Draft Risk Evaluation for Carbon Tetrachloride CCl 4

Draft Risk Evaluation for Carbon Tetrachloride (CCl 4) SACC Peer Review Presentation CCl 4 Workgroup Team Members: Doritza Pagán-Rodríguez (Lead), Tom Bateson, Eliane Catilina, Sandip Chattopadhyay, Jeff Gallagher, Ume Hassan, Tim Mc. Mahon, Claudia Menasche, Amelia Nguyen, Nerija Orentas, Alan Sasso, Molly Shuman-Goodier, Tameka Taylor, Susanna Wegner, Cindy Wheeler, Paul White, Steve Witkin, Edmund Wong Management Lead: Karen Eisenreich Office of Pollution Prevention and Toxics Risk Assessment Division U. S. Environmental Protection Agency February 25 -26, 2020

Overview of Presentation • Background Information on Draft Risk Evaluation • Physical and Chemical Properties of CCl 4 • Scope of the Evaluation – Lifecycle Diagram and Conceptual Model – Conditions of Use and Assessed Exposure Scenarios – Receptors/Populations • Chemistry and Environmental Fate Characterization • Characterization of Environmental Exposure, Hazard, and Risk – Environmental Exposure – Environmental Hazard – Environmental Risk Characterization 2 • Characterization of Human Exposure, Hazard, and Risk – Occupational Exposure – Human Health Hazard – Human Health Risk Characterization

Background Information on Draft Risk Evaluation • With the signing of the Lautenberg Act into law in June of 2016, the scope of the 2019 draft risk evaluation includes: – Application of the TSCA systematic review process and incorporation of new information – Assessment of a wide range of conditions of use – Review, consideration, and evaluation of risks to environmental receptors (e. g. , aquatic organisms), and humans (e. g. , occupational users and non-users) • In problem formulation EPA identified exposure pathways for the general population: 3 – These pathways are covered under other environmental statutes and exposures are managed (i. e. , CAA, SDWA, CWA, and RCRA) – EPA did not evaluate hazards or exposure to general population in this risk evaluation (U. S. EPA, 2018 d) as a result of regulatory nexus with other EPA authorities

Section 1. 1 Physical Chemical Properties of CCl 4 • CASRN 56 -23 -5 • Physical-chemical properties: – Physical form: Colorless liquid; sweet odor (Merck, 1996); (U. S. Coast Guard, 1985) – Melting point: -23 ºC (Lide, 1999) – Boiling point: 76. 8 ºC (Lide, 1999) – Density: 1. 4601 g/cm 3 at 20 ºC (Lide, 1999) – Water solubility: 793 mg/L at 25 ºC (Horvath, 1982) – Vapor pressure: 115 mm. Hg at 25 ºC (Boublík et al. (1984)) 4 – Log Kow: 2. 83 (Hansch et al. (1995))
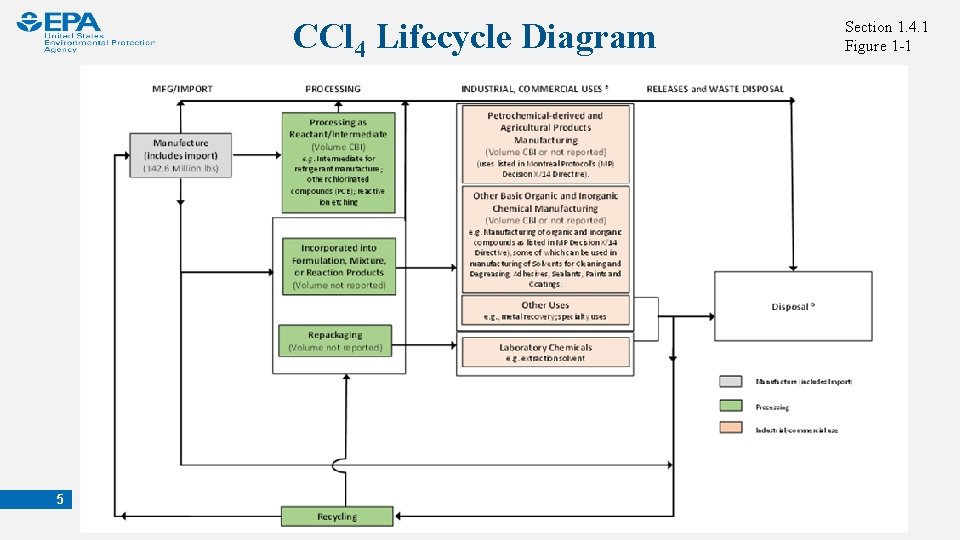
CCl 4 Lifecycle Diagram 5 Section 1. 4. 1 Figure 1 -1
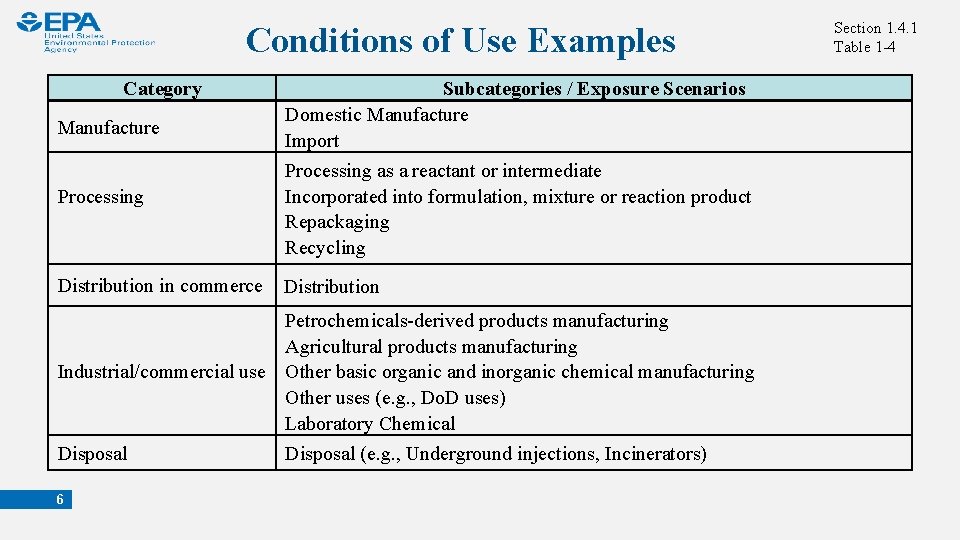
Conditions of Use Examples Category Manufacture Processing Subcategories / Exposure Scenarios Domestic Manufacture Import Processing as a reactant or intermediate Incorporated into formulation, mixture or reaction product Repackaging Recycling Distribution in commerce Distribution Petrochemicals-derived products manufacturing Agricultural products manufacturing Industrial/commercial use Other basic organic and inorganic chemical manufacturing Other uses (e. g. , Do. D uses) Laboratory Chemical Disposal 6 Disposal (e. g. , Underground injections, Incinerators) Section 1. 4. 1 Table 1 -4
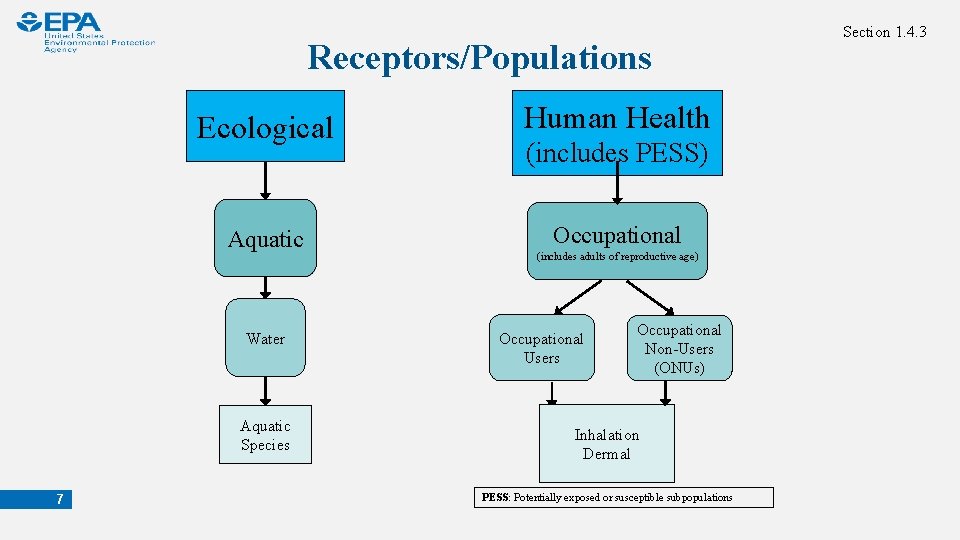
Receptors/Populations Ecological Human Health Aquatic Occupational (includes PESS) Water Aquatic Species 7 (includes adults of reproductive age) Occupational Users Occupational Non-Users (ONUs) Inhalation Dermal PESS: Potentially exposed or susceptible subpopulations Section 1. 4. 3

