Dr Deon Bennett Structure 2 important structural proteins

Dr. Deon Bennett
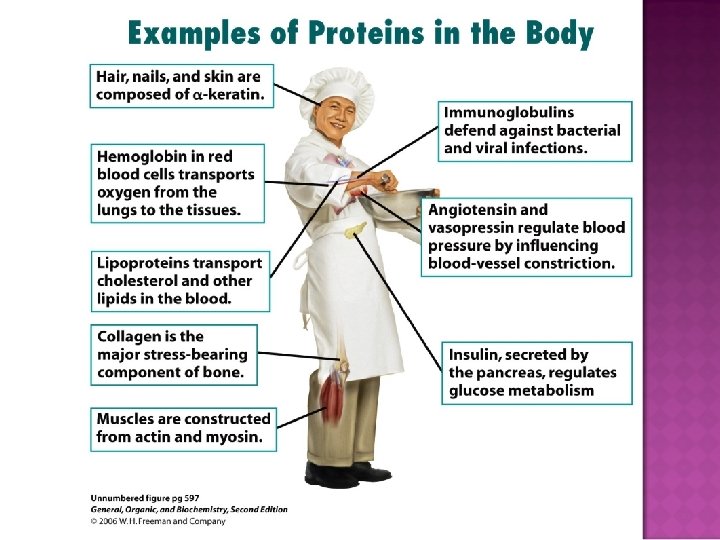

Structure – 2 important structural proteins are collagen and keratin. Catalysis – virtually all reactions are catalyzed by proteins called enzymes. Movement -contractile proteins (e. g. , myosin, actin). Transport – Hb transports O 2 and. CO 2. Other proteins transport molecules across membrane.

Hormones –many hormones are proteins. Protection - antibodies counteract foreign proteins. Blood clotting is another protective function carried out by fibrinogen. Storage – casein in milk and ovalbumin in eggs store nutrients for newborn mammals and birds. Ferritin stores iron in the liver. Regulation – some proteins control gene expression

Amino Acids Ø Amino Acids are the building units of proteins. Proteins are polymers of amino acids linked together by what is called “ Peptide bond” (see latter). Ø There about 300 amino acids occur in nature. Only 20 of them occur in proteins.

Complex molecules Polymers consisting of long chains of smaller units known as amino acids covalently linked through peptide bonds into a chain.

There are 20 different kinds of amino acids that make up the proteins. Proteins may contain hundreds or thousands of amino acids.

Proteins can be classified in two types: 1. Fibrous proteins –insoluble in water and are used mainly for structural purposes. 2. Globular proteins – more or less soluble in water and are mainly for non-structural purposes.
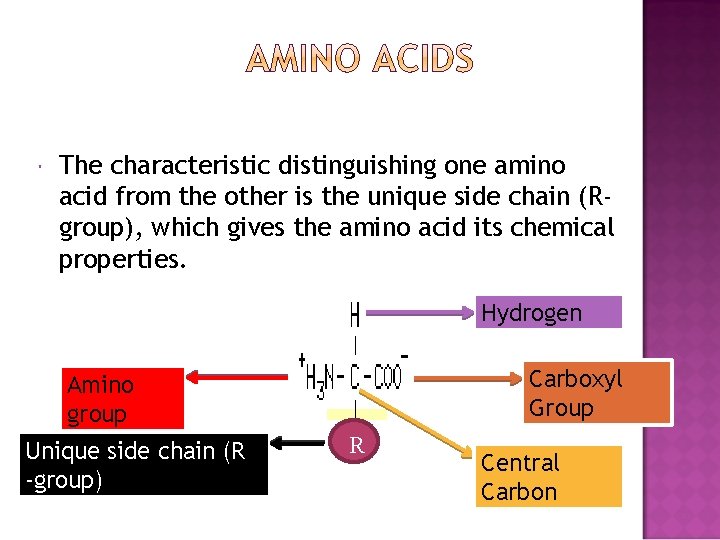
The characteristic distinguishing one amino acid from the other is the unique side chain (Rgroup), which gives the amino acid its chemical properties. Hydrogen Carboxyl Group Amino group Unique side chain (R -group) R Central Carbon
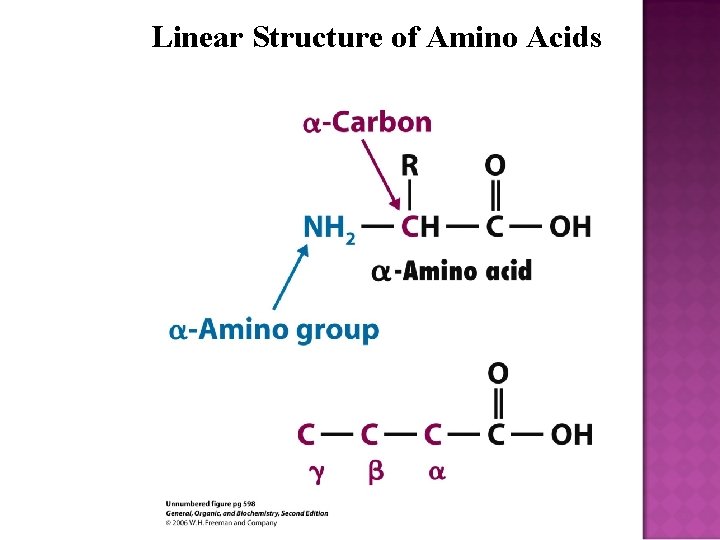
Linear Structure of Amino Acids

