Dr Mohamed Mahmoud Nour Eldein Ph D Biochemistry

Dr/ Mohamed Mahmoud Nour Eldein Ph. D Biochemistry Assistant Professor of Biochemistry Faculty of Medicine Umm AL-Qura University

Three Types of Spectrophotometric Methods • The spectrophotometer measures the absorbance of light of one of the components of a chemical reaction. • Three examples of common types of chemical reactions that are measured by the spectrophotometer are: 1 - endpoint colorimetric 2 - endpoint enzymatic 3 - kinetic reactions
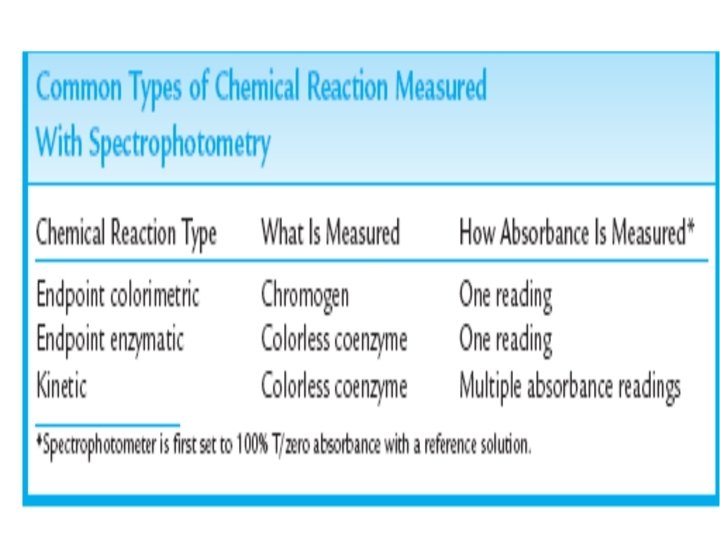

Endpoint Enzymatic Spectrophotometry • Some endpoint methods also use an enzyme to catalyze the chemical reaction. • The final product is often a coenzyme that absorbs light strongly at lower wavelengths in the visible or near-UV spectrum. • The hexokinase method for the measurement of glucose in body fluids is such a procedure; • The analyte in this complex reaction is glucose. That is, glucose is the substance that is to be measured. Glucose absorbs light at many wavelengths. • A test method aimed at directly measuring the absorbance of glucose would be subject to many types of interferences and would not be specific to glucose.

Endpoint Enzymatic Spectrophotometry • Therefore, chemical reactions such as the hexokinase method have been developed in which a chemical reaction involving glucose, the enzyme, and other substances is used to produce NADPH. • NADPH is the substance measured by the spectrophotometer because it absorbs light uniquely at 340 nm and is not subject to many types of interferences. • The concentration of glucose is proportional to the consumption of the coenzyme in the reaction.

Dr/ Mohamed Mahmoud Nour Eldein Ph. D Biochemistry Assistant Professor of Biochemistry Faculty of Medicine Umm AL-Qura University

objectives • To understand the importance of measuring blood glucose level. • To understand the principles of enzymatic estimation of glucose.

Introduction • Estimation of glucose in blood was one of the first biochemical tests to be applied clinically and now it has become a routine in clinical biochemistry lab. • Glucose is formed from carbohydrate digestion • it is primary source of energy for human • The nervous system totally depend on glucose, so it is critical to maintain a steady supply of glucose to tissue • when the concentration Fall, the nervous system incapable to maintain normal function • hormones play big rule in glucose level regulation. ( glucagon, insulin, adrenocorticotropic hormone “ ACTH”, epinephrine, thyroxin)

Sample used 1 - Whole blood 2 - plasma 3 - serum 4 - CSF 5 - urine Whole blood values are 10 -15% lower than plasma. Arterial blood values are higher than venous values. a) Fasting blood Sugar (FBS) : The blood sample is collected after the patient fasts for 6 hours. b) Post-Prandial Blood Sugar (PPBS) : After the patient fasts for 6 hours, a meal is given which contains starch and sugar (approx. 100 gms). Blood is collected 2 hours after the ingestion of the meal. c) Random Sample : Blood is collected any time without prior preparation of the patient.

Collection of Blood Sample • Blood is usually collected from a vein and kept in a tube containing sodium fluoride (Na F) and potassium oxalate mixed at proportion of 1 : 3. • Both the substances act as anticoagulant and Na F prevents glycolysis in RBC's by inhibiting the enzyme 'enolase'. • Glycolysis decrease serum glucose by approximately 5% to 7% per hour ( 5 -10 mg/ dl) in normal uncentrifuged blood at room temperature

Methods of Estimation : 1. Enzymatic : Measure only glucose in blood. a) Glucose Oxidase Method : b) Hexokinase Method Hexokinase glucose + ATP glu-6 -P+ADP G – 6 – P + NAD 6 phosphogluconate + NADH + H+ The rate of formation of NADH is followed spectrophotometrically at 340 mn. 2 - Reduction Methods 1) Alkaline Copper Reduction Methods. • Asatoor & King's method • Folin & Wu method 2) Ortho toludine method : O-Toludine reacts quantitatively with the aldehyde group of glucose to from a glcosylamine and schiff base. The colour development is rapid and stable.