Environmental Fate and Transport Section 2. 1 and Charge Questions 1 and 6 8

Environmental Fate and Transport Approach Section 2. 1, Appendix C EPA evaluated and extracted data using the Systematic Review process for the evaluation of CCl 4 Some environmental fate and transport characteristics were estimated with EPI Suite™, a set of predictive models of physical/chemical and fate and transport characteristics 9 • EPI Suite™ was reviewed by EPA Science Advisory Board in 2007 • Individual models have been peer-reviewed in numerous articles published in technical journals (citations are available in the EPI Suite™ help files)
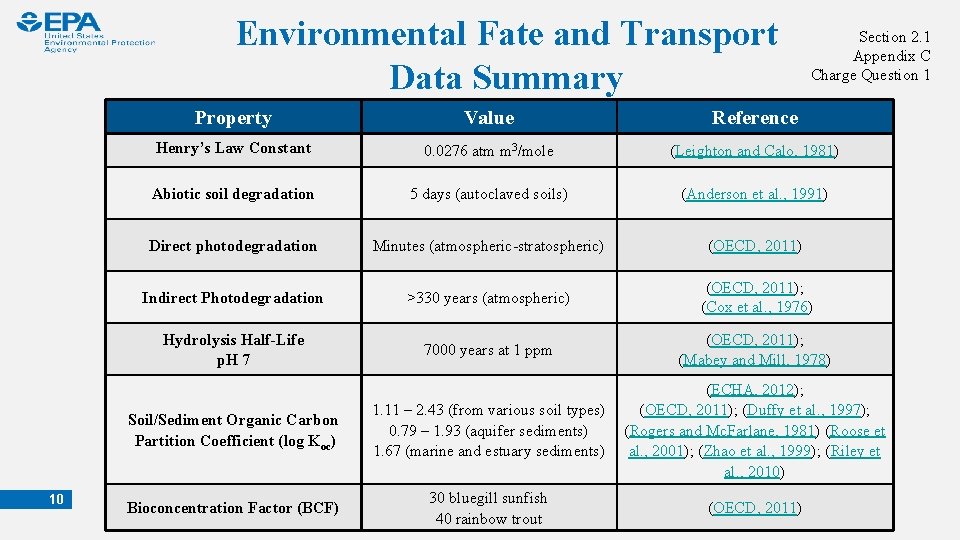
Environmental Fate and Transport Data Summary 10 Section 2. 1 Appendix C Charge Question 1 Property Value Reference Henry’s Law Constant 0. 0276 atm m 3/mole (Leighton and Calo, 1981) Abiotic soil degradation 5 days (autoclaved soils) (Anderson et al. , 1991) Direct photodegradation Minutes (atmospheric-stratospheric) (OECD, 2011) Indirect Photodegradation >330 years (atmospheric) (OECD, 2011); (Cox et al. , 1976) Hydrolysis Half-Life p. H 7 7000 years at 1 ppm (OECD, 2011); (Mabey and Mill, 1978) Soil/Sediment Organic Carbon Partition Coefficient (log Koc) 1. 11 – 2. 43 (from various soil types) 0. 79 – 1. 93 (aquifer sediments) 1. 67 (marine and estuary sediments) (ECHA, 2012); (OECD, 2011); (Duffy et al. , 1997); (Rogers and Mc. Farlane, 1981) (Roose et al. , 2001); (Zhao et al. , 1999); (Riley et al. , 2010) Bioconcentration Factor (BCF) 30 bluegill sunfish 40 rainbow trout (OECD, 2011)

Fate and Transport Overview Section 2. 1 Table 2 -1 Appendix C Charge Questions 1 and 6 Based on physical-chemical properties and environmental fate characteristics, overall, CCl 4: • Has low bioaccumulation potential • Is not likely to adsorb to biosolids, sediments, soil or biota • Is expected to have some volatilization from surface water and soil then degrade by photolysis • May undergo aerobic or anaerobic biodegradation in soil, sediment, and water 11 • EPI Suite module estimates that the half-life of CCl 4 in a model river will be about 1. 3 hours and the half-life in a model lake will be about 5 days

Fate, Transport, and Exposure: Sediment and Biosolids Section 2. 1 Section 4. 1. 3 CCl 4 is expected to be present in the aqueous fraction and pore water of biosolids or sediments • Based on equilibrium partitioning (e. g. , log KOC ) and potential for moderate to rapid anaerobic biodegradation, concentrations in sediments are expected to be lower than those in overlying surface water Concentrations of CCl 4 in biosolids are expected to be similar to or less than concentrations in wastewater treatment plant effluents • If released to land via biosolids, CCl 4 is expected to volatilize to air or 12 migrate to groundwater based on log KOC, water solubility, vapor pressure and Henry’s Law constant

Environmental Risk Assessment Releases, Exposures, Hazards, and Risk Characterization Environmental Release and Exposure Sections 2. 2 and 2. 3, Appendices D and E, and Charge Questions 2 and 6 13

Section 2. 21; Appendix E; Charge Questions 2. 1 and 6 ENVIRONMENTAL RELEASES Environmental releases determined based on reporting to TRI and DMR • An estimated 1, 704 lbs of CCl 4 were reported in TRI to be released to surface water from industrial sources for 2018 (U. S. EPA, 2019) • 21 top annual CCl 4 discharging facilities as reported in DMR used in a screening analysis (U. S. EPA ECHO, 2019) ₋ Average 5 -year (2014 through 2018) releases ranged from 0. 25 pounds/year to 250 pounds/year Application of release data in this Draft Risk Evaluation • Used for screening level evaluation of environmental risks from CCl 4 in surface water 14