The most important aspect of the R group is their polarity. AA can be classified into 4 groups : 1. Non-polar - hydrophobic 2. Polar 3. Acidic 4. Basic

Classification according to polarity of side chain (R): A- Polar amino acids: in which R contains polar hydrophilic group so can forms hydrogen bond with H 2 O. In those amino acids, R may contain: 1 - OH group : as in serine, threonine and tyrosine 2 - SH group : as in cysteine 3 - amide group: as in glutamine and aspargine 4 - NH 2 group or nitrogen act as a base (basic amino acids ): as lysine, arginine and histidine 5 - COOH group ( acidic amino acids): as aspartic and glutamic B- Non polar amino acids: R is alkyl hydrophobic group which can’t enter in hydrogen bond formation. 9 amino acids are non polar ( glycine, alanine, valine, leucine, isoleucine, phenyl alanine, tryptophan, proline and methionine)


C- Basic amino acids: Contain two or more NH 2 groups or nitrogen atoms that act as base i. e. can bind proton. At physiological p. H, basic amino acids will be positively charged. e. g. a- Lysine b- Arginine: contains guanido group c- Histidine: is an example on basic heterocyclic amino acids D Acidic amino acidsat physiological p. H will carry negative charge. e. g. Aspartic acid (aspartate) and Glutamic acid (glutamate).


Building blocks of proteins. Linked together in varying sequences to form a vast variety of proteins. 8 essential amino acids - Isoleucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, and Valine. Arginine and Histidine - nutritionally semiessential, since they can be synthesized but not in quantities sufficient to permit normal growth.
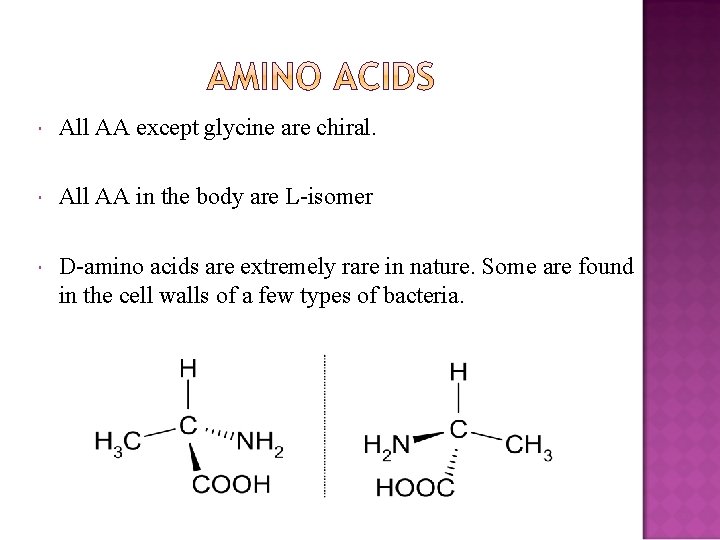
All AA except glycine are chiral. All AA in the body are L-isomer D-amino acids are extremely rare in nature. Some are found in the cell walls of a few types of bacteria.

The amine and carboxylic acid functional groups found in amino acids allow them to have amphiprotic properties. Carboxylic acid groups (-CO 2 H) can be deprotonated to become negative carboxylates (-CO 2 - ), and α-amino groups (NH 2 -) can be protonated to become positive α-ammonium groups (+NH 3 -). This molecular state is known as a zwitterion, from the German Zwitter meaning hermaphrodite or hybrid. Amino acids exist as zwitterions in the solid phase and in solution.

In acidic solution, the carboxylate function (- COO-) accepts a proton and gets converted to carboxyl substituent (- COOH) In basic solution the ammonium substituent (+NH 3) changes to amino group (-NH 2) by losing a proton.

Ionization states of -amino acid at different p. H

p. Ka- negative logarithm of the acid dissociation constant, Ka. Just like the p. H, the p. Ka tells you of the acid or basic properties of a substance. p. Ka <2 means strong acid >2 but <7 means weak acid >7 but <10 means weak base >10 means a strong base

At p. H values greater than the p. Ka of the carboxylic acid group, the negative carboxylate ion predominates. At p. H values lower than the p. Ka of the αammonium group, the nitrogen is predominantly protonated as a positively charged α-ammonium group. Thus, at p. H between 2. 2 and 9. 4, the predominant form adopted by α-amino acids contains a negative carboxylate and a positive α-ammonium group

At p. H values between the two p. Ka values, the zwitterion predominates, but coexists in dynamic equilibrium with small amounts of net negative and net positive ions. At the exact midpoint between the two p. Ka values, the trace amount of net negative and trace of net positive ions exactly balance, so that average net charge of all forms present is zero. This p. H is known as the isoelectric point p. I, so p. I = ½(p. Ka 1 + p. Ka 2). The individual amino acids all have slightly different p. Ka values, so have different isoelectric points.

