DRILL Complete the Table Assume net charge is
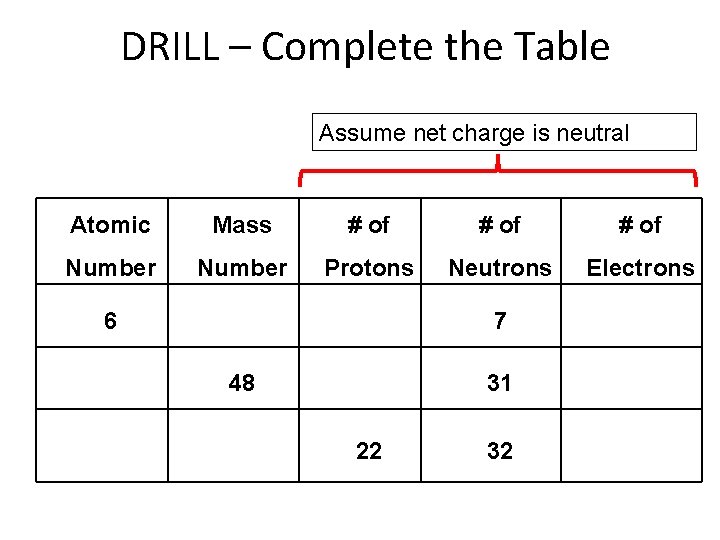
DRILL – Complete the Table Assume net charge is neutral Atomic Mass # of Number Protons Neutrons Electrons 6 7 48 31 22 32

PARTNER CHALLENGE • You have been given a variety of shapes, that have different colors and shapes. • Arrange the shapes in an order that makes sense to you. • Be prepared to defend you organization.

PARTNER WHITEBOARD • Look at the copy of the periodic table you have been given. • List the ways that you think the periodic table has been organized. (Look for the patterns that you see. )

IV. The Periodic Table 4 3. 3 Periodic Table 9. 17. 00 1: 37 PM
The Periodic Table and the Elements Who developed the periodic table ? How much information is on the periodic table ? What trends can we see on the periodic table ? 5 3. 3 Periodic Table 9. 17. 00 1: 37 PM

A. Who developed the periodic table? Answer while watching the following video clips. 1. How did Newlands organize the elements? 2. What was his theory called? 3. Was it accepted? Why or why not? 4. What was his faulty assumption? 5. Who tried next? 6. How did he look for the pattern? 7. How did he arrange the elements? 8. Horizontal rows were called what? 9. What was true about columns? 10. What did he assume the gaps to be? 11. What did he do regarding these gaps? 6 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Video: Newlands 7 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Mendeleevhttp: //www. schooltube. com/video/ 03 d 81990 c 70 d 65720246/Periodic-Table-of. Elements-Chemistry-A-Volatile-History-BBCFour 8 3. 3 Periodic Table 9. 17. 00 1: 37 PM
1. 9 What change did Moseley make? 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Video: Dmitri Mendeleev 10 3. 3 Periodic Table 9. 17. 00 1: 37 PM

A. Who developed the periodic table? How did Newlands organize the elements? By increasing atomic weight 2. What was his theory called? The Law of Octaves 3. Was it accepted? Why or why not? No, too many elements didn’t fit the pattern 4. What was his faulty assumption? He thought all elements had been discovered. 5. Who tried next? Mendeleev 6. How did he look for the pattern? By writing the information for each element on a card & laying them out in a pattern. 1. 11 3. 3 Periodic Table 9. 17. 00 1: 37 PM

7. How did he arrange the elements? By increasing weight and chemical properties 8. Horizontal rows were called what? Periods 9. What was true about columns? They were families with similar properties. 10. What did he assume the gaps to be? Undiscovered elements 11. What did he do regarding these gaps? Predicted the properties of missing elements 12. What change did Moseley make? He put the elements in order of atomic number instead of atomic weight. 12 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Dmitri Mendeleev (1869) In 1869 Mendeleev and Lothar Meyer (Germany) published nearly identical classification schemes for elements known to date. The periodic table is based on the similarity of properties and reactivities exhibited by certain elements. Later, Henri Moseley ( England, 1887 -1915) established that each elements has a unique atomic number, which is how the current periodic table is organized. http: //www. chem. msu. su/eng/misc/mendeleev/welcome. html 13 3. 3 Periodic Table 9. 17. 00 1: 37 PM
14 3. 3 Periodic Table 9. 17. 00 1: 37 PM