Section 4. 1; Appendix E; Charge Questions 2. 1 and 6 ENVIRONMENTAL EXPOSURE PATHWAYS Ecological Surface Water EPA carried out a quantitative assessment comparing available environmental hazard data for aquatic species and estimated aquatic exposure concentrations Aquatic Water Aquatic Species 15 Sediment and Terrestrial (not evaluated) Based on a qualitative consideration of the physicalchemical and environmental fate characteristics, sedimentdwelling and terrestrial organisms are not expected to be exposed to higher concentrations of CCl 4 compared to those estimated for surface water 15

Section 2. 3. 1. 1; Appendix E; Charge Questions 2. 1 and 6 CCl 4 SURFACE WATER CONCENTRATIONS • Screened using the range of environmental releases reported from 2014 through 2018 Discharge Monitoring Reports (DMR) • Modeling assumptions were varied to estimate surface water concentrations using E-FAST 2014: ₋ Release days modeled: 20 days/yr or maximum release frequency (250 days/yr) for direct dischargers. Indirect dischargers (discharging to municipal wastewater treatment facility) used maximum value only 16 § 20 -day release scenarios had modeled median surface water concentration of 19. 6 µg/L (Range = 4. 9 E-05 – 130 µg/L) § Maximum days of release had modeled median surface water concentration of 1. 5 µg/L (Range = 4. 0 E-06 – 10 µg/L)

Environmental Risk Assessment Releases, Exposures, Hazards, and Risk Characterization Environmental Hazard Section 3. 1, Appendix G, and Charge Questions 2. 1 and 6 17

Sections 3. 1 Appendix G Charge Questions 2. 1 and 6 Key Information on Environmental Hazard: Toxicity to Aquatic Organisms 18 14 environmental hazard studies identified for CCl 4 fish, amphibians, aquatic invertebrates, and algae • Acute hazard data: • Amphibian LC 50 = 0. 90 mg/L • Fish LC 50 = 10. 4 mg/L • Aquatic invertebrate EC 50/LC 50 = 11. 1 mg/L • Chronic hazard data: • Amphibian LC 10 = 0. 025 mg/L • Fish LC 50 = 1. 97 mg/L • Aquatic invertebrate chronic value with an acute-to-chronic ratio = 1. 1 mg/L • Algae hazard data: EC 50 = 0. 07 mg/L 18

Section 4. 1. 1; Charge Question 2. 1 Environmental Hazard: Concentrations of Concern (COC) for Aquatic Species • COCs (i. e. , hazard thresholds) for aquatic species were calculated based on the environmental hazard data for CCl 4 • An assessment factor (AF) is applied according to EPA methods (U. S. EPA, 2012 e; U. S. EPA, 2013 b) to account for differences in inter- and intraspecies variability – AF = 10 was used for amphibians and algae • Aquatic COCs were calculated for amphibians and algae 19 – Acute amphibian COC = 90 µg/L – Chronic amphibian COC = 3 µg/L – Algae COC = 7 µg/L

Environmental Risk Characterization Section 4. 1 and Charge Questions 5. 1 – 5. 4 and 6 20
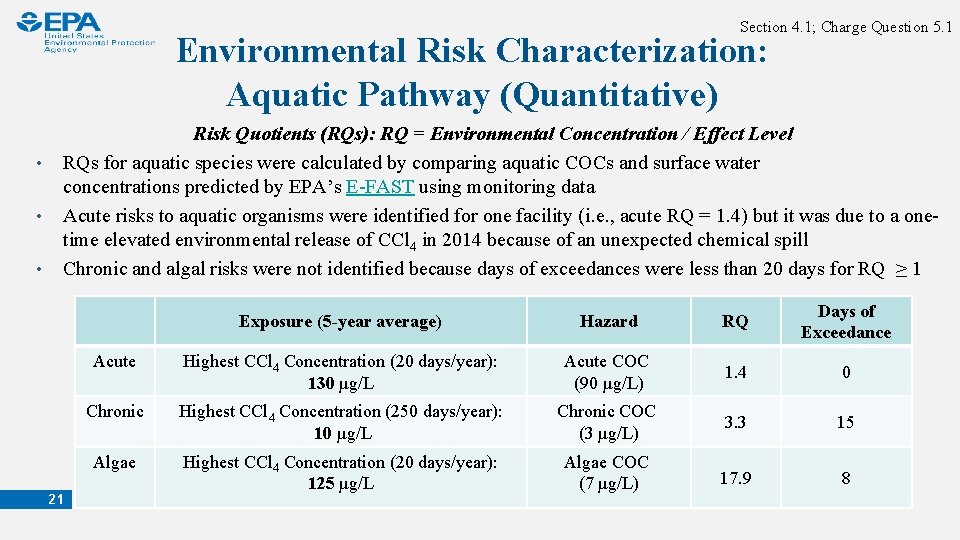
Section 4. 1; Charge Question 5. 1 Environmental Risk Characterization: Aquatic Pathway (Quantitative) • • • Risk Quotients (RQs): RQ = Environmental Concentration / Effect Level RQs for aquatic species were calculated by comparing aquatic COCs and surface water concentrations predicted by EPA’s E-FAST using monitoring data Acute risks to aquatic organisms were identified for one facility (i. e. , acute RQ = 1. 4) but it was due to a onetime elevated environmental release of CCl 4 in 2014 because of an unexpected chemical spill Chronic and algal risks were not identified because days of exceedances were less than 20 days for RQ ≥ 1 21 Exposure (5 -year average) Hazard RQ Days of Exceedance Acute Highest CCl 4 Concentration (20 days/year): 130 µg/L Acute COC (90 µg/L) 1. 4 0 Chronic Highest CCl 4 Concentration (250 days/year): 10 µg/L Chronic COC (3 µg/L) 3. 3 15 Algae Highest CCl 4 Concentration (20 days/year): 125 µg/L Algae COC (7 µg/L) 17. 9 8

Section 4. 1 Charge Questions 5. 1 and 6 Environmental Risk Characterization: Aquatic Sediment / Terrestrial Pathways (Qualitative) • Qualitative consideration of the conditions of use and physical-chemical properties of CCl 4 indicate that risks are not expected to sediment-dwelling or terrestrial organisms o Sediment: CCl 4 is expected to be in the pore water rather than adsorbed to the sediment organic matter, and concentrations of CCl 4 are expected to be lower in pore water than concentrations in the water column o Terrestrial: CCl 4 is not expected to partition to or accumulate in or adsorb to soil; rather, it is expected to volatilize to air or migrate through soil into groundwater. EPA did not include the emission pathways to ambient air from commercial and industrial stationary sources or associated inhalation exposure of terrestrial species, because stationary source releases of CCl 4 to ambient air are adequately assessed any risks effectively managed under the jurisdiction of the Clean Air Act (CAA) 22

Environmental Risk Assumptions and Uncertainties Section 4. 5. 1 Charge Questions 5. 4 and 6 • The use of proxy organisms to represent all organisms within a specific taxa and • • environment can be an oversimplification, possibly underestimating risk The use of Assessment Factors (AFs) is intended to account for interspecies variability in response The use of an acute to chronic ratio is intended to approximate a chronic response based on acute data (aquatic invertebrates) EPA used sub-chronic data, measuring developmental effects in embryo and larvae, to calculate the amphibian chronic COC, which introduces some uncertainty about whether we are overestimating or underestimating chronic risk Environmental exposure data could be over or underestimating exposure to aquatic organisms – E-FAST results: Inputs to the model include DMR data, which cannot be attributed to a specific COU. CCl 4 is a volatile chemical, but E-FAST does not account for volatilization. For identified still water bodies, E-FAST uses a dilution factor as low as 1 23