For amino acids with charged side-chains, the p. Ka of the side-chain is involved. Thus for Asp, Glu with negative side-chains, p. I = ½(p. Ka 1 + p. Ka. R), where p. Ka. R is the side-chain p. Ka. Cysteine also has potentially negative side-chain with p. Ka. R = 8. 14, so p. I should be calculated as for Asp and Glu, even though the side-chain is not significantly charged at neutral p. H. For His, Lys, and Arg with positive side-chains, p. I = ½(p. Ka. R + p. Ka 2).

There is no p. H at which AA have no ionic character. The p. H at which AA have equal number of positive and negative charge is called the isoelectric point (p. I). The dipolar ion exists as a neutral ion and will have zero mobility in electrophoresis. Every AA have a different p. I. The 3 basic AA have higher p. I and the 2 acidic AA have lower p. I.

In zwitter ionic form, amino acids show amphoteric behavior as they react both with acids and bases. Zwitterion – a compound that is both acid and a base. A solution that neutralizes both an acid and a base is called a buffer solution. AA are therefore amphiprotic and aqueous solutions of them are buffers
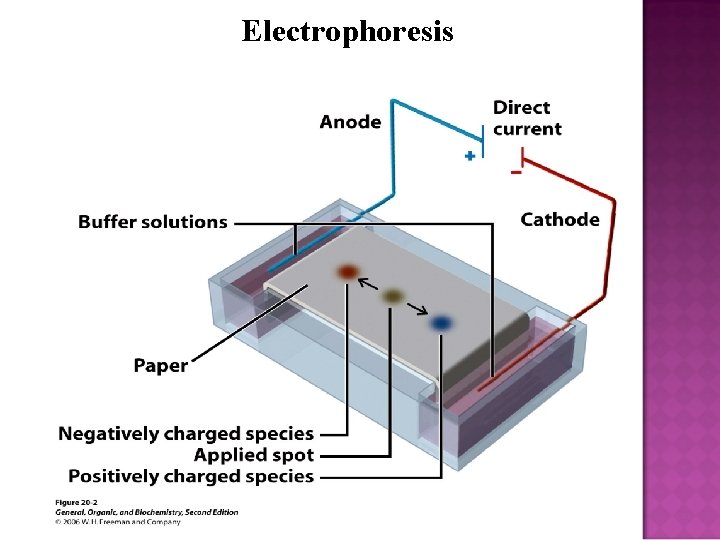
Electrophoresis

AA have both a primary amine group and a primary carboxyl group, and these chemicals can undergo most of the reactions associated with these functional groups. These include nucleophilic addition, amide bond formation and imine formation for the amine group and esterification, amide bond formation and decarboxylation for the carboxylic acid group. The combination of these functional groups allow amino acids to be effective polydentate ligands for metal-amino acid chelates.

The multiple side-chains of amino acids can also undergo chemical reactions. The types of these reactions are determined by the groups on these side-chains and are, therefore, different between the various types of amino acid.

Like AA proteins behave as zwitterions Act like buffers Proteins are least soluble at their isoelectric points and can be precipitated from their solutions.

A peptide bond (amide bond) is a covalent chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other molecule. H 2 O is released and hence the process is a dehydration synthesis reaction (condensation reaction) and usually occurs between amino acids. The resulting C(O)NH bond is called a peptide bond, and the resulting molecule is an amide.
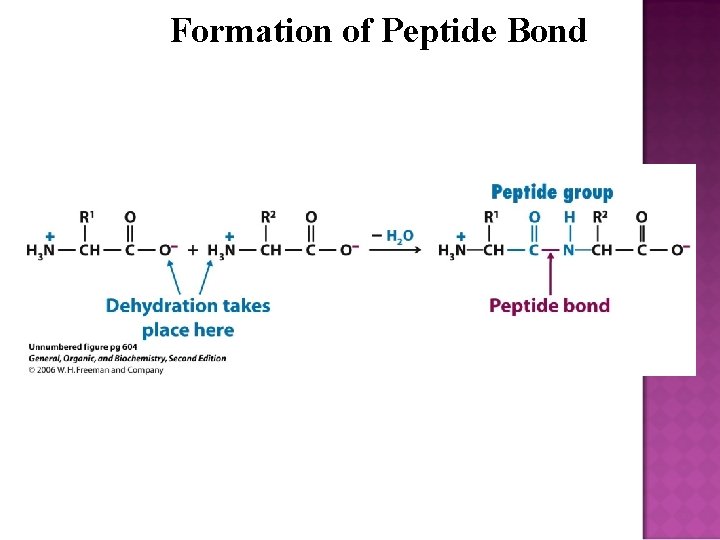
Formation of Peptide Bond

A peptide bond can be broken by amide hydrolysis (the adding of water). The peptide bonds in proteins are metastable, meaning that in the presence of water they will break spontaneously, releasing 2 -4 kcal/mol of free energy, but this process is extremely slow. In living organisms, the process is facilitated by enzymes. Living organisms also employ enzymes to form peptide bonds; this process requires free energy.