HOMEWORK SQ 3 R – Pg. 516 – 524 SHORT QUIZ ON INSERT DATE (This is not the quiz that you need to memorize the names and symbols of elements for. The quiz will cover atomic structure, atomic mass, isotopes, protons, neutrons, electrons, etc. ) 16 3. 3 Periodic Table 9. 17. 00 1: 37 PM

DRILL 17 A) What are two elements that will have properties that are similar to chlorine? B) How do you know they elements will have similar properties? 3. 3 Periodic Table 9. 17. 00 1: 37 PM

At the top of your notes… Date: INSERT DATE Objective: SWBAT explain how elements are organized in the periodic table. 18 3. 3 Periodic Table 9. 17. 00 1: 37 PM
The Periodic Table and the Elements Who developed the periodic table ? How much information is on the table ? What trends can we see on the PT ? 19 3. 3 Periodic Table 9. 17. 00 1: 37 PM
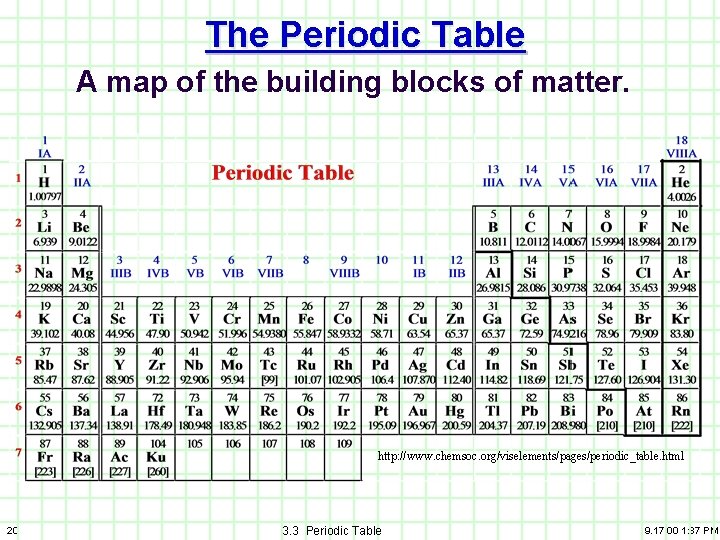
The Periodic Table A map of the building blocks of matter. http: //www. chemsoc. org/viselements/pages/periodic_table. html 20 3. 3 Periodic Table 9. 17. 00 1: 37 PM

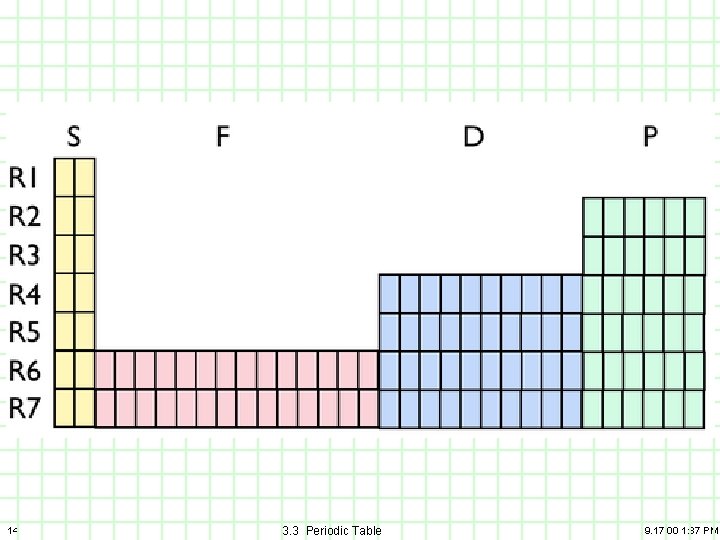
1) Main Classes elements A) B) C) 21 Main or Representative Transition Metals Lanthanides and Actinides (rare earth elements) 3. 3 Periodic Table 9. 17. 00 1: 37 PM
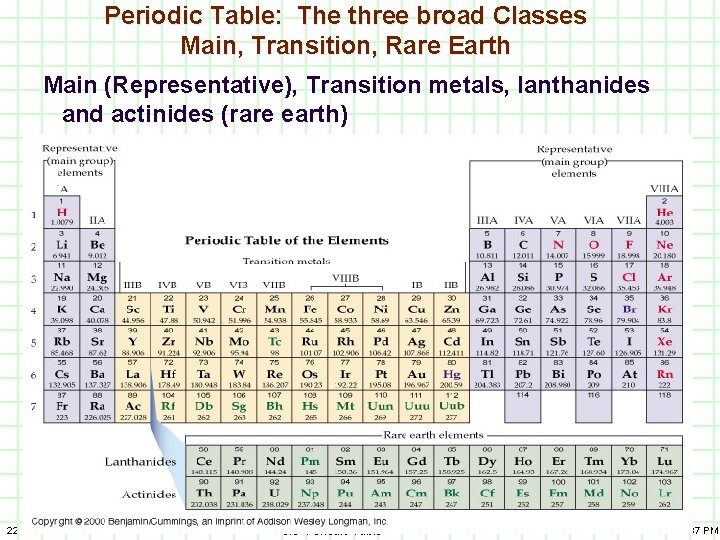
Periodic Table: The three broad Classes Main, Transition, Rare Earth Main (Representative), Transition metals, lanthanides and actinides (rare earth) 22 3. 3 Periodic Table 9. 17. 00 1: 37 PM

