DREAM Diabetes REduction Assessment with ramipril and rosiglitazone

DREAM Diabetes REduction Assessment with ramipril and rosiglitazone Medication

DREAM Prevention of Type 2 Diabetes • Clinical trials have shown that diet & exercise can prevent diabetes by > 50% in people with impaired glucose tolerance (IGT) • Clinical trials have also shown that drugs (e. g. metformin, acarbose) can prevent diabetes to a lesser extent in people with IGT • Growing evidence suggests that – ACE inhibitors may prevent diabetes – Thiazolidinediones may prevent diabetes

DREAM Properties of ACE-Inhibitors • Inhibition of the Renin-Angiotensin system with ACE inhibitors: – Lowers BP – Reduces mortality, MI & strokes in people with – Heart failure – Previous CV events without heart failure – Diabetes plus other CV risk factors • The HOPE trial suggested that the ACE-I ramipril may also reduce DM

DREAM RAS Blockade & New Diabetes (Diabetes - Not Primary Outcome) Study N (no DM) Active Control RRR ACE Inhibitors HOPE 5720 Ramipril 10 OD Placebo 34% PEACE 6174 Trandolapril Placebo 17% EUROPA 10716 Dagenais et al. Lancet 2006; 368: 581 Perindopril 8 mg Placebo 3% Overall Effect (HOPE, EUROPA, PEACE): 0. 86 (0. 78 -0. 95) D-SOLVD 291 Enalapril Placebo 74% Angiotensin Receptor Blockers SCOPE 4368 Candesartan 16/d Placebo 20% CHARM 5436 Candesartan 4 -32/d Placebo 24%

DREAM Do ACE-Inhibitors Prevent Diabetes? Limitations of Previous Reports • Glucose tolerance tests not done at baseline or end… may have missed prevalent diabetes at baseline & new DM on follow-up no ability to detect regression • Different definitions of new DM were used • Participants were of high cardiovascular risk & intermediate diabetes risk (e. g. DM rate ~ 2%/year) • DM prevention was not the primary outcome

DREAM Properties of Thiazolidinediones (TZDs) • Binds to PPAR gamma receptors – Increases insulin sensitivity – Reduces lipolysis – Increases preadipocytes (SC fat) • Possible beta cell protection • Reduces glucose levels if elevated
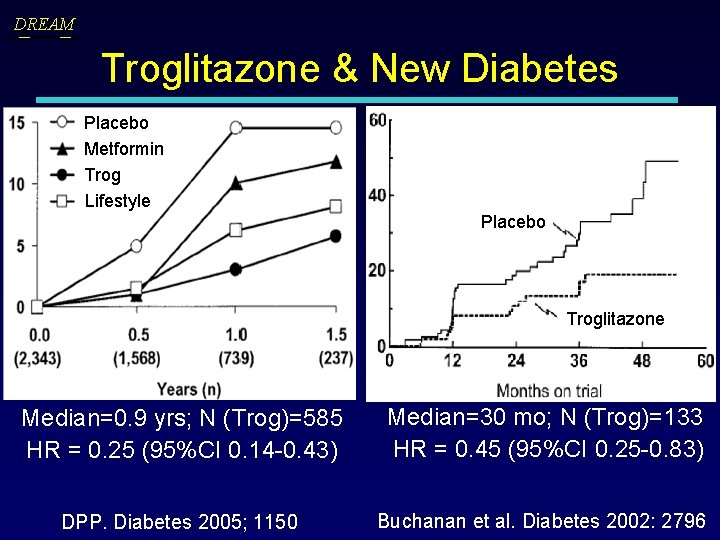
DREAM Troglitazone & New Diabetes Placebo Metformin Trog Lifestyle Placebo Troglitazone Median=0. 9 yrs; N (Trog)=585 HR = 0. 25 (95%CI 0. 14 -0. 43) DPP. Diabetes 2005; 1150 Median=30 mo; N (Trog)=133 HR = 0. 45 (95%CI 0. 25 -0. 83) Buchanan et al. Diabetes 2002: 2796

DREAM The DREAM Trial Aims: Does ramipril 15 mg/d prevent diabetes? Does rosiglitazone 8 mg/d prevent diabetes? Design: 2 X 2 factorial, double-blind RCT Sample: Age 30+; IGT (FPG <7 & 2 hr 7. 8 -11) &/or IFG (FPG 6. 1 -6. 9) Pts: 5269 in 191 sites, 21 countries, & F/U 3 yrs Outcome: Incident DM (confirmed FPG > 7 or 2 hr > 11. 1; or MD diagnosis) or death* *because undiagnosed diabetes may be more frequent in those who die than in those who do not

