Draw a Bohr Model of Chlorine Cl Atomic

Draw a Bohr Model of Chlorine (Cl) • Atomic Number: 17 (# of protons & therefore, same # of electrons) • Atomic Mass: 35. 453 (Atomic mass – Atomic number = # of neutrons) • Halogen: Group 17 (# of valence electrons) • Period 3 (# of energy levels) • Identify the ionic charge

Nonmetals will gain electrons when forming compounds. They become negatively charged because there will be more electrons than protons. A negative ion will be formed. Cl ¯ 17 P 18 N 2 8 7 17 P = +17 18 E= -18 ionic charge: 1 E= -1

Draw a Dot Diagram of Chlorine Need to know for a Dot Diagram: • Element Symbol: Cl • Group Number: 17 (# of valence electrons) • Start drawing valence electrons at the top and go counterclockwise all the way around like compass ( N W S E N)

Available spot for bonding!! Cl ¯ Cl

Draw a Bohr Model of Sodium (Na) • Atomic Number: 11 (# of protons & therefore, same # of electrons) • Atomic Mass: 22. 99 (Atomic mass – Atomic number = # of neutrons) • Alkali: Group 1 (# of valence electrons) • Period 3 (# of energy levels) • Identify the ionic charge

Metals will lose electrons when forming compounds. They become positively charged because there will be more protons than electrons. A positive ion is formed. Na⁺ 11 P 12 N 2 8 1 11 P = +11 10 E = -10 ionic charge: 1 P = +1

Draw a Dot Diagram of Sodium (Na) Need to know for a Dot Diagram: • Element Symbol: Na • Group Number: 1 (# of valence electrons)

Available electron for bonding!! Na⁺ Na

Draw a Bohr Model of Oxygen (O) • Atomic Number: 8 (# of protons & therefore, same # of electrons) • Atomic Mass: 15. 999 (Atomic mass – Atomic number = # of neutrons) • Group 16 (# of valence electrons) • Period 2 (# of energy levels) • Identify the ionic charge
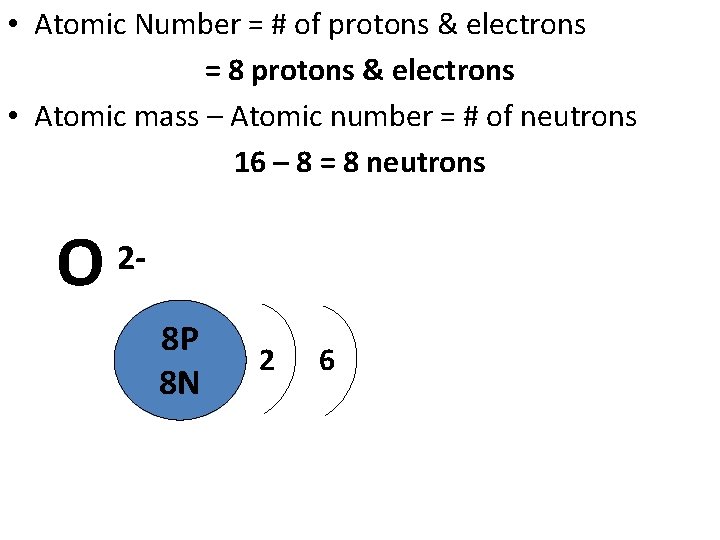
• Atomic Number = # of protons & electrons = 8 protons & electrons • Atomic mass – Atomic number = # of neutrons 16 – 8 = 8 neutrons O 2 - 8 P 8 N 2 6

Draw a Dot Diagram of Oxygen (O) Need to know for a Dot Diagram: • Element Symbol: O • Group Number: 16 (# of valence electrons)
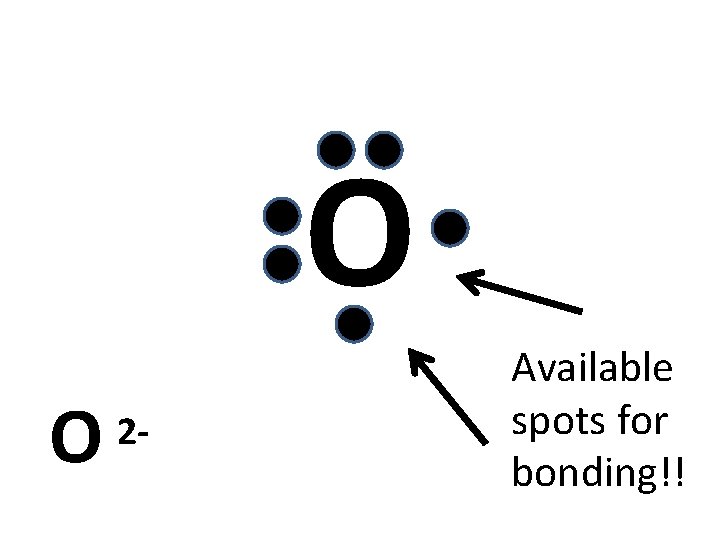
O O 2 - Available spots for bonding!!

Draw a Bohr Model of Beryllium (Be) • Atomic Number: 4 (# of protons & therefore, same # of electrons) • Atomic Mass: 9. 012 (Atomic mass – Atomic number = # of neutrons) • Alkaline: Group 2 (# of valence electrons) • Period 2 (# of energy levels) • Identify the ionic charge

• Atomic Number = # of protons & electrons = 4 protons & electrons • Atomic mass – Atomic number = # of neutrons 9 – 4 = 5 neutrons Be 2+ 4 P 5 N 2 2

Draw a Dot Diagram of Beryllium (Be) Need to know for a Dot Diagram: • Element Symbol: Be • Group Number: 2 (# of valence electrons)
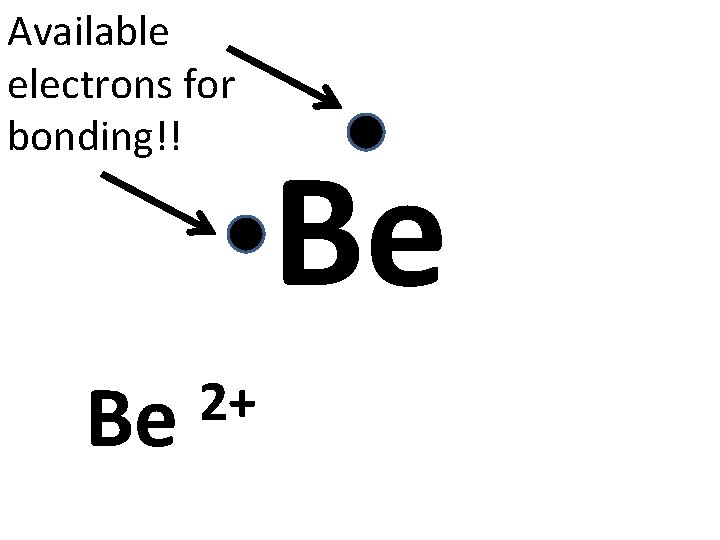
Available electrons for bonding!! Be 2+ Be
- Slides: 17