Dried Blood Spots Acknowledgements Dr Rachanee Chiengasong Kyle

Dried Blood Spots

Acknowledgements Dr. Rachanee Chiengasong Kyle Bond Dr. Marie Downer Dr. Joanne Mei Debbie Kuehl Debbie Candal Dr. Mark Rayfield Dr. Bharat Parekh Dr. Harry Hannon Steve Soroka Dr. Rich Respess Trudy Dobbs

Dried Blood Spots (DBS) l AKA n n l Guthrie cards Filter paper disks Applications n n Antibody testing DNA/RNA Amplifications

Advantages of DBS Easy to collect, store, and transport l Stable l Adaptable to a variety of techniques l Quality protocols already developed l Centralized testing l Whole blood matrix l Safety l

Disadvantages of DBS Skin puncture required l Small sample volume l Dilution for analysis l Suitability for confirmatory method l Clinical sanction of data l

Analytes Measured in Dried Human Blood on Filter Paper ~ 1 Acarboxyprothrombin Acylcarnitine Adenine phosphoribosyl transferase Adenosine deaminase Albumin a-fetoprotein Amino Acids profiles arginine (Krebs cycle) histidine/urocanic acid homocysteine phenylalanine/tyrosine tryptophan Andrenostenedione Antipyrine Arabinitol enantiomers Arginase Benzoylecgonine (cocaine) Biotinidase Biopterin C-reactive protein Carnitine Carnosinase CD 4 Ceruloplasmin Chenodeoxycholic acid Chloroquine Cholesterol Cholinesterase Conjugated 1 -b hydroxycholic acid Cortisol Creatine kinase MM isoenzyme Cyclosporin A D-penicillamine De-ethylchloroquine Dehydroepiandrosterone sulfate DNA (PCR) acetylator polymorphism alcohol dehydrogenase a 1 -antitrypsin cystic fibrosis Duchenne/Becker muscular dystrophy glucose-6 -phosphate dehydrogenase hemoglobinopathies A, S, C, E D-Punjab beta-thalassemia hepatitis B virus HCMV HIV-1 HTLV-1

Analytes Measured in Dried Human Blood on Filter Paper ~2 Leber hereditory optic Glutathione perioxidase neuropathy MCAD m. RNA PKU Plasmodium vivax sexual differentiation 21 -deoxycortisol Desbutylhalofantrine Dihydropteridine reductase Diptheria/tetanus antitoxin Erythrocyte arginase Erythrocyte protoporphyrin Esterase D Fatty acids/acylglycines Free b-human chorionic gonadotropin Free erythrocyte prophyrin Free thyroxine (FT 4) Free tri-iodothyroine (FT 3) Fumarylacetoacetase Galactose/gal-1 -phosphate Galactose-1 -phosphate uridyl transferase Gentamicin Glucose-6 -phosphate dehydrogenase Glutathione Glycocholic acid Glycosylated hemoglobin Halofantrine Hemoglobin variants Hexosaminidase A Human erythrocyte carbonic anhydrase I 17 -a hydroxyprogesterone Hypoxanthine phosphoribosyl transferase Immunoreactive trypsin (CF) Lactate Lead Lipoproteins (a) B/A-1 b Lysozyme Mefloquine Netilmicin Phenobarbitone Phenytoin Phytanic/pristanic acid Progesterone Prolactin Prolidase

