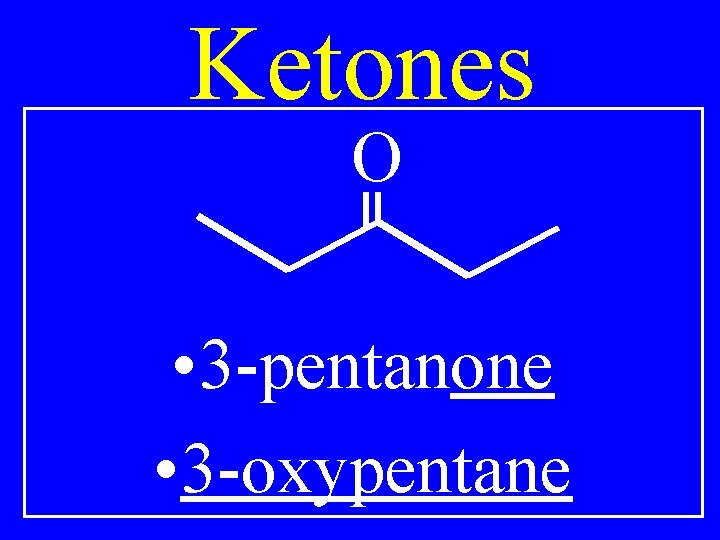
Ketones O • 3 -pentanone • 3 -oxypentane

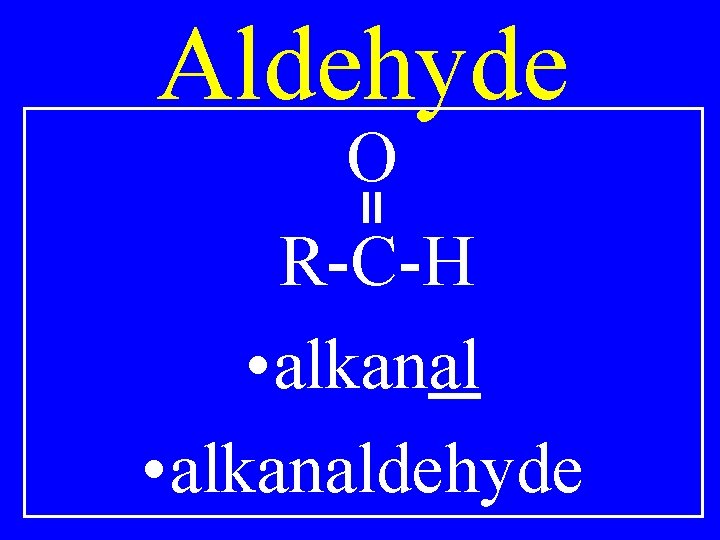
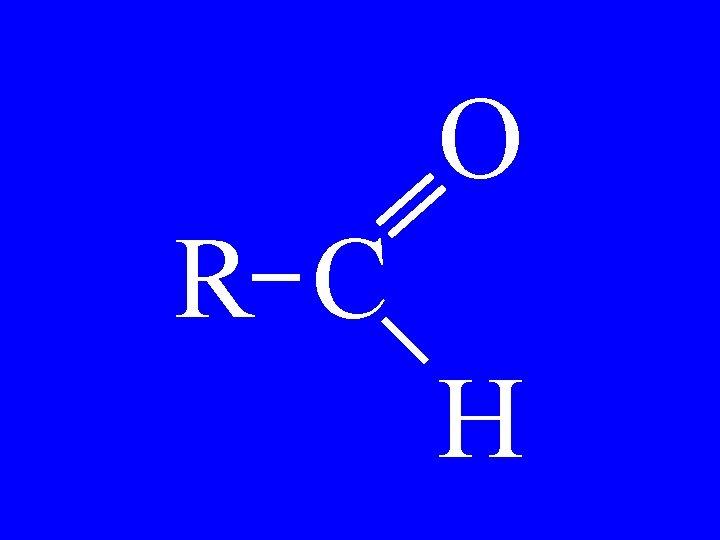
Aldehyde O R-C-H • alkanaldehyde

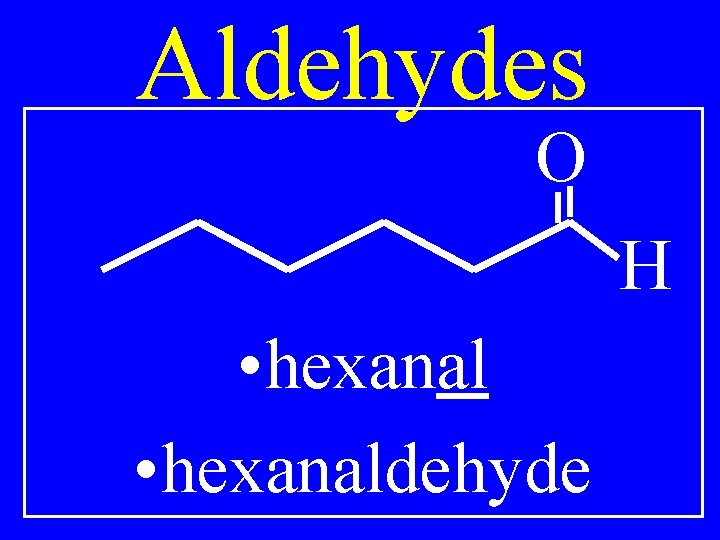
Aldehydes O H • hexanaldehyde

Homework • Draw & Name 5 isomers of C 4 H 8 O

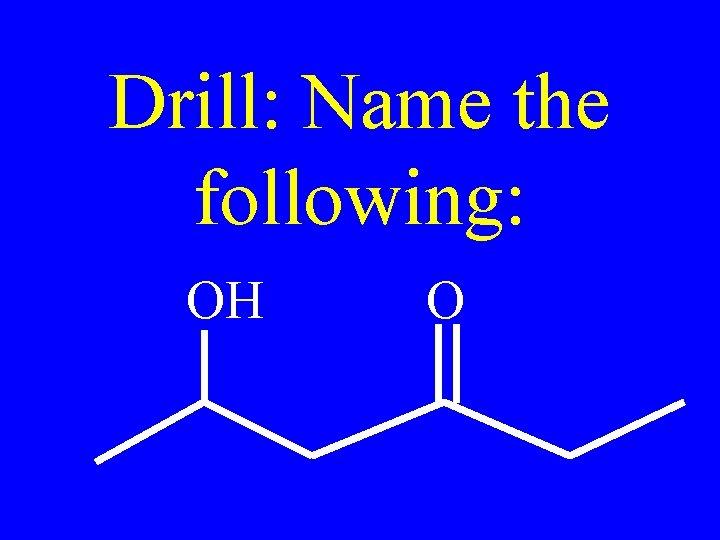
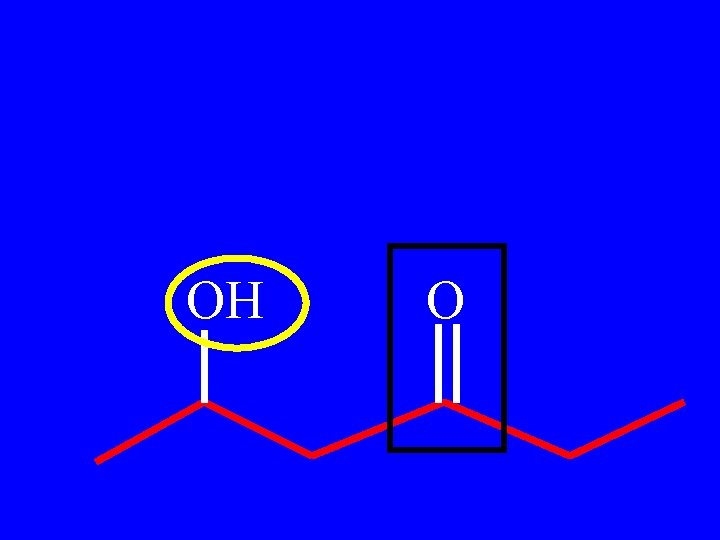
Drill: Name the following: OH O

OH O

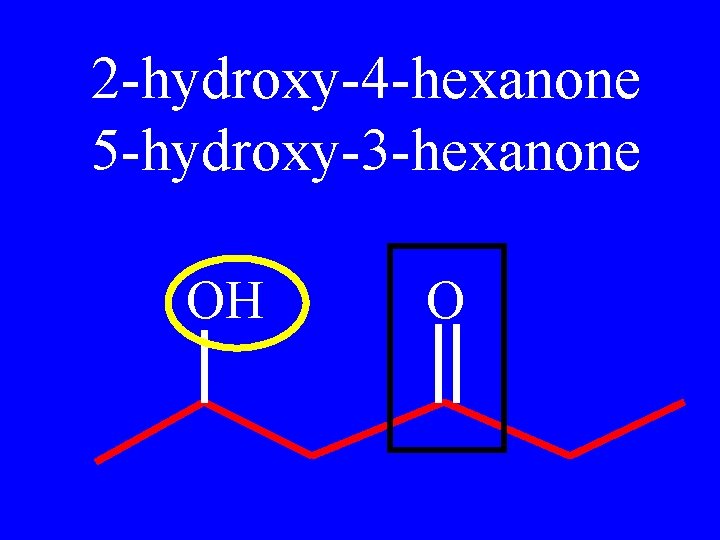
2 -hydroxy-4 -hexanone 5 -hydroxy-3 -hexanone OH O

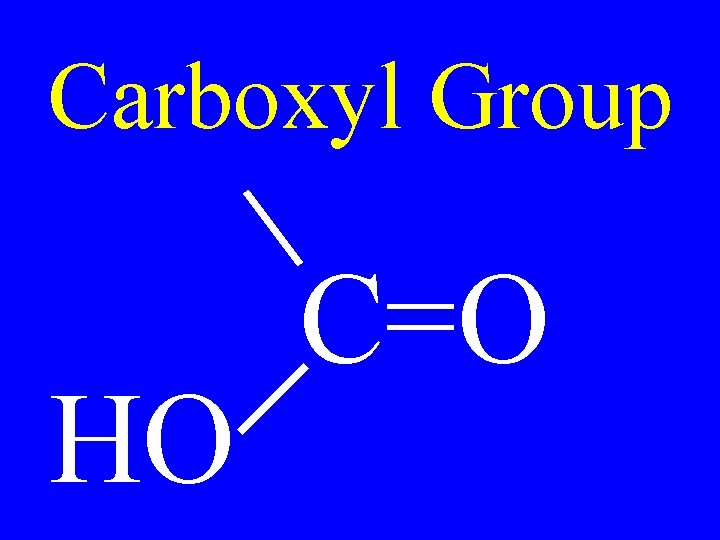
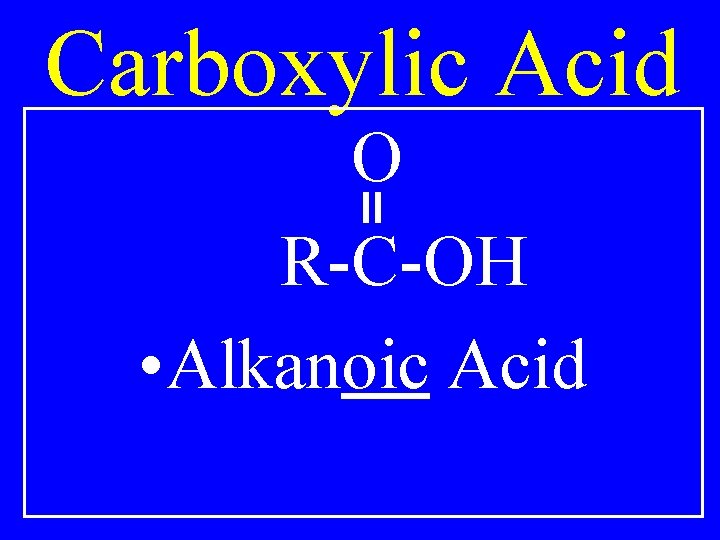
Carboxyl Group HO C=O

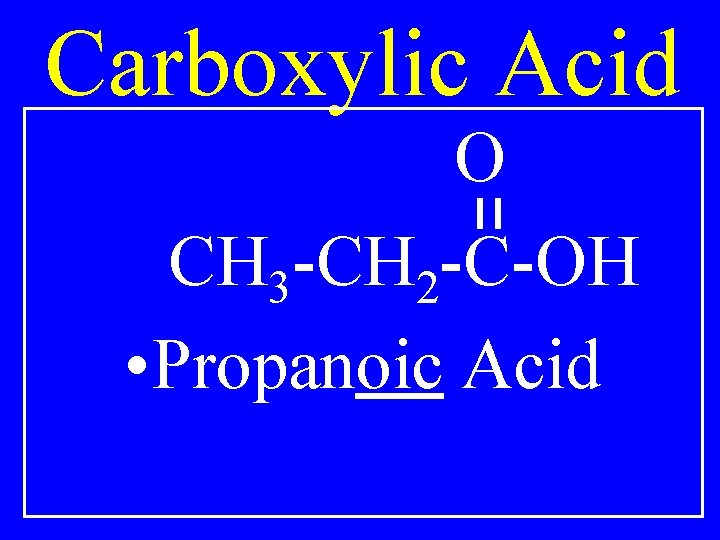
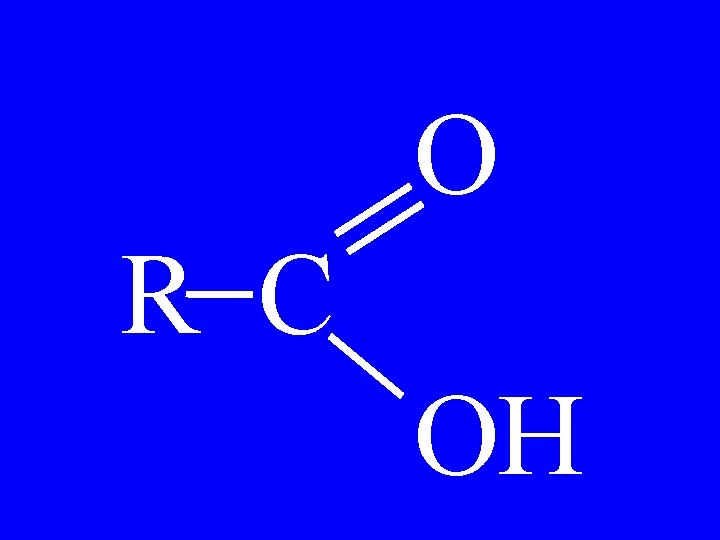
Carboxylic Acid O R-C-OH • Alkanoic Acid

Carboxylic Acid O CH 3 -CH 2 -C-OH • Propanoic Acid

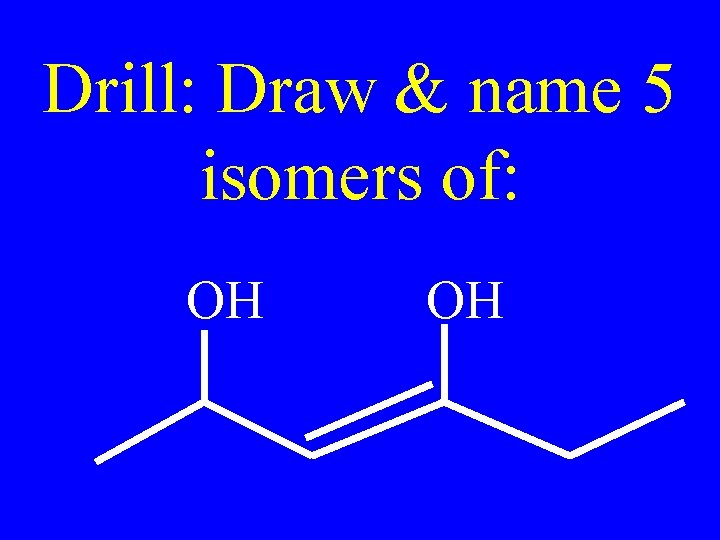
Drill: Draw & name 5 isomers of: OH OH

Functional Isomers: • Isomers with different functional groups: • The suffix changes

