Dr Grard Moulin Dr Catherine Lambert OIE AFSSAANMV

Dr. Gérard Moulin Dr Catherine Lambert OIE, AFSSA/ANMV Collaborating Centre on Veterinary medicinal products BP 90203 - 35302 FOUGERES CEDEX, FRANCE g. moulin@anmv. afssa. fr International approach for veterinary medicinal products: OIE and Codex alimentarius 1

International approach for veterinary medicinal products: OIE and Codex alimentarius Introduction OIE activities related to VMP Codex activities Cooperation between Codex and OIE · Residues in food from animal origin · Antimicrobial resistance Conclusion 2

International approach for veterinary medicinal products: OIE and Codex alimentarius Benefits Animal health and welfare Human nutrition Economic development Risks Risk for animals Risks for humans Risks for the environment 1924 OIE, 1945 FAO, 1948 WHO 3

WTO Agreement on the Application of Sanitary and Phytosanitary Measures (SPS Agreement) · The SPS Agreement calls on countries to harmonize their national standards with “international standards, guidelines or recommendations ”(Article 3. 1). · “international standards, guidelines or recommendations” (Annex A). § Codex Standards for food safety § OIE for animal health and zoonoses § IPPC for plant health Agreement on Technical Barriers to Trade (TBT Agreement) · The TBT Agreement calls on countries to use relevant international standards when they exist (Article 2. 4). 4

OIE's WTO mandate (cont. ) Standards-setting organizations food safety CODEX animal health OIE plant health IPPC Codex = Joint FAO/WHO Codex Alimentarius Commission OIE = World Organization for Animal Health IPPC = International Plant Protection Convention (FAO) 5

• an intergovernmental organisation • founded in 1924 by 28 countries 172 Member Countries • created before the U. N. World Organisation for Animal Health Organisation mondiale de la santé animale Common name adopted by the International Committee on May 2003 6

The 6 Objectives of the OIE ANIMAL HEALTH INFORMATION: to ensure transparency in the global animal disease and zoonosis situation to collect, analyse and disseminate scientific veterinary information ACTIVITIES OF VETERINARY SERVICES to provide expertise and encourage international solidarity in the control of animal diseases to improve the legal framework and resources of national Veterinary Services INTERNATIONAL STANDARD within its WTO mandate, to safeguard world trade by publishing health standards for international trade in animals and animal products to provide a better guarantee of the safety of food of animal origin, and to promote animal welfare, through a science-based 7

INTERNATIONAL COMMITTEE Administrative Commission Director General Specialist Commissions Code, Laboratories, Aquatic animals, Scientific Human Resources and Budget Management Unit Accounts Unit World Animal Health and Welfare Fund Coordination Collaborating Centres Reference Laboratories Regional Commissions Africa, Americas, Europe, Asia- Far East and Oceania, Middle East Central Bureau Deputy Director General Administration, Finances, Human Resources Deputy Director General Animal Health and International Standards Administrative and Management Systems Department International Trade Department Regional Activities Department Scientific and Technical Department Ad hoc Groups Working Groups Publications Department Animal Health Information Department Regional Representations 8

Specialist Commissions (cont. ) Terrestrial Animal Health Standards Commission "Code Commission" Scientific Commission for Animal Diseases "Scientific Commission" Biological Standards Commission "Laboratories Commission" Aquatic Animal Health Standards Commission "Aquatic Animals Commission" 9

OIE International Standards Terrestrial Animal Health Code – mammals, birds and bees Aquatic Animal Health Code – fish, molluscs and crustaceans Manual of Diagnostic Tests and Vaccines for Terrestrial Animals Manual of Diagnostic Tests for Aquatic Animals 10

Working Groups Wildlife Diseases Animal Production Food Safety Animal Welfare Ad’hoc WG on antimicrobial resistance Other… 11

Collaborating Centres Expert Centres on horizontal subjects, for the OIE and Member Countries § Assist in the elaboration of procedures to harmonise animal disease regulations / international standards § Coordinate collaborative studies § Provide technical training § Organise and host scientific 12

AFSSA-ANMV OIE Collaborating centre role Fougères F The ANMV is the French Agency for Veterinary Medicinal Product, Responsibilities in assessment, Authorisation, Monitoring, Control, Inspections ANMV is the OIE collaborating centre for VMP INTERNATIONAL HARMONISATION : - OIE (standards ; methods ; registration of diagnostic assays) - Represent OIE for CODEX meeting related to VMP - Participate to WHO/FAO/OIE workshop, expert groups on VMP - Represent OIE to VICH - Established under the auspices of the OIE - Member of the Steering Committee F RESEARCH and EXPERTISE : Antimicrobial resistance/Benefit/Risk Assessment Pharmaceutical regulation F TECHNICAL ASSISTANCE Bilateral and multilateral cooperation and assistance to OIE member countries (especially developing countries) UEMOA Twinning 13

- C O D E X A L I M E N T A R I US 14

About Codex Alimentarius CODEX ALIMENTARIUS is latin for “food code” The Codex Alimentarius is a collection of food standards, guidelines and codes of practice developed by the Codex Alimentarius Commission June/July 1963 First Session of the Codex Alimentarius Currently 175 Member countries and One Member Organization (EC) 15