DREAM The DREAM Trial Independent Coordination, Data Management & Analysis Population Health Research Institute Mc. Master University & Hamilton Health Sciences Hamilton, Ontario, Canada Funding Canadian Institutes of Health Research Sanofi-Aventis King Pharmaceuticals Glaxo. Smith. Kline
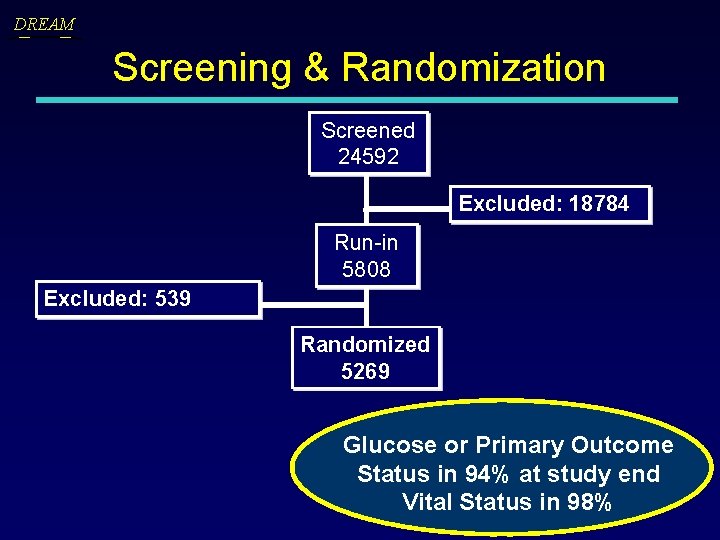
DREAM Screening & Randomization Screened 24592 Excluded: 18784 Run-in 5808 Excluded: 539 Randomized 5269 Glucose or Primary Outcome Status in 94% at study end Vital Status in 98%
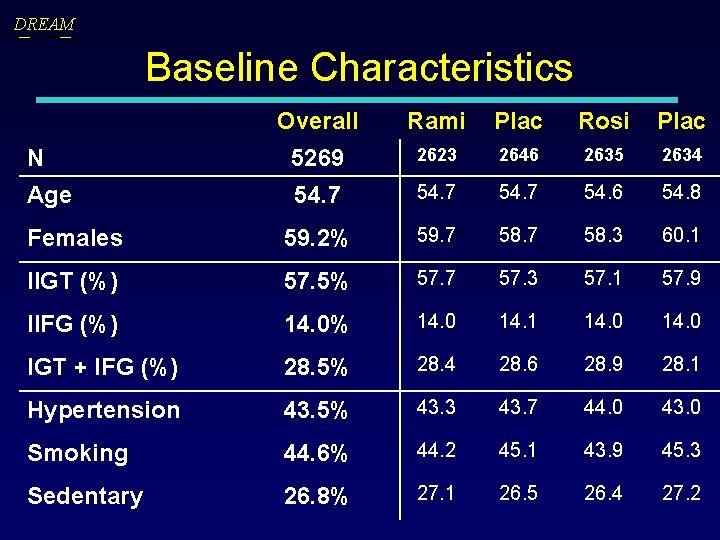
DREAM Baseline Characteristics Overall Rami Plac Rosi Plac N 5269 2623 2646 2635 2634 Age 54. 7 54. 6 54. 8 Females 59. 2% 59. 7 58. 3 60. 1 IIGT (%) 57. 5% 57. 7 57. 3 57. 1 57. 9 IIFG (%) 14. 0% 14. 0 14. 1 14. 0 IGT + IFG (%) 28. 5% 28. 4 28. 6 28. 9 28. 1 Hypertension 43. 5% 43. 3 43. 7 44. 0 43. 0 Smoking 44. 6% 44. 2 45. 1 43. 9 45. 3 Sedentary 26. 8% 27. 1 26. 5 26. 4 27. 2
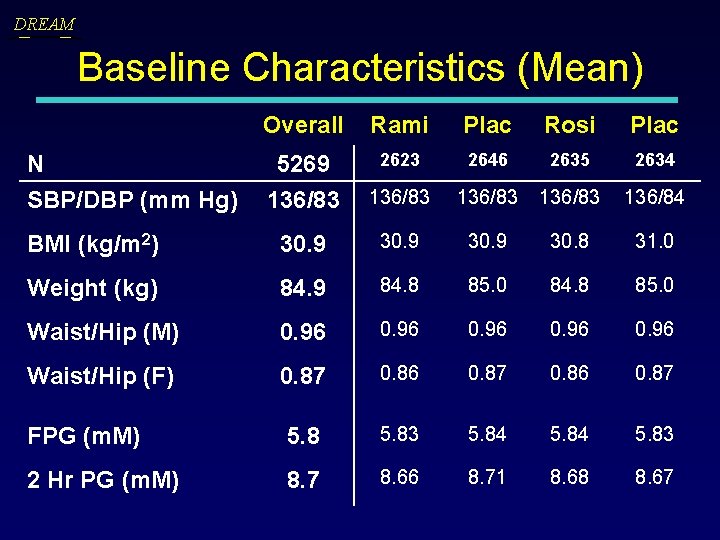
DREAM Baseline Characteristics (Mean) Overall Rami Plac Rosi Plac 5269 136/83 2623 2646 2635 2634 136/83 136/84 BMI (kg/m 2) 30. 9 30. 8 31. 0 Weight (kg) 84. 9 84. 8 85. 0 Waist/Hip (M) 0. 96 Waist/Hip (F) 0. 87 0. 86 0. 87 FPG (m. M) 5. 83 5. 84 5. 83 2 Hr PG (m. M) 8. 7 8. 66 8. 71 8. 68 8. 67 N SBP/DBP (mm Hg)