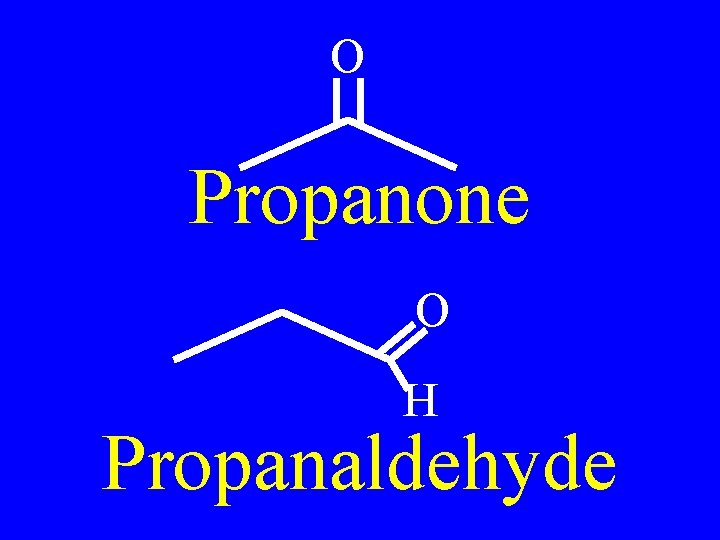
O Propanone O H Propanaldehyde

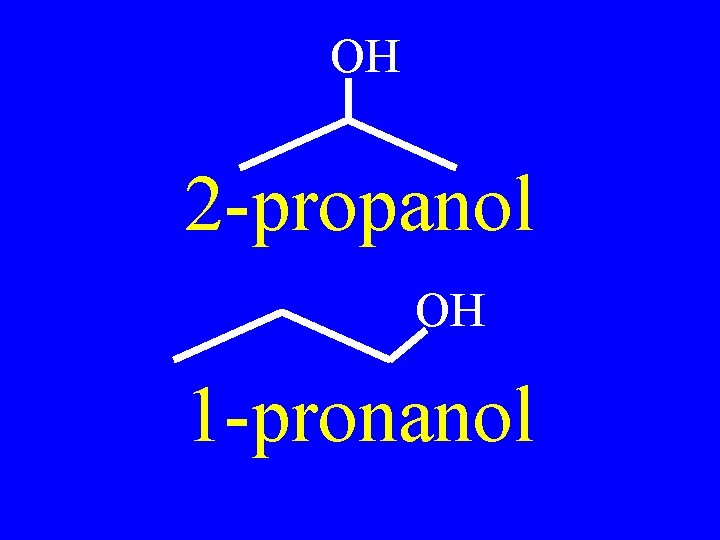
Positional Isomers: • Isomer’s functional group in a new location • The # before the main chain name changes

OH 2 -propanol OH 1 -pronanol

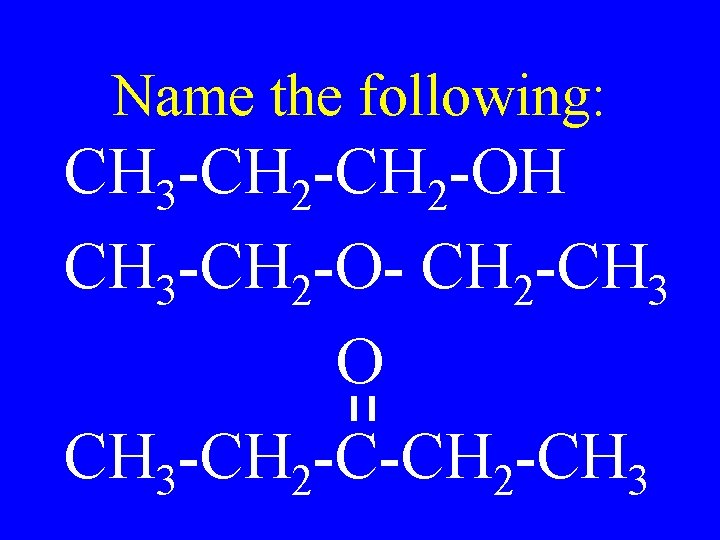
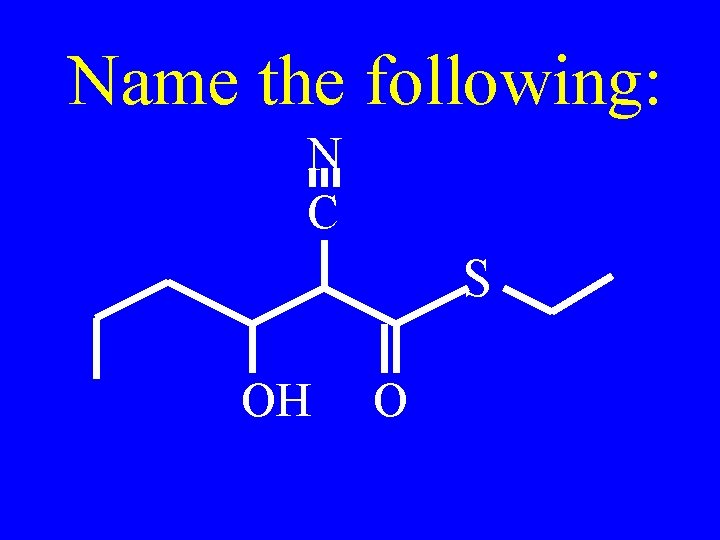
Name the following: CH 3 -CH 2 -OH CH 3 -CH 2 -O- CH 2 -CH 3 O CH 3 -CH 2 -CH 3

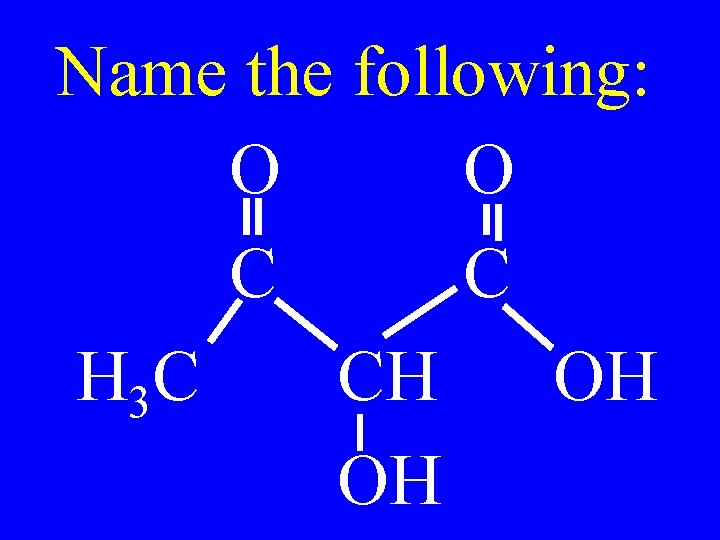
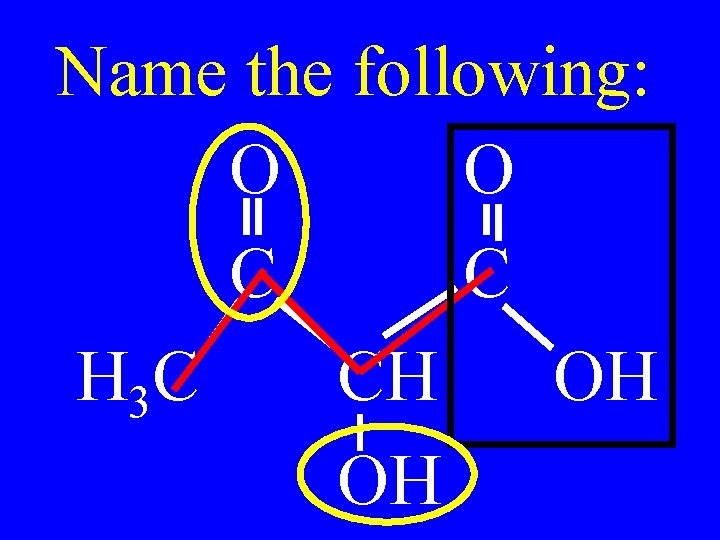
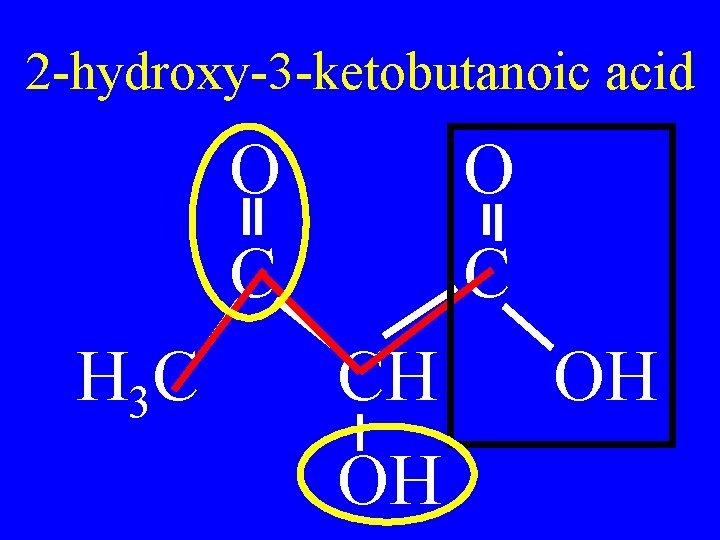
Name the following: O O C C H 3 C CH OH OH

Name the following: O O C C H 3 C CH OH OH

2 -hydroxy-3 -ketobutanoic acid O C H 3 C O C CH OH OH

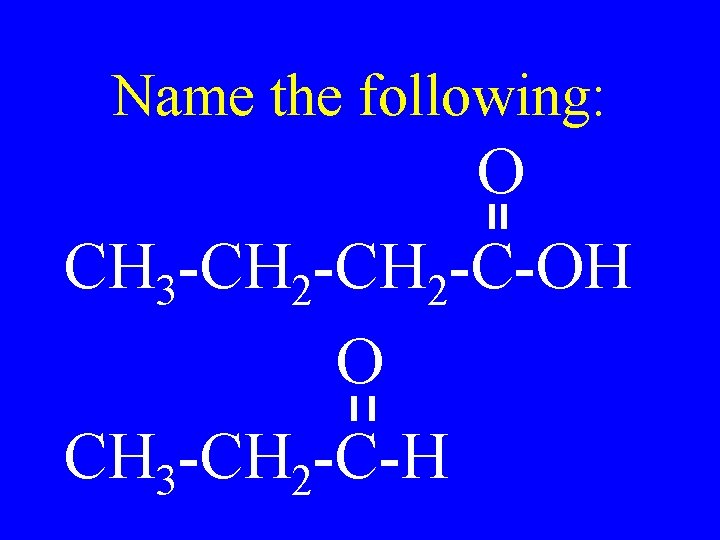
Name the following: O CH 3 -CH 2 -C-OH O CH 3 -CH 2 -C-H

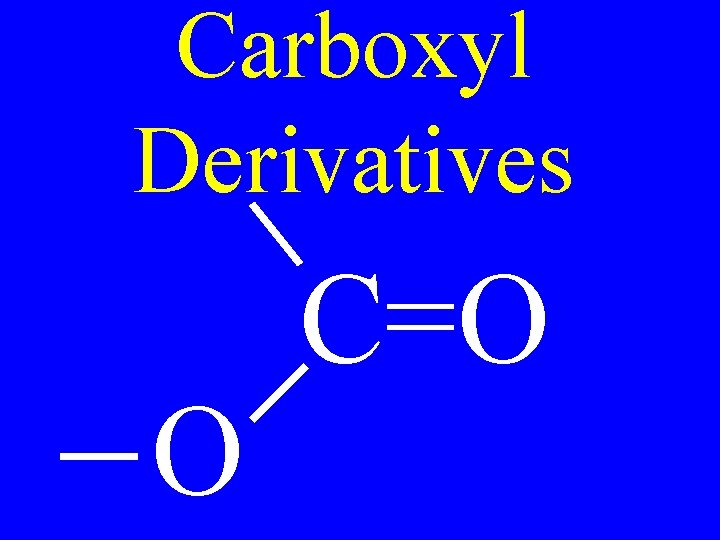
Carboxyl Derivatives O C=O

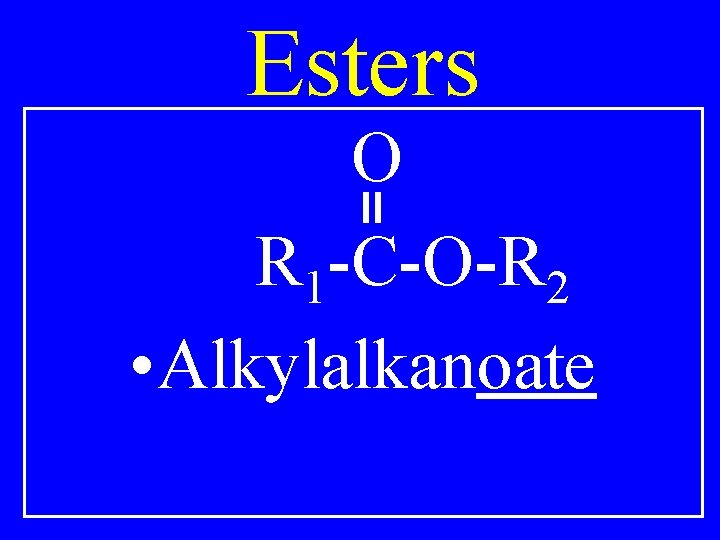
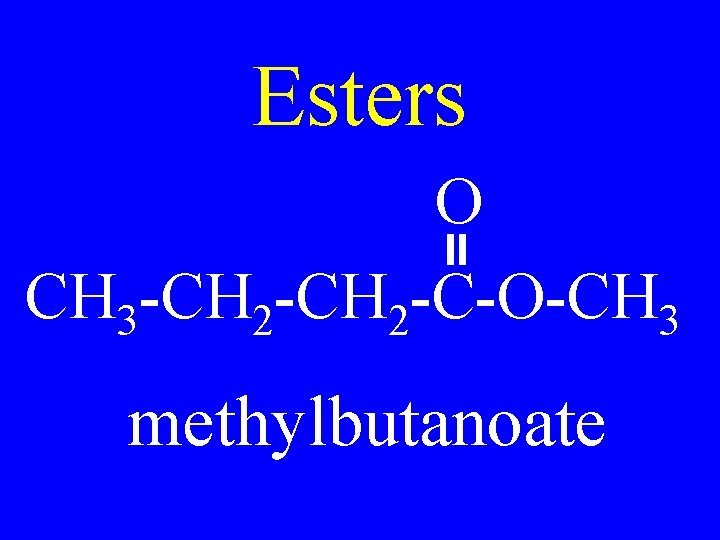
Esters O R 1 -C-O-R 2 • Alkylalkanoate

Esters O CH 3 -CH 2 -C-O-CH 3 methylbutanoate

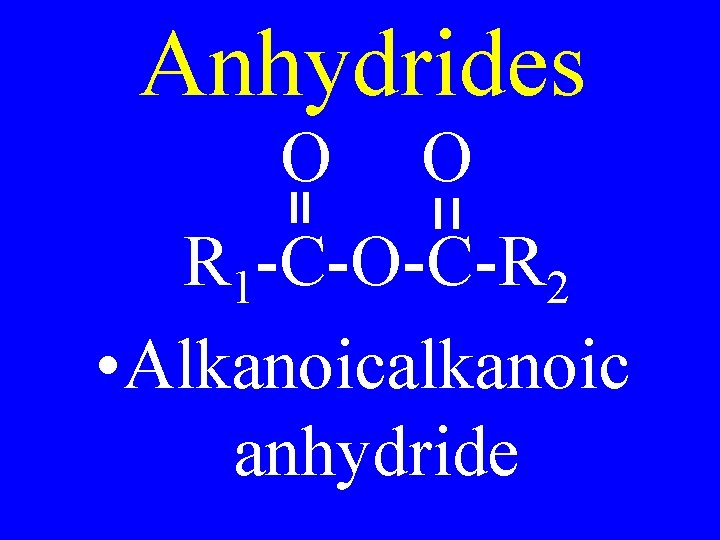
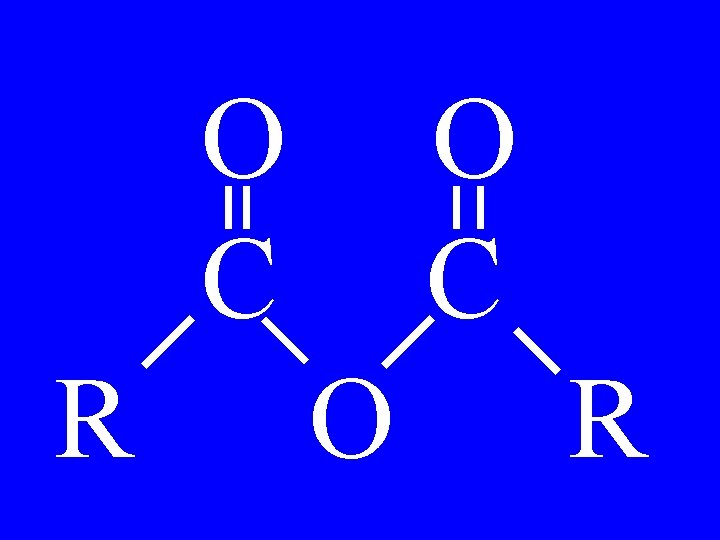
Anhydrides O O R 1 -C-O-C-R 2 • Alkanoicalkanoic anhydride

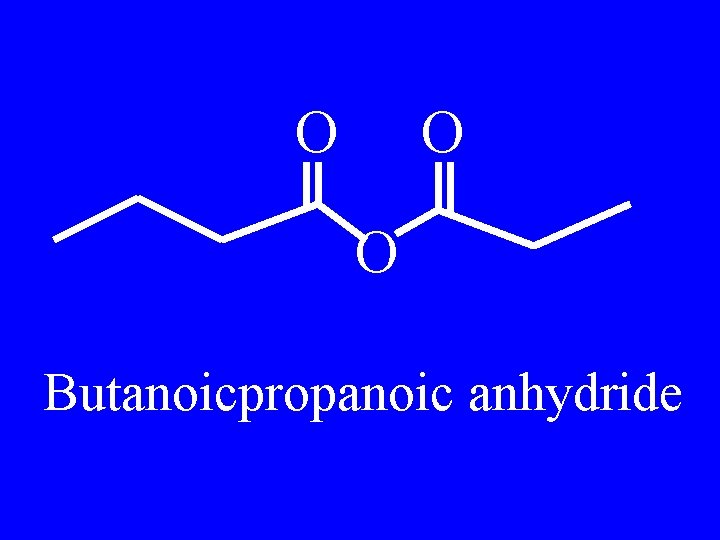
O O O Butanoicpropanoic anhydride

Draw the following: • 2 -hydroxypentanal • Methylpropylether • Trichloroacetic acid • Ethylbutanoate • Aceticformic anhydride

