Drill Draw LDDs for BF 3 NH 3
Drill: Draw LDDs for: BF 3 NH 3 C 3 H 6 C 3 H 7 NO

Review & Collect Drill & HW

CHM II • Test Thursday or Friday

CHM II HW • Read: PP-18 • Complete the attached assignment & turn it in tomorrow.

Solutions

Solution • Homogeneous mixture made up of at least one solute dissolved in the solvent

Solute • Substance being dissolved • Portion in lesser molar amount

Solvent • Substance doing the dissolving • Portion in greatest molar amount
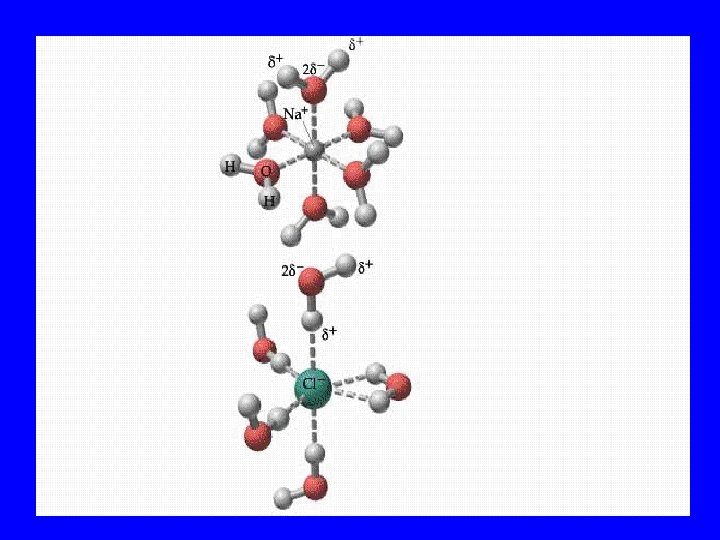

Colloid • Slightly larger particles • Light passes & particles stay suspended

Suspension • Even larger particles • Particles block or reflect light

Tyndall Effect • Because light reflects off suspended particles, the light ray can be seen from the side

Size Comparison • Solution < Colloid • Colloid < Suspension

Soluble • When one substance (solute) dissolves in another (solvent)

Solubility • The max amount of one substance (solute) dissolved in another (solvent)

Concentration • The amount of solute dissolved into solution

Concentrated Solution • A solution with a relatively large amount of solute dissolved

Dilute Solution • A solution with a relatively small amount of solute dissolved

Saturated Solution • A solution with the maximum amount of solute dissolved in the solution

Unsaturated Solution • A solution with less than the maximum amount of solute dissolved in solution

Supersaturated Solution • A solution with greater than the maximum amount of solute dissolved in solution

CHM II HW • Review PP-18 • Complete the worksheet attached to the Poly Website, & turn it in tomorrow.
Drill: Draw LDDs for: C 4 H 8 HNO 3 C 4 H 6

Review Drill & HW

Solution Chemistry

Solution Measures • Concentration: % soln –Molarity –Molality –Mole Fraction

Percent Solution • Mass of one portion per the total mass, all times 100 % • %soln = ma/mtotal x 100 %

Molarity • Moles of solute per liter of solution • M = molessolute/Lsoln

Molality • Moles of solute per kilogram of solvent • mo = molessolute/kgsolvent

Mole Fraction • Moles of one portion per total number of moles in the solution • X = molesa/molessoln

Calculate the molarity of a 250 m. L solution containing 5. 0 g Na. OH dissolved in water

Calculate the molality of 69 g of C 2 H 5 OH dissolved in 500. 0 m. L of water

Calculate the mole fraction of each portion when 92 g of C 2 H 5 OH dissolved in 144 m. L of water

Calculate the molality & mole fraction of a solution containing 46 g of C 2 H 5 OH dissolved in 1782 m. L of water

Drill: • Calculate the mass of KI required to make 250 m. L of 0. 500 M KI.

