Drawing Lewis Structures The Covalent Bond n n

Drawing Lewis Structures
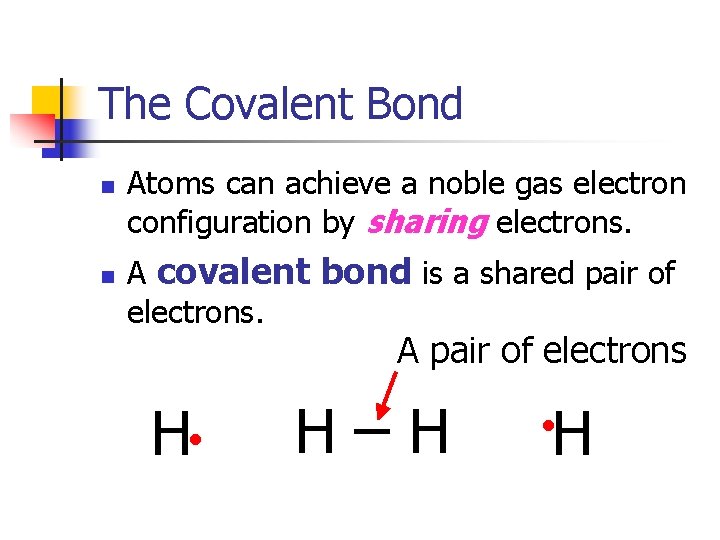
The Covalent Bond n n Atoms can achieve a noble gas electron configuration by sharing electrons. A covalent bond is a shared pair of electrons. A pair of electrons H • H–H H •
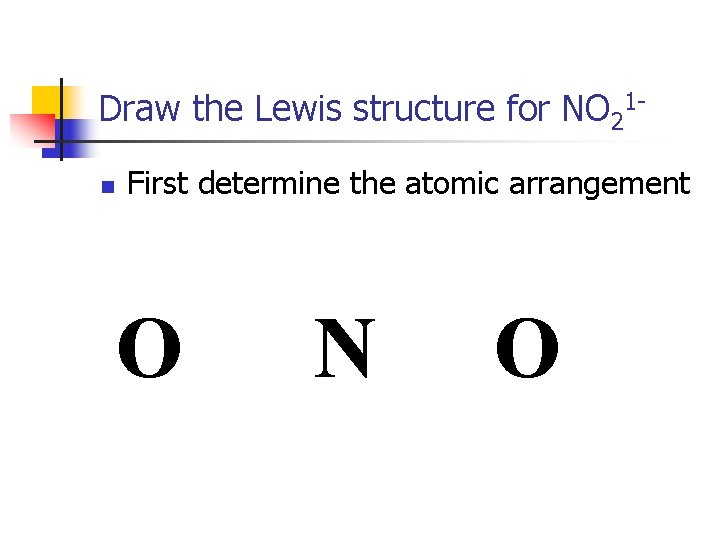
Draw the Lewis structure for NO 21 n First determine the atomic arrangement O N O

Next, determine the total number of valence electrons. 5 electrons from the N 6 electrons from each O for 12 more electrons 1 additional electron because it is a monoanion 18 total valence electrons

Use the electrons to make electron-pair bonds between the central and outer atoms. O N O 18 - 4 = 14 electrons remain
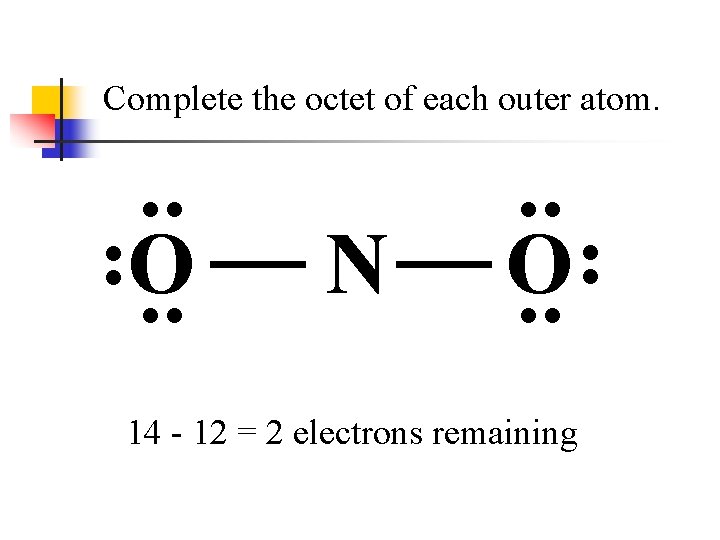
Complete the octet of each outer atom. . . N . . O. . 14 - 12 = 2 electrons remaining