Drill: Draw & Name 5 isomers of: C 3 H 6 O 2

Organic HW • Draw & name 5 isomers (each containing a carboxyl or a carboxyl derivative) of C 5 H 10 O 3

Modeling Lab Monday

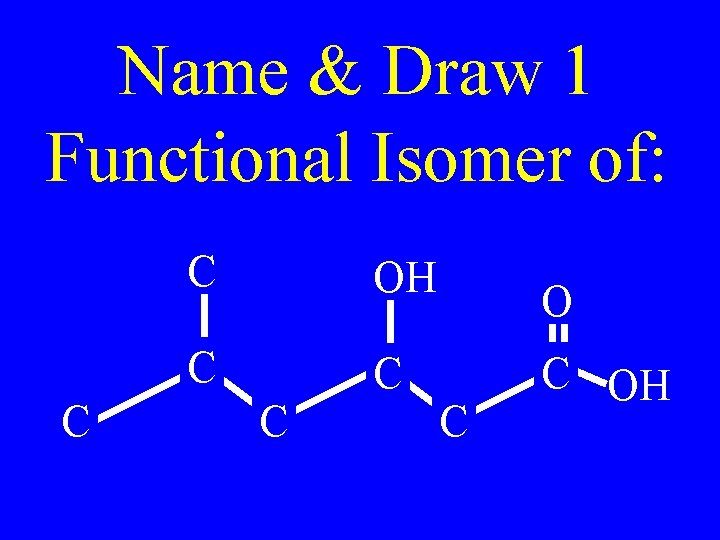
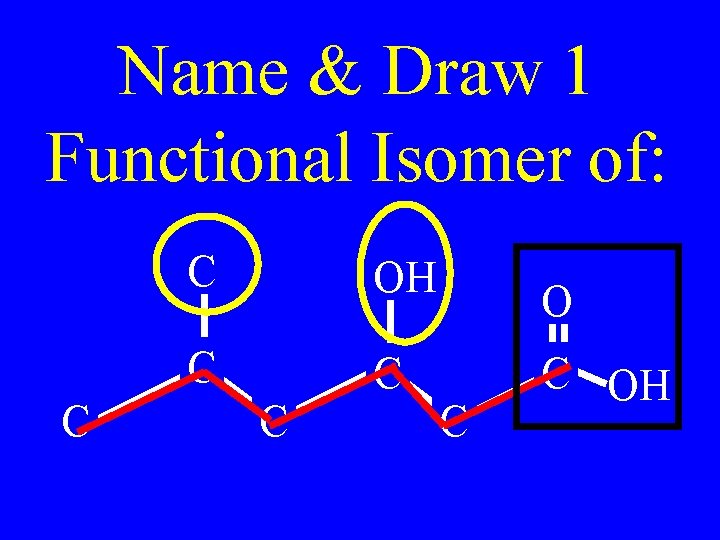
Name & Draw 1 Functional Isomer of: C C OH C C C OH

Name & Draw 1 Functional Isomer of: C C OH C C C OH

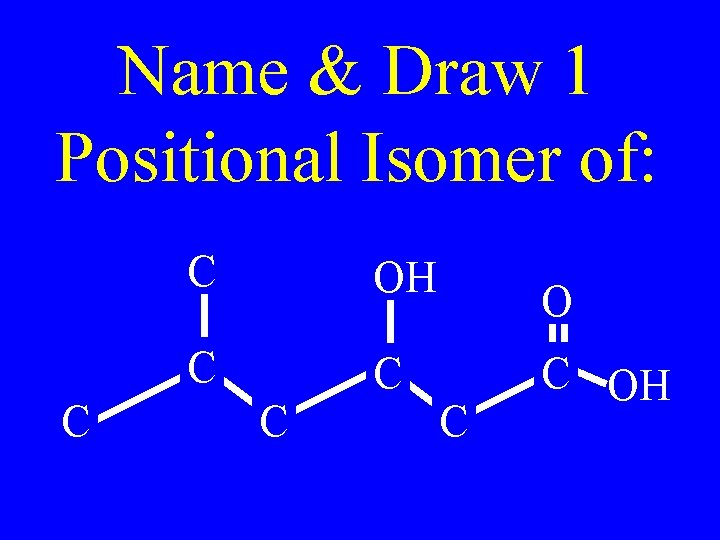
Name & Draw 1 Positional Isomer of: C C OH C C C OH

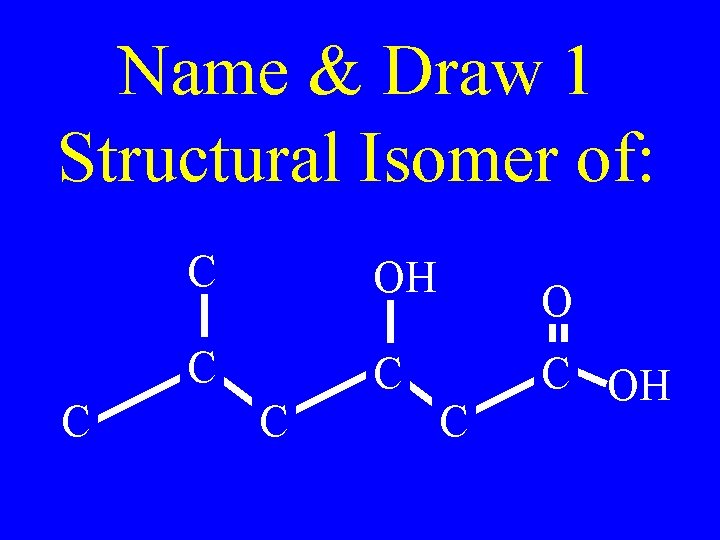
Name & Draw 1 Structural Isomer of: C C OH C C C OH

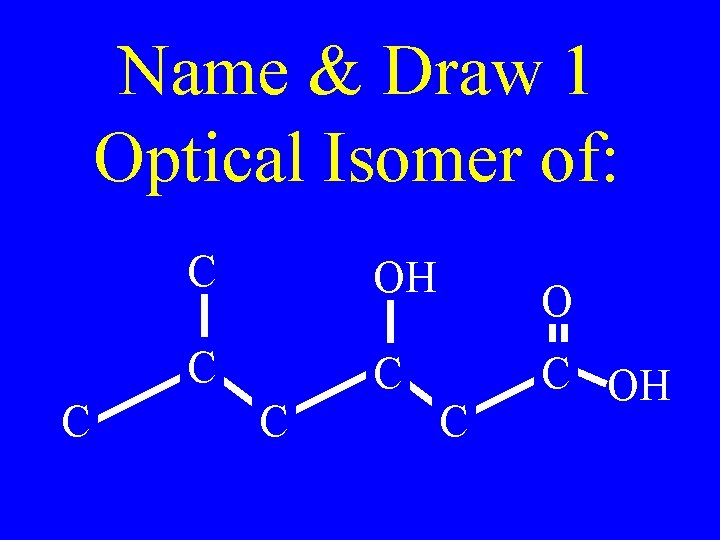
Name & Draw 1 Optical Isomer of: C C OH C C C OH

Draw: • 2 -phenylpropanoic acid • 3 -hydroxy-2 -iodopentanal • 2 -cyclohexyl-3 -octanone • phenylbutanoate

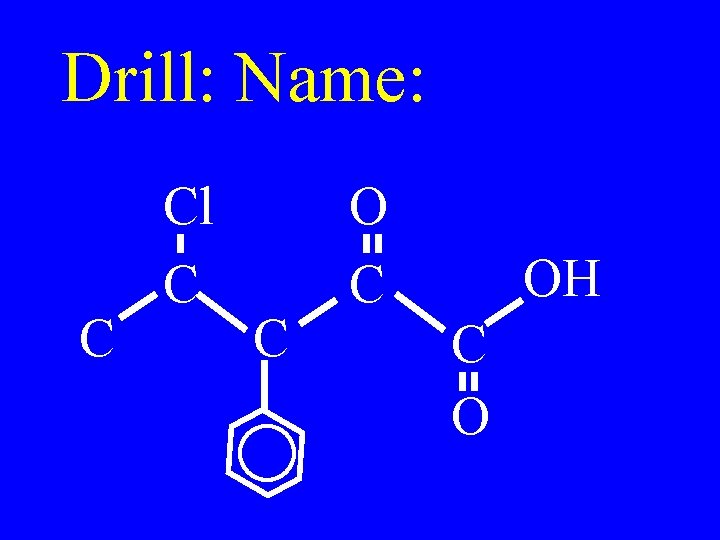
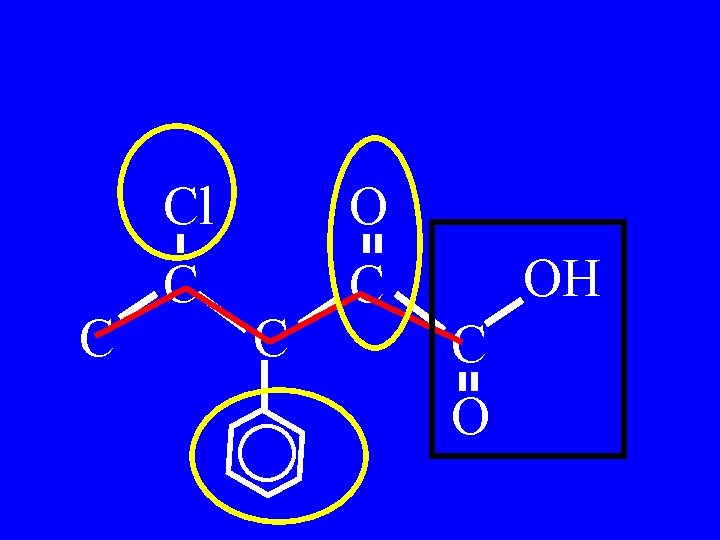
Drill: Name: C Cl C C OH C O

C Cl C C OH C O

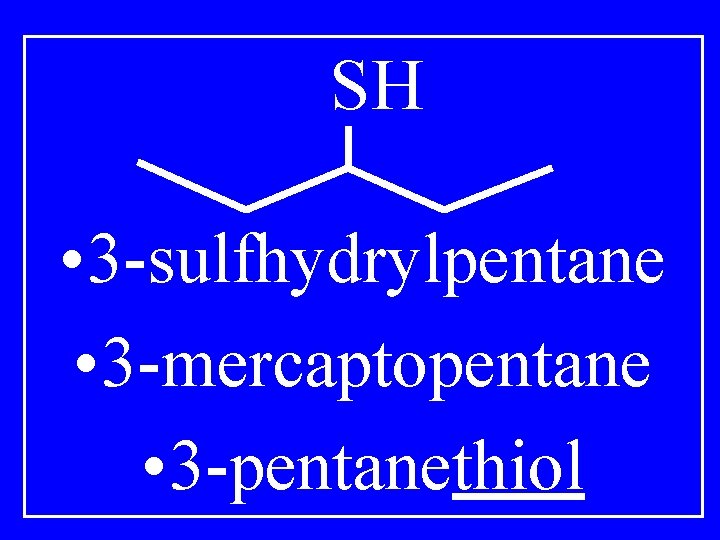
Thiol R-SH • Sulfhydrylalkane • Mercaptoalkane • Alkanethiol

SH • 3 -sulfhydrylpentane • 3 -mercaptopentane • 3 -pentanethiol

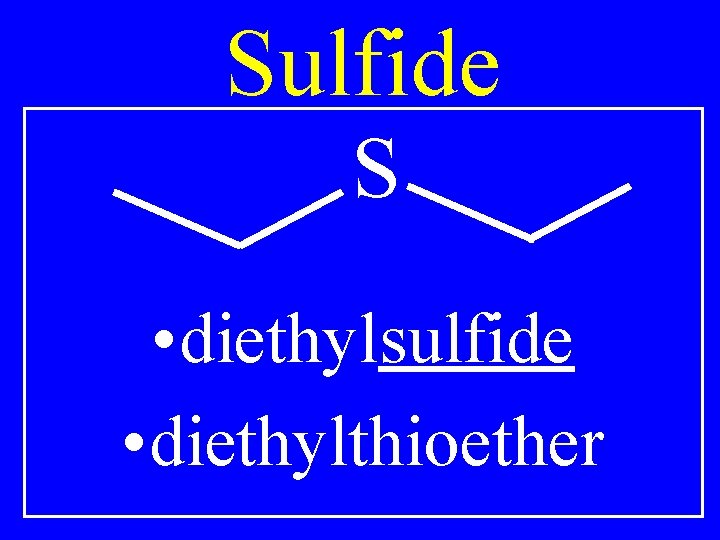
Sulfide R 1 -S-R 2 • Alkylalkylsulfide • Alkylalkylthioether

Sulfide S • diethylsulfide • diethylthioether

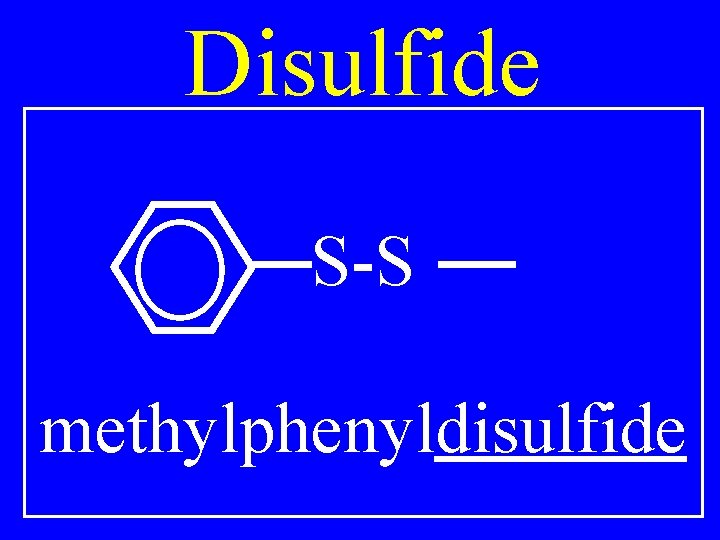
Disulfide R 1 -S-S-R 2 • Alkylalkyldisulfide

Disulfide S-S methylphenyldisulfide

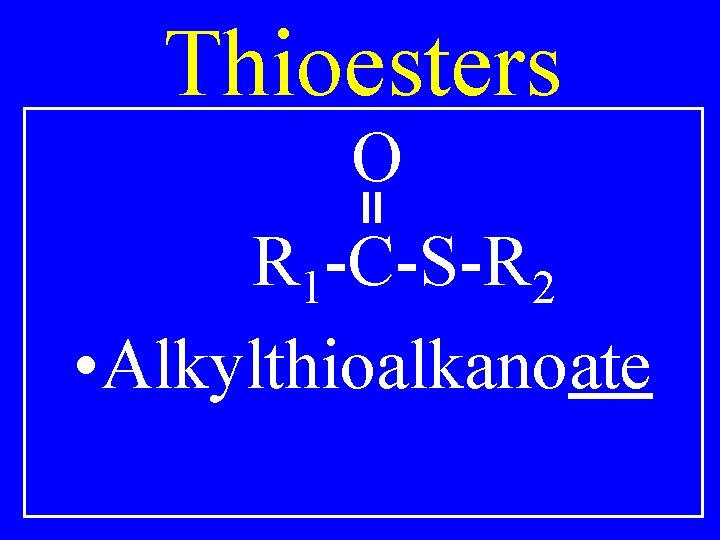
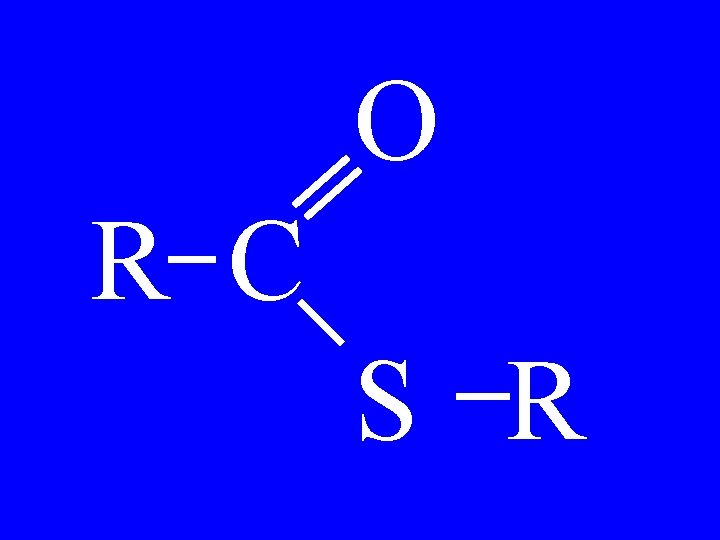
Thioesters O R 1 -C-S-R 2 • Alkylthioalkanoate

