Draw the correct Lewis dot structure for Na




![Write the noble gas configuration for the element rubidium 1 • [Kr]5 s Write the noble gas configuration for the element rubidium 1 • [Kr]5 s](https://slidetodoc.com/presentation_image_h2/52db302c6f4c9d93bd43765029aa64b3/image-7.jpg)




![Write the noble gas configuration for the element chlorine 2 5 • [Ne]3 s Write the noble gas configuration for the element chlorine 2 5 • [Ne]3 s](https://slidetodoc.com/presentation_image_h2/52db302c6f4c9d93bd43765029aa64b3/image-13.jpg)










- Slides: 26

Draw the correct Lewis dot structure for Na. Cl

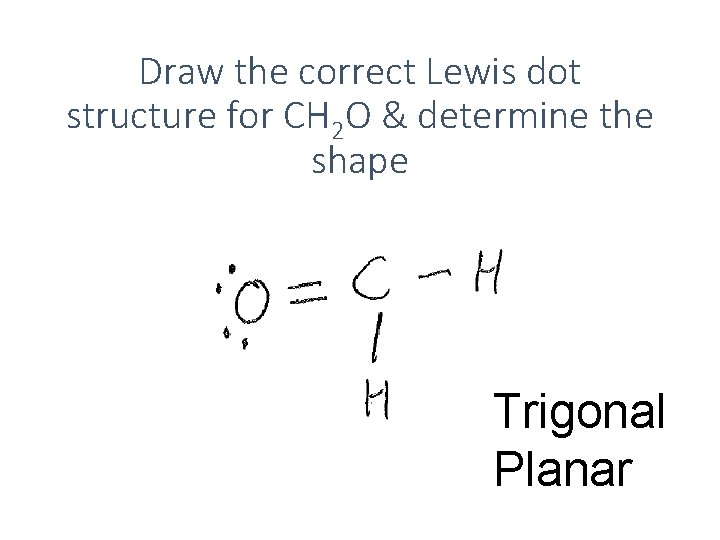
Draw the correct Lewis dot structure for CH 2 O & determine the shape Trigonal Planar

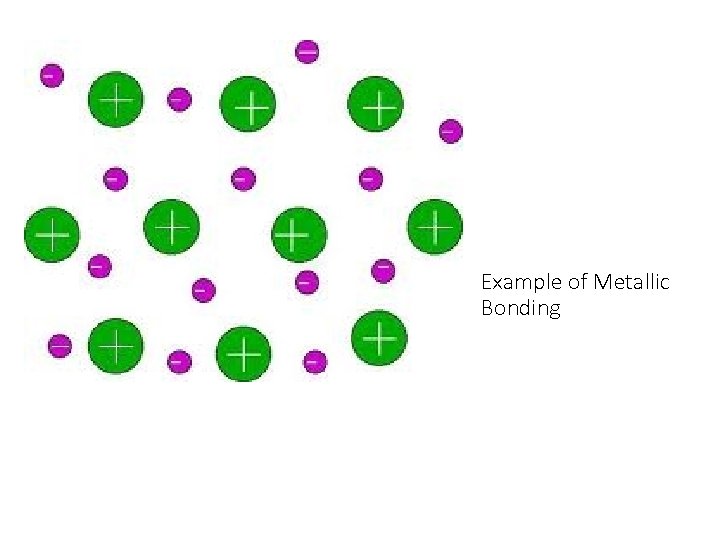
Describe the structure of metallic bonding. • Positive metallic ions surrounded by electrons.

Example of Metallic Bonding

Potassium bromide forms a crystal lattice structure. What type of bonding would you expect it have? • Ionic

Write the electron configuration for the element potassium 2 2 6 1 • 1 s 2 s 2 p 3 s 3 p 4 s
![Write the noble gas configuration for the element rubidium 1 Kr5 s Write the noble gas configuration for the element rubidium 1 • [Kr]5 s](https://slidetodoc.com/presentation_image_h2/52db302c6f4c9d93bd43765029aa64b3/image-7.jpg)
Write the noble gas configuration for the element rubidium 1 • [Kr]5 s

Write the orbital notation for the element carbon ↑↓ ↑↓ 1 s 2 s ↑ ↑ 2 p .

What type of bond exists between chlorine (EN = 3. 0) and bromine (EN = 2. 8)? Give a reason using electronegativity. • Covalent; difference in electronegativity is small (less than 1. 67)

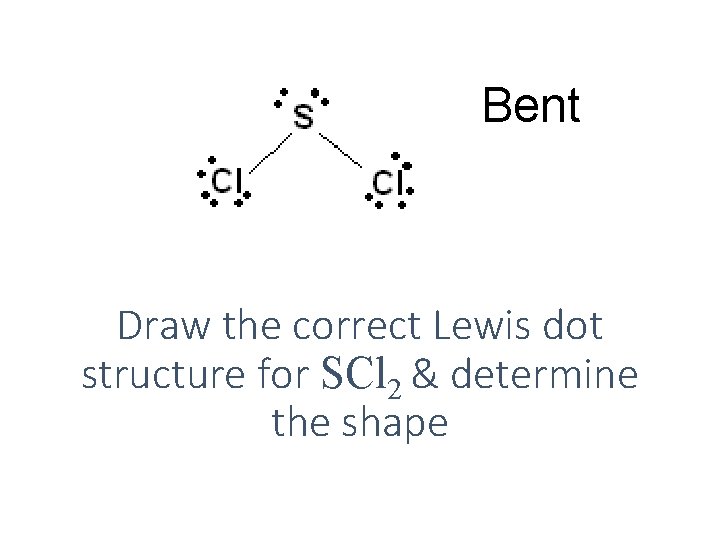
Bent Draw the correct Lewis dot structure for SCl 2 & determine the shape

