Drill and Practice with Free Radical Substitution Mechanism

Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane Name for this step: Cl 2 + h 2 Cl* CHAIN INITIATION What common molecule can stop or slow the free radical substitution reaction ? O 2

Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) What is the second step of the methane chlorination mechanism ? Step 2) Cl* + CH 4 HCl + CH 3* (radical) What is the name of the step(s) that rationalize the high photoyield of the methane chlorination reaction ? CHAIN PROPAGATION

Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) Radical-radical recombination reactions have what effect on the radical chlorination reaction of methane ? TERMINATES THE CHAIN REACTION What step follows: Step 2) Cl* + CH 4 CH 3* + HCl STEP 3) CH 3* + Cl 2 CH 3 Cl + Cl*

Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) Which of the 5 facts is missing ? ? • reaction will only go at >250 o. C in dark or with input of ultraviolet (uv) light. • in xs Cl 2 , a single photon (h ) event causes thousands of halogenations. • O 2 causes halogenation reaction to slow or stop. • Reactivity rate follows: F 2 > Cl 2 > Br 2 >I 2 Wavelength of uv light that initiates reaction is the decomposition wavelength for the halogen
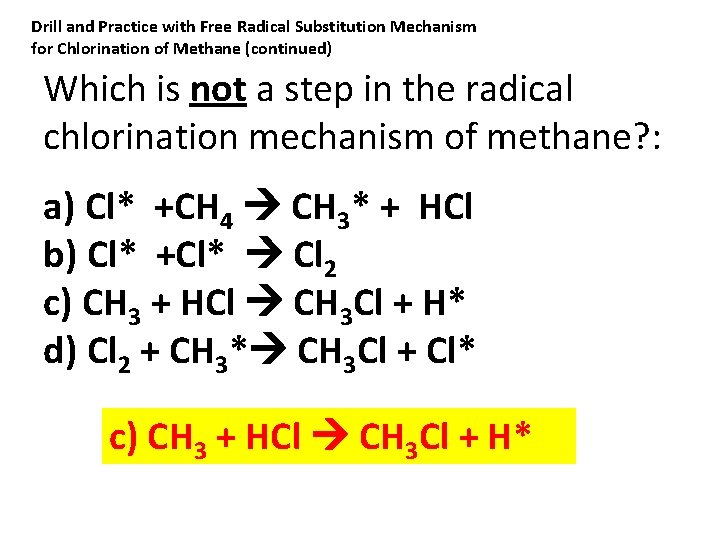
Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) Which is not a step in the radical chlorination mechanism of methane? : a) Cl* +CH 4 CH 3* + HCl b) Cl* +Cl* Cl 2 c) CH 3 + HCl CH 3 Cl + H* d) Cl 2 + CH 3* CH 3 Cl + Cl* c) CH 3 + HCl CH 3 Cl + H*

Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) Chain termination, chain initiation and chain ________ propagation What reaction explains the quenching effect of O 2 on the radical substitution of Cl on methane ? ? CH 3* + O 2 CH 3 OO * methylperoxy radical (~ stable)
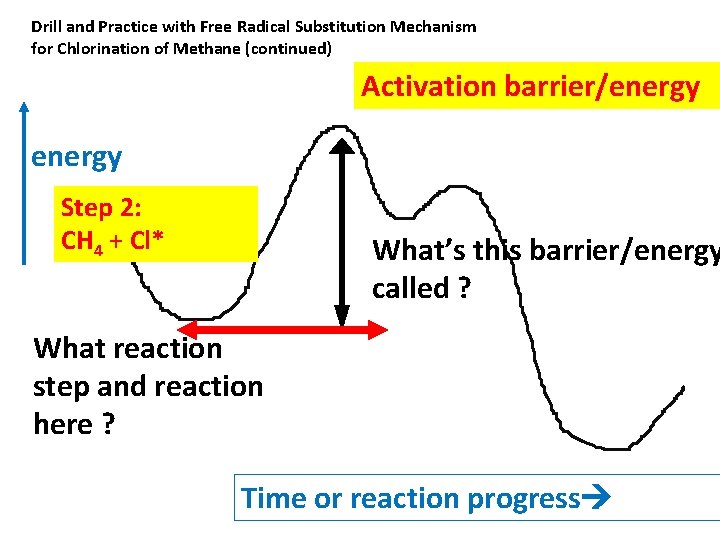
Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) Activation barrier/energy Step 2: CH 4 + Cl* What’s this barrier/energy called ? What reaction step and reaction here ? Time or reaction progress
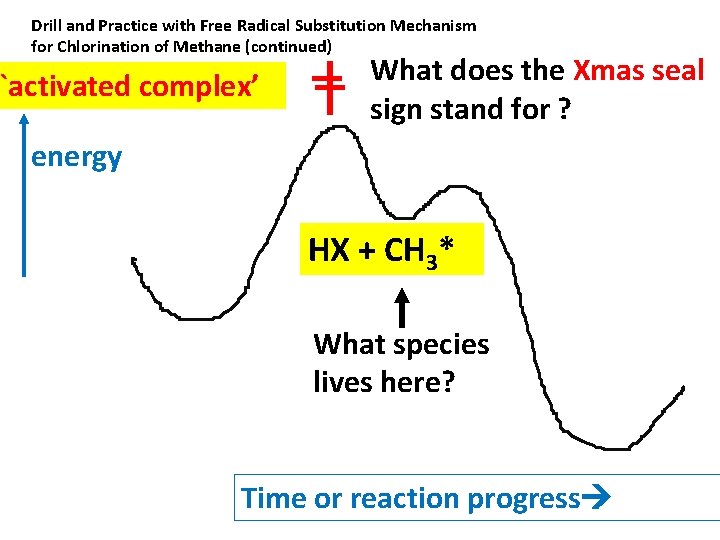
Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) `activated complex’ What does the Xmas seal sign stand for ? energy HX + CH 3* What species lives here? Time or reaction progress
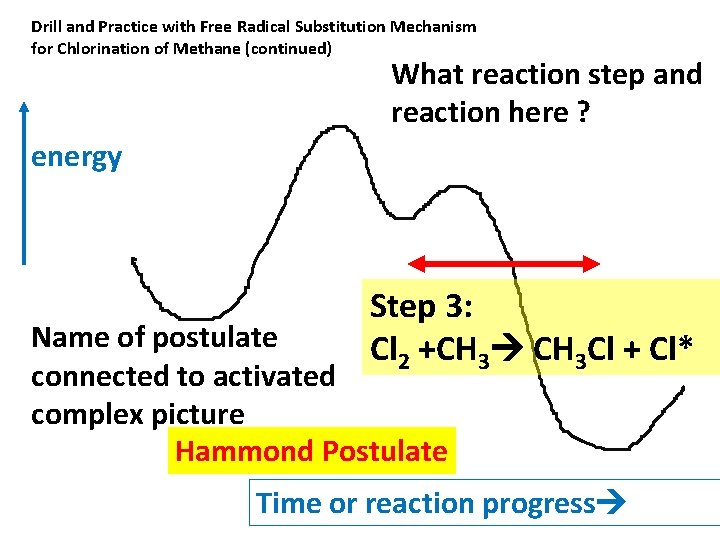
Drill and Practice with Free Radical Substitution Mechanism for Chlorination of Methane (continued) What reaction step and reaction here ? energy Step 3: Cl 2 +CH 3 Cl + Cl* Name of postulate connected to activated complex picture Hammond Postulate Time or reaction progress

ANY DAY DOING ORGANIC CHEMISTRY GOOD IS A ______DAY
- Slides: 10