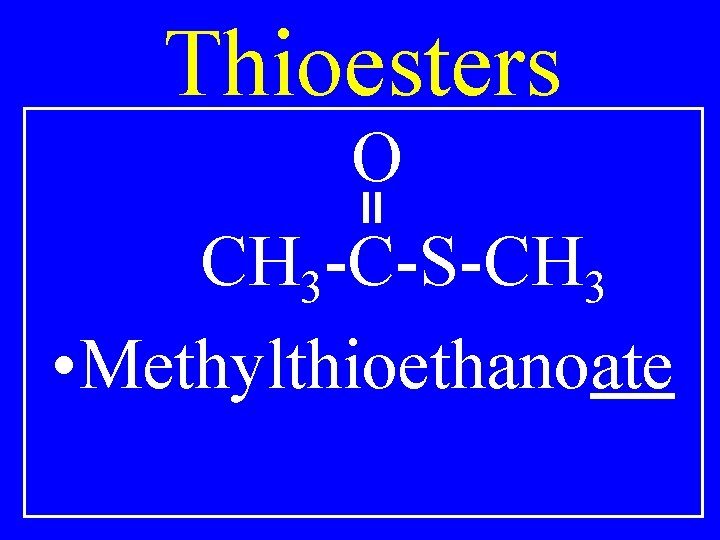
Thioesters O CH 3 -C-S-CH 3 • Methylthioethanoate

Draw the Following: • Methylthiobutanoate • Methylphenyl ether • Diphenyl thioether • 2 -propanethiol

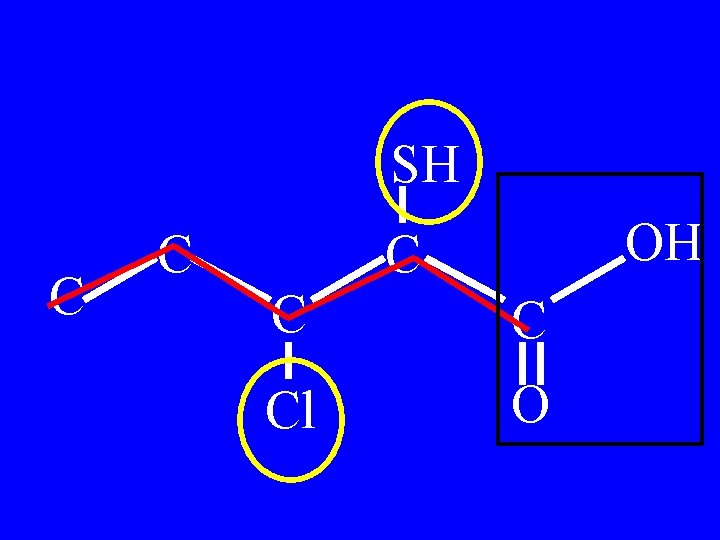
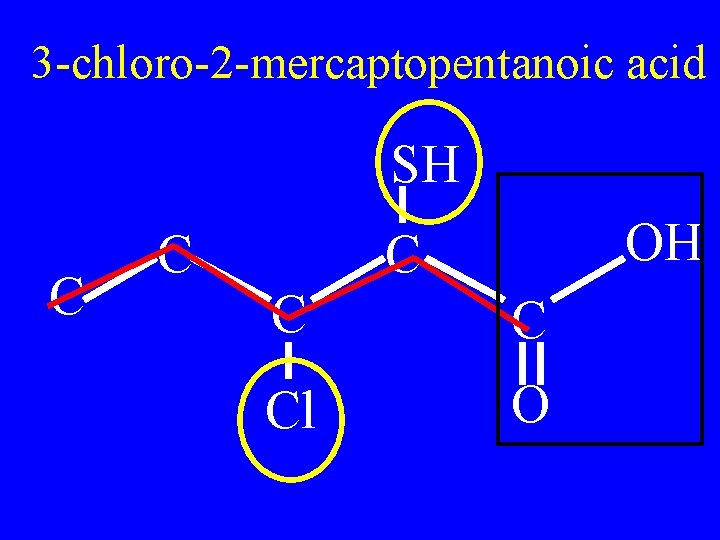
Name: SH C C OH C Cl O

SH C C OH C Cl O

3 -chloro-2 -mercaptopentanoic acid SH C C OH C Cl O

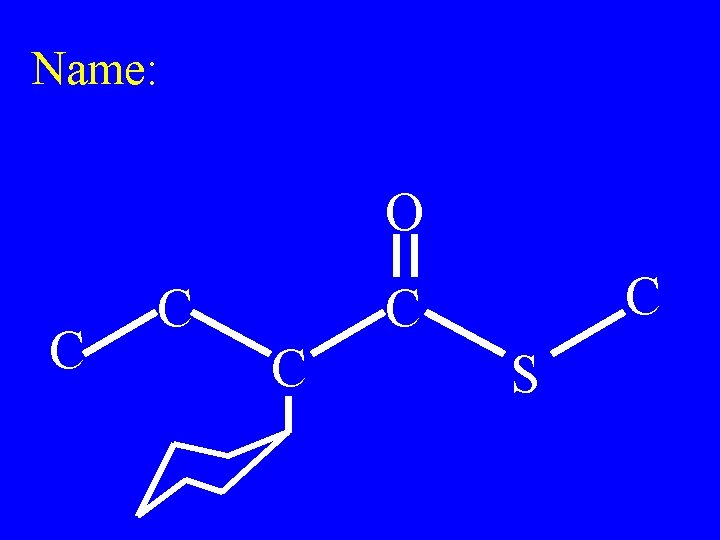
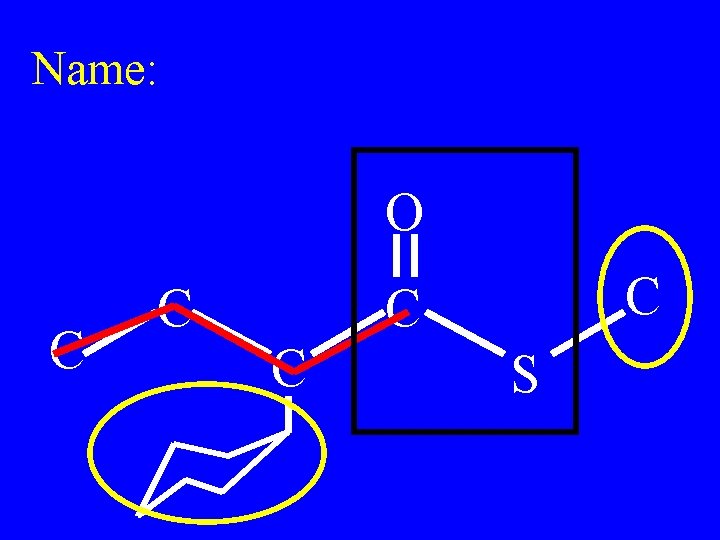
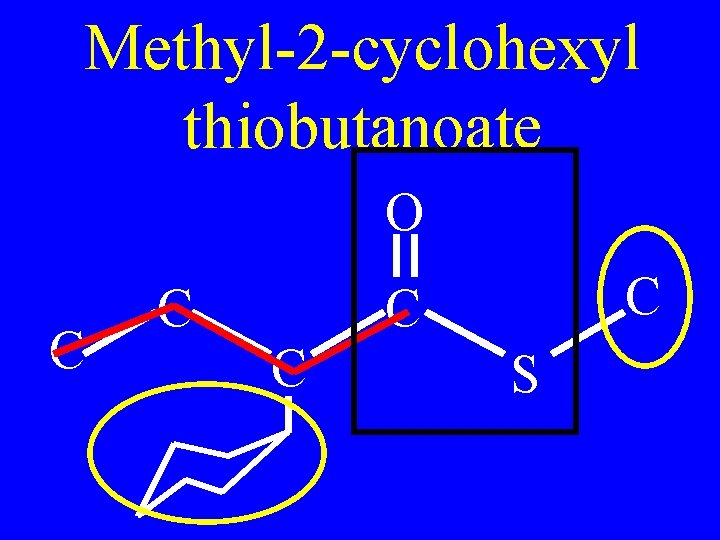
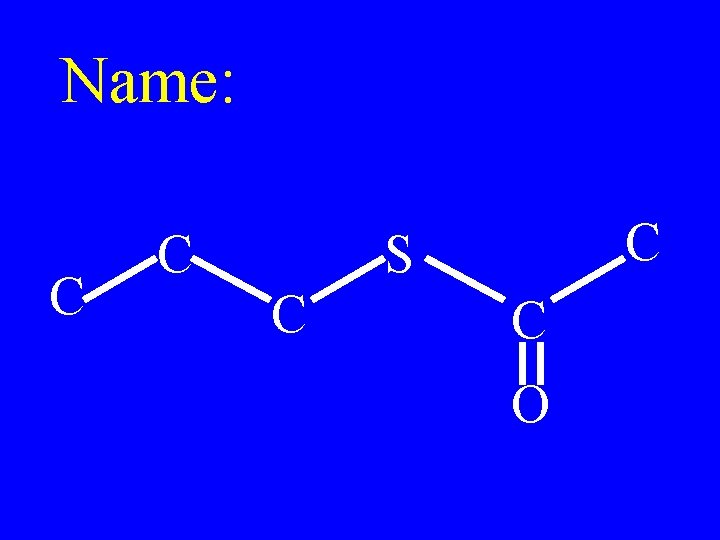
Name: O C C C S

Name: O C C C S

Methyl-2 -cyclohexyl thiobutanoate O C C C S

Organic HW: • Draw & name 5 isomers of C 3 H 6 SO

Draw & name 10 isomers of: C 4 H 8 SO

Drill: Draw the following: • 2 -phenyl-1 -propanethiol • Butylethenyldisulfide

Draw the following: • phenylthioacetate • methylisopropylsulfide • 2 -mercaptobutanal

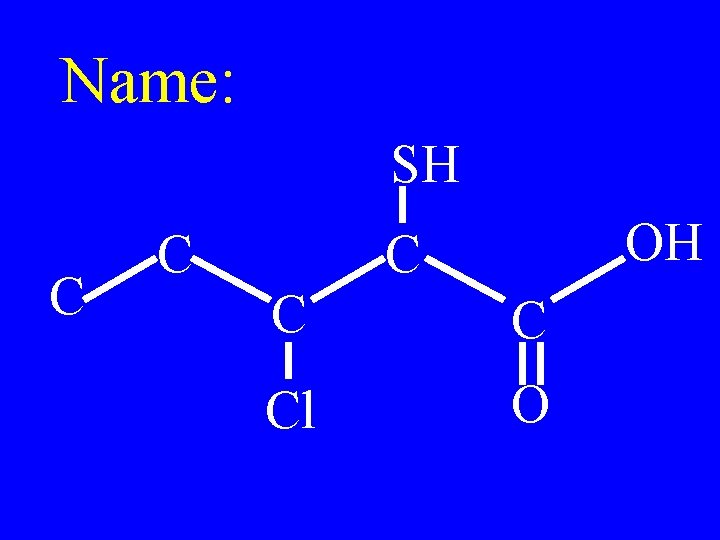
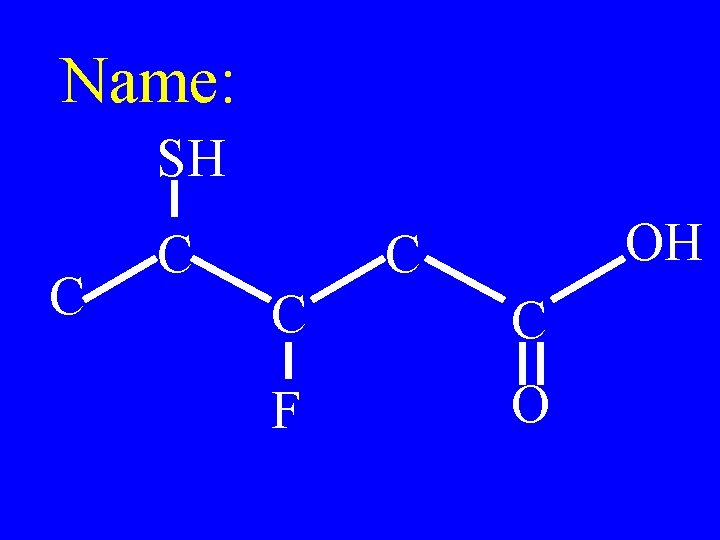
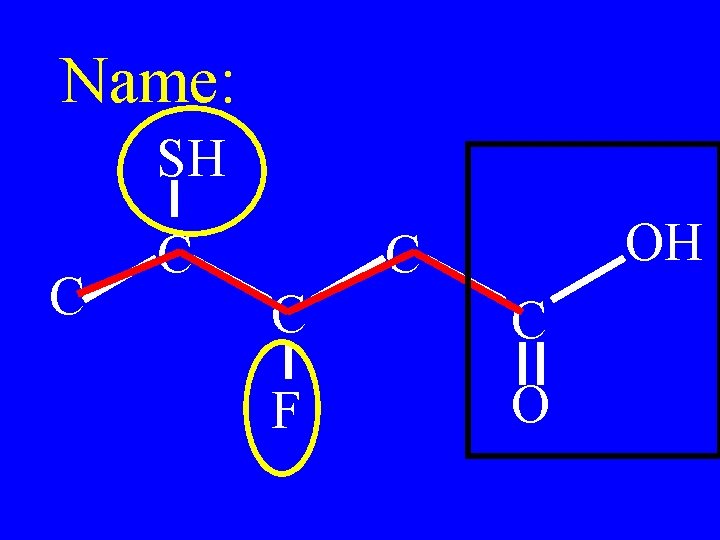
Name: SH C C OH C C C F O

Name: SH C C OH C C C F O

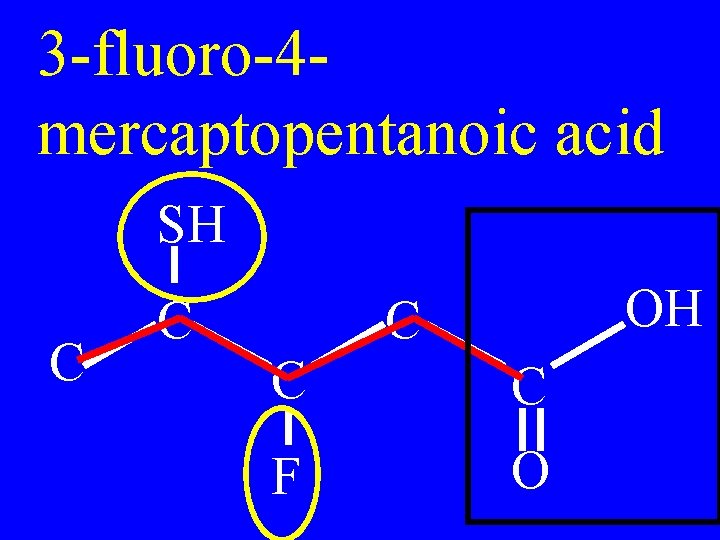
3 -fluoro-4 mercaptopentanoic acid SH C C OH C C C F O

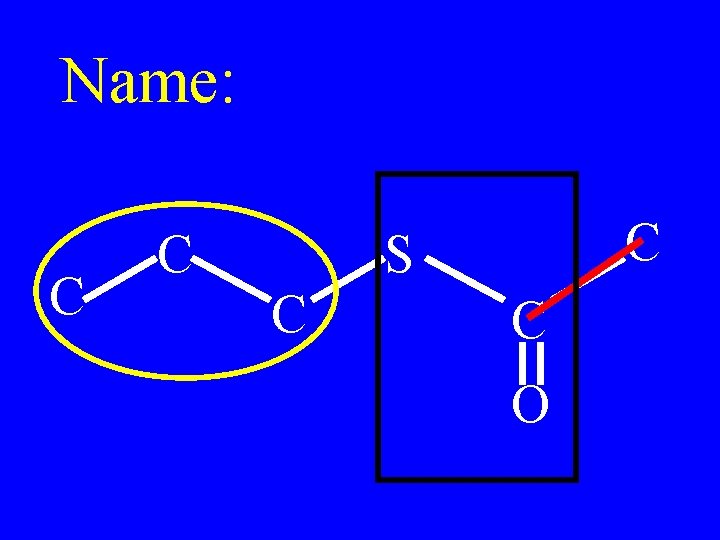
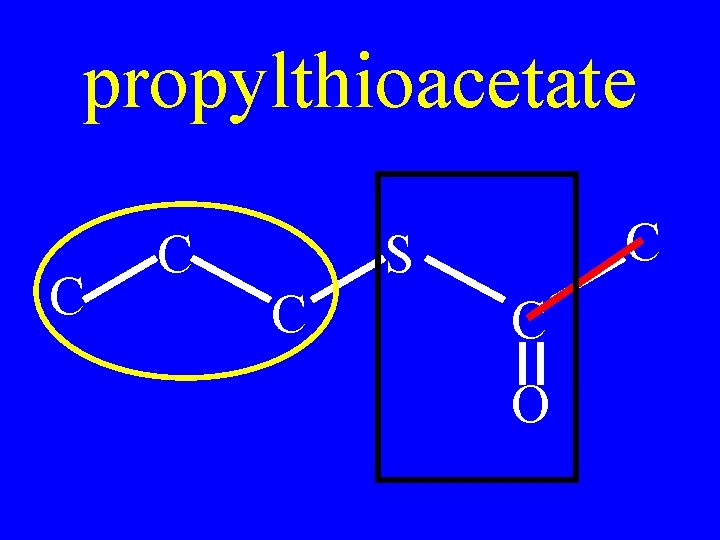
Name: C C C S C C O