2) Types of elements A) B) C) 23 Non-metals Metalloids 3. 3 Periodic Table 9. 17. 00 1: 37 PM
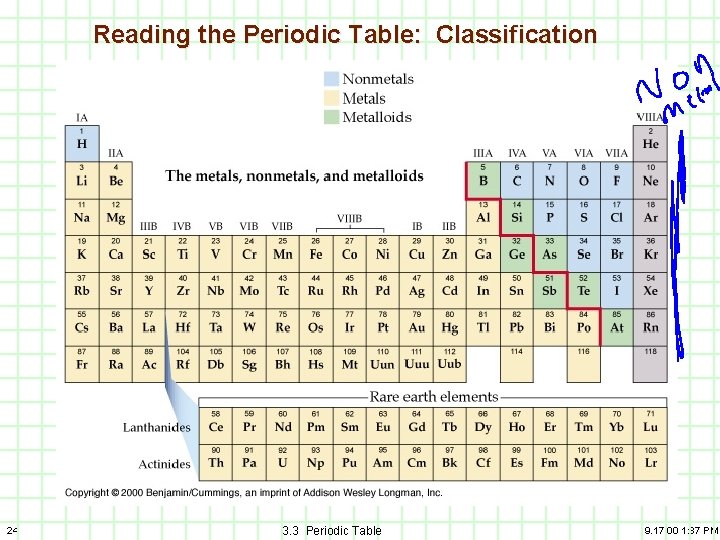
Reading the Periodic Table: Classification Nonmetals, Metalloids, Noble gases 24 3. 3 Periodic Table 9. 17. 00 1: 37 PM
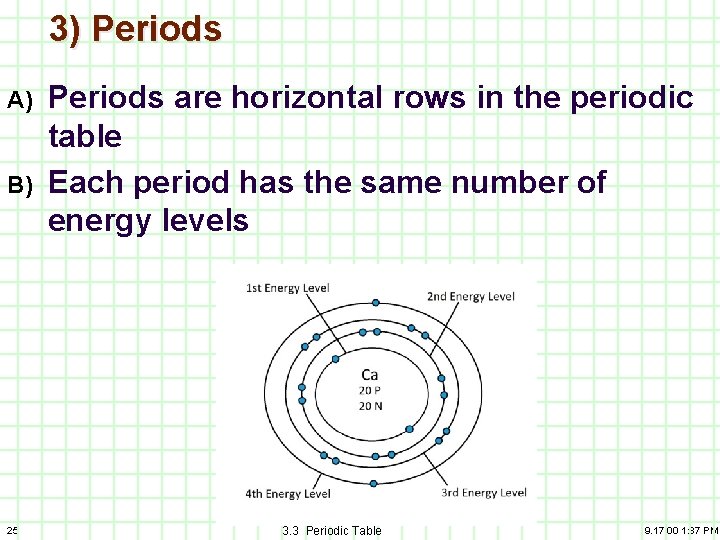
3) Periods A) B) 25 Periods are horizontal rows in the periodic table Each period has the same number of energy levels 3. 3 Periodic Table 9. 17. 00 1: 37 PM
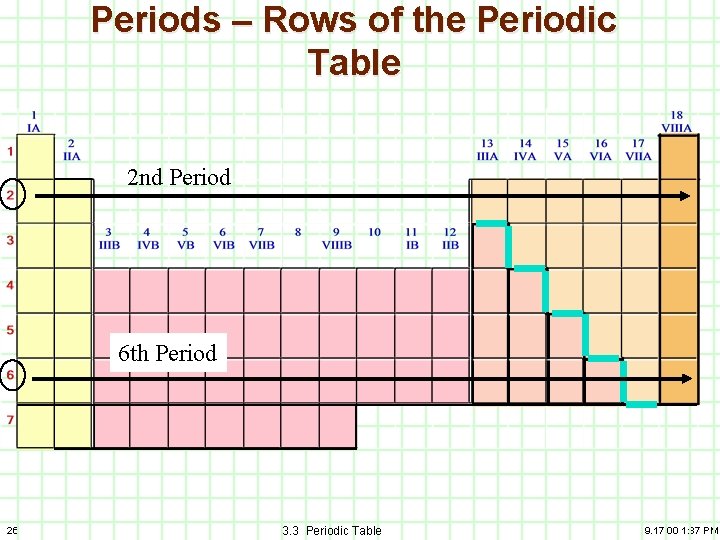