Section 4. 1 Charge Questions 5. 3, 5. 4 and 6 Environmental Risk Conclusions • EPA determined that there are no acute or chronic environmental risks from the TSCA conditions of use of CCl 4 • EPA has high confidence that there are no identified environmental risks from the TSCA conditions of use and exposure pathways within the scope of the risk evaluation for CCl 4 • This is based on EPA using conservative, high end exposures and modeled surface water concentrations and the most conservative (highest toxicity)/environmentally-protective acute and chronic COCs 24

Human Health Assessment Exposures, Hazards, and Risk Characterization Occupational Exposure Section 2. 4. 1 and Charge Questions 3 and 6 25

Section 2. 4. 1 Charge Questions 3 and 6 Occupational Exposure Assessment Objectives • Define Occupational Exposure Scenarios o Determine similarities of occupational users activities among conditions of use o Determine data gaps among conditions of use o Combine/ group subcategories for conditions of use o Example: Import includes the import and repackaging of CCl 4 • Estimate Number of Occupational Users and ONUs for Each Scenario • Estimate Exposures for Each Scenario 26 o Develop inhalation and dermal estimates for occupational users and, where possible, inhalation estimates for ONUs

Section 2. 4. 1; Charge Question 3 Overview of Inhalation Exposure Assessment Objectives ‒ Assess inhalation exposure concentrations for occupational users and ONUs: § Provide central tendency and high-end estimates § Provide estimates for occupational users and, where possible, for ONUs Calculations ‒ Acute concentration (AC), Average Daily Concentration (ADC) 27 ‒ Lifetime average daily concentration (LADC)

Sections 2. 4. 1. 2 and 2. 4. 1. 6 Charge Questions 3 and 6 Inhalation Exposure Assessment Approach 1. Monitoring Data • PBZ monitoring data (8 -hr TWA, and 12 -hr TWA) from directly applicable scenarios • Key sources of data: Industry, NIOSH, OSHA, open literature • All monitoring data were of varying data quality 2. Modeling Approaches • Fills data gaps or supplements existing monitoring data (if available) • Deterministic model developed through relevant literature and consideration of existing EPA exposure models 28 PBZ = personal breathing zone; TWA = time weighted average
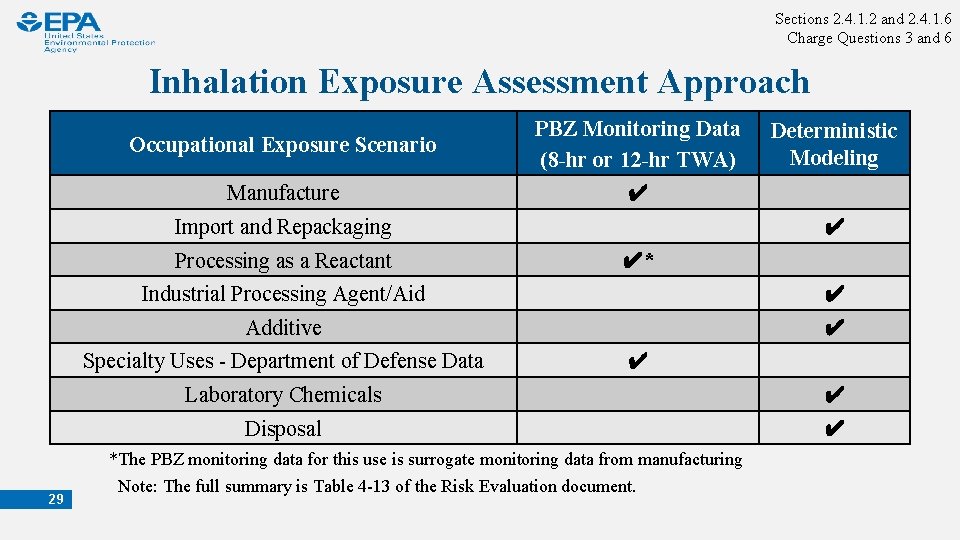
Sections 2. 4. 1. 2 and 2. 4. 1. 6 Charge Questions 3 and 6 Inhalation Exposure Assessment Approach Occupational Exposure Scenario PBZ Monitoring Data (8 -hr or 12 -hr TWA) Manufacture ✔ Import and Repackaging Processing as a Reactant ✔ ✔* Industrial Processing Agent/Aid ✔ Additive ✔ Specialty Uses - Department of Defense Data 29 Deterministic Modeling ✔ Laboratory Chemicals ✔ Disposal ✔ *The PBZ monitoring data for this use is surrogate monitoring data from manufacturing Note: The full summary is Table 4 -13 of the Risk Evaluation document.
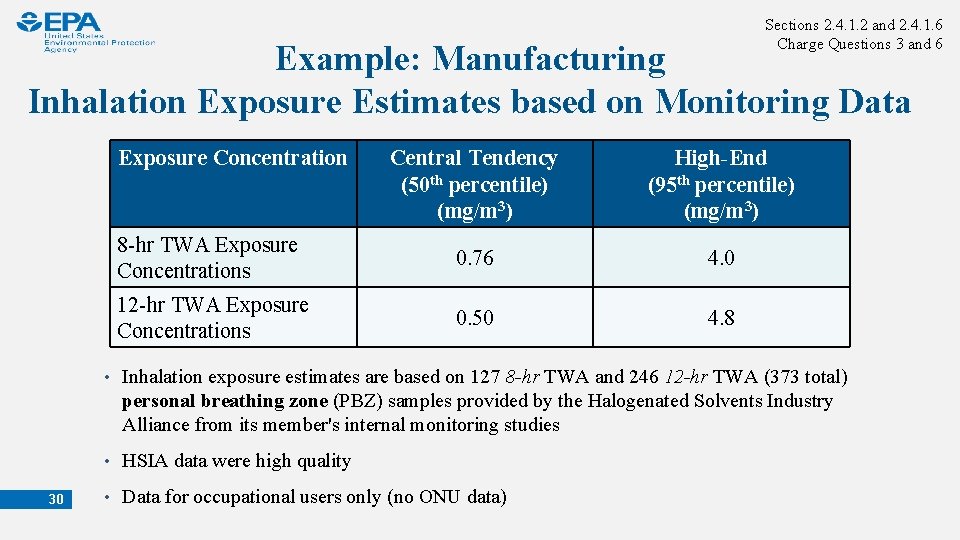
Sections 2. 4. 1. 2 and 2. 4. 1. 6 Charge Questions 3 and 6 Example: Manufacturing Inhalation Exposure Estimates based on Monitoring Data Exposure Concentration Central Tendency (50 th percentile) (mg/m 3) High-End (95 th percentile) (mg/m 3) 8 -hr TWA Exposure Concentrations 0. 76 4. 0 12 -hr TWA Exposure Concentrations 0. 50 4. 8 • Inhalation exposure estimates are based on 127 8 -hr TWA and 246 12 -hr TWA (373 total) personal breathing zone (PBZ) samples provided by the Halogenated Solvents Industry Alliance from its member's internal monitoring studies • HSIA data were high quality 30 • Data for occupational users only (no ONU data)