Name: C C C S C C O

propylthioacetate C C C S C C O

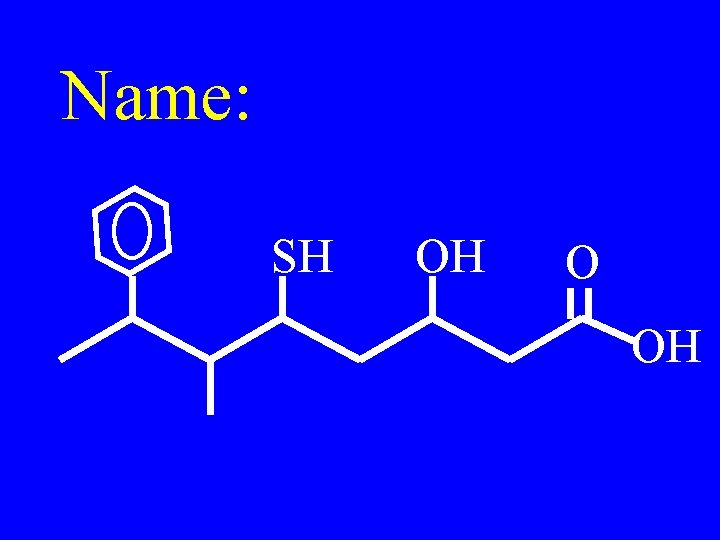
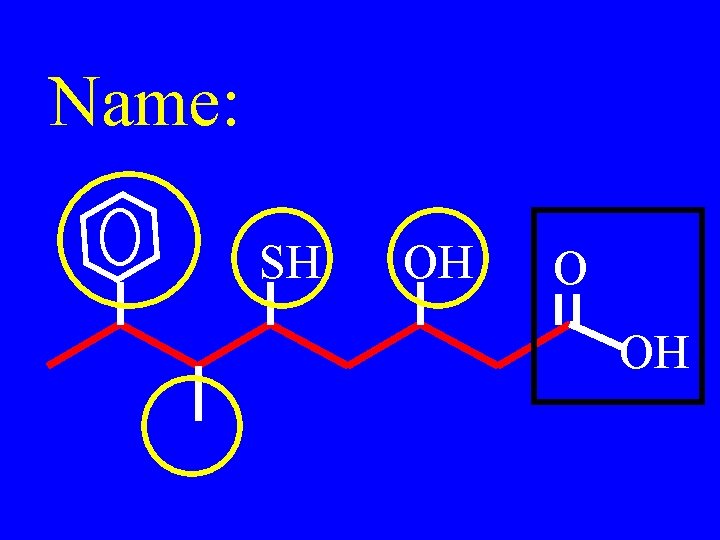
Name: SH OH O OH

Name: SH OH O OH

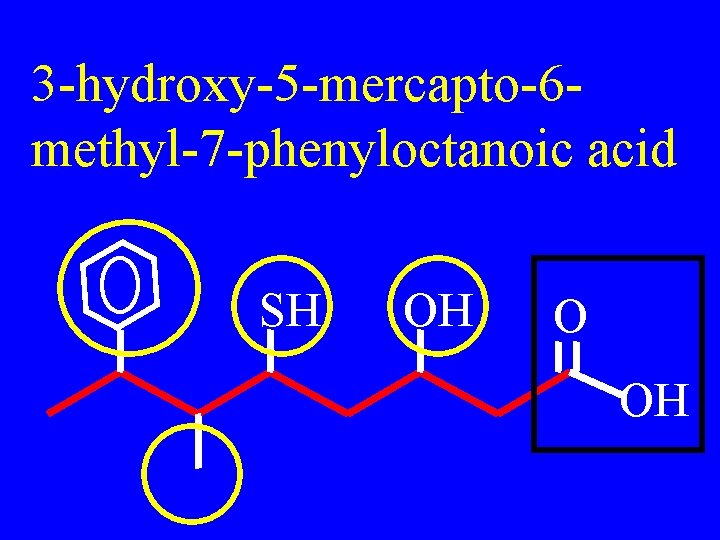
3 -hydroxy-5 -mercapto-6 methyl-7 -phenyloctanoic acid SH OH O OH

Draw & name 10 isomers of: C 4 H 8 SO 2

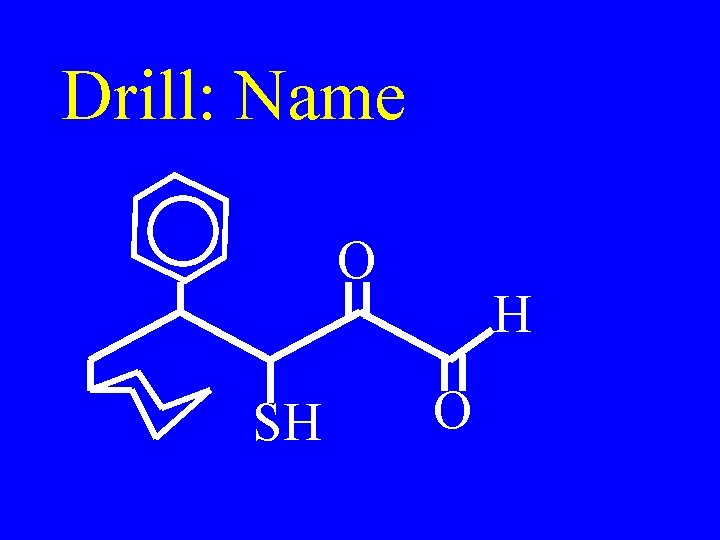
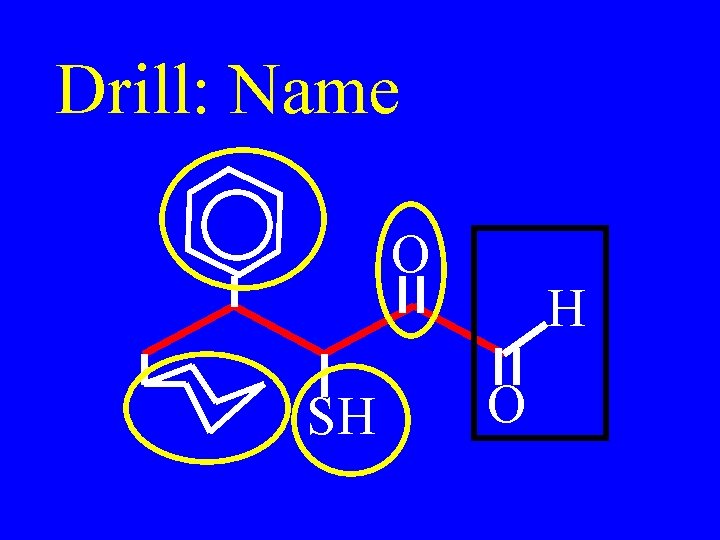
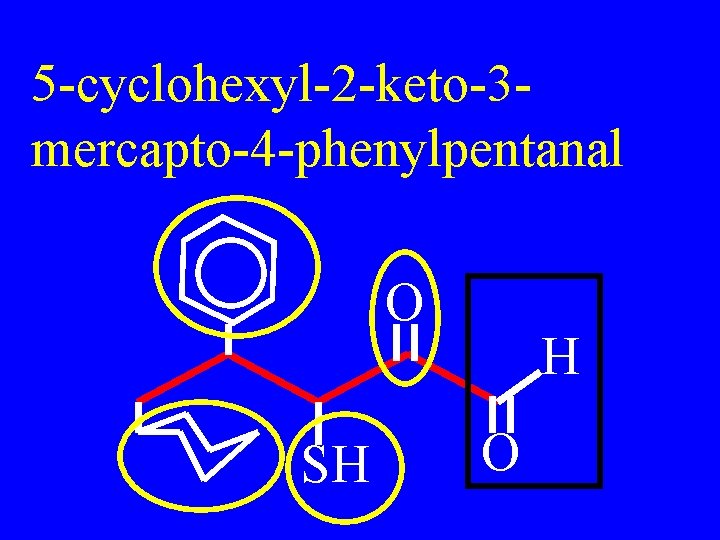
Drill: Name O SH H O

Drill: Name O SH H O

5 -cyclohexyl-2 -keto-3 mercapto-4 -phenylpentanal O SH H O

Amine Group N:

Ammonia : NH 3

Ammonium NH 4 +

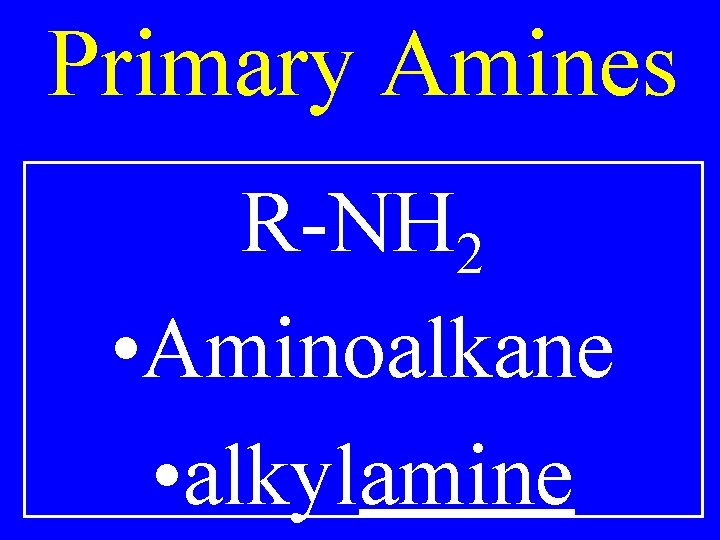
Amines R-NH 2 • Aminoalkane • alkylamine

Primary Amines R-NH 2 • Aminoalkane • alkylamine

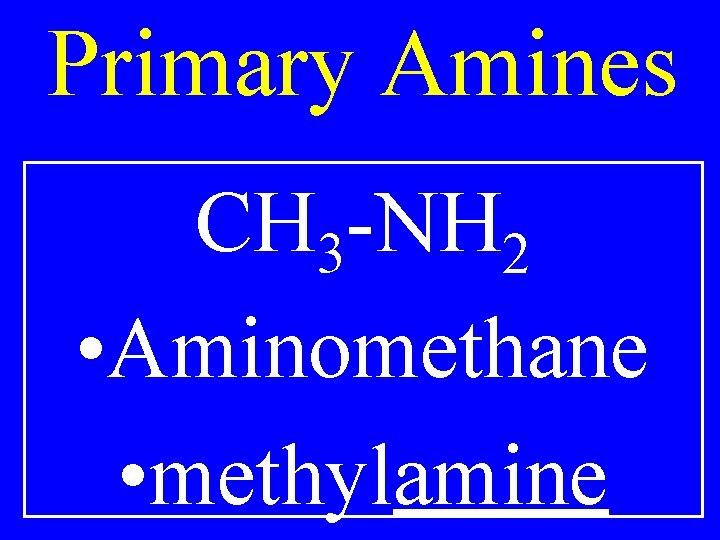
Primary Amines CH 3 -NH 2 • Aminomethane • methylamine

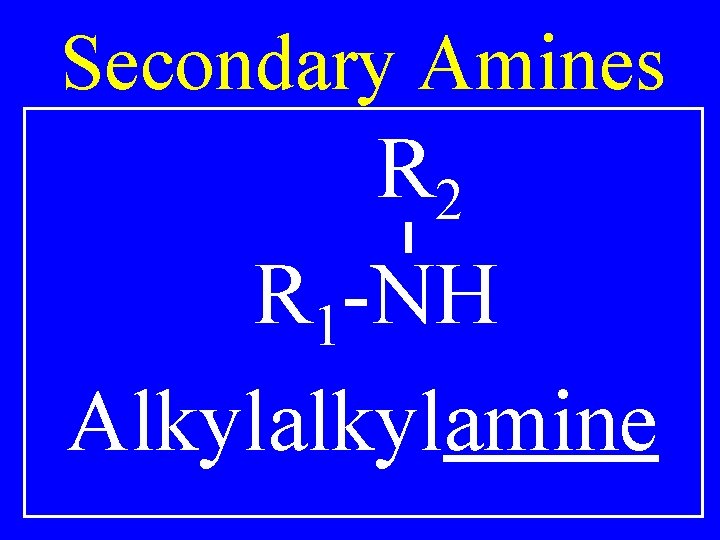
Secondary Amines R 2 R 1 -NH Alkylamine
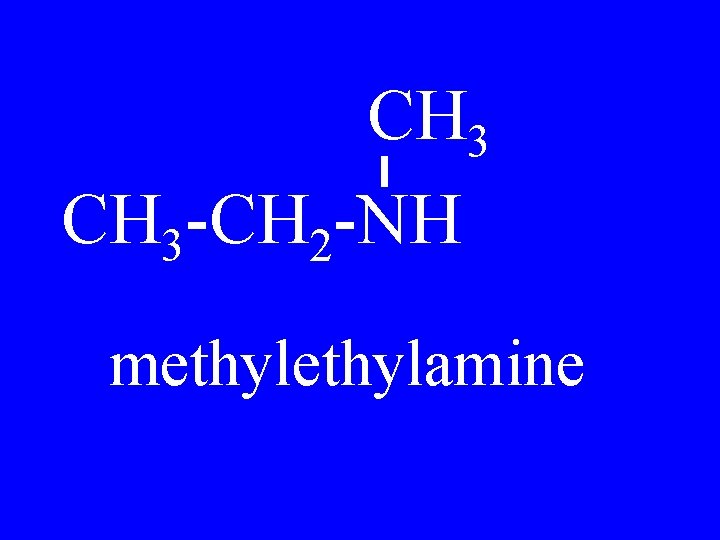
CH 3 -CH 2 -NH methylamine

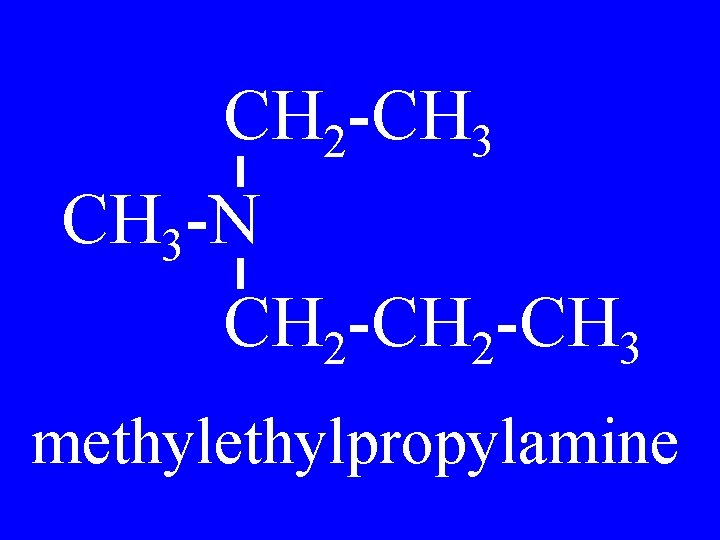
Tertiary Amines R 2 R 1 -N-R 3 Alkylalkylamine

CH 2 -CH 3 -N CH 2 -CH 3 methylpropylamine

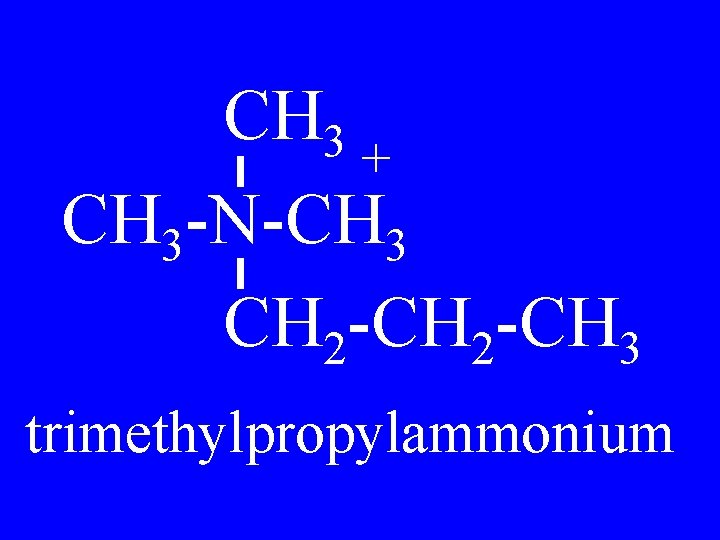
Quaternary Amines R 2 + R 1 -N-R 3 R 4 • Tetraalkylammonium

