Dr J Domenech CALCULATIONS AND PHARMACOKINETICS Objectives To

Dr. J. Domenech CALCULATIONS AND PHARMACOKINETICS

Objectives • To emphasize the importance of calculations • To review important calculation concepts related to – – – – Patient Parameters Measurements Concentrations Electrolyte Solutions Clinical Laboratory Tests IV infusions flow rates Parenteral Nutrition • To practice using the reviewed calculation concepts • To review important pharmacokinetic concepts 1

WHY SHOULD YOU CARE? • You can’t escape it • You need it to pass the NAPLEX • It can be applied to any pharmacy-related specialty • It may save your patient’s money • It may save your patient’s life (and your license) 2

Important Calculation Concepts PATIENT PARAMETERS 3

BSA and IBW • Body Surface Area (BSA), m 2 – BSA = Weight (kg)0. 425 x Height (cm)0. 725 x 0. 007184 – BSA = • Ideal Body Weight, kg – Males: 50 +( 2. 3 x inches greater 60) – Females: 45. 5 + (2. 3 x inches greater than 60) 4

BODY MASS INDEX (BMI) • Measure of body fat to help categorize patients and assess risk of morbidity for certain diseases • BMI = weight(kg) ÷ [height (m)2] BMI Classification < 18. 5 Underweight 18. 5 to <25 Normal 25 to <30 Overweight >30 Obese 5

Creatinine Clearance (Cr. Cl) • Formula used to estimate renal function • Not useful when renal function is fluctuating rapidly • Used in the dosing of several medications – Tamiflu, Antibiotics, Low-molecular weight heparins Multiply by 0. 85 if female Cr. Cl (m. L/min) Interpretation 60 to 90 Mild renal impairment 30 to < 60 Moderate Renal impairment 15 to <30 Severe Renal impairment < 15 Kidney failure 6

EXAMPLE Mack “Big Mack” Donald is a 55 year old man recently admitted to your service after choking on a Zebra Cake. He is 6’ 2” and 980 pounds. Calculate his BSA and BMI. 7

EXAMPLE Body Surface Area • Height in centimeters: 6’ 2” = 74 inches – 74 inches x 2. 54 cm/inch = 187. 96 cm • Weight in kilograms: 980 lbs – 980 lbs x 1 kg/2. 2 lbs = 445. 45 kg • Answer: 4. 82 Body Mass Index • Height in meters = 1. 88 m • Answer: 126 • How do we classify Big Mack’s BMI? – – Underweight Normal Overweight Obese 8

EXAMPLE Mack had a DVT during admission. The doctors wish to start him on Enoxaparin (Lovenox), a renally dosed medication. His SCr is 2. 3. Calculate his IBW and Creatinine Clearance 9

EXAMPLE Ideal Body Weight • Inches over 60 – 74 – 60 = 14 inches • IBW = 50 kg + 2. 3 (14) • Answer: 82. 2 kg Creatinine Clearance • Use IBW of 82. 2 kg • Answer: 42. 19 m. L/min – Dose of Lovenox is 50% if Cr. Cl is less than 30 m. L/min 10

Important Calculation Concepts MEASUREMENTS 11

The Basics Of Measurement • Irrelevant information is often given • Fundamental information is often left out – How many grams are in a kilogram? – How many milliliters are in a fluid ounce? • Start each problem by assessing what you know and what you want to know • Double check! 12

EXAMPLE A cough syrup contains 10 mg of dextromethorphan per 5 m. L. Your pharmacy has 6 bottles left in stock. Each bottle contains 120 m. L of the syrup. How many grams of the drug are in one bottle? • What information is irrelevant? • What fundamental information do you need to know to complete this problem? • What do you know? • What do you want to know? 13

EXAMPLE A cough syrup contains 10 mg of dextromethorphan per 5 m. L. Your pharmacy has 6 bottles left in stock. Each bottle contains 120 m. L of the syrup. How many grams of the drug are in one bottle? 10 mg/5 m. L 120 m. L/ 1 bottle 1 gram/1000 mg 0. 24 grams/ 1 bottle 14

Specific Gravity Definition • A ratio of the weight of any substance in relation to the weight of an equal volume of water • Water is used because 1 g of water is equal to 1 m. L Equations • Grams = m. L x SG • m. L = Grams ÷ SG • SG = Grams ÷ m. L – Do not confuse with density or concentration – If SG > 1 the substance is heavier than water – If SG < 1 the substance is lighter 15

EXAMPLE There is 10 grams of glycerin in a 500 m. L solution. 300 m. L of glycerin weighs 165 grams. What is the specific gravity of glycerin? • Specific Gravity = grams / m. L • 10 g / 500 m. L ? • 165 g/ 300 m. L ? • Think of SG as a conversion factor • 10 grams of glycerin does not EQUAL 500 m. L of glycerin • 165 grams of Glycerin EQUALS 300 m. L of glycerin 16

EXAMPLE There is 10 grams of glycerin in a 500 m. L solution. 300 m. L of glycerin weighs 165 grams. What is the specific gravity of glycerin? 165 grams 300 m. L 0. 55 17