Estimation of Blood Glucose by glucose oxidase method Principle : Glucose oxidase catalyses the oxidation of glucose to gluconic acid and hydrogen peroxide. This H 2 O 2 is broken down to water and oxygen by a peroxidase in the presence of an oxygen acceptor which itself is converted to a coloured compound, the amount of which can be measured colorimetrically. This method is used in various autoanalyzers. Glucose Oxidase Glucose + H 2 O 2 + O 2 Gluconic Acid+H 2 O 2 Peroxidase H 2 O 2 + 4 - aminophenazone + phenol quinonemine + H 2 O O 2 acceptor (Measured) The intensity of the color formed is proportional to glucose concentration in the sample.
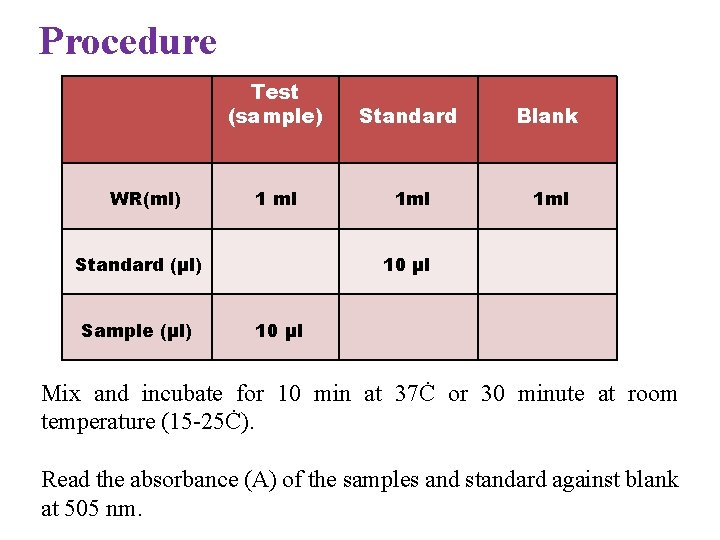
Procedure WR(ml) Test (sample) Standard Blank 1 ml 1 ml Standard (µl) Sample (µl) 10 µl Mix and incubate for 10 min at 37Ċ or 30 minute at room temperature (15 -25Ċ). Read the absorbance (A) of the samples and standard against blank at 505 nm.

Calculation (A) Sample (A) Standard × 100 ( Standard conc. ) = mg/dl glucose in the sample Conversion Factor mg/dl × 0. 0555 = mmol/l Reference Value (Normal Range) The normal fasting blood glucose varies from 70 -110 mg/dl and post prandial from 110 -140 mg/dl. The appear limit increase with age and during pregnancy.

Interpretation 0 f Results Hyperglycemia : Causes are : 1. Diabetes mellitus – Fasting blood sugar raised. Values of 140 mg/dl on more than one occasion or post prandial level of 200 mg/dl confirms the diagnosis. 2. Hyperactivity of thyroid, pituitary, or adrenal gland (Cushing's disease). 3. Surgical removal of pancrease, pancreatitis, carcinoma of pancrease, fibrocystic disease. 4. Intracranial diseases like meningitis, encephalitis, tumors and hemorrhage show a moderate hyperglycemia. 5. Drug induced eg : thiazide diuretics, steroids, ACTH, thyroid extracts.

Diabetes Mellitus Diabetes mellitus is a chronic disease, the commonest cause of hyperglycemia and its early detection is of vital importance. Clinical Biochemical finding in diabetes 1) Presence of large amount of glucose in urine. 2)Large volume of urine & increased frequency (Polyuria) 3) Polyphagia i. e. eats more frequently. 4) Increased catabolism of fat so there is increase in free fatty acid level in blood & liver. 5) Increased acetyl co. A is seen which further lead to increase formation of cholesterol & hence at formation of atherosclerosis. 6) Increased ketone bodies in blood & its appearance in urine leads to acidosis. 7) Increased catabolism of tissue protein for energy requirement lead to loss of weight & increased level of amino acid in blood & more formation of urea by deamination of amino acid.

Diabetes Mellitus (a) Juvenile Diabetes or. Type I Diabetes or Insulin dependent Diabetes Mellitus (IDDM) Less Frequent Occurs before the age of 15 years. Due to less production of insulin from β cells of langerhans (b) Maturity onset diabetes or. Type II diabetes or Non-insulin dependent Diabetes mellitus (NIDDM) More frequent in population. Occurs at middle age. associated with obesity. due to insulin resistance

Hypoglycemia When blood glucose falls below 60 mg/dl. 1. Most commonly seen due to overdosage of insulin in treatment of diabetes mellitus. 2. Hypothyroidism – cretinism, myxedema. 3. Insulin secreting tumours of pancreas – rare. 4. Hypogonadism (Addison's disease) 5. Hypopituitrism. 6. Severe exercise. 7. Starvation. 8. Chronic alcoholism 9. Congenital disease like – glycogen storage disorders
- Slides: 18