Analytes Measured in Dried Human Blood on Filter Paper ~ 3 Purine nucleoside phosphorylase Quinine Reverse tri-iodothyronine (r. T 3) Selenium Serum pancreatic lipase Sissomicin Somatomedin C Specific antibodies adenovirus anti-nuclear antibody anti-zeta antibody arbovirus Aujeszky’s disease virus dengue virus Dracunculus medinensis Echinococcus granulosus Entamoeba histolytica enterovirus Giardia duodenalisa Helicobacter pylori hepatitus B virus herpes virus HIV-1 Ig. E (atopic disease) influenza virus Leishmania donovani leptospira measles/mumps/rubella Mycobacterium leprae Mycoplasma pneumoniae Onchocerca volvulus parainfluenza virus Plasmodium falciparum poliovirus Pseudomonas aeruginosa respiratory syncytial virus rickettsia (scrub typhus) Schistosoma mansoni Toxoplasma gondii Trepenoma pallidium Trypanosoma cruzi/rangeli vesicular stomatis virus Wuchereria bancrofti yellow fever virus Spectic antigens hepatitis B virus HIV-1 Succinylacetone Sulfadoxine Theophylline Thyrotropin (TSH) Throxine (T 4) Thyroxine-binding globulin Trace elements Transferrin UDP-galactose-4 -epimerase Urea Uroporphyrinogen I synthase Vitamin A White blood cells Zinc protoporphyrin

Applications of DBS for HIV testing Surveillance l Quality control for HIV rapid testing l Quantitation of HIV viral load l Identification of HIV infected infants l

Dried Blood Spots (DBS) Characteristics Fingerstick or whole blood draw l Placed onto special collection papers l Dried properly l Stored appropriately l Inspected for quality l

Tenderfoot® Lancet for Heel Sticks l l l Safe for obtaining blood samples from heels of infants. A surgical blade incises to a standardized depth and length An incision created to allow blood to flow freely A higher quality blood sample is collected and bruising is diminished. Blade permanently retracts after use for safety Available in three incision depths for preemies, toddlers and full term
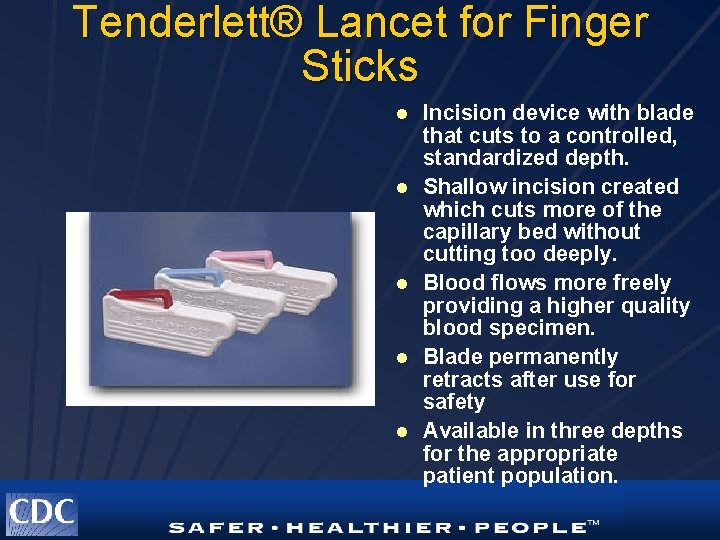
Tenderlett® Lancet for Finger Sticks l l l Incision device with blade that cuts to a controlled, standardized depth. Shallow incision created which cuts more of the capillary bed without cutting too deeply. Blood flows more freely providing a higher quality blood specimen. Blade permanently retracts after use for safety Available in three depths for the appropriate patient population.

Instructions for Specimen Collection: l l l l l Do not touch any of the filter paper circle before or after collection. Select puncture site and cleanse with 70% isopropanol. Use a sterile, disposable lancet with 2. 0 mm, or less, point Keep heel in down position at or below heart level. Wipe away first blood drop. Use second LARGE blood drop to apply to surface of filter paper circle. If not completely filled, add a second LARGE drop immediately. FILL all required circles completely. FILL from only one side of the filter paper. Dry specimen at room temperature 3 -4 hours in HORIZONTAL position.

Tips for Specimen Collection: l l l Complete each item on the collection form. Closely follow the collection instructions on the request form. Warm heel with a warm towel and hold heel at or below the heart. Fill one circle at a time. If capillaries are used to transfer blood from heel to paper: n Capillaries must be heparinized (Do not use EDTA). n Mix capillaries well before applying blood to filter paper. n Apply blood to filter paper immediately after filling.

