Dr Areefa Al Bahri Chapter 3 Complications During

Dr. Areefa Al Bahri Chapter 3 Complications During Pregnancy Complications that arise during pregnancy are often challenging and demand the perinatal nurse’s skills, knowledge, and expertise, combined with the nursing process, to fi rst identify the pregnant patient at risk and then formulate, implement, and evaluate an appropriate, holistic plan of care.

Early Pregnancy Complications Ø Hyperemesis gravidarum Ø Spontaneous abortion/ miscarriage Ø Gestational trophoblastic disease Ø Ectopic pregnancy Ø Perinatal loss- Fetal death Less than perfect child

Miscellaneous Complications ØMultiple gestation Ø Premature rupture of membranes Ø Preterm labor ØIncompetent cervix

Endocrine Complication ØDiabetes Type I & II ØHyper/hypothyroid Cardiovascular/Hematological Øcardiomyopathy ØHematological • Sickle cell anemia • Thalassemia

Hypertensive disorders • Chronic hypertension • Preeclampsia/eclampsia Proteinuria, edema, CNS alterations, HELLP • Chronic hypertension with preeclampsia • Gestational hypertension

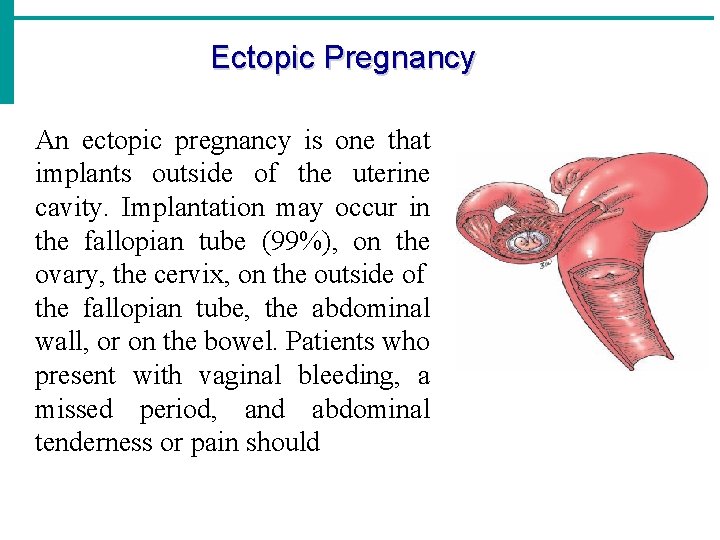
Ectopic Pregnancy An ectopic pregnancy is one that implants outside of the uterine cavity. Implantation may occur in the fallopian tube (99%), on the ovary, the cervix, on the outside of the fallopian tube, the abdominal wall, or on the bowel. Patients who present with vaginal bleeding, a missed period, and abdominal tenderness or pain should

A number of factors of ectopic pregnancy which include: • History of STI or pelvic inflammatory disease • Prior ectopic pregnancy • Previous tubal, pelvic, or abdominal surgery • Endometriosis • In vitro fertilization or other method of assisted reproduction • Use of an intrauterine device

Diagnosis ectopic pregnancy should be diagnosed before the onset of hypotension, bleeding, pain, and overt rupture. The patient’s history (e. g. , unilateral, bilateral or diffuse abdominal pain, missed period) and physical exam (a palpable mass is present on bimanual examination in approximately 50% of women) should alert the health care professional to the possible presence of an ectopic pregnancy. Diagnostic laboratory tests include a beta-human chorionic gonadotropin (h. CG). Transvaginal ultrasonography should be performed to confirm intrauterine or tubal pregnancy (Farquhar, 2005). Ultrasonographic identification of an intrauterine pregnancy rules out the presence of an ectopic pregnancy in most women (Murray, Baakdah, Bardell, & Tulandi, 2005).

Management Salpingectomy (removal of the ruptured fallopian tube) by laparotomy (surgical procedure in which the abdomen is opened to visualize the abdominal organs) has long offered an almost 100% cure for the treatment of an ectopic pregnancy. using a laparoscope inserted into the pelvic cavity through a small incision in the abdomen), salpingostomy (incision into the fallopian tube to remove the pregnancy) and partial salpingectomy are replacing laparotomy as the treatment mode of choice.

GESTATIONAL TROPHOBLASTIC DISEASE Gestational trophoblastic disease (GTD) is a clinical diagnosis that includes the histologic diagnoses of hydatidiform mole (“molar pregnancy”), locally invasive mole, metastatic mole, and choriocarcinoma. It is a disease characterized by an abnormal placental development that results in the production of fluid-filled grapelike clusters (instead of normal placental tissue) and a vast proliferation of trophoblastic tissue. It is associated with loss of the pregnancy and rarely, the development of cancer. GTD occurs in 1200 pregnancies (Berman, Di. Saia, & Tewari, 2004).