Which of these elements would make a covalent bond with nitrogen? Al Mg Ba • C C

Which element has the following electron configuration: 1 s 22 p 63 s 23 p 64 s 23 d 6 • Iron, Fe
![Write the noble gas configuration for the element chlorine 2 5 Ne3 s Write the noble gas configuration for the element chlorine 2 5 • [Ne]3 s](https://slidetodoc.com/presentation_image_h2/52db302c6f4c9d93bd43765029aa64b3/image-13.jpg)
Write the noble gas configuration for the element chlorine 2 5 • [Ne]3 s 3 p

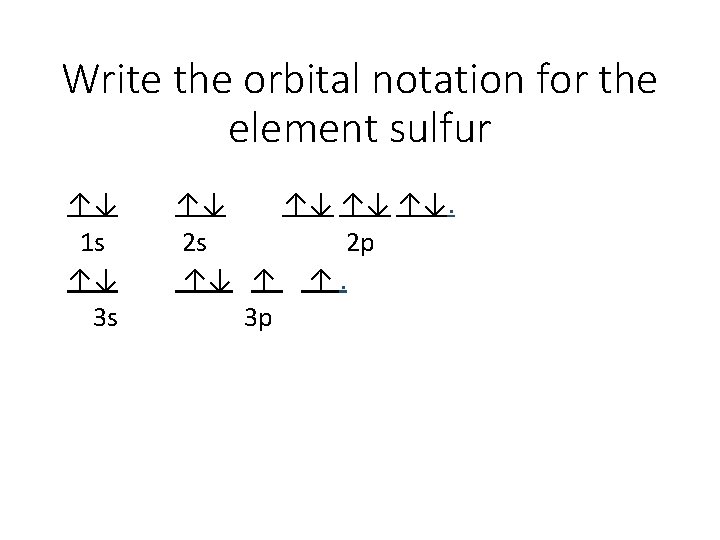
Write the orbital notation for the element sulfur ↑↓ 1 s ↑↓ 3 s ↑↓ ↑↓. 2 s 2 p ↑↓ ↑ ↑. 3 p

Which of these elements would make an ionic bond with potassium? Mg N Sr • N & Cl Cl

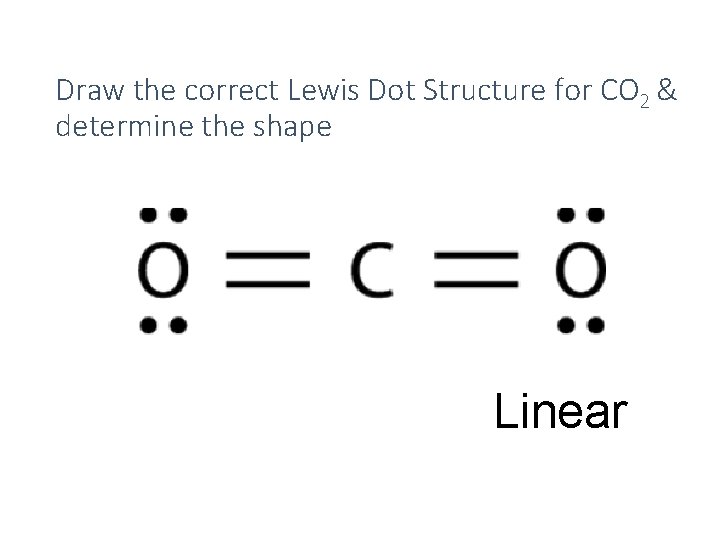
Draw the correct Lewis Dot Structure for CO 2 & determine the shape Linear

Which of these elements would make an ionic bond with chlorine? Ni F Li • Ni and Li Xe

Write the electron configuration for the element fluorine • 1 s 22 p 5

Which element has the following electron configuration: 1 s 22 p 63 s 23 p 64 s 23 d 104 p 3 • Arsenic, As

What type of bond exists between nitrogen (EN = 3. 0) and calcium(EN = 1. 00)? Give a reason using electronegativity. • ionic; difference in electronegativity is big (more than 1. 67)

Write the electron configuration for the element silicon • 1 s 22 p 63 s 23 p 2

Write the electron configuration for the element titanium 2 2 6 2 2 • 1 s 2 s 2 p 3 s 3 p 4 s 3 d

Write the Lewis Dot structure for the following atoms

Aluminum

Phosphorus

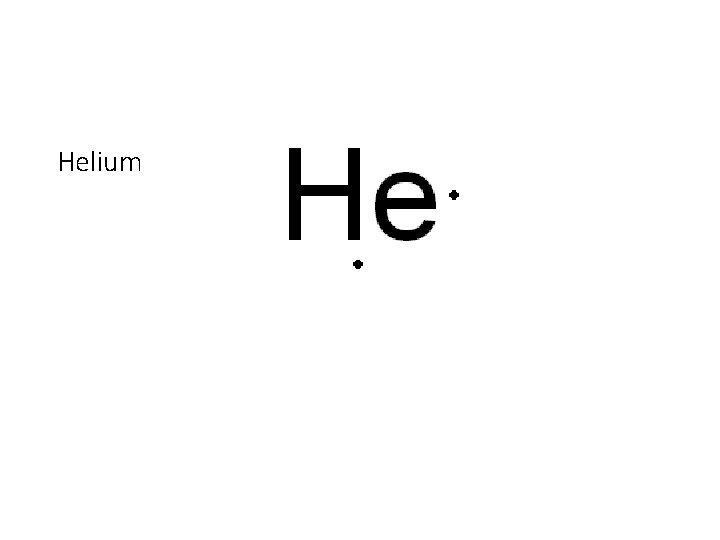
Helium