CH 3 + CH 3 -N-CH 3 CH 2 -CH 3 trimethylpropylammonium

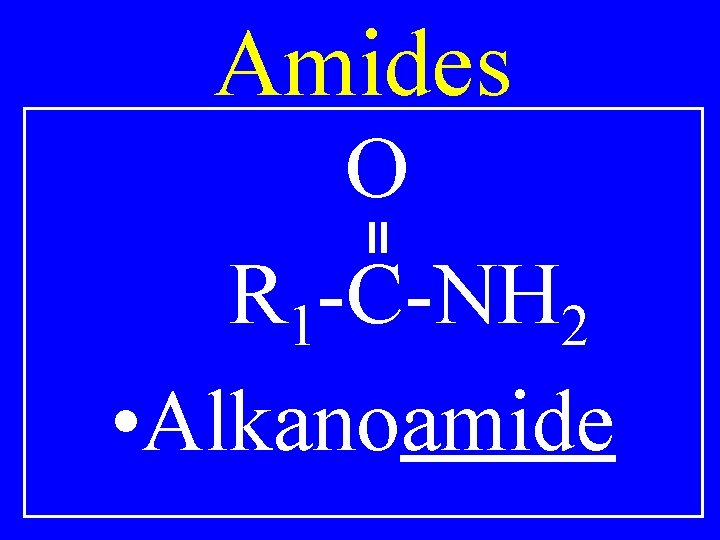
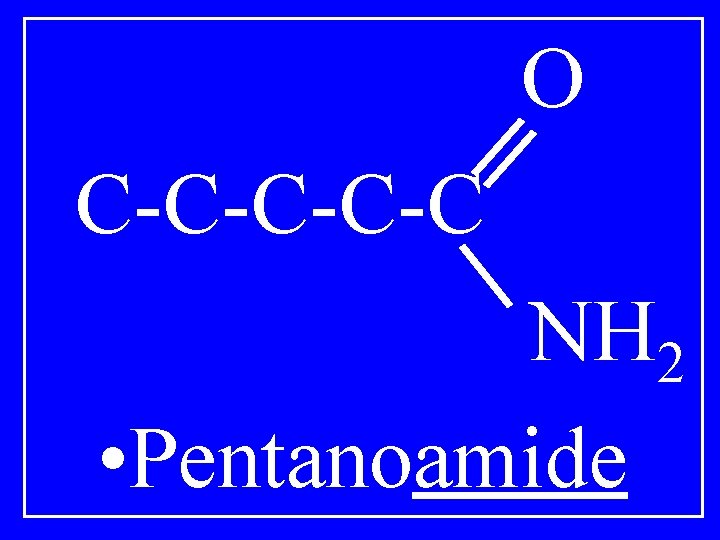
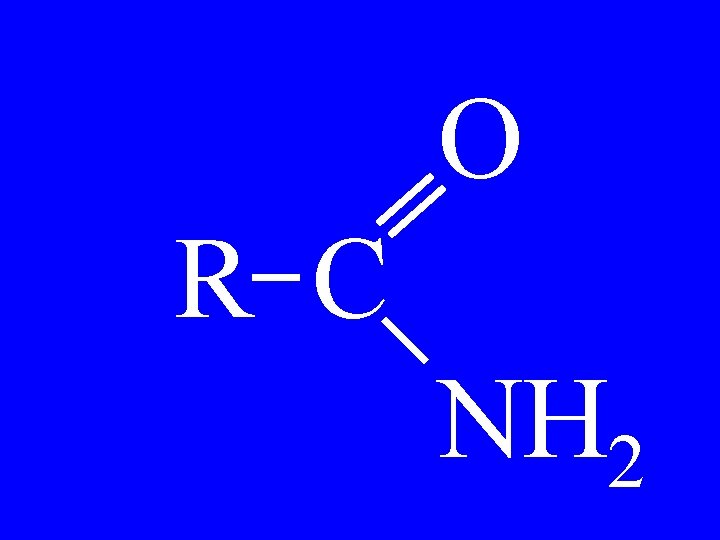
Amides O R 1 -C-NH 2 • Alkanoamide

O C-C-C NH 2 • Pentanoamide

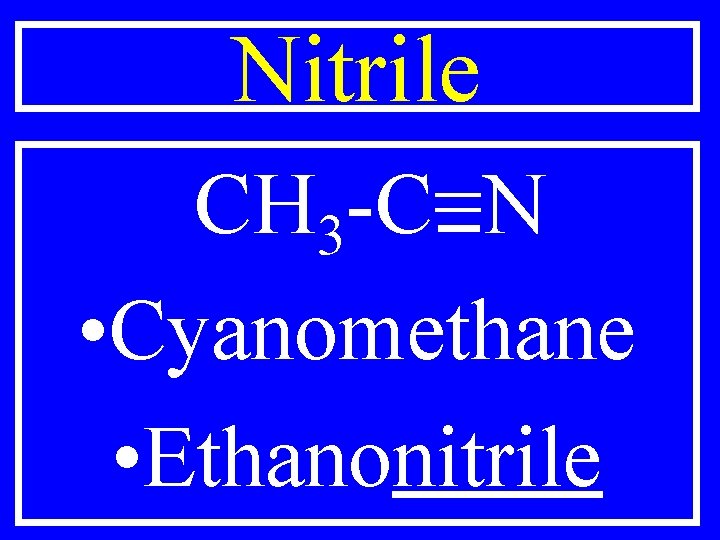
Nitrile R-C=N • Cyanoalkane • Alkanonitrile

Nitrile CH 3 -C=N • Cyanomethane • Ethanonitrile

Homework • Draw & Name 5 isomers of C 4 H 9 NO 2

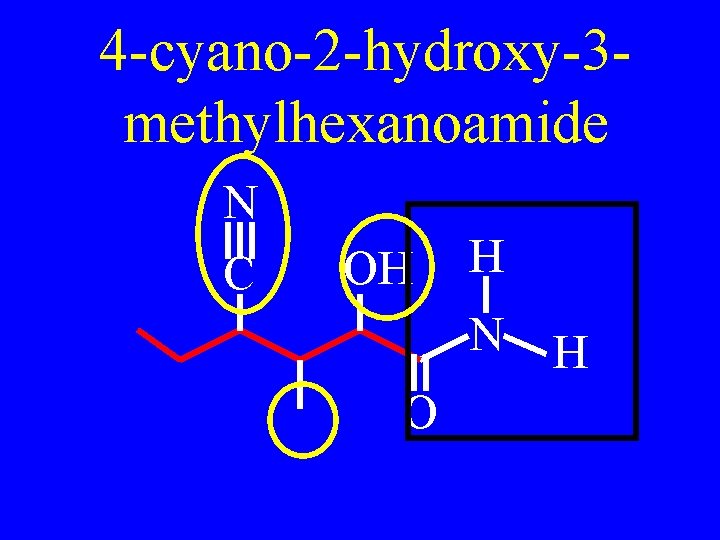
Name N C OH H N O H

Name N C OH H N O H

4 -cyano-2 -hydroxy-3 methylhexanoamide N C OH H N O H

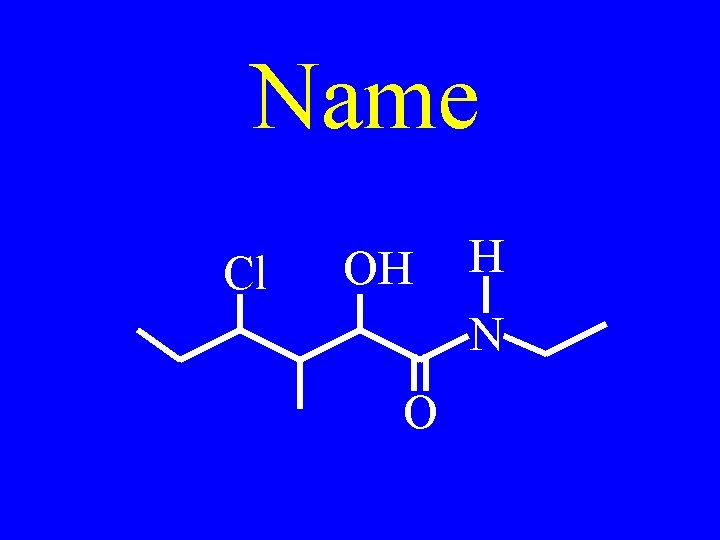
Name Cl OH H N O

Name Cl OH H N O

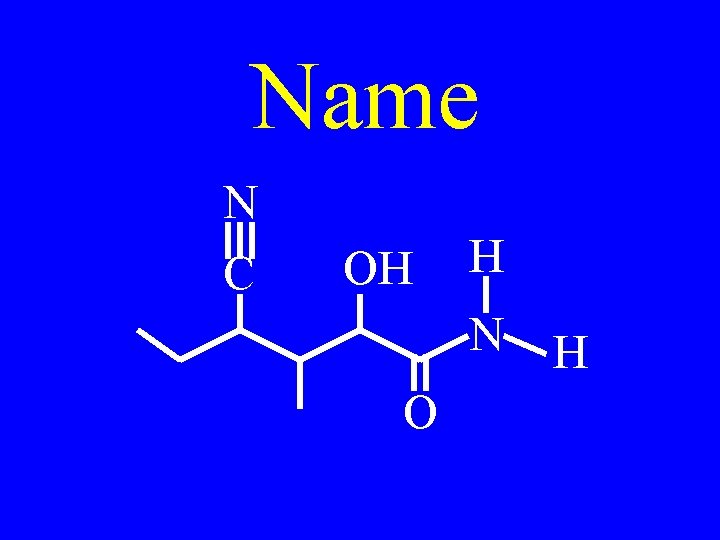
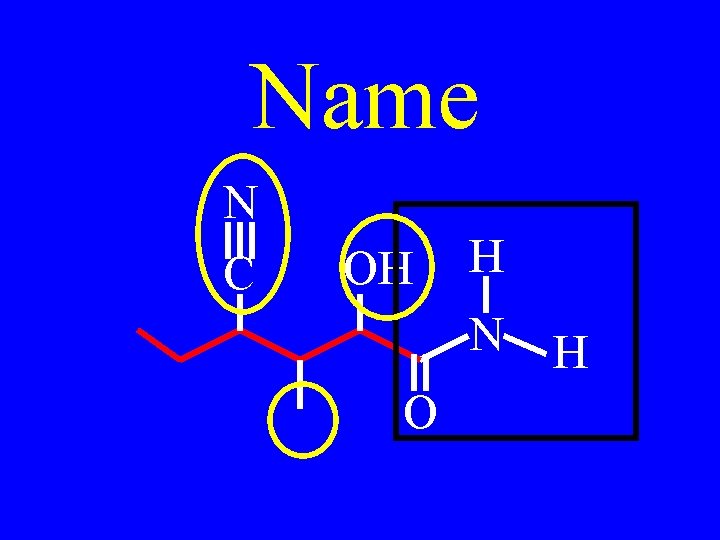
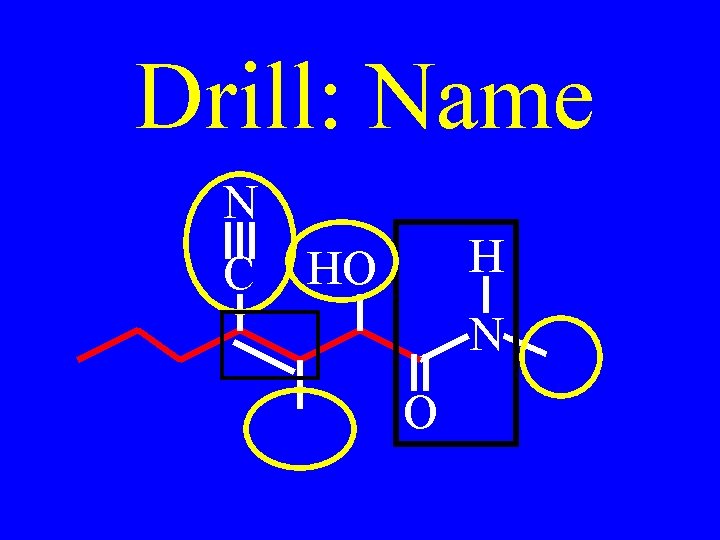
Drill: Name N C OH H N O

