Dr Ketki K MBBS MD Assistant Professor Department

Dr. Ketki K, MBBS, MD Assistant Professor Department of Biochemistry

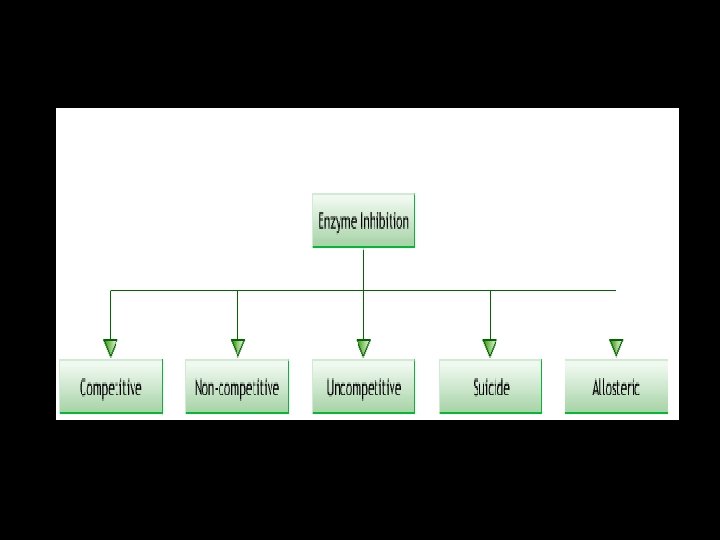
Enzyme Inhibitions

Effect of enzyme inhibition Inhibitors • cause a loss of catalytic activity • Change the protein structure of an enzyme • May be competitive or noncompetitive

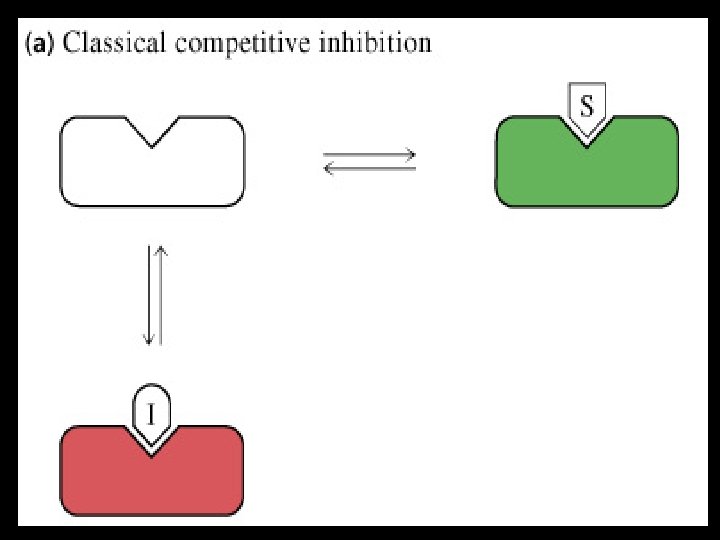
competitive inhibition Inhibitors • Has a structure similar to substrate • Occupies active site • Competes with substrate for active site • Effect is reversed by increasing substrate concentration • Reduces velocity
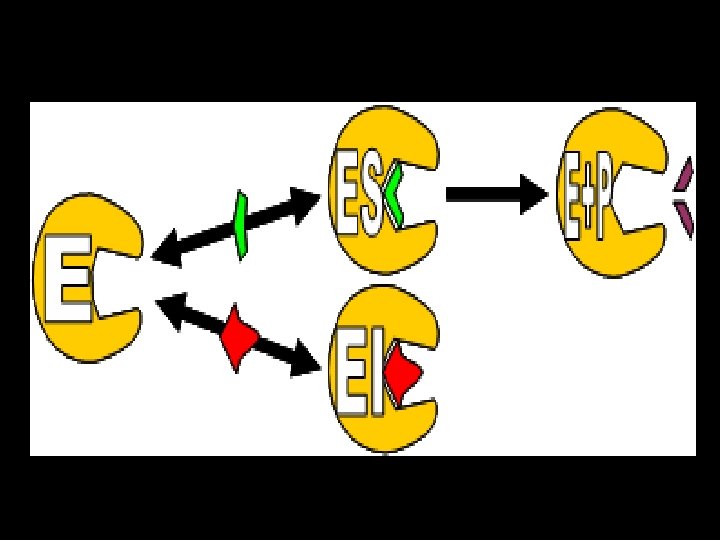
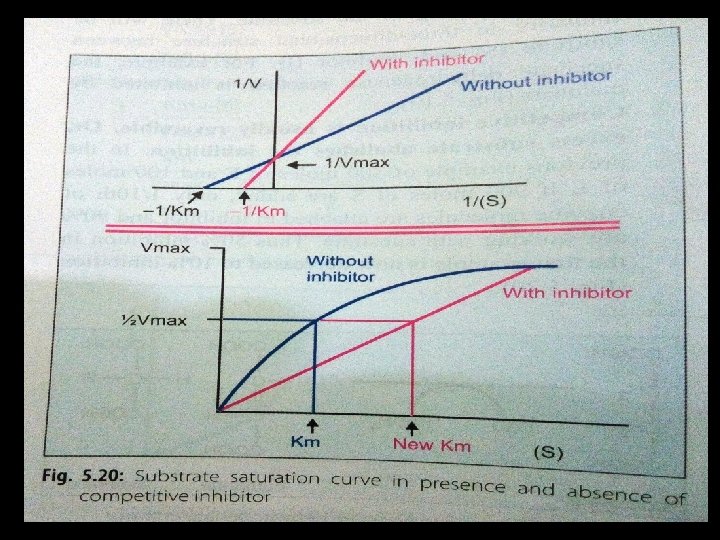