Sections 2. 4. 1. 2 and 2. 4. 1. 6 Charge Questions 3 and 6 Inhalation Exposure Estimates from Deterministic Exposure Modeling • Applicable for any volatile chemical with conditions of use where unloading or loading of the chemical is the main occupational users interaction with the chemical • Assumes CCl 4 is unloaded into tank trucks and railcars and transported and distributed in bulk • Considers fugitive emissions from: o Displacement of saturated air containing CCl 4 as the container/truck is filled with liquid o Emissions of saturated air containing CCl 4 that remains in the loading arm, transfer hose, and related equipment o Emissions from equipment leaks from processing units such as pumps, seals and valves • Input parameters (e. g. , concentration of chemical in air, vapor generation rate, outdoor ventilation rate, air speed, mixing factor, and environmental parameters) defined using data from literature 31

Section 2. 4. 1. 2 Charge Question 3 and 6 Example: Inhalation Exposure Estimates for Import and Repackaging based on Modeling Condition Occupational User 8 -hr TWA Exposure Concentrations Central Tendency (50 th percentile) (mg/m 3) High-End (95 th percentile) (mg/m 3) 0. 06 0. 30 Model Assumptions: • At least one person will be near the loading rack during the entire duration of the loading operation and one container is loaded/unloaded per shift • Central tendency scenario: one tank truck per shift • High-end scenario: one railcar per shift 32

Sections 2. 4. 1. 2, 2. 4. 1. 6 and 4. 4. 1 Charge Questions 3, 5. 2 and 6 Uncertainties and Limitations: Inhalation Exposure • Monitoring Data ‒ ‒ PBZ data are available for 3 OESs. However, certain OESs or job categories had limited or no data Representativeness of PBZ data is uncertain Data provided by limited sources Many data are reported below the limit of detection • Modeling Approaches ‒ Modeling approach captures variability in input parameters. However, there is uncertainty associated with each parameter, which is determined based on review of available literature ‒ Model assumes at least one occupational users will be near the loading rack during the entire duration of the loading operation and one container is loaded/unloaded per shift § central tendency scenario: one tank truck per shift § high-end scenario: one railcar per shift ‒ Representativeness of modeling is uncertain 33

Section 2. 4. 1 Charge Questions 3 and 6 Dermal Exposure Assessment Approach • ONUs are not exposed via the dermal route • Occluded dermal exposures for occupational users are unlikely • The Dermal Exposure to Volatile Liquids Model is used to estimate dermal retained dose for occupational users ‒ Modified the EPA/OPPT 2 -Hand Dermal Exposure to Liquids 34 Model • Evaporation accounted for by Fraction absorbed term, based on experimental measurement from Frasch et al. , (2011) • Potential glove use accounted by Protection Factor term
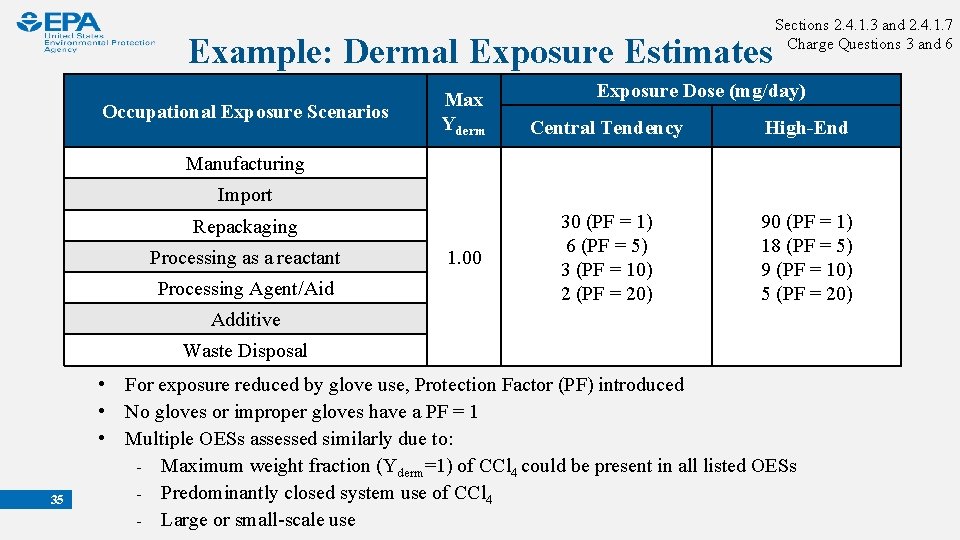
Example: Dermal Exposure Estimates Occupational Exposure Scenarios Max Yderm Sections 2. 4. 1. 3 and 2. 4. 1. 7 Charge Questions 3 and 6 Exposure Dose (mg/day) Central Tendency High-End 30 (PF = 1) 6 (PF = 5) 3 (PF = 10) 2 (PF = 20) 90 (PF = 1) 18 (PF = 5) 9 (PF = 10) 5 (PF = 20) Manufacturing Import Repackaging Processing as a reactant Processing Agent/Aid 1. 00 Additive Waste Disposal 35 • For exposure reduced by glove use, Protection Factor (PF) introduced • No gloves or improper gloves have a PF = 1 • Multiple OESs assessed similarly due to: ₋ Maximum weight fraction (Yderm=1) of CCl 4 could be present in all listed OESs ₋ Predominantly closed system use of CCl 4 ₋ Large or small-scale use

Sections 2. 4. 1. 3, 2. 4. 1. 7 and 4. 4. 1 Charge Questions 3, 5. 2 and 6 Uncertainties and Limitations: Dermal Exposure • Modeling accounts for evaporation and absorption • Absorption may depend on skin loading conditions, rather than fixed fraction • Assumes surface areas of two full hands for high-end and one full hand for central tendency • Assumes a single exposure event per day and does not address variability in exposure duration and frequency 36

Human Health Assessment Exposures, Hazards, and Risk Characterization Human Health Hazard Section 3. 2 and Charge Questions 4 and 6 37

Section 3. 2. 3 Hazard Identification, Weight of Evidence (WOE) Human health hazard domains based on May 2018 problem formulation (U. S. EPA, 2018 d) and recent literature search: Neurotoxicity * Liver Toxicity * Kidney Toxicity Reproductive/Developmental Toxicity Irritation/Sensitization Genetic Toxicity Carcinogenicity * * Based on WOE/data quality evaluation, several domains carried to dose-response. • • 38 Acceptable studies mostly available by inhalation and oral routes, limited by dermal route.