Collection Problems

DBS -- HIV Antibody detection Quality of collection l Antibody elution l Optimization of enzyme immunoassay (EIA) -- washing, temperature, elution, mixing l Development of miniaturized Western blot for confirmation l

Assay Optimization l l l Optimal antibody elution Determination of effective specimen dilution Assessment of antibody detection n Strongly reactive samples n Weakly reactive samples, seroconversion n Non-reactive samples n Others – HIV-2, subtypes? Assay modifications Number of tests to perform
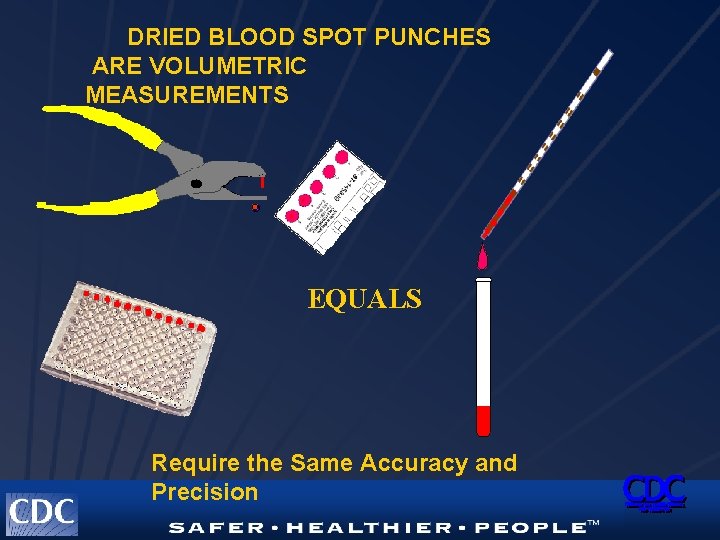
DRIED BLOOD SPOT PUNCHES ARE VOLUMETRIC MEASUREMENTS EQUALS Require the Same Accuracy and Precision
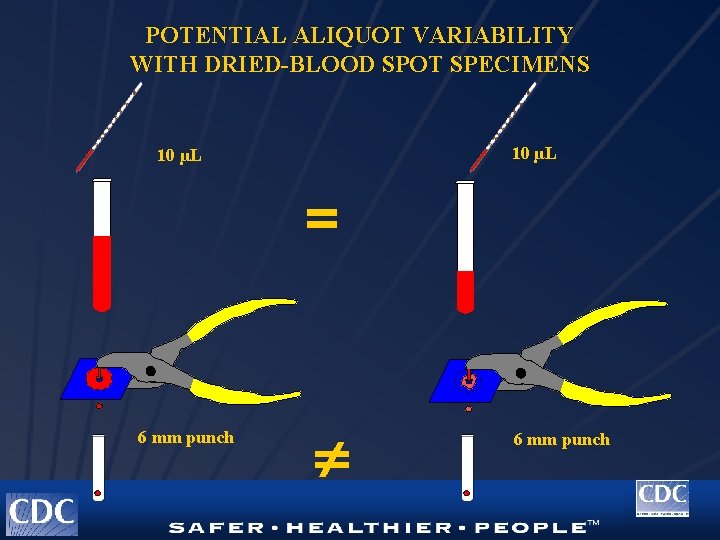
POTENTIAL ALIQUOT VARIABILITY WITH DRIED-BLOOD SPOT SPECIMENS 10 µL = 6 mm punch
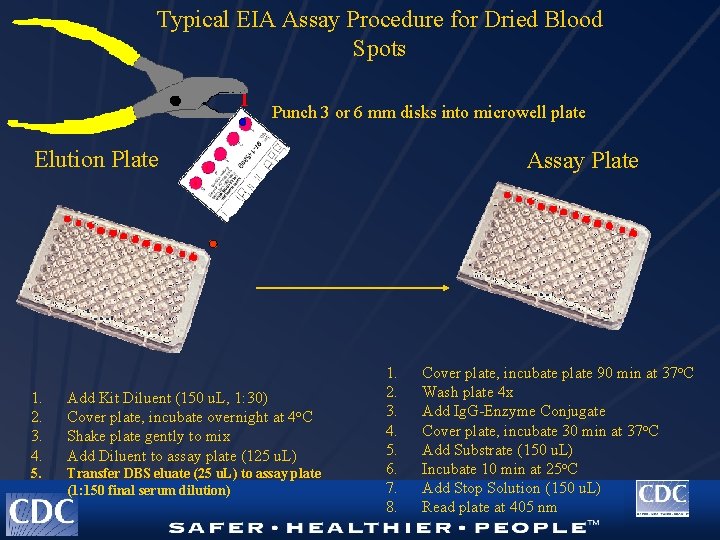
Typical EIA Assay Procedure for Dried Blood Spots Punch 3 or 6 mm disks into microwell plate Elution Plate 1. 2. 3. 4. Add Kit Diluent (150 u. L, 1: 30) Cover plate, incubate overnight at 4 o. C Shake plate gently to mix Add Diluent to assay plate (125 u. L) 5. Transfer DBS eluate (25 u. L) to assay plate (1: 150 final serum dilution) Assay Plate 1. 2. 3. 4. 5. 6. 7. 8. Cover plate, incubate plate 90 min at 37 o. C Wash plate 4 x Add Ig. G-Enzyme Conjugate Cover plate, incubate 30 min at 37 o. C Add Substrate (150 u. L) Incubate 10 min at 25 o. C Add Stop Solution (150 u. L) Read plate at 405 nm