DREAM Results of the Ramipril Arm

DREAM Adherence/Adverse Effects Ramipril Placebo On Study Drug at 1 year 86. 6% 89. 9% at 2 years 81. 3% 84. 8% at 3 years 75. 4% 80. 9% Participant Refusal 17. 4% 17. 7% MD advice 2. 3% 2. 5% Cough 9. 7% 1. 8% Hypotension 0. 8 % 0. 4% Reasons for Stopping Study Drug
DREAM Ramipril’s Effect on Blood Pressure Systolic BP Placebo Ramipril Mean Final Ramipril Placebo P Systolic BP 128. 3 (17. 3) 132. 3 (17. 2) <0. 0001 Diastolic BP 78. 0 (10. 8) 80. 3 (10. 4) <0. 0001 Diastolic BP Base 2 6 12 24 36 48 Final Months

DREAM ALT (U/l) Ramipril’s Effect on ALT Placebo Ramipril P = 0. 04
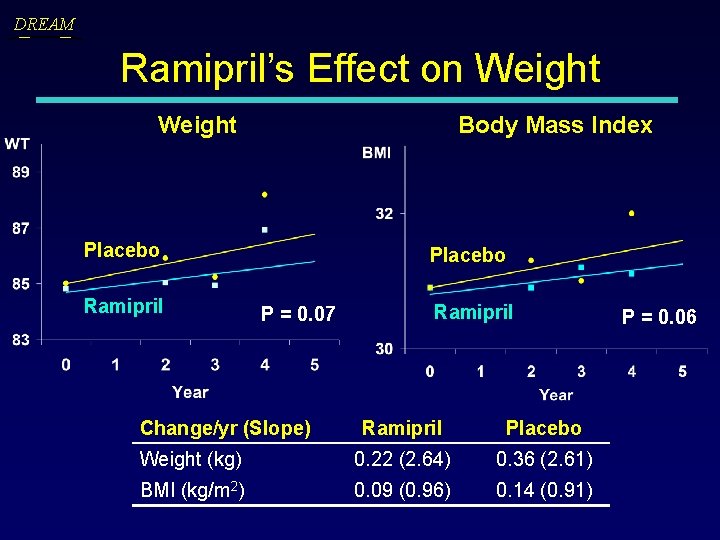
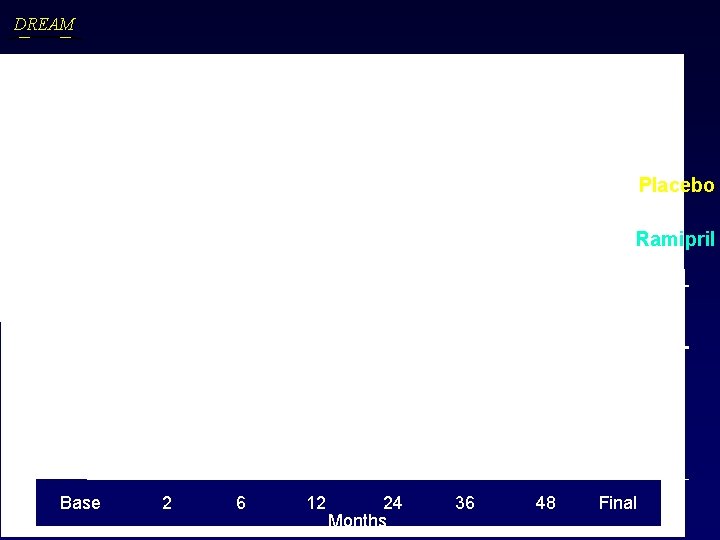
DREAM Ramipril’s Effect on Weight Body Mass Index Placebo Ramipril Placebo P = 0. 07 Change/yr (Slope) Ramipril Placebo Weight (kg) 0. 22 (2. 64) 0. 36 (2. 61) BMI (kg/m 2) 0. 09 (0. 96) 0. 14 (0. 91) P = 0. 06
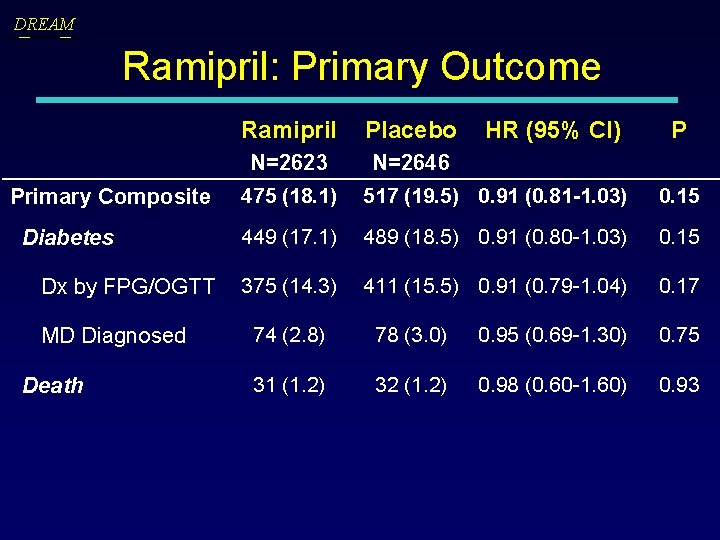