Section 3. 2. 4. 1. 1 Charge Questions 4. 1 and 6 Inhalation Hazard from Acute Exposure - WOE Findings from animal studies are consistent with those associated with human exposures (NRC, 2014) • Neurotoxicity, central nervous system (CNS) depression o Immediate effect in acute toxicity studies in animals and humans exposed by inhalation to relatively high concentrations of CCl 4 (U. S. EPA, 2010) (NRC, 2014) o Liver and kidney toxicity are also considered primary end points in acute inhalation studies (animals and humans) • Reproductive/Developmental Toxicity, endpoint not considered an acute hazard for CCl 4 based on animal data. There are no reasonably available human data 39
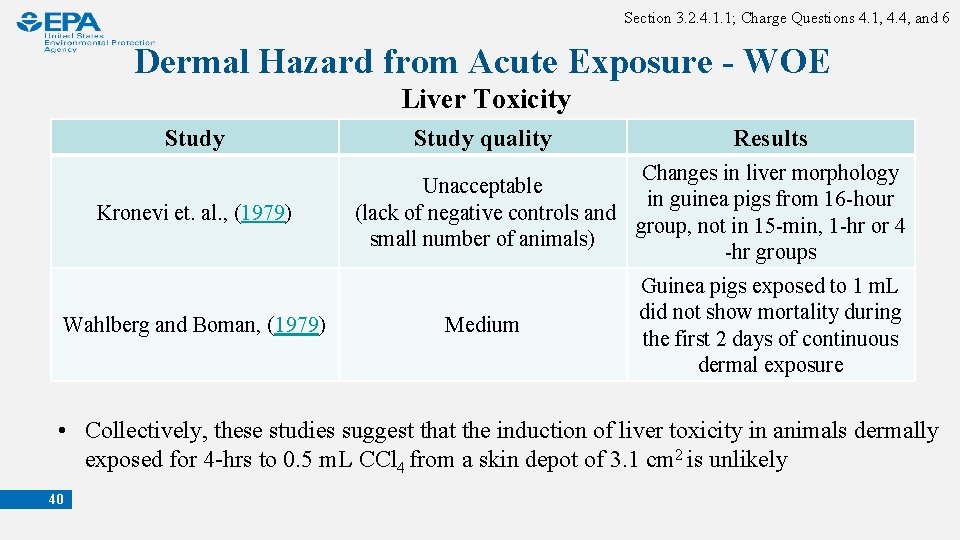
Section 3. 2. 4. 1. 1; Charge Questions 4. 1, 4. 4, and 6 Dermal Hazard from Acute Exposure - WOE Liver Toxicity Study Kronevi et. al. , (1979) Wahlberg and Boman, (1979) Study quality Results Changes in liver morphology Unacceptable in guinea pigs from 16 -hour (lack of negative controls and group, not in 15 -min, 1 -hr or 4 small number of animals) -hr groups Medium Guinea pigs exposed to 1 m. L did not show mortality during the first 2 days of continuous dermal exposure • Collectively, these studies suggest that the induction of liver toxicity in animals dermally exposed for 4 -hrs to 0. 5 m. L CCl 4 from a skin depot of 3. 1 cm 2 is unlikely 40

Sections 3. 2. 4. 1. 2 and 3. 2. 4. 2 Charge Questions 4. 1, 4. 2, 4. 3 and 6 Inhalation Hazard from Chronic Exposure – WOE • Non-cancer liver toxicity ₋ Sensitive effect in multiple inhalation and oral animal studies and human data • Cancer ₋ Under the Guidelines for Carcinogen Risk Assessment (U. S. EPA, 2005 b), EPA classified CCl 4 as "likely to be carcinogenic to humans" based on: 41 § Adequate evidence of carcinogenicity in humans (e. g. , brain cancer) § Sufficient evidence in animals by oral and inhalation exposure, i. e. , hepatic tumors in multiple species (e. g. , rat, mouse, and hamster) and pheochromocytomas (e. g. , adrenal gland tumors) in mice (U. S. EPA, 2010) ₋ Systematic review identified 11 epidemiological studies with acceptable data quality

Sections 3. 2. 5. 1 and 3. 2. 5. 3 Charge Questions 4 and 6 Endpoints, Studies Used to Evaluate Risks Acute/Short Term Inhalation Exposure: Neurotoxicity (Davis, 1934). • Immediate and temporary CNS effects, consisting of escape-impairing symptoms (i. e. , dizziness) • Low quality study Chronic Exposure Non-Cancer: Liver toxicity (Nagano et al. , 2007 a) • High quality chronic study • Increased incidence and severity of nonneoplastic liver lesions (e. g. , fatty change, fibrosis, cirrhosis) at 25 and 125 ppm in male and female rats • Fatty change was selected as the specific endpoint indicative of cellular damage/ most sensitive endpoint Cancer: Liver and adrenal tumors (Nagano et al. , 2007 a) • High quality chronic animal study • Statistically significant increase in hepatocellular adenomas and carcinomas in rats and mice of both sexes, and adrenal pheochromocytomas in mice of both sexes 42

Section 3. 2. 5. 2. 1; Charge Question 4. 1 Dose-Response -Acute Inhalation POD Derivation NOEC of 76 ppm (480 mg/m 3) for CNS effects (i. e. , dizziness) in humans exposed for 4 hrs (Davis, 1934) • Adult human POD adjusted from experimental duration: ₋ 8 -hr and 12 -hr exposure durations ₋ Cn x T = K - modified Haber’s Law, with n = 2. 5 from Ten Berge et al. (1986) • Uncertainty factors for benchmark margin of exposure (MOE) based on EPA guidance (U. S. EPA, 2002): ₋ UFA = interspecies UF (value = 1 based on use of human data) ₋ UFH = intraspecies UF (value = 10) 43
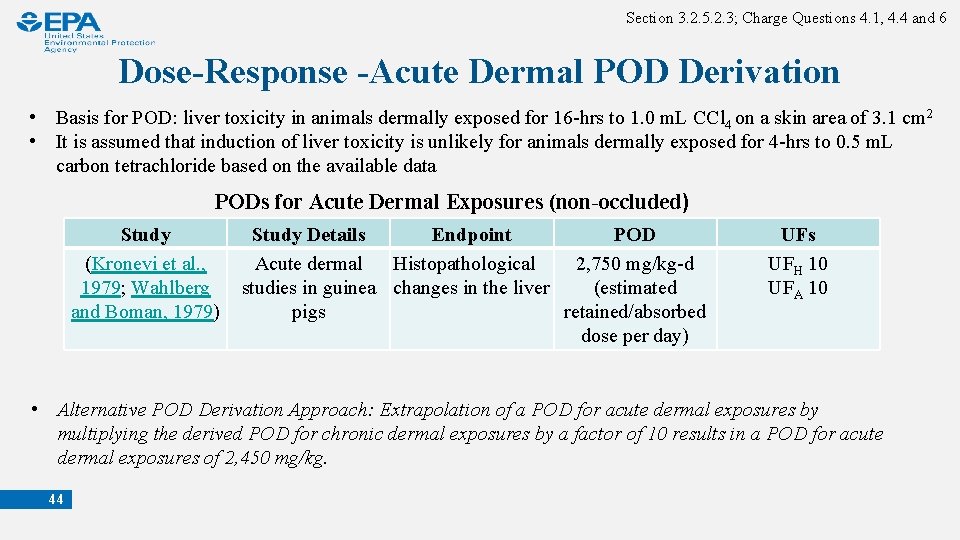
Section 3. 2. 5. 2. 3; Charge Questions 4. 1, 4. 4 and 6 Dose-Response -Acute Dermal POD Derivation • Basis for POD: liver toxicity in animals dermally exposed for 16 -hrs to 1. 0 m. L CCl 4 on a skin area of 3. 1 cm 2 • It is assumed that induction of liver toxicity is unlikely for animals dermally exposed for 4 -hrs to 0. 5 m. L carbon tetrachloride based on the available data PODs for Acute Dermal Exposures (non-occluded) Study (Kronevi et al. , 1979; Wahlberg and Boman, 1979) Study Details Endpoint POD Acute dermal Histopathological 2, 750 mg/kg-d studies in guinea changes in the liver (estimated pigs retained/absorbed dose per day) UFs UFH 10 UFA 10 • Alternative POD Derivation Approach: Extrapolation of a POD for acute dermal exposures by multiplying the derived POD for chronic dermal exposures by a factor of 10 results in a POD for acute dermal exposures of 2, 450 mg/kg. 44