Review Drill Review HW

We will have a test on LDDs & Solution Chemistry by the end of this week.

CHM II HW • Review PP-18 • Complete the attached problem (CP-1) & turn it in tomorrow.

Colligative Properties • Properties dependent only on the concentration of particles in solution

Examples • Vapor pressure • Boiling & Freezing points • Osmotic pressure
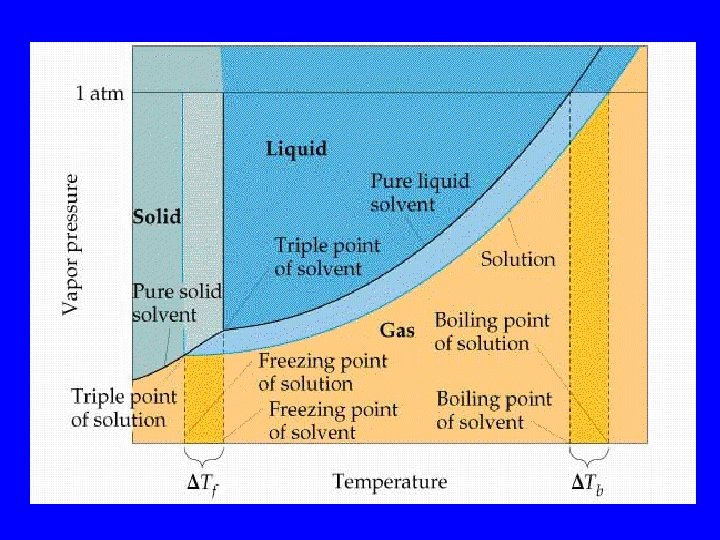

Vapor Pressure • VPsolution = (VPsolvent)(Xsolvent) • X = mole fraction • VP = vapor pressure

Boiling & Freezing DT = imo. K DT = change in BP or FP i = ionic activity K = BP or FP constant

Osmotic Pressure p = i. MRT p = osmotic pressure i = ionic activity M = Molarity

Calculate the vapor pressure of a solution containing 150 g C 5 H 10 O 5 in 162 m. L of o water at 30 C

Calculate BP & FP of 60. 0 g of Na. OH in 250 m. L water o KBP = 0. 512 C/mo o KFP = -1. 86 C/mo

Calculate the osmotic pressure of a solution containing 12 g of Na. OH dissolved in 250 m. L solution at o 27 C

Drill: • Calculate the VP of a solution containing 36 % m/m glucose (C 6 H 12 O 6) in o water at 29 C: • (VPwater = 30. 0 mm Hg)

Review Drill & Check HW

CHM II HW • Review PP-18 • Test: Thursday

Test Review

Classify the type of Bonding: Fe-Cl C-Cl S-O Fe-Cr H-Br K-I

Classify the type of IM Force: H 2 & Cl 2 HF & HF H 2 & HCl HBr & HI CH 4 & C 2 H 6
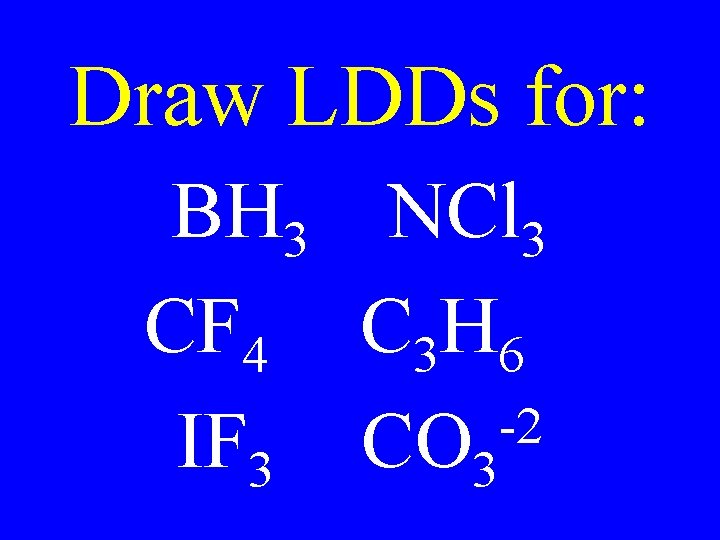
Draw LDDs for: BH 3 NCl 3 CF 4 C 3 H 6 -2 IF 3 CO 3