Inhibitory peptides are also used in clinical research to examine the effects of peptides on the inhibition of cancer proteins and other diseases. Peptides are used in imaging where, specific peptides in the fluorescing material help with early detection of potentially lifethreatening cancers. Peptides are used in skin care to increase the amount of collagen being produced in the skin, stimulate fibroblast growth, and stimulate the production of keratinocytes. The zero-calorie sweetener found in many diet beverages is a synthesized peptide.
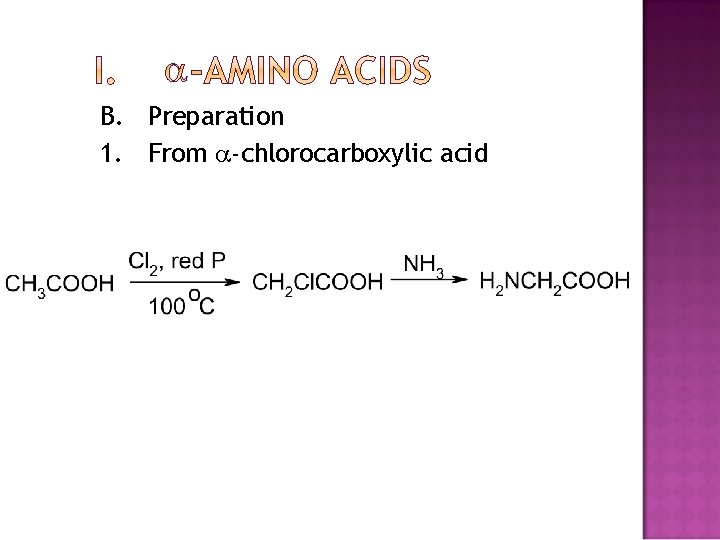
B. Preparation 1. From -chlorocarboxylic acid
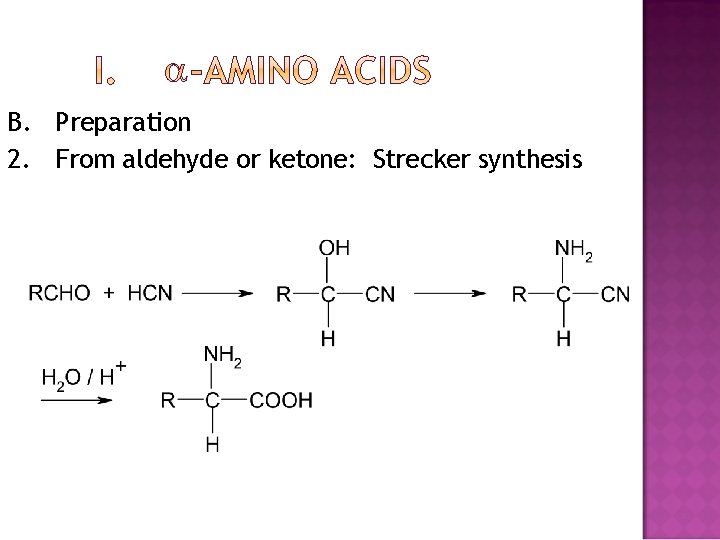
B. Preparation 2. From aldehyde or ketone: Strecker synthesis
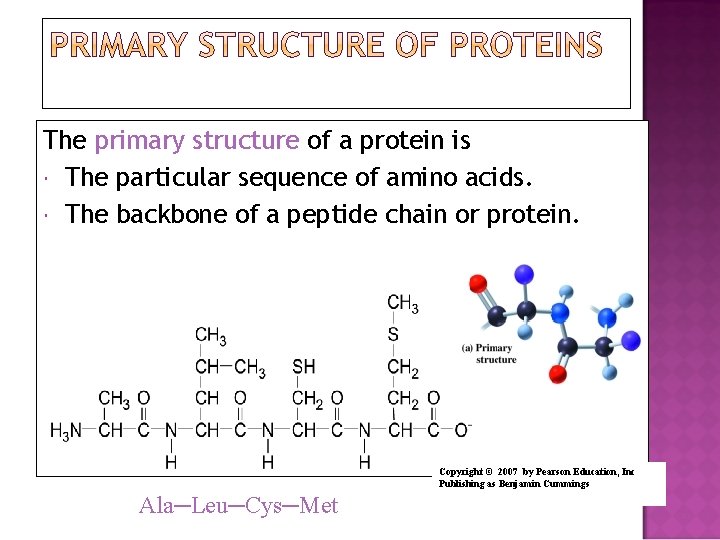
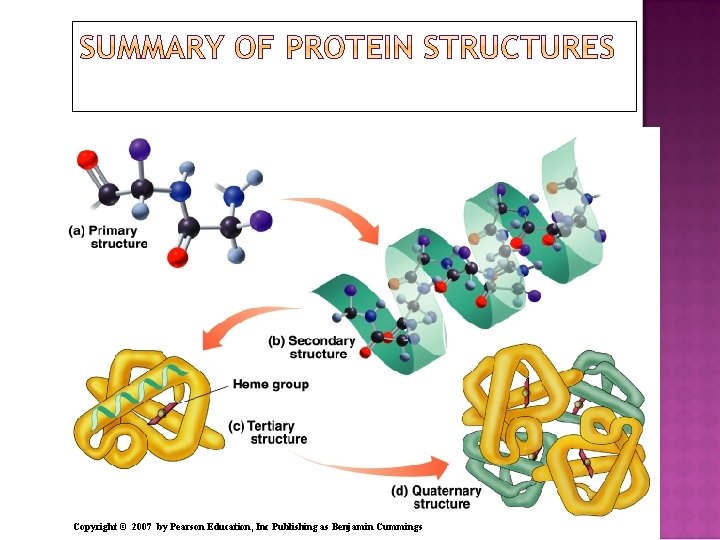
The primary structure of a protein is The particular sequence of amino acids. The backbone of a peptide chain or protein. Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings Ala─Leu─Cys─Met
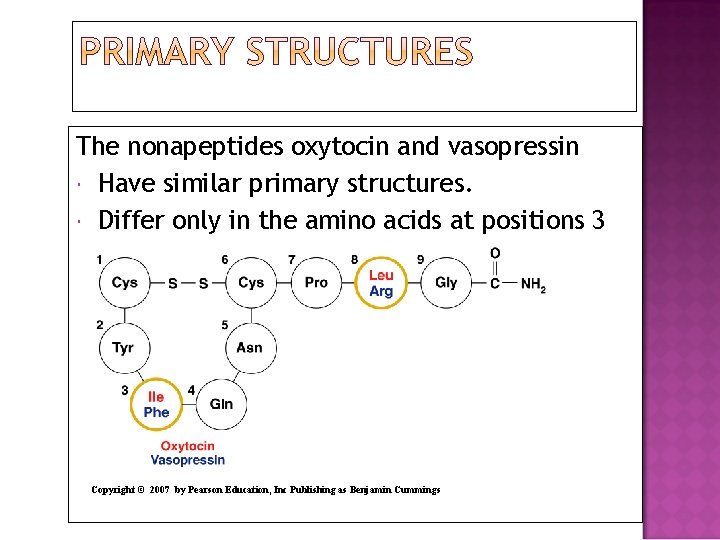
The nonapeptides oxytocin and vasopressin Have similar primary structures. Differ only in the amino acids at positions 3 and 8. Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings

The secondary structures of proteins indicate three-dimensional spatial arrangements of the polypeptide chains. An alpha helix has A coiled shape held in place by hydrogen bonds between the amide groups and the carbonyl groups of the amino acids along the chain. Hydrogen bonds between the H of a –N-H group and the O of C=O of the fourth amino acid down the chain.
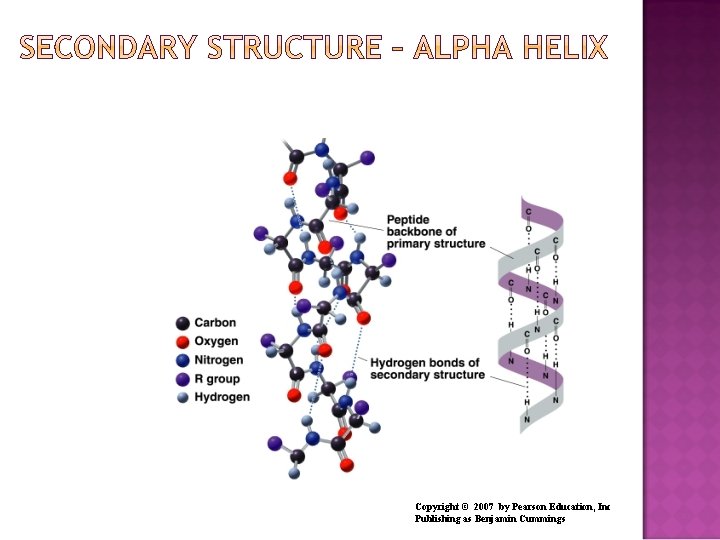
Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings

A beta-pleated sheet is a secondary structure: Consists of polypeptide chains arranged side by side. Has hydrogen bonds between chains. Has R groups above and below the sheet. Is typical of fibrous proteins such as silk.
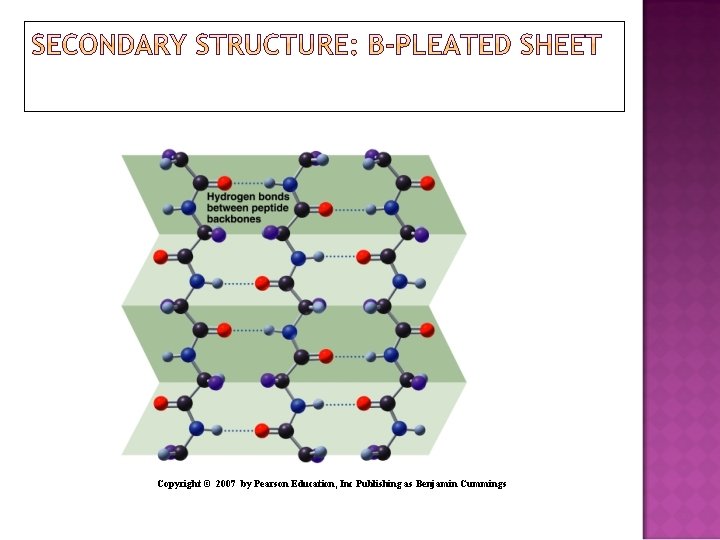
Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings
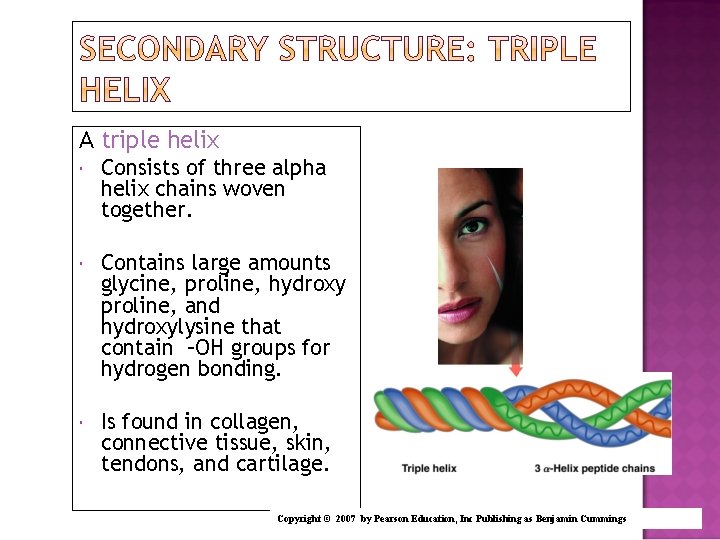
A triple helix Consists of three alpha helix chains woven together. Contains large amounts glycine, proline, hydroxy proline, and hydroxylysine that contain –OH groups for hydrogen bonding. Is found in collagen, connective tissue, skin, tendons, and cartilage. Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings
Protein Structure: Tertiary and Quaternary Levels Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings

The tertiary structure of a protein Gives a specific three dimensional shape to the polypeptide chain. Involves interactions and cross links between different parts of the peptide chain. Is stabilized by Hydrophobic and hydrophilic interactions. Salt bridges. Hydrogen bonds. Disulfide bonds.
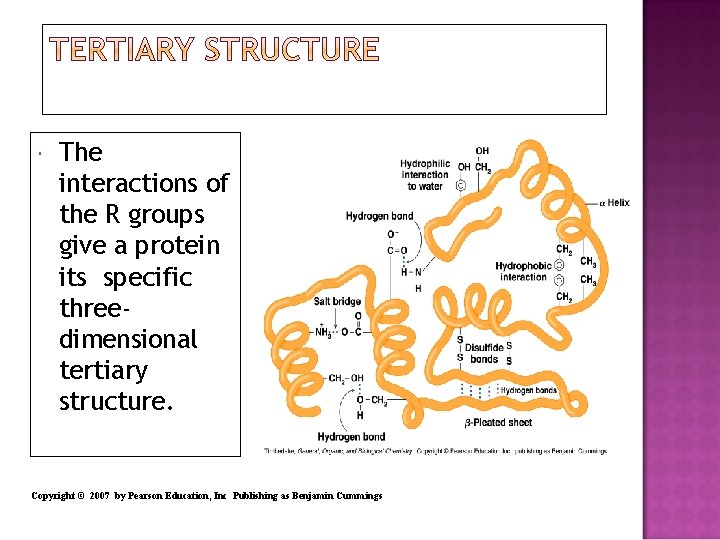
The interactions of the R groups give a protein its specific threedimensional tertiary structure. Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings
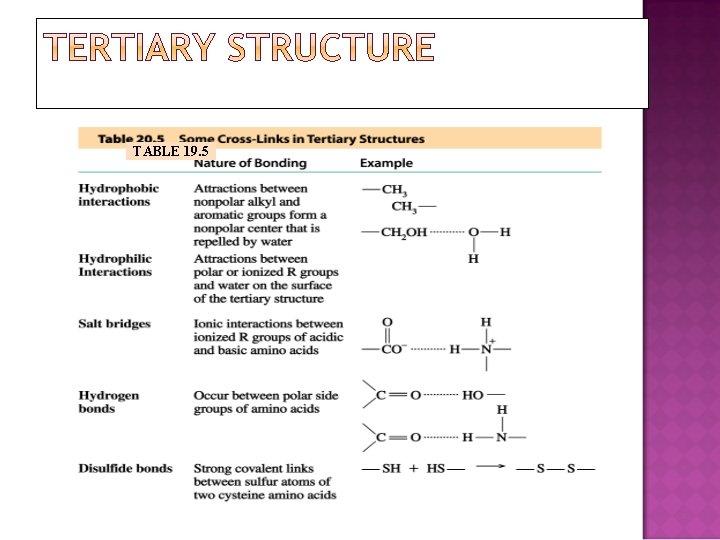
TABLE 19. 5
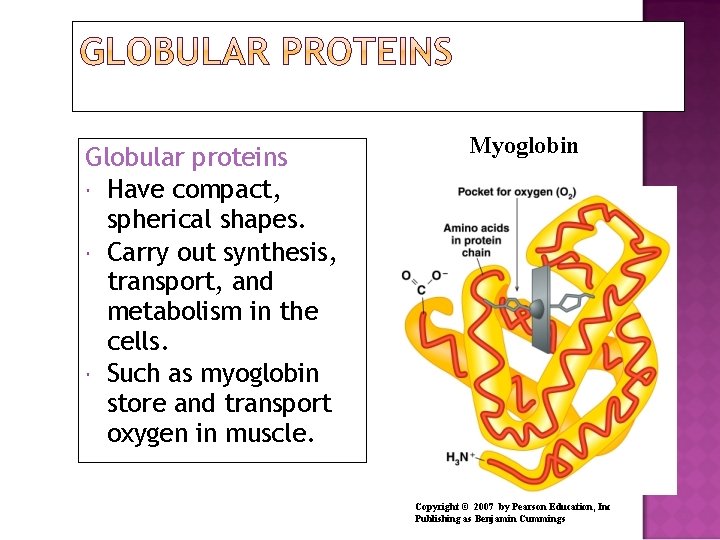
Globular proteins Have compact, spherical shapes. Carry out synthesis, transport, and metabolism in the cells. Such as myoglobin store and transport oxygen in muscle. Myoglobin Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings

Fibrous proteins Consist of long, fiber-like shapes. Such as alpha keratins make up hair, wool, skin, and nails. Such as feathers contain beta keratins with large amounts of beta-pleated sheet structures. Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings
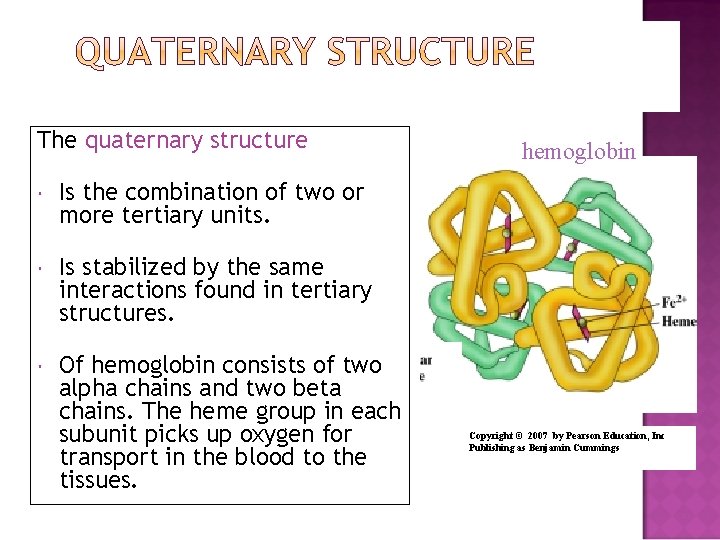
The quaternary structure Is the combination of two or more tertiary units. Is stabilized by the same interactions found in tertiary structures. Of hemoglobin consists of two alpha chains and two beta chains. The heme group in each subunit picks up oxygen for transport in the blood to the tissues. hemoglobin Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings

Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings

In the adult human, Hb (tetramer) consists of 4 polypeptide chains (globins) – 2 identical α chains (141 residues) and 2 identical β chains (146 residues). Each globin chain surrounds an iron-containing heme. Conjugated proteins – proteins that contains non-amino acid portion. Prosthetic group – the non-amino acid portion of a conjugated protein. The globins are the amino acid portion and the prosthetic groups are the heme units.

Myoglobin consist of 1 polypeptide chain, 153 residues long. Both the α and β chains of Hb are similar to myoglobin. Many of the AA of the α and β chains of Hb are similar to myoglobin. The heme group is the same in both Hb and myoglobin

1 myoglobin binds to 1 O 2 1 Hb can bind to 4 O 2 The binding of oxygen to Hb Oxygen binding to exhibit positive cooperativity myoglobin does not exhibit (when 1 oxygen molecule is bound it becomes easier for positive cooperativity the other oxygen to bind Hb functions in oxygen transport. It must bind strongly to oxygen and be able to release it easily Heamoglobin Myoglobin has the function of oxygen storage in muscles Myoglobin

The amino acid sequence of any polypeptide created by a cell, is in turn determined by the stretches of DNA called genes. In all proteins, it is the amino acid sequence, which determines the protein's chemical properties and function. The amino acid sequences of the globin proteins in hemoglobins usually differ between species, although the differences grow with the evolutionary distance between species.

For example, the most common hemoglobin sequences in humans and chimpanzees are nearly identical, differing by only one amino acid in both the alpha and the beta globin protein chains. These differences grow larger between less closely related species. Variations in hemoglobin amino acid sequences, as with other proteins, may be adaptive. For example, the genetic difference between lowland mice and highland mice enables highland mice to make more efficient use of their oxygen, since less oxygen is available at higher altitudes.

Denaturation - proteins lose their tertiary structure and secondary structure by application of a chemical or physical agent. Denaturation leaves the primary structure between the amino acids intact. If proteins in a living cell are denatured, this results in disruption of cell activity and possibly cell death.

Denatured proteins can exhibit a wide range of characteristics, from loss of solubility to communal aggregation. Communal aggregation is the phenomenon of aggregation of the hydrophobic proteins to come closer and form the bonding between them, so as to reduce the total area exposed to water. Since all structural levels of the protein determines its function, the protein can no longer perform its function once it has been denatured.