• Increased Km since affinity of enzyme for substrate is decreased in presence of competitive inhibitors. In other words, in the presence of competitive inhibitor, more substrate is needed to achieve ½ vmax • Vmax is not changed.

Effect of CI on the Lineweaver-Burk Double reciprocal plot: Ø Plot of inhibited and uninhibited reactions intersect on the y axis at 1/ Vmax (Vmax IS UNCHANGED) Ø inhibited and uninhibited reactions show different x axis intercepts Ø Indicates that Km is increased in presence of competitive inhibitor because -1/Km moves closer to zero from a negative value


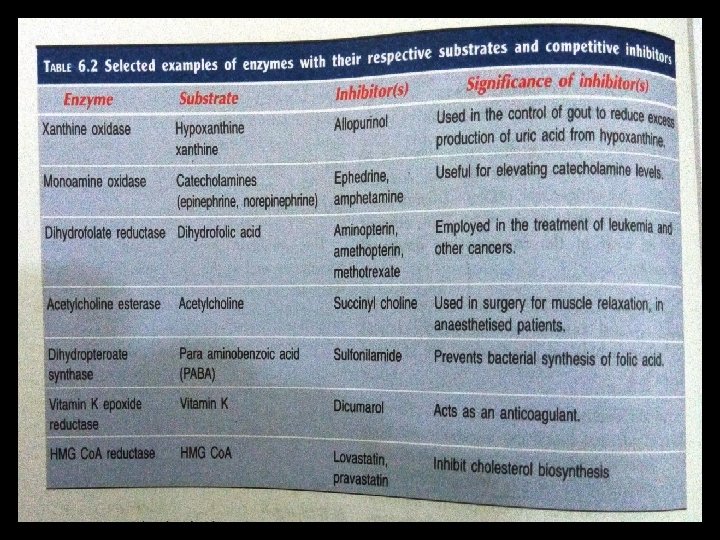

Non competitive inhibition Inhibitors Not similar to substrate/ Not a structural analogue Does not bind to active site Changes the shape of enzyme and active site Substrate binds but no catalysis occurs in the presence of inhibitor • Usually irreversible (covalently) • Effect is not reversed by adding substrate • Variety of poisons, like iodoacetate, heavy metals(lead, mercury) acts as irreversible non competitive inhibitor • •
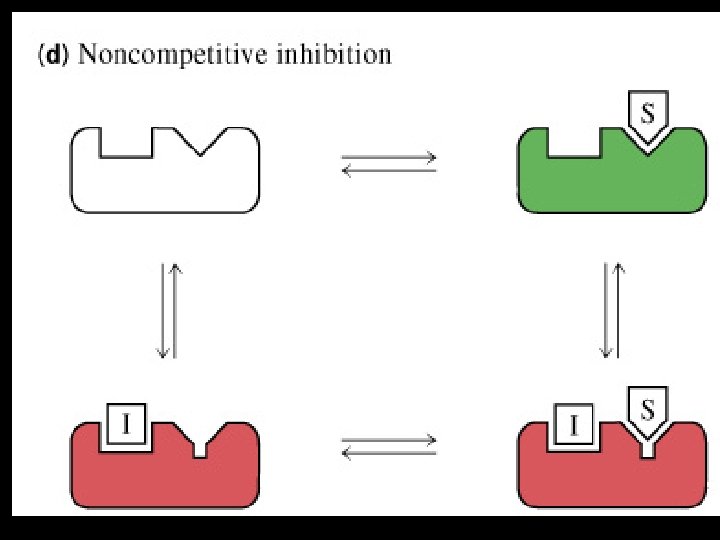
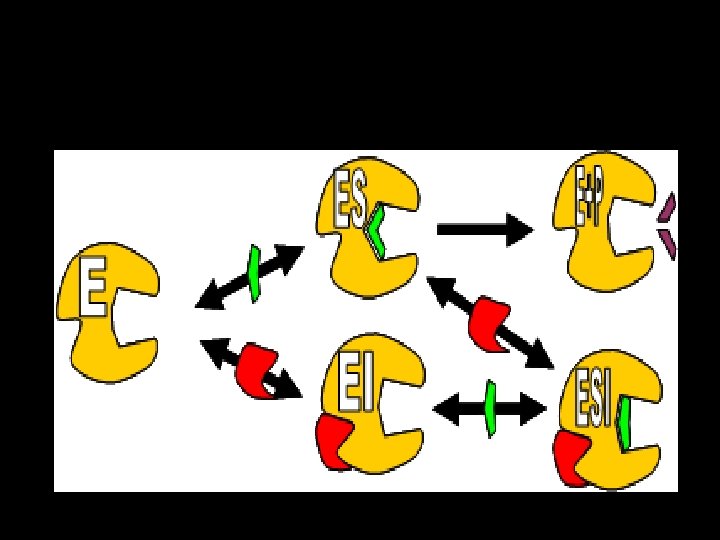

• Does not alter the km • V max reduced