Periods – Rows of the Periodic Table 2 nd Period 6 th Period 26 3. 3 Periodic Table 9. 17. 00 1: 37 PM
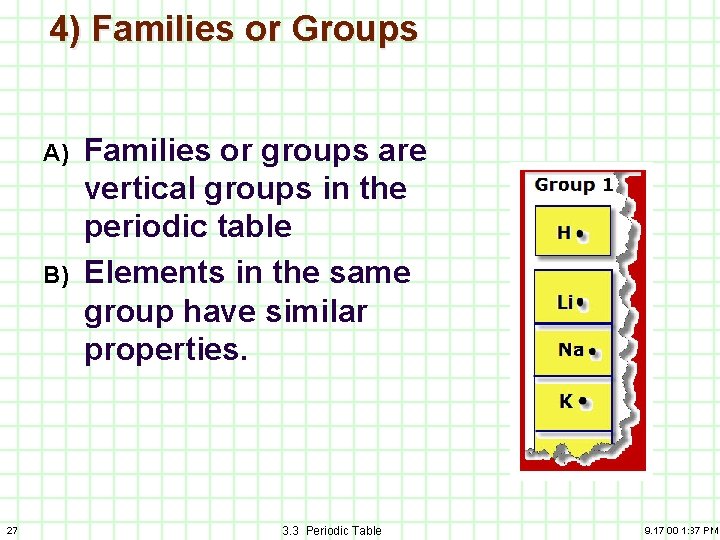
4) Families or Groups A) B) 27 Families or groups are vertical groups in the periodic table Elements in the same group have similar properties. 3. 3 Periodic Table 9. 17. 00 1: 37 PM
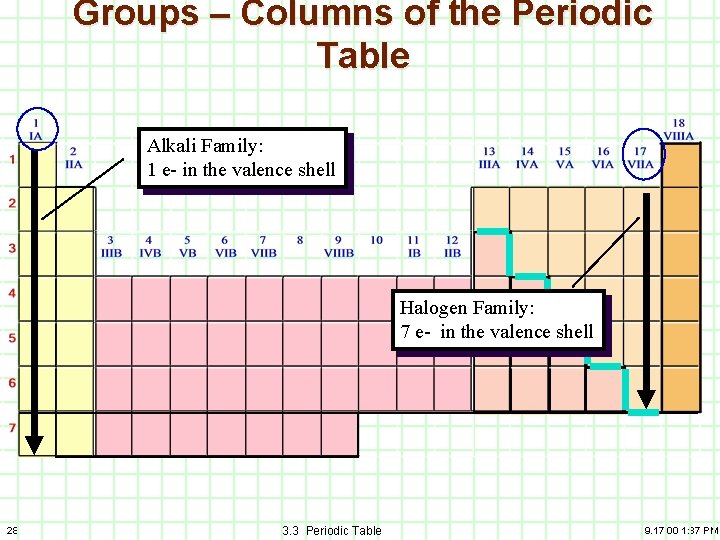
Groups – Columns of the Periodic Table Alkali Family: 1 e- in the valence shell Halogen Family: 7 e- in the valence shell 28 3. 3 Periodic Table 9. 17. 00 1: 37 PM