Thermal denaturation p. H Denaturation Ionic Strength

Knowing the quantity of a protein after each separation step is useful in checking the progress of purification and evaluating the technique's efficiency. The concentration of a protein can be measured by immunological techniques such as ELISA or Western Blotting Activity can be measured using fluorescent techniques. Mass spectrometry is another accurate analytical technique for determining protein mass. In this technique, atoms are ionized through a machine and passed through a vacuum into the detector.

Denatured protein - used in the formation of geletin, which is used in many foods, in the pharmaceutical and cosmetic industries. Collagen has been widely used in cosmetic surgery, as a healing aid for burn patients for reconstruction of bone and a wide variety of dental, orthopedic and surgical purposes. Collagens are also used in the construction of artificial skin substitutes used in the management of severe burns. Corn gluten is used in feed concentrates and in small amounts as extenders for commercial resins and glues. Corn gluten is sometimes destarched by acid or enzyme hydrolysis to yield a product that is almost entirely protein and is used in paints. Sometimes it is broken down by complete hydrolysis to yield monosodium glutamate, a seasoning for food.

Electrophoresis - used to separate complex mixtures of proteins and to purify the protein for subsequent applications. Western blotting is used to identify a specific protein in a complex mixture of proteins. The technique exploits both the efficiency of SDS-PAGE to separate a mixture of proteins into distinct protein bands, and the ability of immunochemical reagents to interact specifically with a given protein antigen. Column Chromatography can be used to fractionate proteins. The mixture of proteins in solution is passed through a column containing a porous solid matrix. The 3 types of matrices used are 1) Ion-exchange chromatography, 2) gel-filtration chromatography and 3) affinity chromatography.

The size of a synthesized protein can be measured by the number of amino acids it contains and by its total molecular mass, which is normally reported in units of daltons or the derivative unit kilodalton (k. Da). Yeast proteins are on average 466 amino acids long and 53 k. Da in mass. The largest known proteins are the titins, a component of the muscle sarcomere, with a molecular mass of almost 3, 000 k. Da and a total length of almost 27, 000 amino acids.

Proteins may be positive, negative, neutral, or polar in nature, and together give a protein its overall charge. At a p. H below their p. I, proteins carry a net positive charge; above their p. I they carry a net negative charge. Proteins can, thus, be separated according to their isoelectric point (overall charge) on a polyacrylamaide gel using a technique called isoelectric focusing, which uses a p. H gradient to separate proteins. Isoelectric focusing is also the first step in 2 -D gel polyacrylamide gel electrophoresis.

The isoelectric point, p. I, is the p. H of an aqueous solution of an amino acid (or peptide) at which the molecules on average have no net charge. The positively charged groups are exactly balanced by the negatively charged groups. For simple amino acids such as alanine, the p. I is an average of the p. Ka's of the carboxyl (2. 34) and ammonium (9. 69) groups. Thus, the p. I for alanine is calculated to be: (2. 34 + 9. 69)/2 = 6. 02, the experimentally determined value.
Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings
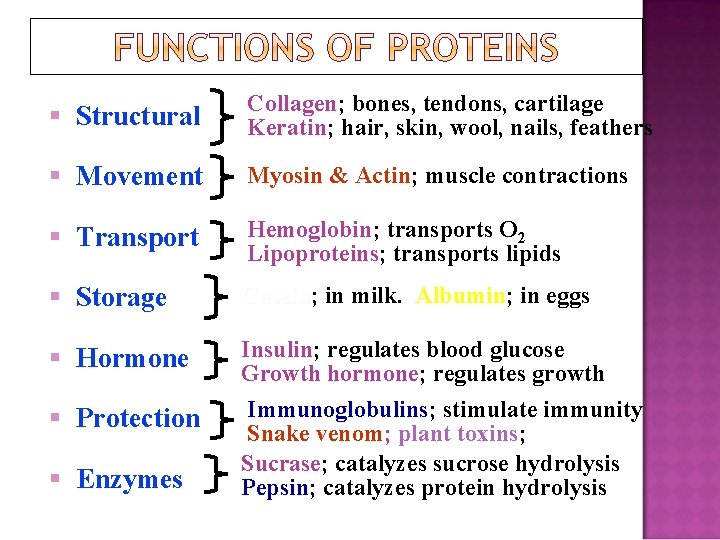
§ Structural Collagen; Collagen bones, tendons, cartilage Keratin; Keratin hair, skin, wool, nails, feathers § Movement Myosin & Actin; Actin muscle contractions § Transport Hemoglobin; Hemoglobin transports O 2 Lipoproteins; Lipoproteins transports lipids § Storage Casein; Casein in milk. Albumin; Albumin in eggs § Hormone Insulin; Insulin regulates blood glucose Growth hormone; hormone regulates growth Immunoglobulins; Immunoglobulins stimulate immunity Snake venom; plant toxins; toxins Sucrase; Sucrase catalyzes sucrose hydrolysis Pepsin; Pepsin catalyzes protein hydrolysis § Protection § Enzymes

1. Lecture was created by Dr. Mellissa Sanderson and Dr. Andrew Lamm lecture material and modified by Dr. D. Bennett. 2. Copyright © 2007 by Pearson Education, Inc Publishing as Benjamin Cummings
- Slides: 68