DREAM Ramipril: Primary Outcome Ramipril Placebo Primary Composite N=2623 475 (18. 1) N=2646 517 (19. 5) 0. 91 (0. 81 -1. 03) 0. 15 Diabetes 449 (17. 1) 489 (18. 5) 0. 91 (0. 80 -1. 03) 0. 15 Dx by FPG/OGTT 375 (14. 3) 411 (15. 5) 0. 91 (0. 79 -1. 04) 0. 17 MD Diagnosed Death HR (95% CI) P 74 (2. 8) 78 (3. 0) 0. 95 (0. 69 -1. 30) 0. 75 31 (1. 2) 32 (1. 2) 0. 98 (0. 60 -1. 60) 0. 93
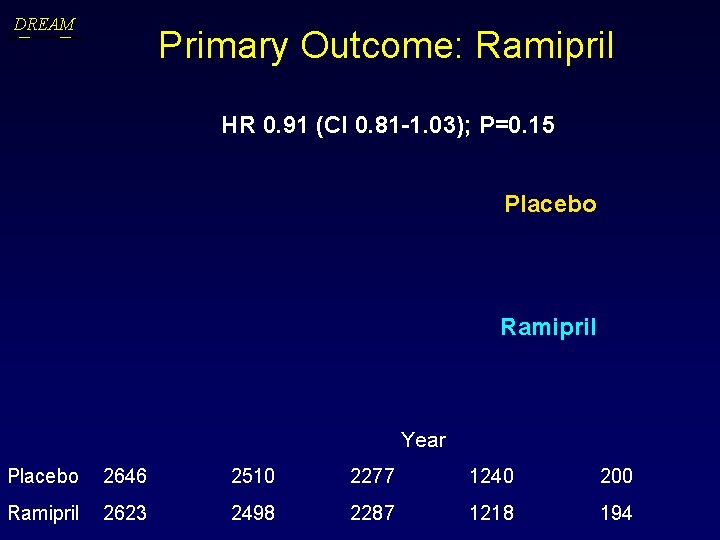
DREAM Primary Outcome: Ramipril HR 0. 91 (CI 0. 81 -1. 03); P=0. 15 Placebo Ramipril Year Placebo 2646 2510 2277 1240 200 Ramipril 2623 2498 2287 1218 194
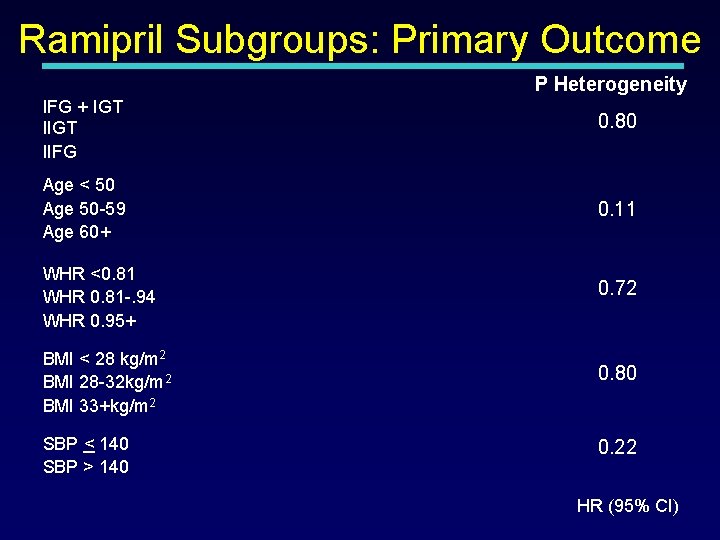
Ramipril Subgroups: Primary Outcome P Heterogeneity IFG + IGT IIFG 0. 80 Age < 50 Age 50 -59 Age 60+ 0. 11 WHR <0. 81 WHR 0. 81 -. 94 WHR 0. 95+ 0. 72 BMI < 28 kg/m 2 BMI 28 -32 kg/m 2 BMI 33+kg/m 2 SBP < 140 SBP > 140 0. 80 0. 22 HR (95% CI)

DREAM Cumulative Hazard Regression: Ramipril HR 1. 16 (1. 07 -1. 27); P=0. 001 Ramipril Placebo 0 1 2 3 4 Ramipril 2623 2487 2060 791 127 Placebo 2646 2494 2090 876 145

DREAM Effect on Glucose Category: Ramipril HR = 1. 16; P = 0. 001 HR = 1. 17; P = 0. 002 HR = 0. 91; P = 0. 15 (ADA Cutoff)