5) Valence Electrons A) B) C) 29 Families/Groups have the same number of electrons in their outermost energy level. These electrons are called valence electrons. The valence electrons will be particularly of interest when we talk about bonding. 3. 3 Periodic Table 9. 17. 00 1: 37 PM
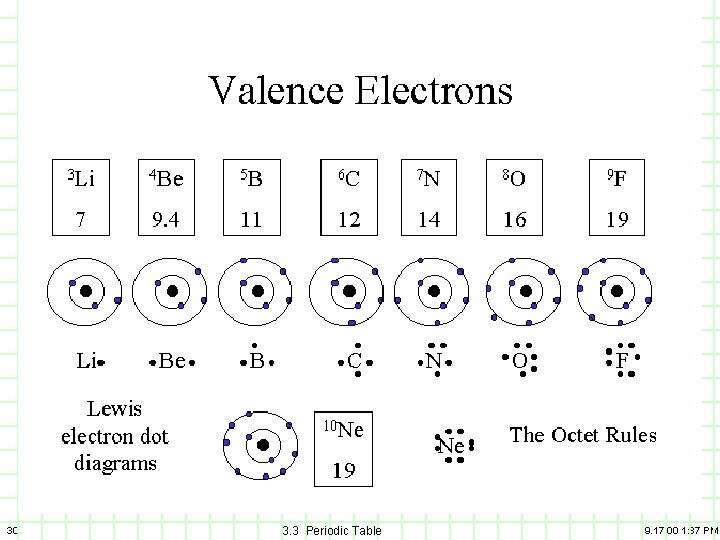
30 3. 3 Periodic Table 9. 17. 00 1: 37 PM
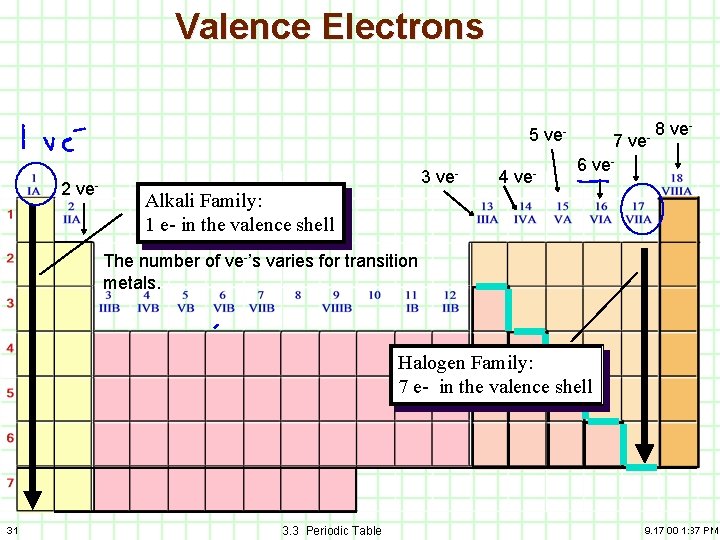
Valence Electrons 5 ve 2 ve- 3 ve- 4 ve- 7 ve- 8 ve- 6 ve- Alkali Family: 1 e- in the valence shell The number of ve-’s varies for transition metals. Halogen Family: 7 e- in the valence shell 31 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Summary Periodic Table: Table Map of the Building blocks of matter Types of Elements: Elements Metal, metalloid and Nonmetal Groupings: Groupings Representative or main, transition and Lanthanide/Actinides Family/Group: Family/Group Elements in the same column have similar chemical properties because of similar valence electrons Period: Elements in the same row have valence electrons in the same energy level. 32 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element is in period 2 and group 15? 33 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element is in period 4 and group 8? 34 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element has 11 protons? 35 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element has an atomic number of 17? 36 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element has 3 energy levels and 4 valence electrons? 37 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element has 5 energy levels and 2 valence electrons? 38 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element is in period 3 and the same group as helium? 39 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt What element as an atomic mass of 10. 81 amu? 40 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt Name an element that would have similar properties to lithium. 41 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt Name an element that would have similar properties to fluorine. 42 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt An atom has a mass number of 17. The atom has 9 neutrons. What element is it? 43 3. 3 Periodic Table 9. 17. 00 1: 37 PM

Periodic Table Scavenger Hunt An atom has a mass number of 24. The atom has 13 neutrons. What element is it? 44 3. 3 Periodic Table 9. 17. 00 1: 37 PM

HOMEWORK Worksheet: Who Am I? QUIZ ON INSERT DATE Atomic structure Protons, neutrons, electrons Atomic number, atomic mass, isotopes 45 3. 3 Periodic Table 9. 17. 00 1: 37 PM
- Slides: 45