Variable Affecting Measurements for Specimens Collected on Filter Paper Homogeneity within a production lot l Homogeneity among production lots l Variance among manufacturers l Variance within a collection card l Cutting and printing process l

Variable Affecting Measurements for Specimens Collected on Filter Paper Handling and storage of paper l Humidity condition of paper l Volume of blood collected l Hematocrit level of blood donor l Absorption time for blood l
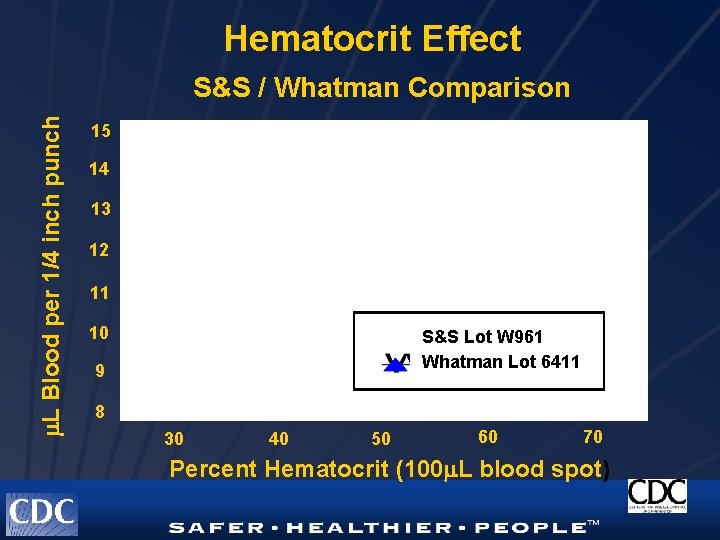
Hematocrit Effect L Blood per 1/4 inch punch S&S / Whatman Comparison 15 14 13 12 11 10 S&S Lot W 961 Whatman Lot 6411 9 8 30 40 50 60 70 Percent Hematocrit (100 L blood spot)

L Blood per 1/4 inch punch Spot Volume S&S / Whatman Comparison 14 13 12 S&S Lot W 961 Whatman Lot 6411 11 25 50 75 100 Blood Spot Voulme ( L) (55% hematocrit) 125