Section 3. 2. 5. 2. 2; Charge Question 4. 1 Dose-Response - Chronic Non-Cancer POD Derivation • Basis for chronic inhalation PODs: 104 -weeks inhalation study in rats (Nagano et al. , 2007 b), in which the lowest exposure concentration in this study, 5 ppm, was considered a NOAEC based on liver and kidney toxicity at ≥ 25 ppm • BMD modeling o A human PBPK model was used in the IRIS Assessment to estimate HEC (in mg/m 3) consisting of calculated BMDL 10 for fatty changes of the liver of 14. 3 mg/m 3 for continuous exposures o Conservatively, the BMDL 10 was extrapolated to shorter exposure durations using the equation Cn × t = k, where an empirical value of n was determined to be 2. 5 on the basis of rat lethality data (Ten Berge et al. , 1986) 45
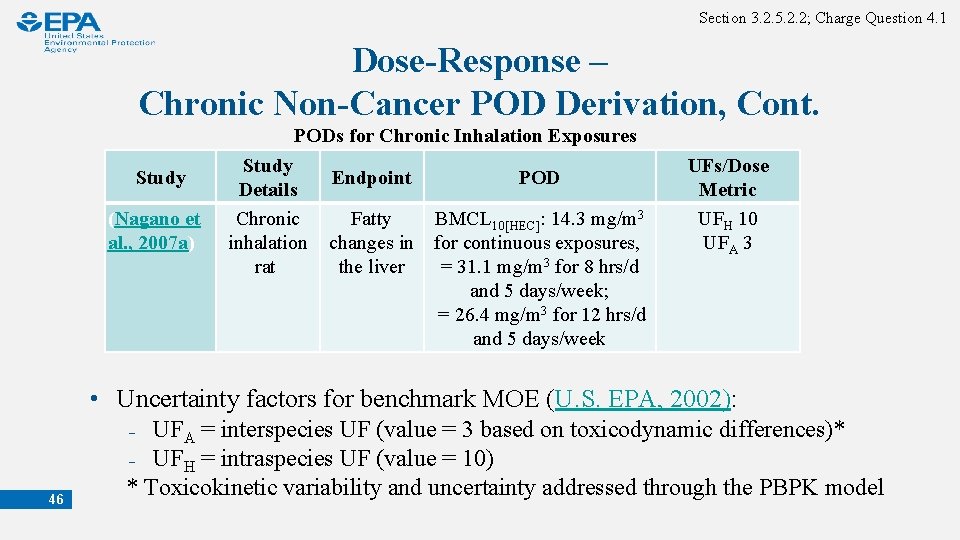
Section 3. 2. 5. 2. 2; Charge Question 4. 1 Dose-Response – Chronic Non-Cancer POD Derivation, Cont. Study (Nagano et al. , 2007 a) PODs for Chronic Inhalation Exposures Study Endpoint POD Details Chronic Fatty BMCL 10[HEC]: 14. 3 mg/m 3 inhalation changes in for continuous exposures, rat the liver = 31. 1 mg/m 3 for 8 hrs/d and 5 days/week; = 26. 4 mg/m 3 for 12 hrs/d and 5 days/week UFs/Dose Metric UFH 10 UFA 3 • Uncertainty factors for benchmark MOE (U. S. EPA, 2002): 46 ₋ UFA = interspecies UF (value = 3 based on toxicodynamic differences)* ₋ UFH = intraspecies UF (value = 10) * Toxicokinetic variability and uncertainty addressed through the PBPK model

Section 3. 2. 5 Charge Questions 4. 1, 4. 2, 4. 3 and 6 Dose-Response - Cancer Inhalation Unit Risk (IUR) EPA (U. S. EPA, 2010) classifies CCl 4 as "likely to be carcinogenic to humans" based on: • inadequate evidence of carcinogenicity in humans • sufficient evidence in animals by oral and inhalation exposure, i. e. , hepatic tumors in multiple species (rat, mouse, and hamster) and pheochromocytomas (adrenal gland tumors) in mice IRIS Toxicological Review of CCl 4 (U. S. EPA, 2010) Conclusions: • low dose linear cancer risk model, which is EPA’s default approach to risk assessment when the MOA is unknown IUR of 6 × 10 -6 per μg/m 3 based on incidence of pheochromocytoma in male mice. • Data for male mouse adrenal tumors yielded the highest estimate of the IUR of those data sets modeled including the liver tumors in rats and mice of both sexes and pheochromocytomas in mice of both sexes. 47

Sections 3. 2. 1. 2. 4 and 3. 2. 5 Charge Questions 4. 1, 4. 4 and 6 Dose-Response – Chronic Dermal POD and Cancer Slope Factor • Inadequate dermal information ₋ No repeated-dose toxicity studies ₋ No PBPK/PD models for dermal route or relevant dermal kinetic, metabolic data • Dermal human equivalent doses (or HEDs) from inhalation PODs ₋ Chronic: inhalation rate of 10 m 3/day and BW of 80 kg • Extrapolated from inhalation toxicity studies: ₋ Inhalation studies already in risk evaluation ₋ Toxic moieties for liver toxicity and cancer are metabolites and both routes lack first pass metabolism • POD not adjusted for absorption ₋ Handled through dermal exposure estimates 48
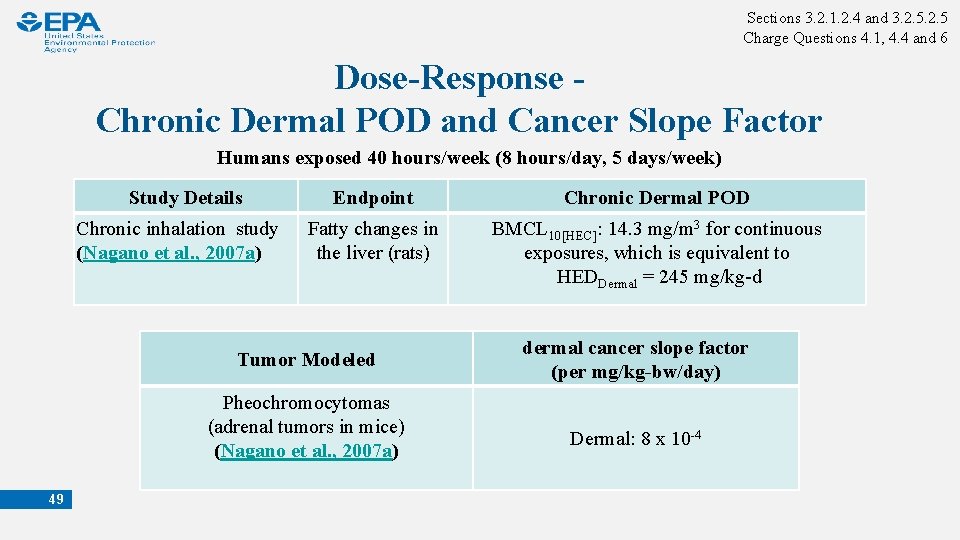
Sections 3. 2. 1. 2. 4 and 3. 2. 5 Charge Questions 4. 1, 4. 4 and 6 Dose-Response - Chronic Dermal POD and Cancer Slope Factor Humans exposed 40 hours/week (8 hours/day, 5 days/week) Study Details Chronic inhalation study (Nagano et al. , 2007 a) 49 Endpoint Chronic Dermal POD Fatty changes in the liver (rats) BMCL 10[HEC]: 14. 3 mg/m 3 for continuous exposures, which is equivalent to HEDDermal = 245 mg/kg-d Tumor Modeled dermal cancer slope factor (per mg/kg-bw/day) Pheochromocytomas (adrenal tumors in mice) (Nagano et al. , 2007 a) Dermal: 8 x 10 -4