DREAM Ramipril & Median Glucose Fasting PG (m. M) 2 Hour PG (m. M) P = 0. 07 P = 0. 01 Placebo Ramipril Median Final Ramipril Placebo Fasting PG (mmol/L) 5. 70 5. 74 2 Hr PG (mmol/L) 7. 50 7. 80
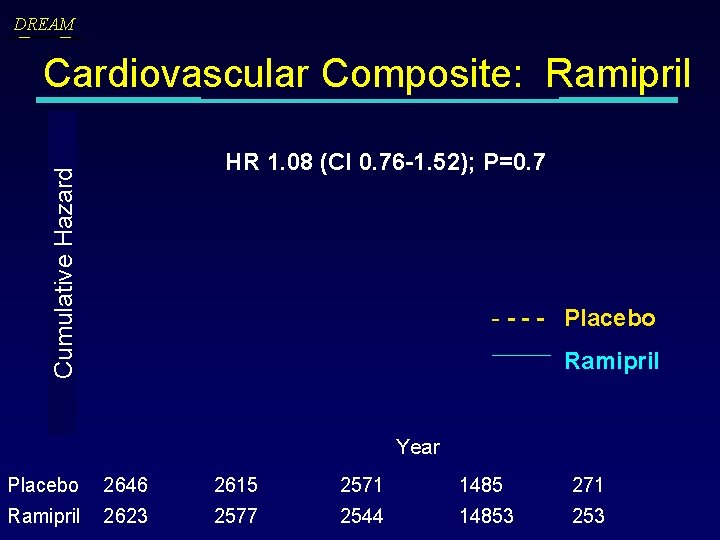
DREAM Cardiovascular Composite: Ramipril Cumulative Hazard HR 1. 08 (CI 0. 76 -1. 52); P=0. 7 - - Placebo Ramipril Year Placebo 2646 2615 2571 1485 271 Ramipril 2623 2577 2544 14853 253

DREAM Summary & Conclusions: Ramipril • Modestly improves glycemic status in IFG/IGT – A nonsignificant 9% DM reduction – Significant 16% increase in regression to normal glucose levels by at least 2 yrs – Reduced 2 hr glucose by 0. 3 m. M by study end • Significantly reduces BP in IGT / IFG • Small, favourable effect on liver function

DREAM vs. Previous Trials • Diabetes was the primary outcome in DREAM • People with undiagnosed diabetes were excluded • Regression was a predefined secondary outcome • People were low vs. high CV risk so: a) may have had a less activated RAS b) controls were less likely on drugs that raise glucose c) there was low power to detect differences in CVD events (short duration, low risk participants)

DREAM Summary & Conclusions: Ramipril • The DREAM results provide the best estimate of the effect of ACE-Is on diabetes prevention in people with IFG / IGT & no previous CV disease • Ramipril cannot currently be recommended for DM prevention • However, in people in whom there is an indication for ACE inhibitors (high BP, CHF, vascular disease, high risk DM) the favourable effects on glucose may be of added benefit

DREAM Results of the Rosiglitazone Arm
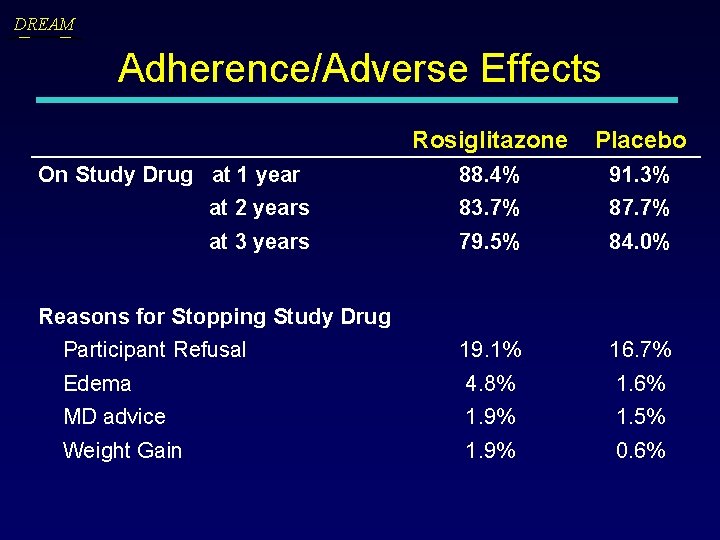
DREAM Adherence/Adverse Effects Rosiglitazone Placebo On Study Drug at 1 year 88. 4% 91. 3% at 2 years 83. 7% 87. 7% at 3 years 79. 5% 84. 0% Participant Refusal 19. 1% 16. 7% Edema 4. 8% 1. 6% MD advice 1. 9% 1. 5% Weight Gain 1. 9% 0. 6% Reasons for Stopping Study Drug