Schleicher and Schuell Grade 903 Filter Paper Lysed Red Blood Cells 1. 7 Serum Volume per 1/8” Punch ( L) 1. 6 99% 1. 5 95% 1. 4 _ X 1. 3 1. 2 95% 1. 1 99% 1. 0 0. 9 W 2 1 W 2 2 W 3 1 W 3 2 W 4 1 W 5 2 W 8 5 3 W 8 7 1 W 8 7 2 W 8 8 1 W 8 9 1 W 9 0 1 Lot Numbers In Chronological Order W 9 2 1 W 9 3 2 W 9 4 1 W 9 6 1 W 9 8 1

Optimization of DBS EIA N=108

DBS EIA Performance l l Sensitivity -- 100% Specificity -- 99. 8% False positive rate -- 0. 15% Repeat reactive specimens WB confirmed -- 47%

Gold Standard Positive Negative Positive True positives A False negatives B Negative False positives C True negatives D New Test l Establishment of performance characteristics l l l Sensitivity = A / A+C Specificity = D / B+D PVP = A / A+B NVP = D / C+D Efficiency = A+D / A+B+C+D Prevalance = A+C / A+B+C+D

DBS EIA problems Insufficient washing l Spot quality l Incubation temperature l Elution/air bubbles l Splashing l

DBS Western Blot Performance l l Miniaturization of methods for low sample volumes Reduction of background reactivity Assay optimization Presence of non-viral bands
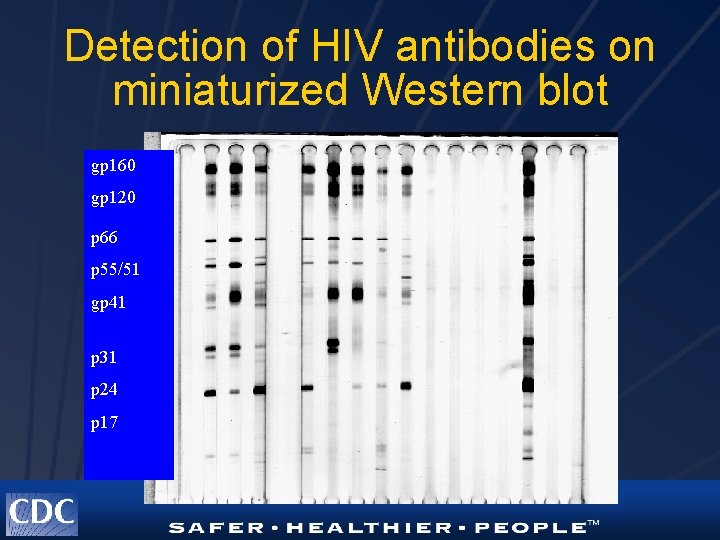
Detection of HIV antibodies on miniaturized Western blot gp 160 gp 120 p 66 p 55/51 gp 41 p 31 p 24 p 17

Quality Control for DBS l l l Evaluation of kit controls DBS controls performed in duplicate All DBS controls must be properly classified Low DBS controls monitor EIA performance and eluate stability High DBS controls may be used as Western blot controls High DBS controls should be included with frozen specimens to monitor long-term stability