Examples of irreversible Noncompetitive inhibitors • Cyanide inhibits cytochrome oxidase • Fluroide inhibits enolase • Iodoacetate inhibits enzymes having –SH group at active site. eg Papain, G 3 PDH • BAL, Dimercaprol used as antidote for heavy metal poisoning

• DFP(diisopropyl fluorophosphates) inhibits enzymes containing serine at the active site. eg: serine proteases, acetylcholine esterase • Malathion(insecticide): inhibits enzyme acetylcholine esterase

• Disulfiram (Antabuse) : drug used in treatment of chronic alcoholism • It irreversibly inhibits the enzyme aldehyde dehydrogenase, acetaldehyde accumulates, toxic effects Ethanol ADH Acetaldehyde ADH Acetic acid Disulfiram inhibits

Reversible Non competitive • Trypsin inhibitor: ascariasis • Alpha 1 antitrypsin


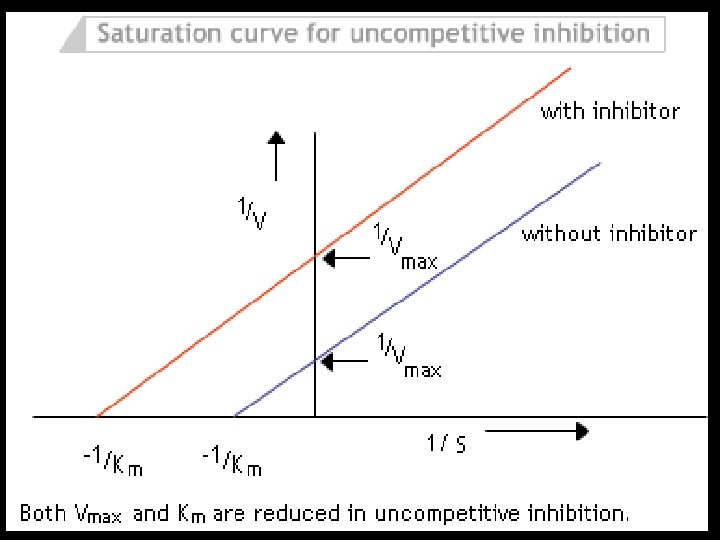
Uncompetitive Inhibitors • Inhibitor does not have any affinity for free enzymes • Inhibitor(I) binds to enzyme-substrate(ES) complex to form ESI, but not to free enzyme • ESI complex enhance the affinity of enzyme towards substrate, this decreases Km, so decreased product formation hence decrease in Vmax • Example: Inhibition of Placental alkaline phosphatase by Phenylalanine
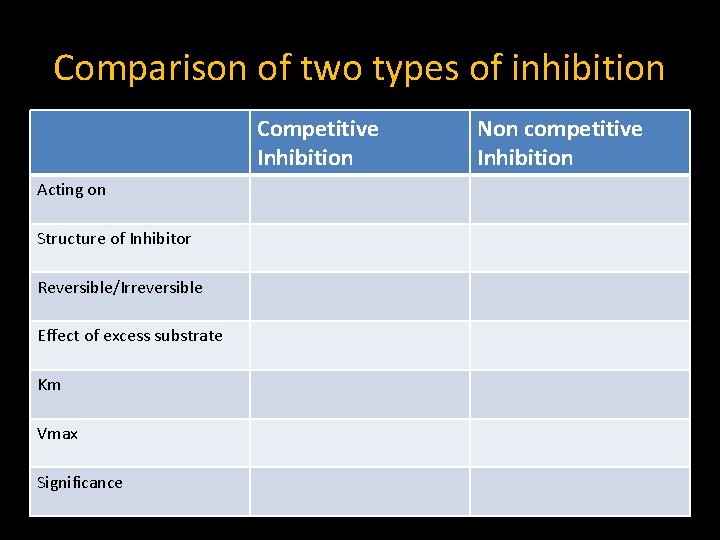
Comparison of two types of inhibition Competitive Inhibition Acting on Structure of Inhibitor Reversible/Irreversible Effect of excess substrate Km Vmax Significance Non competitive Inhibition