If there any remaining electrons, put them on the central atom. . . N . . O. . All of the valence electrons have now been used.
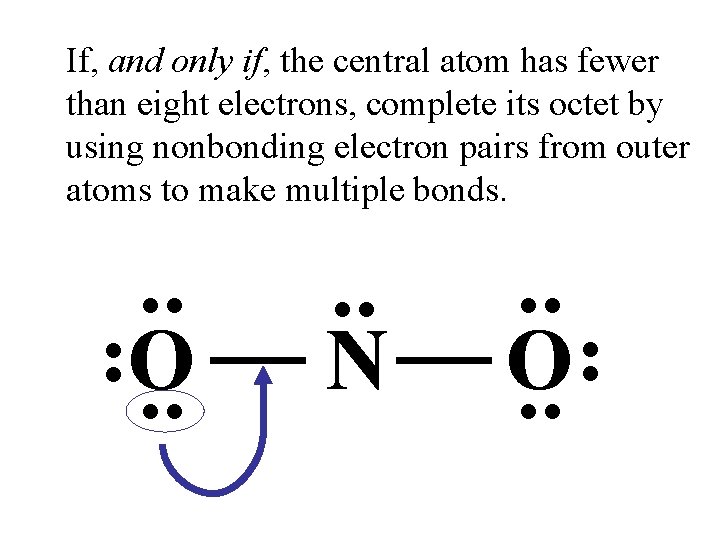
If, and only if, the central atom has fewer than eight electrons, complete its octet by using nonbonding electron pairs from outer atoms to make multiple bonds. . . N . . O. .
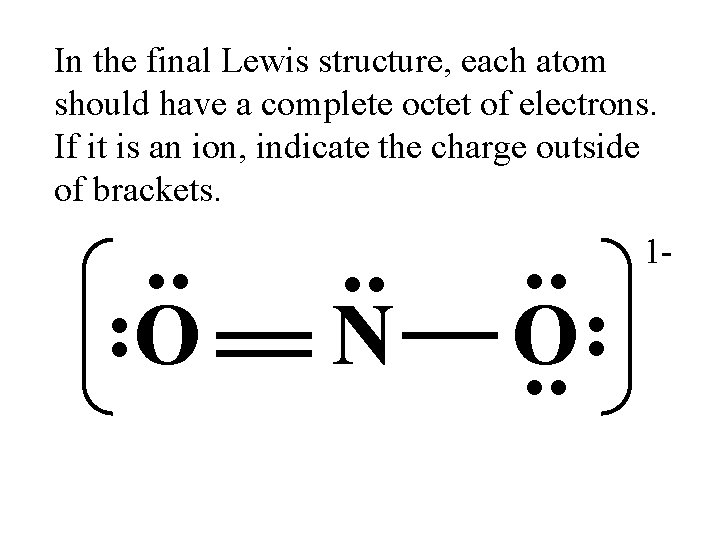
In the final Lewis structure, each atom should have a complete octet of electrons. If it is an ion, indicate the charge outside of brackets. . . N . . O. . O . . 1 -

Examples n n n PH 3 SO 3 Cl. O 31 O 3 PO 43 -
- Slides: 10