Quality Control for DBS (Cont. ) Specimens should be separated from the filter paper if testing will not be completed immediately l Specimens should be stored in microvolume tubes with caps equipped with rubber gaskets. l Store short-term at 4 o. C and long-term and – 20 o. C l
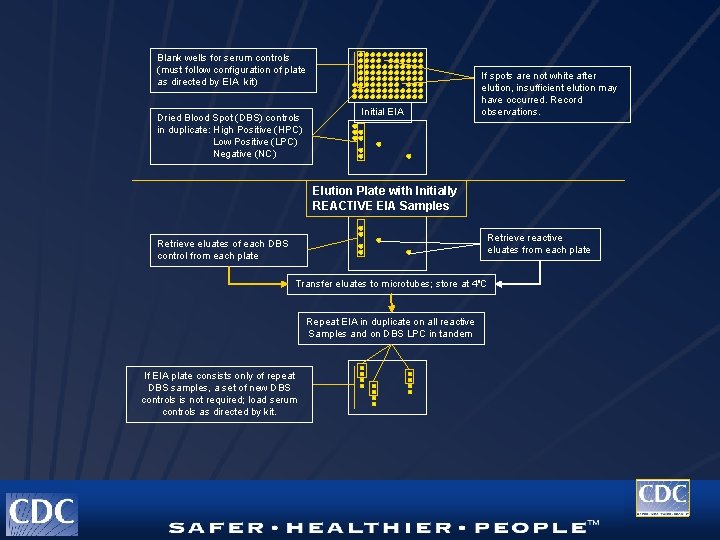
Blank wells for serum controls (must follow configuration of plate as directed by EIA kit) Dried Blood Spot (DBS) controls in duplicate: High Positive (HPC) Low Positive (LPC) Negative (NC) Initial EIA If spots are not white after elution, insufficient elution may have occurred. Record observations. Elution Plate with Initially REACTIVE EIA Samples Retrieve reactive eluates from each plate Retrieve eluates of each DBS control from each plate Transfer eluates to microtubes; store at 4 o. C Repeat EIA in duplicate on all reactive Samples and on DBS LPC in tandem If EIA plate consists only of repeat DBS samples, a set of new DBS controls is not required; load serum controls as directed by kit.
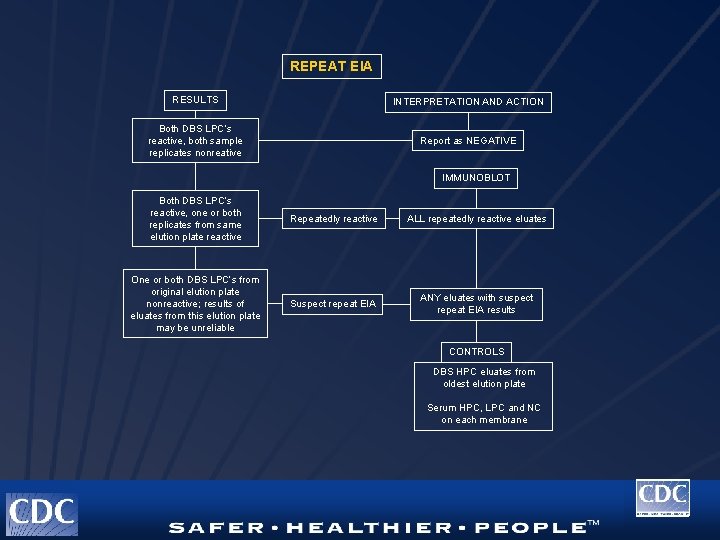
REPEAT EIA RESULTS INTERPRETATION AND ACTION Both DBS LPC’s reactive, both sample replicates nonreative Report as NEGATIVE IMMUNOBLOT Both DBS LPC’s reactive, one or both replicates from same elution plate reactive Repeatedly reactive ALL repeatedly reactive eluates One or both DBS LPC’s from original elution plate nonreactive; results of eluates from this elution plate may be unreliable Suspect repeat EIA ANY eluates with suspect repeat EIA results CONTROLS DBS HPC eluates from oldest elution plate Serum HPC, LPC and NC on each membrane






DBS Collection Forms

Detection of antibodies to other viruses in DBS eluates HTLV-I l Hepatitis A, B, and C l Measles, mumps and rubella l Dengue l

Detection of Viral Nucleic Acid on DBS l l l Extraction procedures n Boiling n Phenol/Chloroform n Glass beads/powder Amplification protocols n Single step DNA PCR n Nested DNA PCR n RT-PCR Precautions n Contamination n Removal of inhibitors u Chelex 100 u Proteinase K

Detection of Viral Nucleic Acid on DBS - Applications HIV-1 n Subtypes l CMV l Hepatitis C l HTLV-I l
- Slides: 45