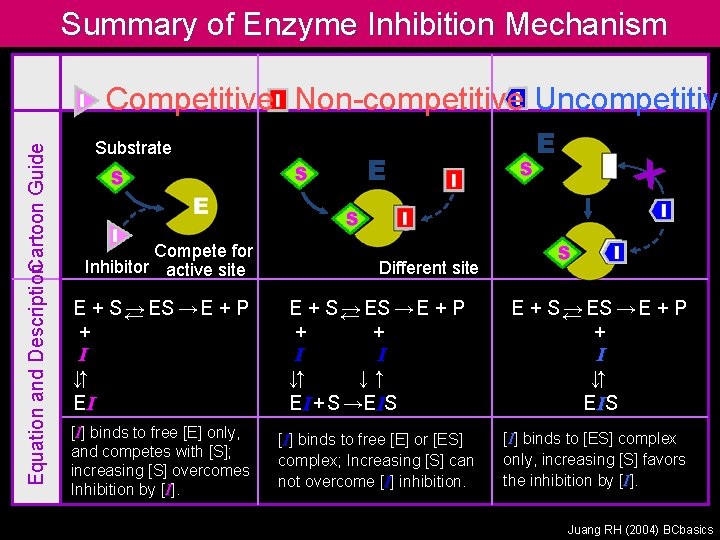
Summary of Enzyme Inhibition Mechanism Equation and Description. Cartoon Guide Competitive Non-competitive Uncompetitive Substrate Compete for Inhibitor active site E E X Different site E + S← → ES → E + P + I ↓↑ EI E + S← → ES → E + P + + I I ↓↑ ↓↑ EI + S →EIS E + S← → ES → E + P + I ↓↑ E IS [I] binds to free [E] only, and competes with [S]; increasing [S] overcomes Inhibition by [I] binds to free [E] or [ES] complex; Increasing [S] can not overcome [I] inhibition. [I] binds to [ES] complex only, increasing [S] favors the inhibition by [I]. Juang RH (2004) BCbasics

Suicide Inhibition • Special type of irreversible inhibition • Also k/a mechanism based inactivation. (inhibitor makes use of enzyme’s own reaction mechanism to inactivate it) • Original inhibitor(Structural analogue) is converted to more effective inhibitor with the help of enzyme to be inhibited

• So formed inhibitor(new product) binds irreversibly with the enzyme, In contrast to the original inhibitor which binds reversibly Example: 1) Allopurinol inhibits xanthine oxidase Hypoxanthine XO Xanthine XO Uric acid Allopurinol, structural analogue of Hypoxanthine, Allopurinol XO Alloxanthine, more effective inhibitor of XO

2) Aspirin as anti-inflammatory action Arachidonic acid Cyclo-oxygenase Prostaglandin Aspirin acetylates serine residues in the active center of cyclo-oxygenase, leading to decreased PG synthesis, reduced inflammation.