EXAMPLE • What is the weight, in grams of a 2 fluid ounces of a liquid with a specific gravity of Grams = m. L x SG 1. 118? 2 fl oz 30 m. L/ 1 fl oz 1. 118 m. L SG 67. 08 grams 18

DOUBLE CHECK • DOES THE ANSWER MAKE SENSE? • If the SG is 1. 118, the substance is – Heavier than water? – Lighter than water? • 2 fluid ounces of water (60 m. L) = 60 grams • The answer is 67. 08 grams which is > 60 grams • Any answer below 60 would be WRONG 19

Important Calculation Concepts CONCENTRATIONS 20

PERCENTAGE PREPARATIONS Weight in volume • Amount of grams in 100 m. L of solution Volume in volume • Amount of m. L in 100 m. L of solution Weight in weight • Amount of grams in 100 grams of substance 21

EXAMPLE Weight in Volume How many grams of dextrose are required to prepare 4000 m. L of a 5% solution? • 5% Dextrose by definition means 5 grams in 100 m. L 4000 m. L 5 grams/ 100 m. L 200 grams 22

ALLIGATION • Alligation is a method of solving problems that involves the mixing of solutions or substances with different percentage strengths • Alligation alternate is a method to calculate the number of parts of two or 23

EXAMPLE ALLIGATION What is the percentage of zinc oxide in an ointment prepared by mixing 200 grams of a 10% ointment, 50 grams of a 20% ointment, and 100 grams of a 5% ointment 0. 10 x 200 g = 20 grams 0. 20 x 50 g = 10 grams 0. 05 x 100 g 350 g = 5 grams 35 grams ÷ 350 grams = 0. 10 x 100% = 10% 24

EXAMPLE ALLIGATION ALTERNATE A pharmacist needs to prepare 50 m. L of 3% hydrogen peroxide solution. He has 30% and 1. 5% solutions in stock. How many m. L of each should he use? • A–C=Y Percent Available Percent Desired A Proportions required X C B Y • C–B=X • X and Y are proportions of A and B (respectively) needed for the entire preparation 25

EXAMPLE ALLIGATION ALTERNATE A pharmacist needs to prepare 50 m. L of 3% hydrogen peroxide solution. He has 30% and 1. 5% solutions in stock. How many m. L of each should he use? Percent Available Percent Desired 30% Proportions required 1. 5 3% 1. 5% 27 • 1. 5 parts of 30% • 27 parts of 1. 5% • Total Parts: 28. 5 26

EXAMPLE ALLIGATION ALTERNATE A pharmacist needs to prepare 50 m. L of 3% hydrogen peroxide solution. He has 30% and 1. 5% solutions in stock. How many m. L of each should he use? • We need 1. 5 of all 28. 5 parts to contain 30% Hydrogen peroxide • 1. 5/28. 5 = x/50 m. L • X = 2. 63 m. L of 30% • We need 27 parts of all 28. 5 parts to contain 3% Hydrogen peroxide • 27/28. 5 = x/50 m. L • X = 47. 37 m. L of 1. 5% 27

Important Calculation Concepts ELECTROLYTE SOLUTIONS 28

MILLIEQUIVALENTS • Unit of measure related to the total number of ionic charges in a solution – Measures the chemical activity of an electrolyte relative to 1 mg of hydrogen • 1 m. Eq represents the milligrams equal to its equivalent weight, taking into account the valency 29

MILLIEQUIVALENTS Ion Abbr. Valence Atomic Weight Milliequivalent weight Aluminum Al 3+ 3 27 9 Magnesium Mg 2+ 2 24 12 Sodium Na+ 1 23 23 Bicarbonate HCO 32 - 2 60 ? ? ? 30 30

EXAMPLE What is the concentration, in milligrams per milliliter, of a solution containing 2 m. Eq of Na. Cl per m. L? • • Molecular weight of Sodium = 23 Molecular weight of Chloride = 35. 5 Molecular weight of Sodium chloride = 58. 5 Valency = 1 (Na+, Cl+) 117 mg 31

MOLARITY How many milligrams would 3 mmol of monobasic sodium phosphate (MW = 138) weigh? • Millimoles – 1 mol = molecular weight in grams, therefore… – mmol = molecular weight in milligrams • Molarity is the number of mmols in a solution 3 mmol 138 mg/ 1 mmol 414 mg 32

OSMOLARITY • m. Osmoles represent the number of particles in a solution when the substance dissociates – Na. Cl = 2 m. Osmol – Ca. Cl 2 = 3 m. Osmol – Anhydrous dextrose = 1 m. Osmol • Osmolality is the milliosmoles of solute in a solution 33

Important Calculation Concepts CLINICAL LABORATORY TESTS 34

CALCIUM-ALBUMIN • Almost 50% of calcium is bound to plasma proteins • If levels of proteins are low, then the serum calcium may be inaccurate • If albumin is low, calcium will appear to be low, when it levels are actually within normal limits – Correct levels when Albumin is less than 4 g/d. L • Corrected Ca 2+ = Ca 2+ + 0. 8 [4 – 35