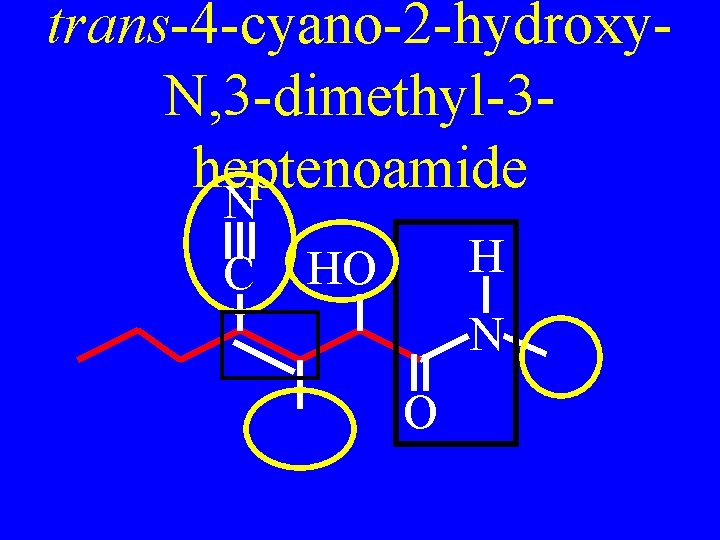
Drill: Name N C H HO N O

trans-4 -cyano-2 -hydroxy. N, 3 -dimethyl-3 heptenoamide N C H HO N O

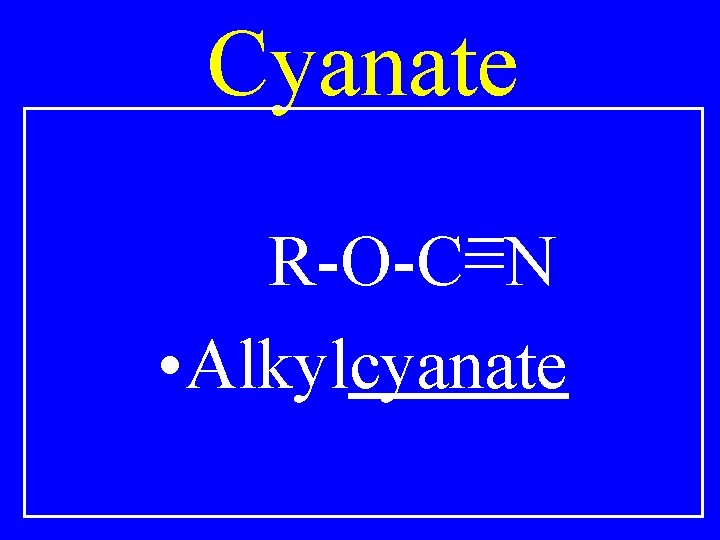
Cyanate R-O-C=N • Alkylcyanate

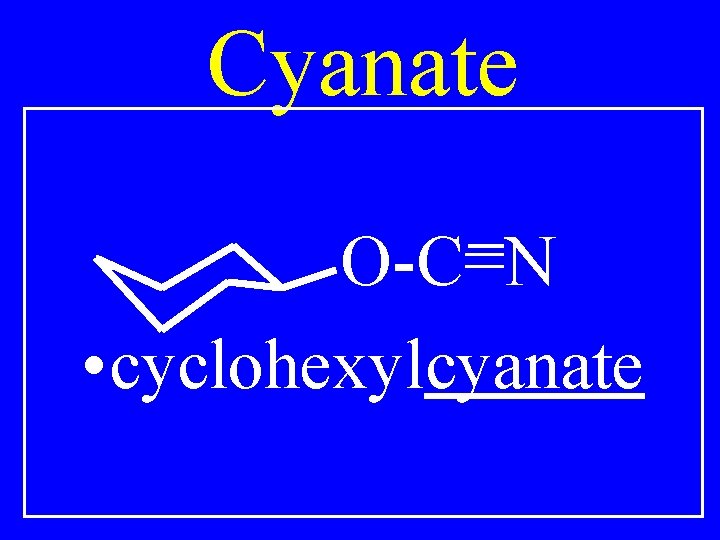
Cyanate O-C=N • cyclohexylcyanate

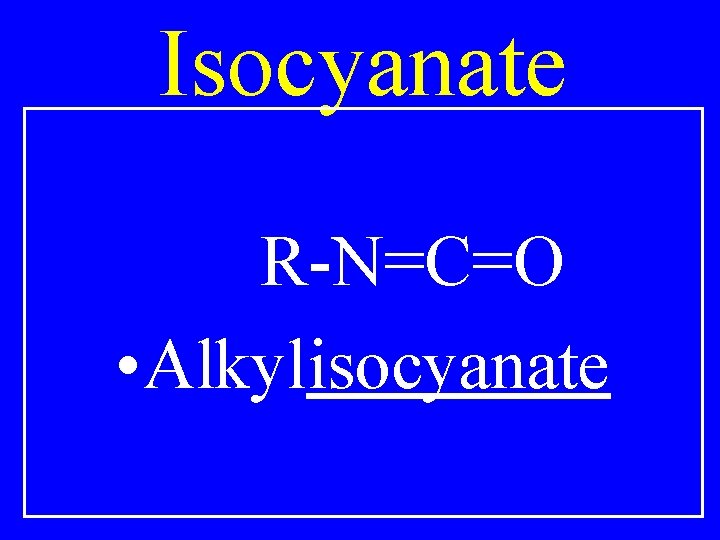
Isocyanate R-N=C=O • Alkylisocyanate

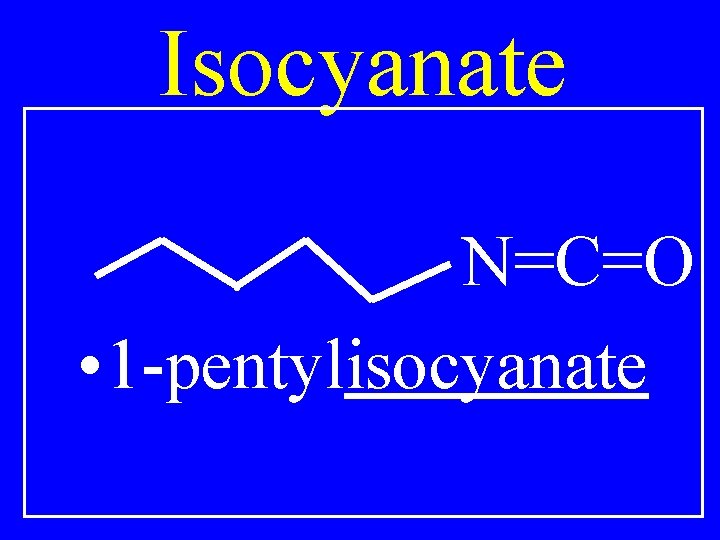
Isocyanate N=C=O • 1 -pentylisocyanate

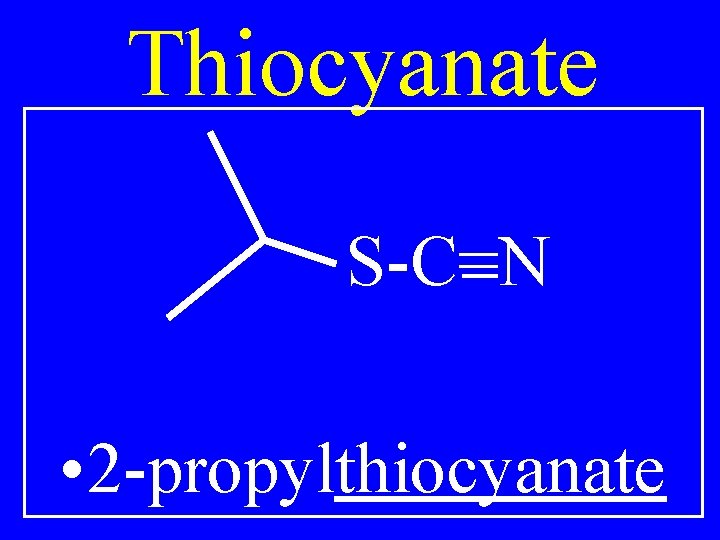
Thiocyanate R-S-C=N • Alkylthiocyanate

Thiocyanate S-C=N • 2 -propylthiocyanate

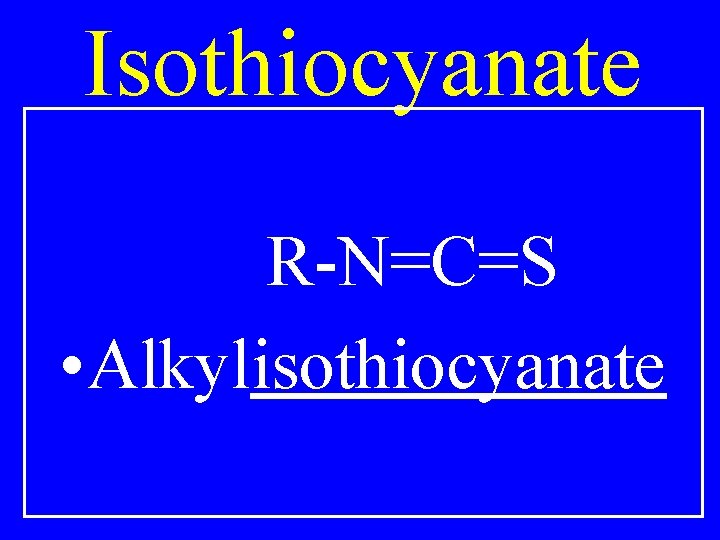
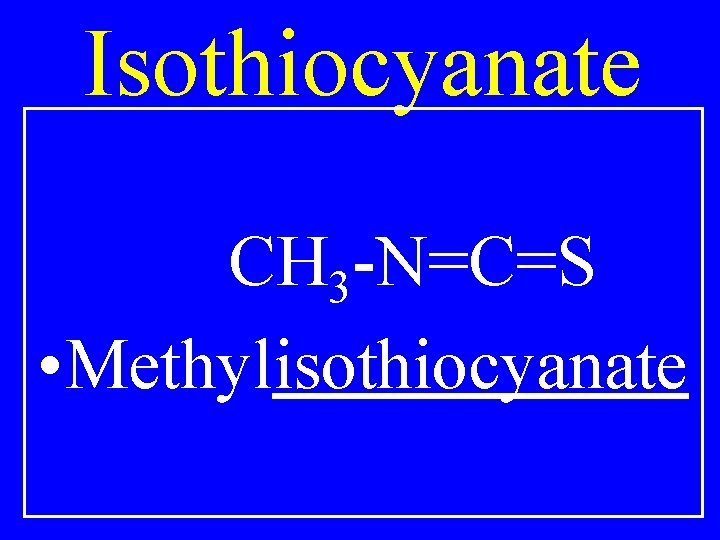
Isothiocyanate R-N=C=S • Alkylisothiocyanate

Isothiocyanate CH 3 -N=C=S • Methylisothiocyanate

Lab: 05 • Functional Groups Containing Nitrogen

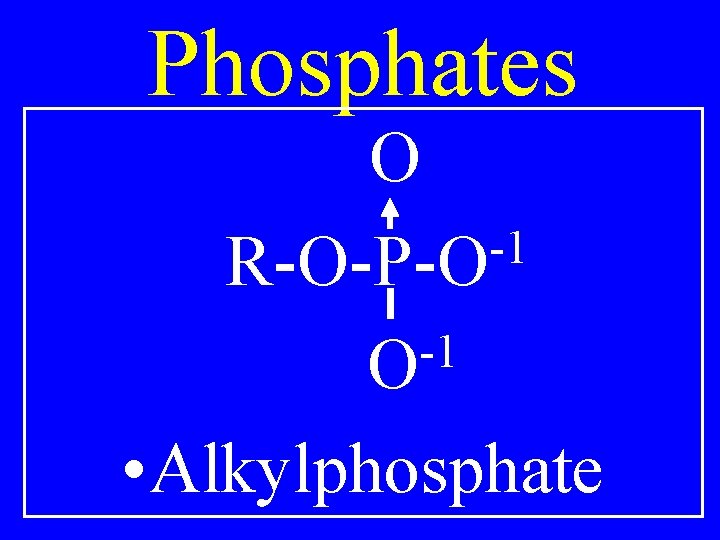
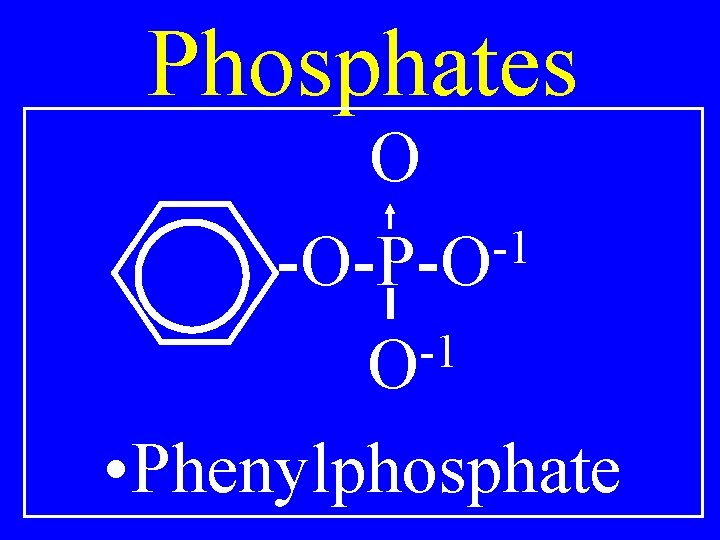
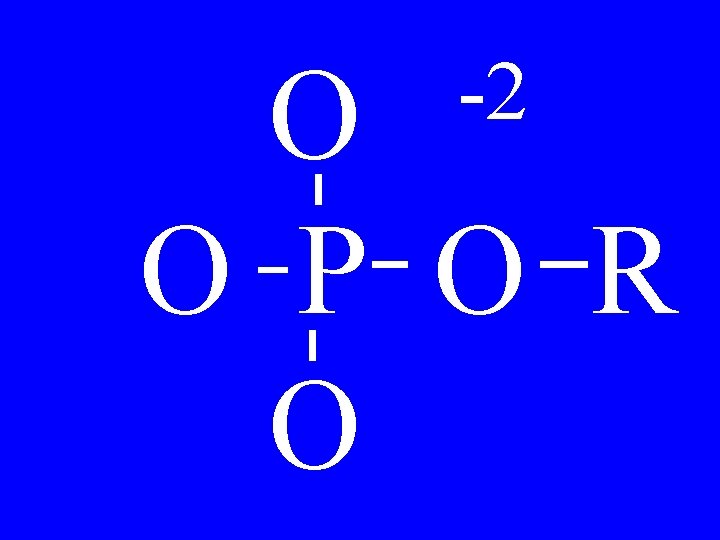
Phosphates O -1 R-O-P-O -1 O • Alkylphosphate

Phosphates O -1 -O-P-O -1 O • Phenylphosphate

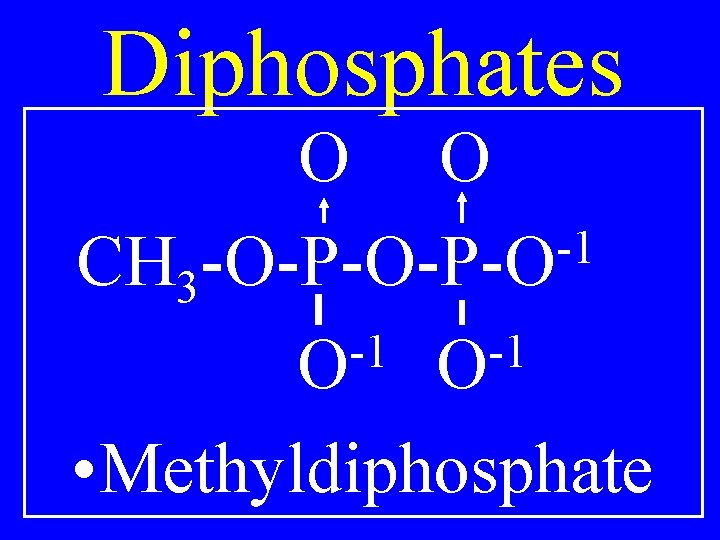
Diphosphates O O -1 R-O-P-O -1 -1 O O • Alkyldiphosphate

Diphosphates O O -1 CH 3 -O-P-O -1 -1 O O • Methyldiphosphate

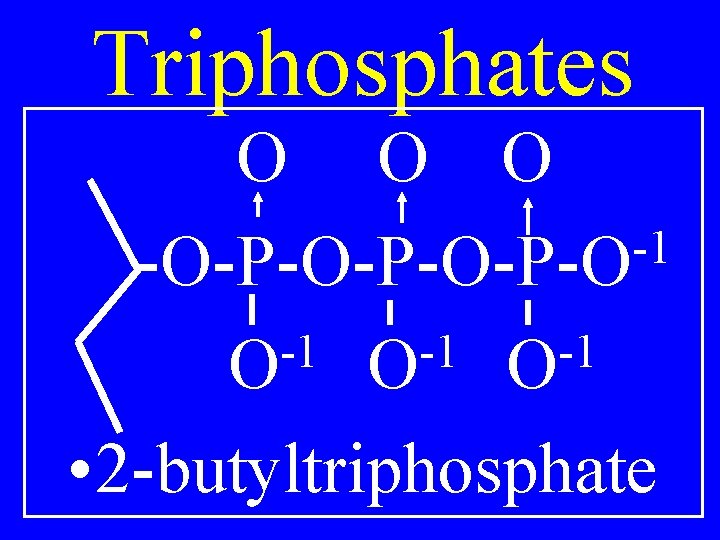
Triphosphates O O O -1 R-O-P-O-P-O -1 -1 -1 O O O • Alkyltriphosphate

Triphosphates O O O -1 -O-P-O-P-O -1 -1 -1 O O O • 2 -butyltriphosphate

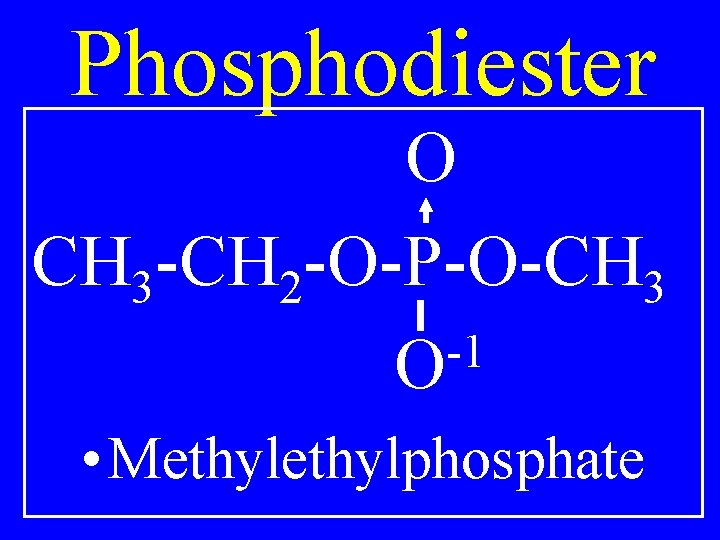
Phosphodiester O CH 3 -CH 2 -O-P-O-CH 3 -1 O • Methylphosphate

Drill: Draw & Name 5 isomers of: C 3 H 7 NO 2

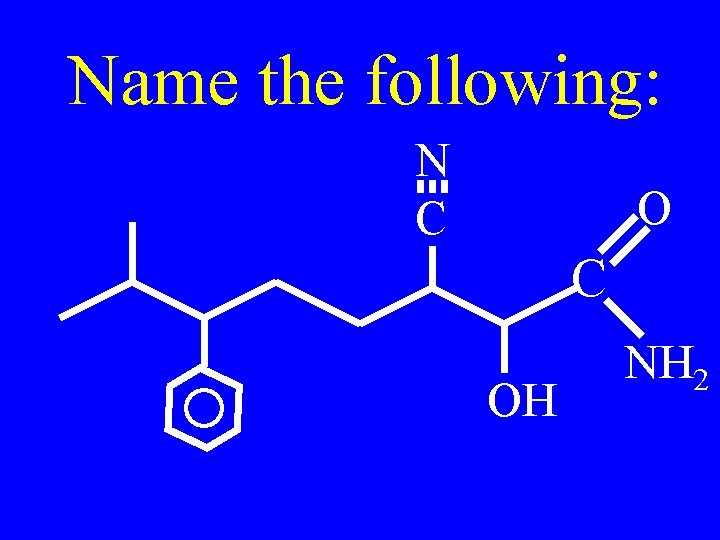
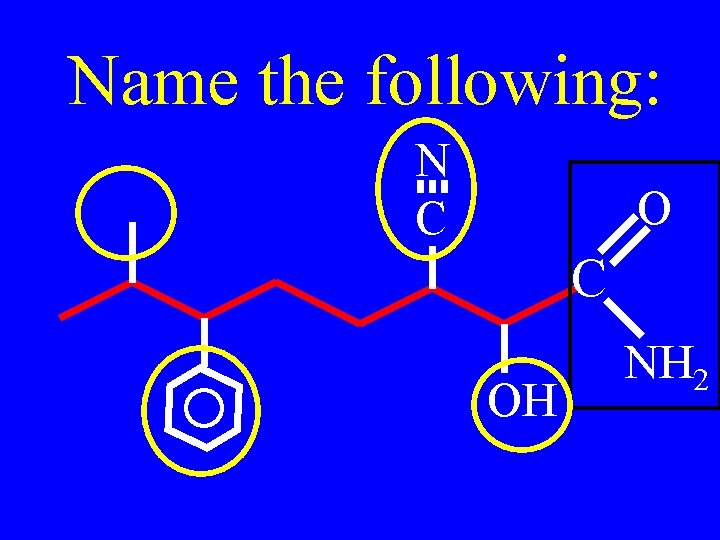
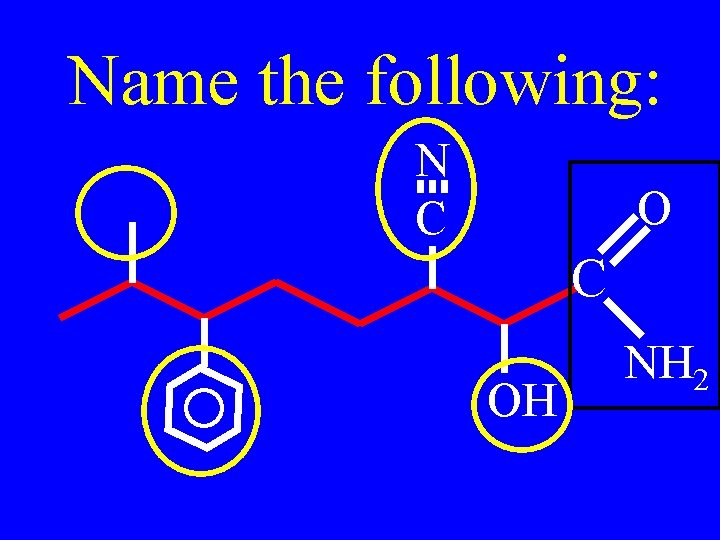
Name the following: N C OH NH 2