3) Purine and pyrimidine analogues in cancer therapy Analogue, 5 fluorouracil Thymidylate synthase Fluorodeoxy uridylate (5 fd. UMP) Fluorodeoxy uridylate inhibits the enzyme thymidylate synthase and thus inhibits nucleotide synthesis (d. UMP to d. TMP)

• Some of Mahesh's problems have arisen from product inhibition of liver alcohol dehydrogenase by NADH. As ethanol is oxidized in liver cells, NAD+ is reduced to NADH and the NADH/NAD+ ratio rises. • NADH is an inhibitor of alcohol dehydrogenase, competitive with respect to NAD+, so the increased NADH/NAD+ ratio slows the rate of ethanol oxidation and ethanol clearance from the blood. • NADH is also a product inhibitor of enzymes in the pathway that oxidizes fatty acids. Consequently, these fatty acids accumulate in the liver, eventually contributing to the alcoholic fatty liver.

• A year after recovering from salicylate poisoning, Rahul was playing in his grandfather's basement. Rahul drank an unknown amount of the insecticide malathion, which is sometimes used for killing fruit flies and other insects. Sometime later, when he was not feeling well, Rahul told his grandfather what he had done. His grandfather retrieved the bottle and rushed Rahul to the emergency room of the local hospital. On the way, Rahul vomited repeatedly and complained of abdominal cramps. At the hospital, he began salivating and had an uncontrollable defecation. • In the emergency room, physicians passed a nasogastric tube for stomach lavage, started intravenous fluids, and recorded vital signs. Rahul’s pulse rate was 48 beats/minute (slow), and his blood pressure was 78/48 mm Hg (low). The physicians noted involuntary twitching of the muscles in his extremities.

• Rahul survived his malathion intoxication because he had ingested only a small amount of the chemical, vomited shortly after the agent was ingested, and was treated rapidly in the emergency room. • Lethal doses of oral malathion are estimated at 1 g/kg of body weight for humans. Emergency room physicians used a drug (oxime) to reactivate the acetylcholinesterase in Rahul before the aged complex formed. • They also used intravenous atropine, an anticholinergic (antimuscarinic) agent, to antagonize the action of the excessive amounts of acetylcholine accumulating in cholinergic receptors throughout his body.

• After several days of intravenous therapy, the signs and symptoms of acetylcholine excess abated, and therapy was slowly withdrawn. Rahul made an uneventful recovery. • The symptoms experienced by Rahul resulted from inhibition of acetylcholinesterase. Acetylcholinesterase cleaves the neurotransmitter acetylcholine to acetate and choline in the postsynaptic terminal, thereby terminating the transmission of the neural signal. • Once it has been ingested, the liver converts malathion to the toxic reactive compound, malaoxon, by replacing the sulfur with an oxygen. Malaoxon then binds to the active site of acetylcholinesterase and reacts to form the covalent intermediate.

• This initial acylenzyme intermediate is reversible. However, with time, the enzyme-inhibitor complex “ages” (dealkylation of the inhibitor and enzyme modification) to form an irreversible complex • As a result, acetylcholine accumulates and overstimulates the autonomic nervous system (the involuntary nervous system, including the heart, blood vessels, and glands), thereby accounting for Rahul's vomiting, abdominal cramps, salivation, and sweating. • Acetylcholine is also a neurotransmitter for the somatic motor nervous system, where its accumulation resulted in Rahul's involuntary muscle twitching (muscle fasciculations).

• Tania was diagnosed with acute gouty arthritis involving her right great toe. The presence of insoluble urate crystals within the joint space confirmed the diagnosis. Several weeks after her acute gout attack subsided, Ms. Tania was started on allopurinol therapy in an oral dose of 150 mg twice per day

• Ms Tania, within several days of starting allopurinol therapy, her serum uric acid level began to decrease. Several weeks later, the level in her blood was normal. However, while Tania was adapting to allopurinol therapy, she experienced a mild gout attack, which was treated with a low dose of colchicine. • Tania is being treated with allopurinol for gout, which is caused by an accumulation of sodium urate crystals in joints and joint fluid, particularly in the ankle and great toe.

• Allopurinol is a suicide inhibitor of the enzyme xanthine oxidase, which is involved in the degradation of purine nucleotides AMP and GMP to uric acid (urate). • Although hypoxanthine levels increase in the presence of allopurinol, hypoxanthine does not participate in urate crystal formation and precipitation at this concentration. It is excreted in the urine.

• • "Educationisisthe thebest friend. An Aneducatedpersonisis respectedeverywhere. Educationbeatsthe thebeauty and andthe theyouth. " Chanakya quotes (Indian politician, strategist and writer, 350 BC-275 BC)

THANK YOU
- Slides: 42