Human Health Hazard – Strength of the Assessment • EPA considered adverse effects for CCl 4 across organ systems • CNS depression and neurotoxicity observed in multiple case reports and human experiments ₋ Human experiments → > 317 ppm for 30 minutes ₋ Chosen POD considers dose-response leading to more severe effects • Noncancer liver toxicity from chronic exposure ₋ Consistently seen in chronic and subchronic inhalation, oral animal studies • Adrenal tumors 50 ₋ Seen in mice (both sexes) in inhalation studies ₋ Tumor (e. g. , adrenal and liver) risks in the male mice could not be combined because the liver cancer IUR was too uncertain. ₋ Upper bound combination of the risks in female mice was still lower than just the adrenal tumors in male mice and thus would not have affected the bottom-line

Human Health Hazard – Assumptions, Uncertainties and Limitations Section 4. 4. 4 • Assumptions ₋ For the acute and chronic endpoints, EPA used a default approach - a modification to Haber’s law - to convert the POD to other exposure durations assuming the relationship of C 2. 5 x T (Ten Berge et al. , 1986) • 51 Uncertainties and Limitations ₋ Acute Inhalation POD – transient CNS effects (i. e. , dizziness) - is of limited severity § Considered appropriate given potential for impairment/more serious harm at increasing concentrations/exposure durations § Associated with increasing workplace accidents ₋ Acute Dermal POD – based on limited dermal data; acute inhalation POD endpoint is influenced by direct access to CNS tissue ₋ EPA derived dermal chronic HED and cancer slope factor by extrapolating from chronic inhalation PODs (i. e. , liver toxicity) and IUR

Human Health Assessment Exposures, Hazards, and Risk Characterization Human Health Risk Characterization Section 4. 2 and Charge Questions 5. 1 - 5. 8 and 6 52

Human Health Risk – Non-Cancer Approach Human Health (includes PESS) Occupational (includes adults of reproductive age) Occupational Users Inhalation Dermal ONUs • Benchmark MOE = Total Uncertainty Factors • Exposure reduction by PPE use was applied to occupational users • Risks were not aggregated across routes Inhalation Risks to occupational users and ONUs populations were independently estimated for acute and chronic exposures 53 *PESS: Potentially exposed or susceptible subpopulations Section 4. 2. 1

Section 4. 2. 1; Charge Questions 5. 2, 5. 5, 6. 1 Cancer Risk Approach - Benchmarks Cancer Risk = Human Exposure X Slope Factor Where: • Human exposures used were central tendency or high end (e. g. , 50 th or 95 th percentile) and route specific the inhalation LADC and dermal LADD. • For most COUs, occupational users are assumed to be exposed 8 hr/day, 250 day/yr for 31 or 40 years (central tendency or high-end). • Slope factors were exposure route specific for inhalation IUR and dermal slope factor. • Slope factors were calculated for occupational users i. e. , for an 8 hr/day, 250 day/yr. 1 x 10‐ 4 : 1 chance in 10, 000 of an individual developing cancer (equivalent to 100 cancer cases in 1 million) 54 LADC: lifetime average daily concentration LADD: lifetime average daily dose IUR: Inhalation Unit Risk

Section 4. 2; Table 4 -13 Charge Questions 5 and 6 Human Health Risk Summary Following Acute and Chronic Exposures Risk Estimates Indicating No Risk For Cancer and/or Non-cancer Effects: • Inhalation exposures to ONUs, no risk for non-cancer effects only • Inhalation exposures to occupational users without/with consideration of PPE • Dermal exposures to occupational users without/with consideration of PPE − All COUs Risk Estimates Indicating Cancer Risks: • Inhalation exposures to ONUs − Domestic manufacture − Processing as a reactant or intermediate in the production of HCFCs, HFOs, and 55 perchloroethylene − Processing for incorporation into formulation, mixtures or reaction products (other basic organic and inorganic chemical manufacturing) − Industrial/commercial use in the manufacture of other chemicals (including chlorinated compounds used in solvents, adhesives, asphalt, and paints and coatings)

Strengths of the Draft Risk Evaluation The current draft risk evaluation has been refined to include the: • Application of the TSCA systematic review process and incorporation of new information • Assessment of a wide range of conditions of use • Review, consideration, and evaluation of risks to environmental receptors (e. g. , aquatic organisms) • Review, consideration, and evaluation of the risks to humans from dermal exposures • Increased transparency with summary of risk estimates in risk characterization table for each condition of use 56

Section 4. 4 Charge Questions 5. 1 -5. 5, 5. 8 and 6 Human Health Risk – Assumptions and Key Sources of Uncertainties • The human populations considered include individuals of both sexes (> 16 and older, including pregnant females) for occupational settings • EPA extrapolated dermal chronic HEDs from inhalation PODs due to lack of: – dermal toxicity data – PBPK model with data to inform a dermal compartment • Overall risks may be underestimated by not aggregating across routes of exposure • For occupational scenarios, EPA has high or medium confidence in the exposure estimates for occupational users from monitoring data and modeling approaches • PPE for occupational users use assumes proper training, fitting and use during work activity 57

Sections 3. 2. 5. 4 and 4. 3 Charge Questions 5. 6 and 5. 7 Potentially Exposed or Susceptible Subpopulations (PESS) • EPA identified groups having greater exposure than the general population: − Occupational users and ONUs: Adults and youth (≥ 16 to 20 years old), including men and women of reproductive age • EPA identified groups having greater susceptibility than the general population: − Genetic polymorphisms in metabolizing enzymes − Nutritional status, age, pre-existing disease (particularly liver disease) − Co-exposure to other chemicals that induce p 450 enzymes (i. e. , alcohol) • Accounting for PESS in Risk Characterization: − Occupational Users: risk estimates for adult occupational users, including men and women of reproductive age; use of intraspecies variability UF of 10 58 Procedures for Chemical Risk Evaluation Under the Amended TSCA. 82 FR 33726 Frank R. Lautenberg Chemical Safety for the 21 st Century Act. 15 U. S. C 2602(12)
![Section 4 Risk Characterization Considerations – Human and Environmental [40 CFR § 702. 43(b)] Section 4 Risk Characterization Considerations – Human and Environmental [40 CFR § 702. 43(b)]](http://slidetodoc.com/presentation_image_h/0300e28ae1d272b8ec7c468b37ea5571/image-59.jpg)
Section 4 Risk Characterization Considerations – Human and Environmental [40 CFR § 702. 43(b)] When considering risk, EPA will: 1. Integrate the hazard and exposure assessments into quantitative and /or qualitative estimates of risk for the identified populations (including potentially exposed or susceptible subpopulations) 2. Describe whether aggregate or sentinel exposures under the conditions of use were considered and the basis for their consideration 3. Not consider costs or other non-risk factors 4. Take into account, where relevant, the likely duration, intensity, frequency, and number of exposures under the conditions of use of the chemical substance 5. Describe the weight of the scientific evidence for the identified hazards and exposures 59
- Slides: 59