Codex Alimentarius Commission Mandate To protect the health of consumers To ensure fair practices in international food trade To coordinate all food standardization work done by international organizations 16

Codex Alimentarius Commission Work organization Work is based on independent scientific advice provided by FAO/WHO in accordance with Risk Analysis principles Consensus whenever possible to ensure broad acceptance of standards - Votes are possible but rare Frequency: Committees/ task forces: every 12 to 24 months - Executive Committee: twice annually Commission: annually 17
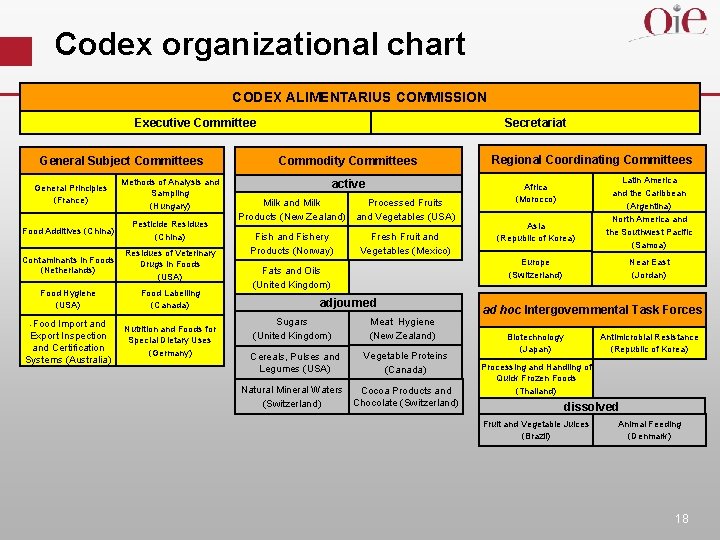
Codex organizational chart CODEX ALIMENTARIUS COMMISSION Secretariat Executive Committee General Subject Committees General Principles (France) Methods of Analysis and Sampling (Hungary) Food Additives (China) Pesticide Residues (China) Contaminants in Foods (Netherlands) Residues of Veterinary Drugs in Foods (USA) Food Hygiene (USA) Food Labelling (Canada) -Food Import and Export Inspection and Certification Systems (Australia) Nutrition and Foods for Special Dietary Uses (Germany) Commodity Committees active Milk and Milk Processed Fruits Products (New Zealand) and Vegetables (USA) Fish and Fishery Products (Norway) Fresh Fruit and Vegetables (Mexico) adjourned Cereals, Pulses and Legumes (USA) Africa (Morocco) Asia (Republic of Korea) Latin America and the Caribbean (Argentina) North America and the Southwest Pacific (Samoa) Europe (Switzerland) Fats and Oils (United Kingdom) Sugars (United Kingdom) Regional Coordinating Committees Meat Hygiene (New Zealand) Vegetable Proteins (Canada) Natural Mineral Waters Cocoa Products and Chocolate (Switzerland) Near East (Jordan) ad hoc Intergovernmental Task Forces Biotechnology (Japan) Antimicrobial Resistance (Republic of Korea) Processing and Handling of Quick Frozen Foods (Thailand) dissolved Fruit and Vegetable Juices (Brazil) Animal Feeding (Denmark) 18
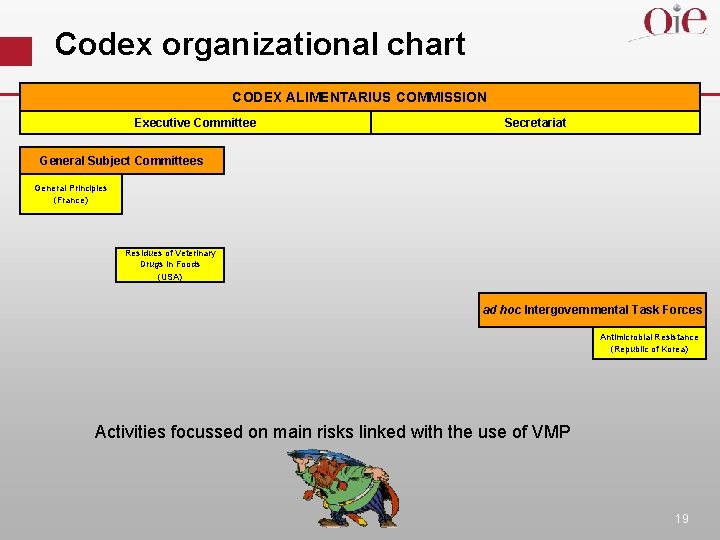
Codex organizational chart CODEX ALIMENTARIUS COMMISSION Executive Committee Secretariat General Subject Committees General Principles (France) Residues of Veterinary Drugs in Foods (USA) ad hoc Intergovernmental Task Forces Antimicrobial Resistance (Republic of Korea) Activities focussed on main risks linked with the use of VMP 19

COLLABORATION BETWEEN World Organisation for Animal Health Organisation mondiale de la santé animale AND - C O D E X A L I M E N T A R I US 20