DREAM ALT (U/l) Rosiglitazone’s Effect on ALT Placebo P <0. 0001 Rosiglitazone Months
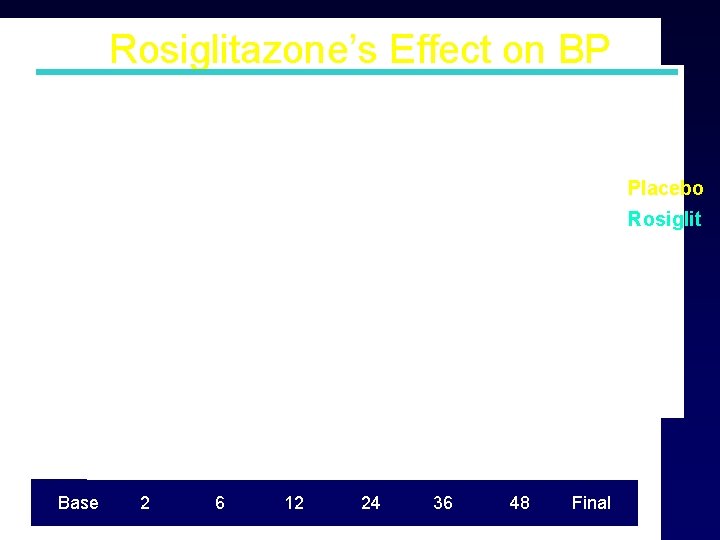
Rosiglitazone’s Effect on BP Systolic BP P=0. 0001 Placebo Rosiglit Mean Final Rosiglitazone Placebo P Systolic BP (mm) 129. 4 (17. 0) 131. 1 (17. 5) 0. 0001 Diastolic BP (mm) 78. 4 (10. 7) 79. 8 (10. 5) <0. 0001 P<0. 0001 Diastolic BP Base 2 6 12 24 36 48 Final

DREAM Rosiglitazone & Weight, BMI Weight (Kg) BMI (Kg/m 2) Rosiglitazone Placebo P < 0. 0001 Change/yr (Slope) P < 0. 0001 Rosiglitazone Placebo Weight (kg) 0. 67(2. 77) -0. 09 (2. 41) BMI (kg/m 2) 0. 25 (1. 01) -0. 01 (0. 84)
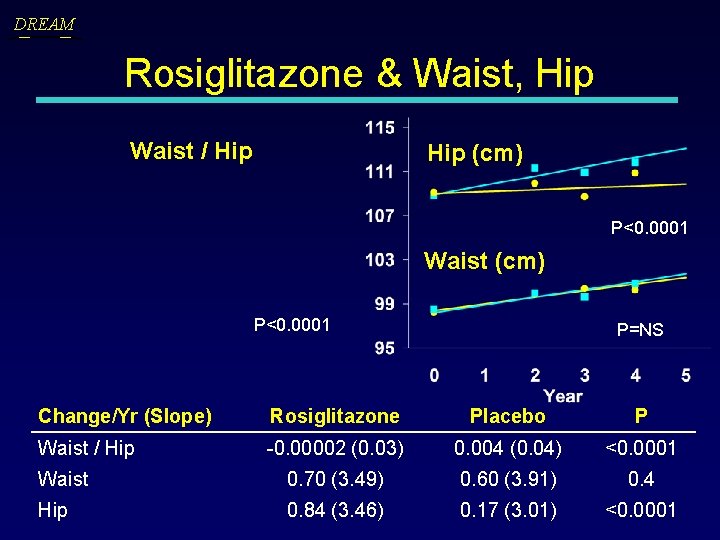
DREAM Rosiglitazone & Waist, Hip Waist / Hip (cm) P<0. 0001 Waist (cm) P<0. 0001 P=NS Change/Yr (Slope) Rosiglitazone Placebo P Waist / Hip -0. 00002 (0. 03) 0. 004 (0. 04) <0. 0001 Waist 0. 70 (3. 49) 0. 60 (3. 91) 0. 4 Hip 0. 84 (3. 46) 0. 17 (3. 01) <0. 0001
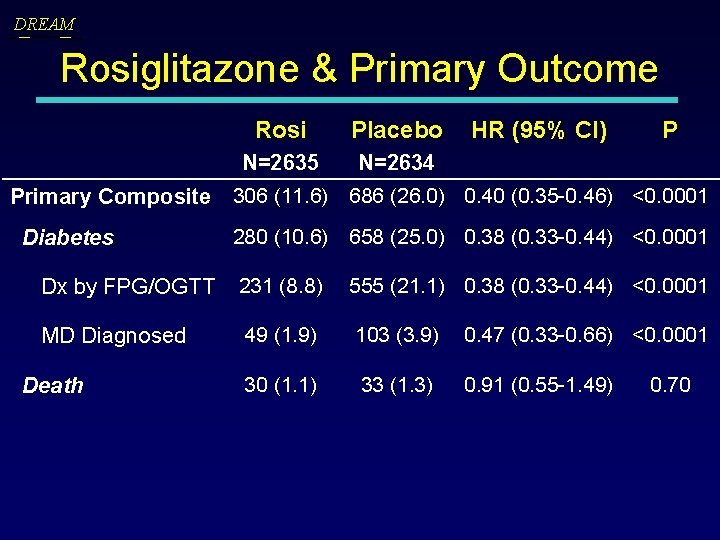
DREAM Rosiglitazone & Primary Outcome Rosi Placebo HR (95% CI) P N=2635 N=2634 Primary Composite 306 (11. 6) 686 (26. 0) 0. 40 (0. 35 -0. 46) <0. 0001 Diabetes 280 (10. 6) 658 (25. 0) 0. 38 (0. 33 -0. 44) <0. 0001 Dx by FPG/OGTT 231 (8. 8) 555 (21. 1) 0. 38 (0. 33 -0. 44) <0. 0001 MD Diagnosed 49 (1. 9) 103 (3. 9) 0. 47 (0. 33 -0. 66) <0. 0001 30 (1. 1) 33 (1. 3) 0. 91 (0. 55 -1. 49) Death 0. 70
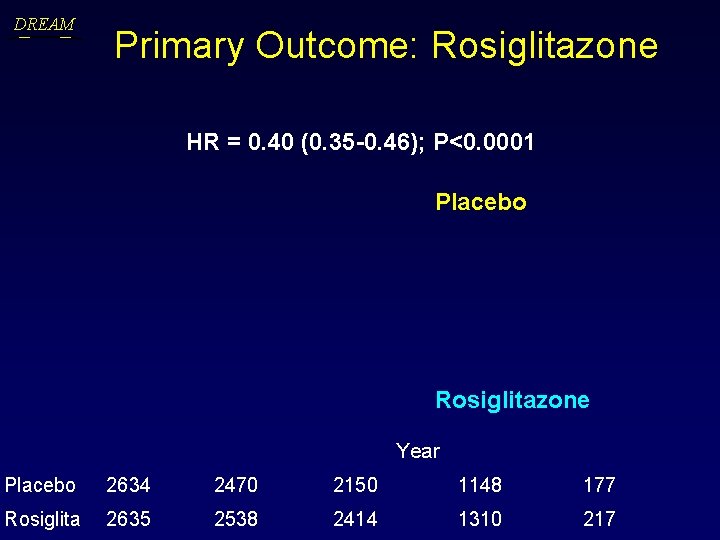
DREAM Primary Outcome: Rosiglitazone HR = 0. 40 (0. 35 -0. 46); P<0. 0001 Placebo Rosiglitazone Year Placebo 2634 2470 2150 1148 177 Rosiglita 2635 2538 2414 1310 217