Name the following: N C OH NH 2

Name the following: N C OH NH 2

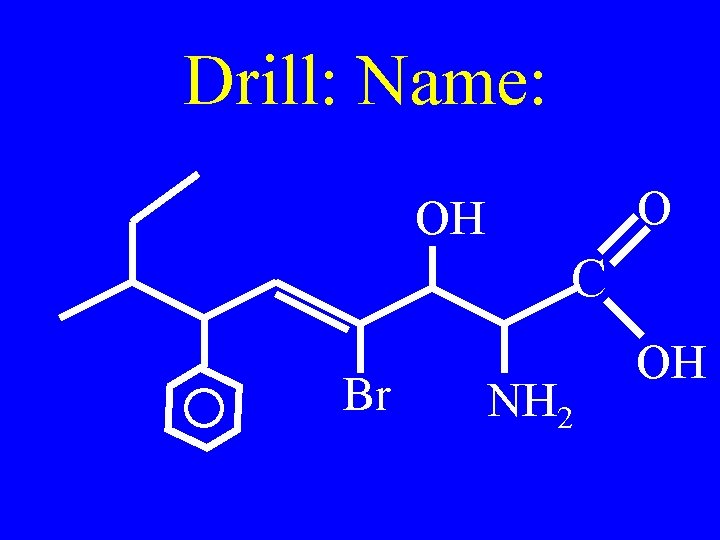
Drill: Name: O OH C Br NH 2 OH

Name the following: O OH C Br NH 2 OH

Organic HW • Draw & Name 5 isomers of: C 3 H 6 SO

Drill: Draw & Name 5 Isomers of: C 4 H 7 NO

Review: Today Lab: Tomorrow Test : Monday

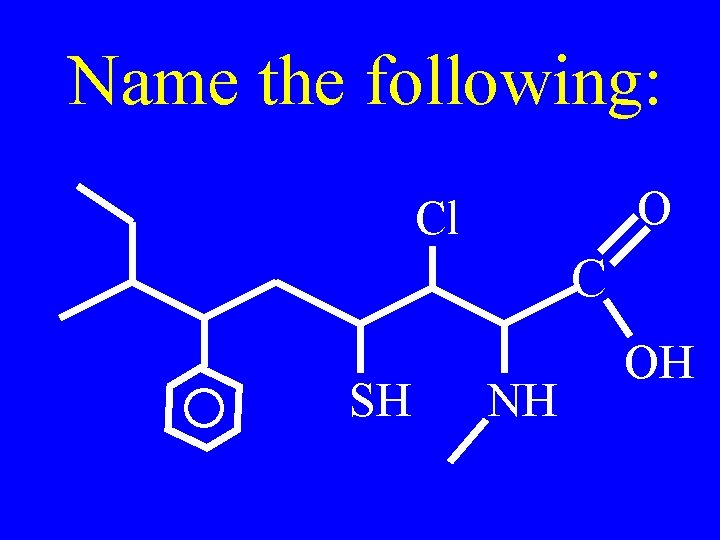
Name the following: O Cl C SH NH OH

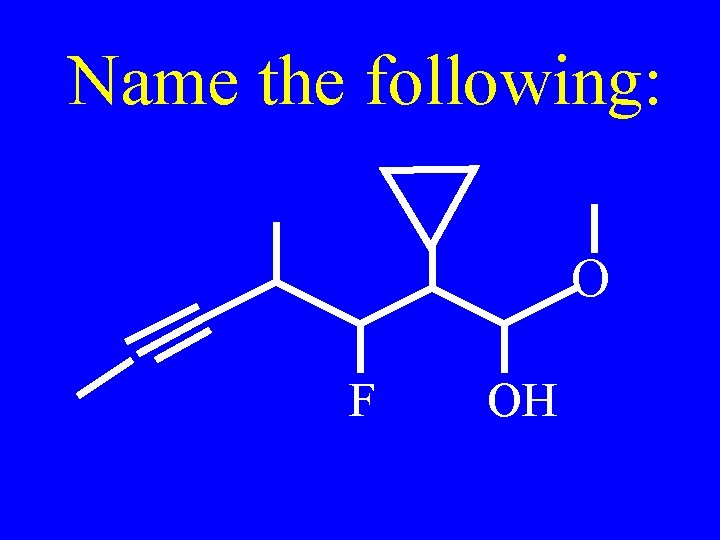
Name the following: O F OH

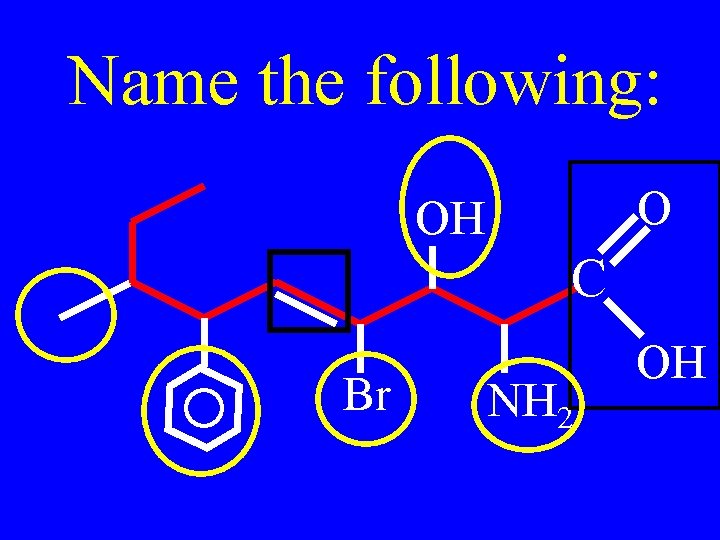
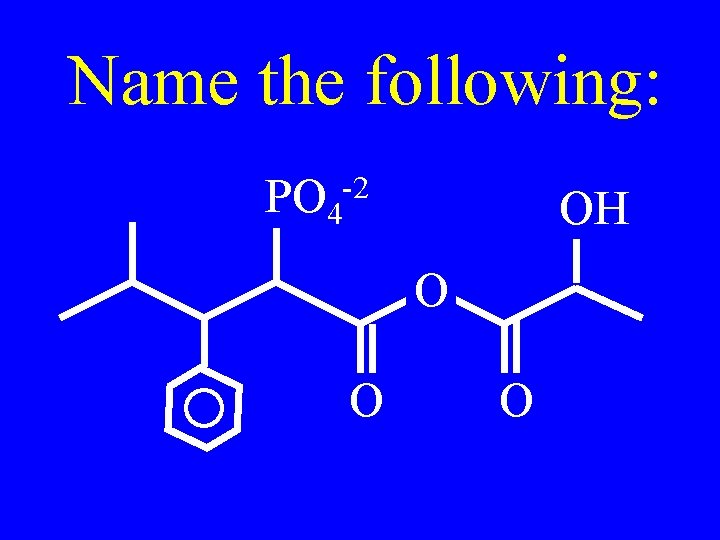
Name the following: PO 4 -2 OH O O O

Draw: • 3 -mercaptohexanoamide • Methyl-2 -hydroxythioacetate • Para-iodophenylformic acid • Phenylpropylamine • 2 -amino-3 -ethyl-4 -keto-Nmethylhexanaldehyde

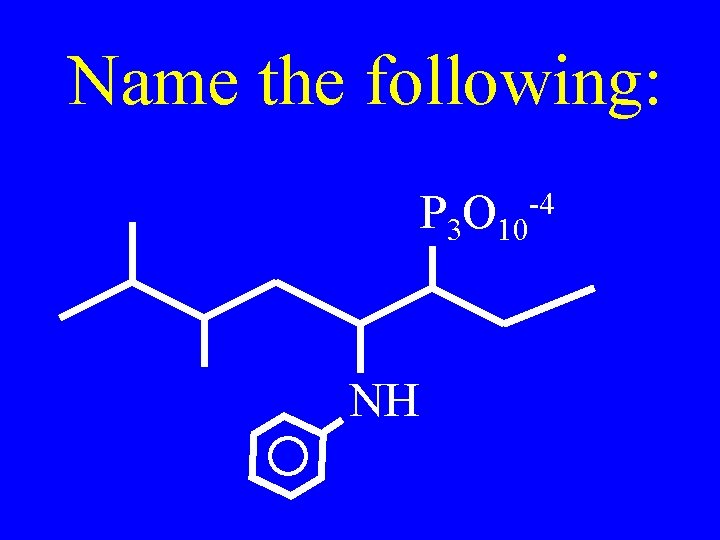
Name the following: P 3 O 10 -4 NH

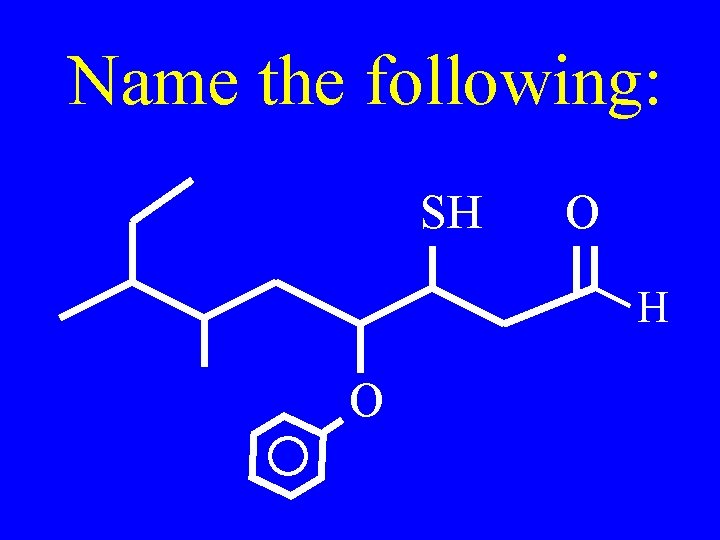
Name the following: SH O

Drill: Name: N C NH 2 O OH OH

Name the following: N C S O O Br

Name the following: O Cl OH

Drill: Name the following: O O F O

Name the following: N C S OH O

Draw the Following: • 3 -methyl-5 -propyloctanoamide • Octylbutanoate

Draw the Following: • Aceticpentanoic anhydride • 2 -cyano-3 mercaptohexanoic acid

Drill Draw & name 5 isomers of: • C 4 H 7 NO

Draw: • Benzoic acid
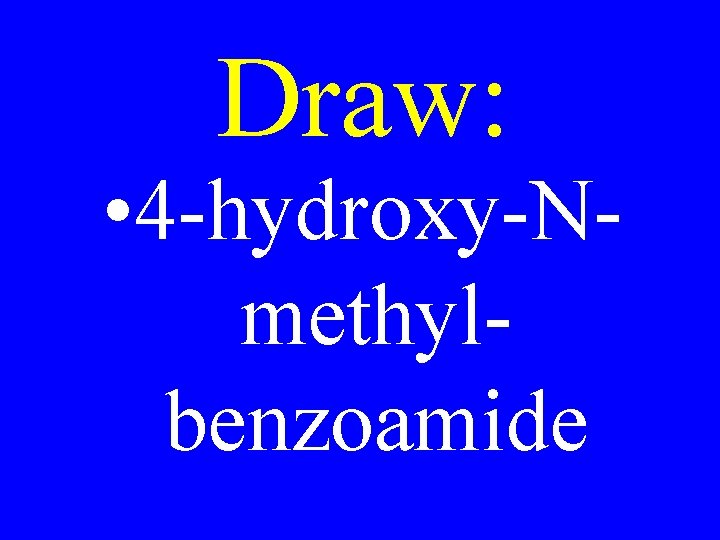
Draw: • 4 -hydroxy-Nmethylbenzoamide

Drill: Draw: 4 -hydroxy-2 mercapto pentanoic acid

Review

C=C

O R C O R
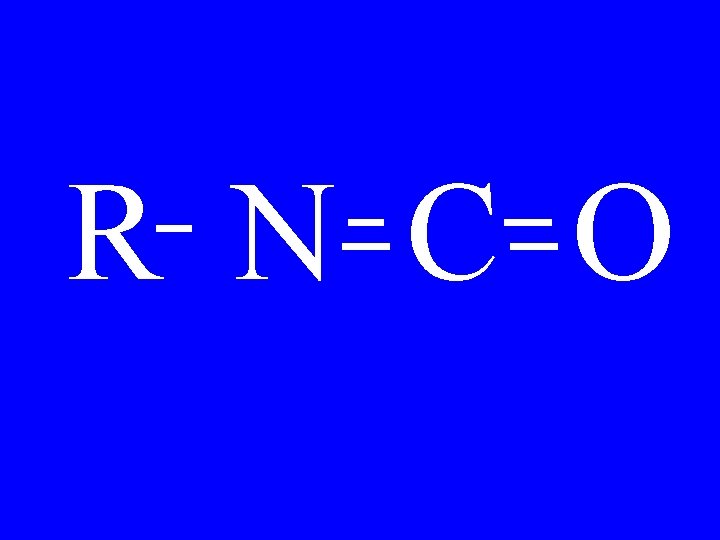
R N C O

R-SH

R-NH 2

O R C OH

O C R R

R-OH

O C R O C O R

R C N

H : N R R

O R C NH 2

O R C H

RO C N

R 1 -O-R 2

R-I

R + R N R R

C=C

R + R NH R

R 1 -S-R 2

O R C S R

R S C N

O O P O R O -2

R : N R R

R-S-S-R
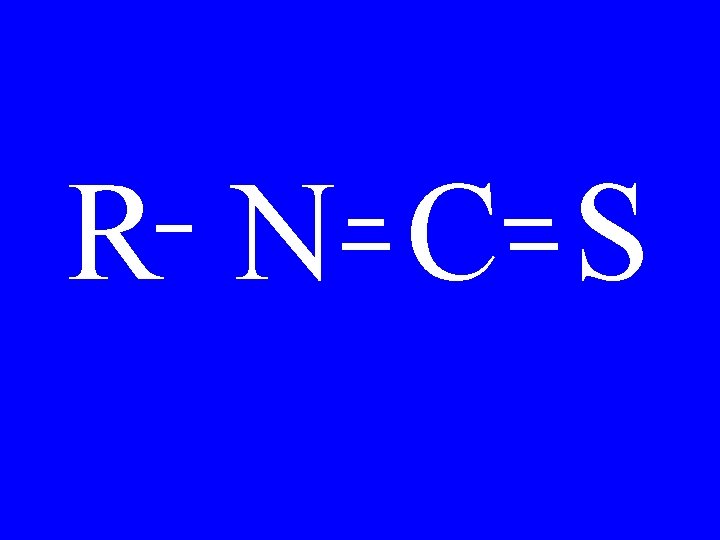
R N C S

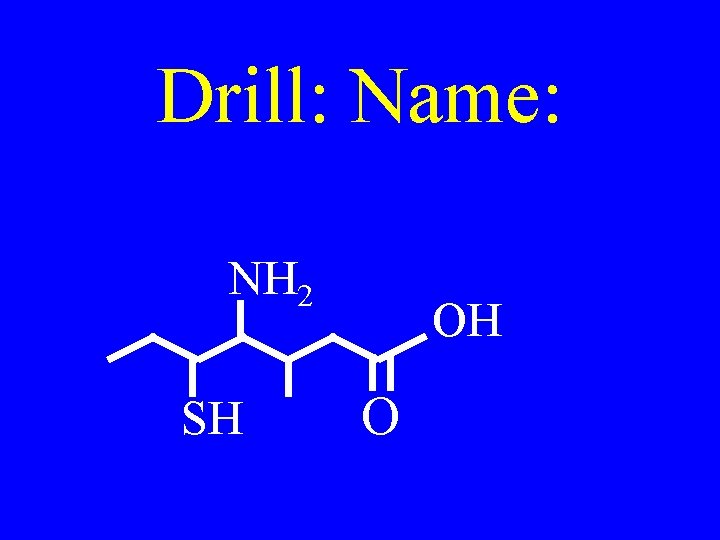
Drill: Name: NH 2 SH OH O

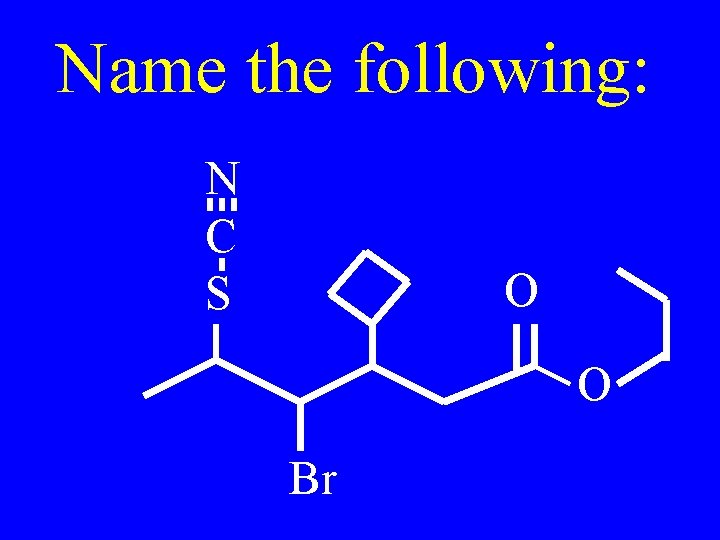
Name the compounds on the board

Draw the compounds on the board

Draw & Name 10 isomers of C 4 H 7 NO