SODIUM-GLUCOSE • In patients with hyperglycemia, glucose does not enter the cell, causing a shift of fluid from intracellular to extracellular • The shift of fluid dilutes the concentration of sodium in the extracellular fluid • This type of hyponatremia (translational) does not need to be treated, instead control of glucose levels is indicated • Corrected Na+ = Na+ + 0. 016(Serum Glucose – 100) 36

Important Calculation Concepts INFUSION FLOW RATES 37
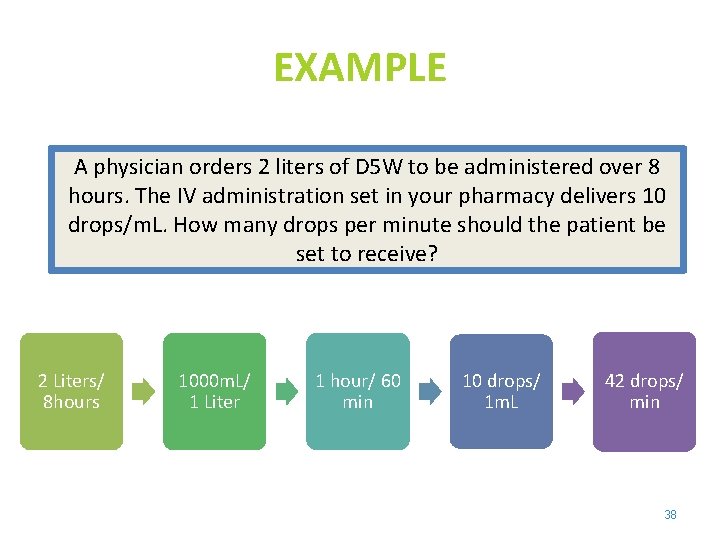
EXAMPLE A physician orders 2 liters of D 5 W to be administered over 8 hours. The IV administration set in your pharmacy delivers 10 drops/m. L. How many drops per minute should the patient be set to receive? 2 Liters/ 8 hours 1000 m. L/ 1 Liter 1 hour/ 60 min 10 drops/ 1 m. L 42 drops/ min 38

Important Calculation Concepts TOTAL PARENTERAL NUTRITION 39

TOTAL PARENTERAL NUTRUTION • Provides a patient with all nutritional requirements • Composition: – Fluids – Carbohydrates (Dextrose) – Protein (Amino acids) – Fats – Vitamins, minerals, trace elements – Electrolytes 40

TPN HIGHLIGHTS • Fluid Requirements: – Generally 30 – 40 m. L/kg/day – Calculated: 1500 m. L + 20 m. L (kg over 20) • Protein Requirements – Ambulatory: 0. 8 – 1 g/kg/day – Hospitalized: 1. 2 – 2 g/kg/day • Non-Protein Requirements (Total Energy Expenditure) – Male: 66. 47 + 13. 75(kg) + 5. 0(cm) – 6. 76 (yrs) – Female: 655. 1 + 9. 6(kg) + 1. 85(cm) – 4. 68(yrs) – Multiply by 1. 2 if confined to a bed, and 1. 3 if out of bed 41

TPN HIGHLIGHTS • Each TPN component provides a defined amount of calories • COMMIT THESE TO MEMORY!! Component Units Dextrose 3. 4 kcal per gram Amino Acids 4 kcal per gram Lipids 9 kcal per gram 10% lipid emulsion 1. 1 kcal per m. L 20% lipid emulsion 2 kcal per m. L 42

PHARMACOKINETICS Important Calculation Concepts 43

PHARMACOKINETCS • Pharmacokinetics describes what happens to a drug or substance inside of the body Absorption Distribution Metabolism Excretion 44

FIRST-ORDER KINETICS • The amount of drug given is proportional to the change in concentration – The change in drug concentration with respect to time will create a rate constant (k) • C = C 0 e-kt 45

HALF-LIFE • The half-life (t 1/2) is the time required for the concentration of the drug to decrease by onehalf – t 1/2 = 0. 693/k (k is the rate constant) A patient is receiving an antibiotic for the treatment of a respiratory infection. The initial concentration of the drug was 17. 9 mg/L. The drug has an elimination half-life of 2 hours. How much of the drug is present after 8 hours? 46

EXAMPLE A patient is receiving an antibiotic for the treatment of a respiratory infection. The initial concentration of the drug was 17. 9 mg/L. The drug has an elimination half-life of 2 hours. How much of the drug is present after 8 hours? • C = C 0 e-kt SOLVE FOR “C” • t 1/2 = 0. 693/k (k is the rate constant) – 2 h = 0. 693/k – k = 0. 347 h-1 • C = 17. 90 e-0. 347(8) = 1. 11 mg/L 47

QUESTIONS? 48

THANK YOU! • Joy A. Awoniyi • Email your questions, comments or concerns to Joy. awoniyi@va. gov 49
- Slides: 50