DREAM Effect on Glucose Category: Rosiglitazone HR 1. 71; P < 0. 0001 HR 1. 83; P < 0. 0001 HR 0. 38; P < 0. 0001
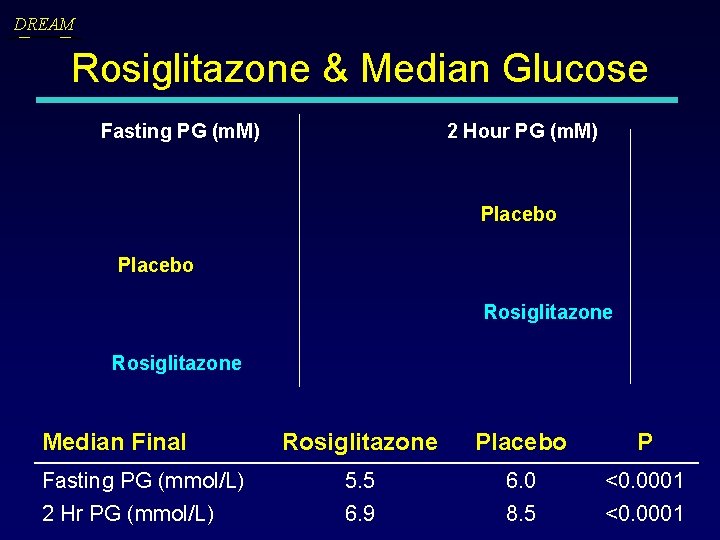
DREAM Rosiglitazone & Median Glucose Fasting PG (m. M) 2 Hour PG (m. M) Placebo Rosiglitazone Median Final Rosiglitazone Placebo P Fasting PG (mmol/L) 5. 5 6. 0 <0. 0001 2 Hr PG (mmol/L) 6. 9 8. 5 <0. 0001
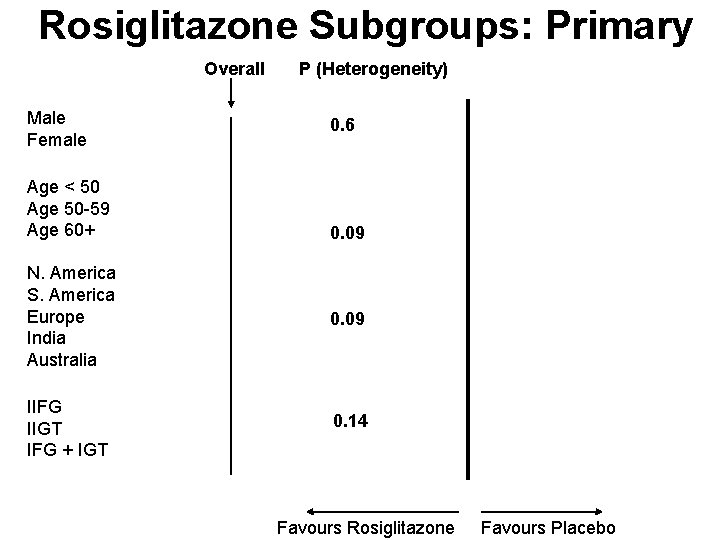
Rosiglitazone Subgroups: Primary Overall Male Female Age < 50 Age 50 -59 Age 60+ P (Heterogeneity) 0. 6 0. 09 N. America S. America Europe India Australia 0. 09 IIFG IIGT IFG + IGT 0. 14 Favours Rosiglitazone Favours Placebo
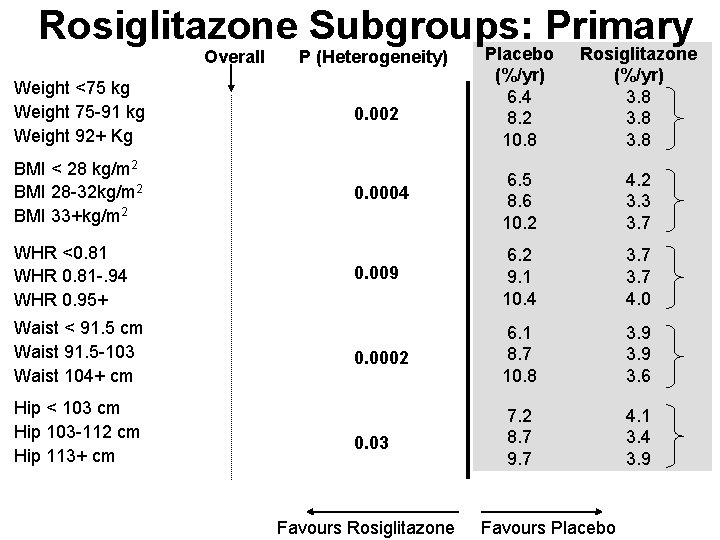
Rosiglitazone Subgroups: Primary Overall P (Heterogeneity) Placebo (%/yr) 6. 4 8. 2 10. 8 Rosiglitazone (%/yr) 3. 8 Weight <75 kg Weight 75 -91 kg Weight 92+ Kg 0. 002 BMI < 28 kg/m 2 BMI 28 -32 kg/m 2 BMI 33+kg/m 2 0. 0004 6. 5 8. 6 10. 2 4. 2 3. 3 3. 7 0. 009 6. 2 9. 1 10. 4 3. 7 4. 0 0. 0002 6. 1 8. 7 10. 8 3. 9 3. 6 0. 03 7. 2 8. 7 9. 7 4. 1 3. 4 3. 9 WHR <0. 81 WHR 0. 81 -. 94 WHR 0. 95+ Waist < 91. 5 cm Waist 91. 5 -103 Waist 104+ cm Hip < 103 cm Hip 103 -112 cm Hip 113+ cm Favours Rosiglitazone Favours Placebo