Calculate the FP & BP of a solution containing 120 g C 3 H 7 OH in 250 m. L of water at its BP. o KBP = 0. 512 C/mo o KFP = -1. 86 C/mo

Drill: Calculate the molarity of 33. 1 g of Pb(NO 3)2 dissolved in 250 m. L of solution.

Review & Collect Drill & HW

CHM II HW • Review for the test covering tomorrow. • The test covers Bonding, LDDs, & Solution Chemistry (PP 15 & 18).
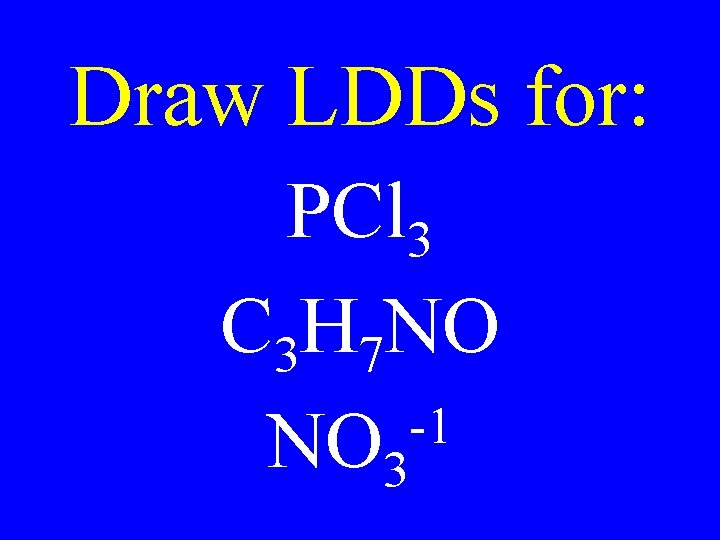
Draw LDDs for: PCl 3 C 3 H 7 NO -1 NO 3

Calculate the osmotic pressure of a solution containing 12 g of C 4 H 8 O 4 dissolved in o 50. 0 m. L solution at 27 C

Calculate the vapor pressure of a solution containing 12. 0 g C 3 H 8 O in 14. 4 m. L of water at its BP. VPsolvent = 120 k. Pa

Calculate the mass of lead(II)nitrate required to make 250 m. L of 0. 40 M Pb(NO 3)2

Calculate the BP of a solution containing 29. 9 g of Co. Br 3 dissolved in 75 m. L of water. o KBP = +0. 512 C/mo

Calculate the molality of a solution that is 33. 1 % by mass Pb(NO 3)2 in water

Drill: Calculate the osmotic pressure of 0. 88 g of CO 2 in 750 m. L o of soln at 27 C

30. 0 g C 3 H 6 NF was dissolved in 50. 0 g C 6 H 12 O (VP = 20. 0 k. Pa) o at 27 C making a 0. 800 g/m. L solution. Calculate: X, mo, M, p, & VP.

180 g C 3 H 8 O was dissolved in 180 m. L o H 2 O at 27 C making a 1. 5 g/m. L solution. Calculate X, mo, M, p, VP, BP, & FP.

3 g/cm A 1. 2 aqueous solution is 20. 0 % by o mass Na. OH at 27 C. Calculate: X, mo, M, p, & MP

Calculate the molecular mass of a covalent compound dissolved in an aqueous solution to make it 25 % by mass when it o boils at 102. 048 C
- Slides: 69