Collaboration between OIE and Codex alimentarius No formal agreement with Codex Separate Agreement between OIE and FAO and OIE and WHO (2004) Current Status of OIE in Codex: Observer On going discussion to strengthen the link with Codex (CAC July 2007, Alinorm 05/28/41) 21
CCRVDF Veterinary drug 22

CCRVDF FAO/WHO ID No CX-730 Acronym CCRVDF Document Reference CX/RVDF Name Codex Committee on Residues of Veterinary Drugs in Foods Terms of Reference (a)to determine priorities for the consideration of residues of veterinary drugs in foods; (b)to recommend maximum levels of such substances; MRL (c)to develop codes of practice as may be required; and, (d) to consider methods of sampling and analysis for the determination of veterinary drug residues in foods. Status Host Government Active United States of America 23

JECFA RISK ASSESSMENT The Joint FAO/WHO Expert Committee on Food Additives (JECFA) is an international expert scientific committee that is administered jointly by the FAO) and WHO. It has been meeting since 1956, initially to evaluate the safety of food additives. Its work now also includes the evaluation of contaminants, naturally occurring toxicants and residues of veterinary drugs in food. To date, JECFA has evaluated more than 1500 food additives, approximately 40 contaminants and naturally occurring toxicants, and residues of approximately 90 veterinary drugs. The Committee has also developed principles for the safety assessment of chemicals in food that are consistent with current thinking on risk assessment and take account of recent developments in toxicology and other relevant scientific areas such as microbiology, biotechnology, exposure assessment, food chemistry including analytical chemistry and assessment of maximum residue limits for veterinary drugs. JECFA normally meets twice a year with individual agendas covering either (i) food additives, contaminants and naturally occurring toxicants in food or (ii) residues of veterinary drugs 24 in food.

Antimicrobial resistance activities Resistant bacteria 25

OIE antimicrobial resistance related activities OIE ad’hoc group on antimicrobial resistance Five guidelines adopted by the OIE international session and published as standards (2003/2004) : Terrestrial animal health code Section - 3. 9 : antimicrobial resistance · l l l Appendix 3. 9. 1 : Guidelines for the harmonisation of antimicrobial resistance surveillance and monitoring programmes Appendix 3. 9. 2 Guidelines for the monitoring of the quantities of antimicrobials used in animal husbandry Appendix 3. 9. 3 : Guidelines for the responsible and prudent use of antimicrobial agents in veterinary medicine Appendix 3. 9. 4: Risk assessment for antimicrobial resistance arising from the use of antimicrobials in animals Manual of Diagnostic Tests and vaccines for Terrestrial Animals · Laboratory methodologies for bacterial antimicrobial susceptibility testing 26

OIE antimicrobial resistance related activities International meetings: · FAO/WHO/OIE experts consultation (Geneva) on non human antimicrobial usage and antimicrobial resistance · FAO/WHO/OIE experts consultation (Oslo) on non human antimicrobial usage and antimicrobial resistance · Participation to the WHO expert meeting on CIA (Canberra) · FAO/WHO/OIE Expert Consultation on Antimicrobial Use in Aquaculture and Antimicrobial Resistance (Seoul) · Participation to the WHO meeting on CIA (Copenhagen) · Participation to the Codex Task Force on AMR (Seoul) · FAO/WHO/OIE expert consultation on CIA and VCIA (Roma) 27

Codex task Forcehttp: //codextfamr. kfda. go. kr 28

Codex task Force - Results Creation of Three Working groups: · Risk assessment (Chair Canada) · Risk profile (Chair US) · Risk management (Chair DK/FR) § Recommendation that risk management should be adapted to level of development of countries · Meeting of the three WG (May 2008, Brussels) 29

ANTIMICROBIAL WORK IN OIE List of critically important antimicrobial agents VCIA After 2 joint FAO-WHO-OIE experts consultation (Geneva 2003, Oslo 2004) on non human antimicrobial usage and antimicrobial resistance It was recommended that the concept of critically important” classes of antimicrobial agents, both in human and veterinary medicine should be developed 30

List of veterinary critically important antimicrobial agents In May 2006, OIE International committee adopted Resolution n° XXXIII asking to refine the list · The OIE Ad hoc group on Antimicrobial resistance met in September 2006 and proposed a new list In May 2007 OIE International committee adopted the List of Antimicrobials of Veterinary Importance (Resolution n° XXVII) The list can be found at: http: //www. oie. int/downld/Antimicrobials/OIE_list_antimic robials. pdf 31

Rome meeting November 2007 32

CONCLUSION 33

Conclusion International standards are useful tools for countries to be used · Public health · International trade OIE cooperate actively with Codex in the field of Veterinary medicine particularly in CCRVDF and in Codex Task force on antimicrobial resistance The ongoing cooperation between FAO, WHO, Codex and OIE is essential, OIE Working Group on Food Safety (created to coordinate and manage the animal production food safety activities) will continue to work for a strengthened cooperation with Codex. 34
No Resistant Bacteria No Residues 35

36
- Slides: 36