DREAM Cardiovascular Outcomes: Rosiglitazone Composite HR 1. 37 (0. 97 -1. 94): P=0. 08 MI Stroke CV Death 14 (0. 5%) vs. 2 (0. 1%); P=0. 01 CHF New Angina Revascularized LOG HR (95% CI)

DREAM Summary & Conclusions: Rosiglitazone • A dose of 8 mg/day reduces new DM by > 60% in people with IGT or IFG • Promotes regression to normal FPG & 2 hr PG by >70% • Effective in all regions of the world • Eliminates the gradient of DM risk with increasing weight • ~ 3% increase in body weight, but a favourable effect on waist/hip ratio • Reduces ALT

DREAM Summary & Conclusions: Rosiglitazone • Modestly lowers systolic BP & diastolic BP • Increases the risk of CHF • Too few events to draw any conclusions re the effect on other CV events or death For every 1000 people treated with rosiglitazone for ~ 3 years, 144 cases of DM will be prevented with an excess of ~ 4 cases of CHF

DREAM Conclusions of the DREAM Trial • Rosiglitazone has a substantial benefit on prevention of diabetes & regression to normoglycaemia • Ramipril has a modest benefit on regression to normoglycaemia • The durability of the glycaemic effect of these drugs is being assessed in a washout phase DREAM Slides: 2 DREAM Papers: www. phri. ca/dream NEJM & Lancet - online

DREAM TEAM International Leaders H. C. Gerstein S. Yusuf R. Holman J. Bosch S. Anand A. Avezum A. Budaj J. Chiasson I. Conget G. Dagenais M. Davis R. Diaz N. Dinccag M. Enjalbert A. Escalante G. Fodor M. Hanefeld T. Hedner B. Hoogwerf K. Jolly M. Keltai M. Laakso F. Lanas E. Lonn M. Mc. Queen V. Mohan A. Phillips L. Piegas V. Pirags J. Probstfield I. Schmid J. Shaw K. Teo P. Zimmet B. Zinman Statisticians: P. Sheridan, J. Pogue TMC: D. Sackett; D. Altman; C. Clark; P. Bennett; R. Hamman; L. Ryden
- Slides: 44