Pathophysiology The cause is unknown, but it is thought that complete moles result from the fertilization of an empty ovum (one whose nucleus is missing or nonfunctional) by a normal sperm. Since the ovum contains no maternal genetic material. A complete mole is characterized by trophoblastic proliferation and the absence of fetal parts. Most fetuses associated with incomplete moles being spontaneously aborted. Incomplete moles are almost always benign and have a much lower malignancy potential than complete moles. Choriocarcinoma is invasive, malignant trophoblastic disease that is usually metastatic and can be fatal (Berman et al. , 2004).

Signs and Symptoms Øvaginal bleeding More than 95% of patients experience which may be scant or profuse ØUterine enlargement results from the rapidly proliferating trophoblastic tissue and the large accumulation of clotted blood. Øexcessive nausea and vomiting (hyperemesis gravidarum) and abdominal pain caused by uterine distention. ØPreeclampsia may occur, usually between 9 and 12 weeks of gestation but any symptoms of gestational hypertension before 24 weeks of gestation may be indicative of hydatidiform mole. Clinical and laboratory findings include an absence of fetal heart sounds. Øa markedly elevated quantitative serum h. CG (may be 100, 000 m. IU/m. L), and very low levels of maternal serum fetoprotein (MSAFP).

Management Øremoval of the uterine contents ØThe h. CG levels should be assessed every 1 to 2 weeks until h. CG is undetectable on two consecutive Øh. CG should be measured every 1 to 2 months for at least a year ØEffective contraception is needed. Øcounsel the patient about different methods of contraception Øavoiding pregnancy for a year

Prognosis Invasive moles are generally not metastatic and respond well to single-agent chemotherapy. Choriocarcinoma spreads to the lungs, vagina, pelvis, brain, liver, intestines, and kidneys. Since choriocarcinoma can occur weeks to years after any type of gestation, patients usually present with signs and symptoms of active metastases. The long term prognosis depends on the degree of metastases and the patient’s response to the chemotherapy.

SPONTANEOUS ABORTION Not all conceptions result in a live-born infant. Of all clinically recognized pregnancies, 10% to 20% are lost, and approximately 22% of pregnancies detected on the basis of h. CG assays are lost before the appearance of any clinical signs or symptoms (White & Bouvier, 2005). By definition, an early pregnancy loss occurs before 12 weeks of gestation; a late pregnancy loss is one that occurs between 12 and 20 weeks of gestation.

Spontaneous abortions may be classified as the following: • Abortus: Fetus lost before 20 weeks of gestation, wt. less than 500 g) • Complete abortion: Complete expulsion before 20 weeks of gestation • Incomplete abortion: Partial expulsion of some but not all before 20 weeks of gestation • Inevitable abortion: No expulsion of products, but bleeding and dilation of the cervix such that a pregnancy is unlikely • Threatened abortion: Any intrauterine bleeding before 20 weeks of gestation, without dilation of the cervix or expulsion • Missed abortion: Death of the embryo or fetus before 20 weeks with complete retention these often proceed to a complete abortion within 1 to 3 weeks but occasionally they are retained much longer.

Etiology It is estimated that 60% to 80% of all SABs in the first trimester are associated with chromosomal abnormalities Infections (e. g. , bacteriuria and Chlamydia trachomatis), maternal anatomical defects, and immunological and endocrine factors have also been identified as causes of early pregnancy loss, although many have no obvious cause. Second trimester spontaneous abortions (12 to 20 weeks) have been linked to chronic infection, recreational drug use, maternal uterine or cervical anatomical defects, maternal systemic disease, exposure to fetotoxic agents, and trauma (Cunningham et al. , 2005).

Diagnosis A woman who is experiencing a spontaneous abortion usually presents with bleeding and may also complain of cramping, abdominal pain, and decreased symptoms of pregnancy; cervical changes (dilation) may be present on vaginal examination. An ultrasound is performed for placental evaluation and to determine fetal viability. Laboratory tests include a quantitative level of -h. CG, which should show a lower value than when associated with a viable pregnancy. hemoglobin and hematocrit, blood type and Rh status determination, and indirect Coombs’ screen (Cunningham et al. , 2005).

Management Incomplete, inevitable, and missed abortions are usually managed via a dilatation and curettage (D & C: the cervix is dilated and a curette is inserted and used to scrape the uterine walls and remove the uterine contents). In the case of an incompetent cervix, an emergent cerclage (placement of ligature to close the cervix) may be performed. An unsensitized, Rhnegative woman should be given Rho(D) immune globulin (Rho. GAM) to prevent

INCOMPETENT CERVIX Patients with cervical incompetence usually present with painless dilation and effacement of the cervix, often during the second trimester of pregnancy. They frequently give a history of repeated second trimester losses with no apparent etiology. Incompetent cervix is estimated to cause approximately 15% of all second trimester losses (Cunningham et al. , 2005).

Obstetric Causes of Vaginal Bleeding PLACENTA PREVIA Placenta previa is an implantation of the placenta in the lower uterine segment, near or over the internal cervical os. This condition accounts for 20% of all antepartal hemorrhages. There are three recognized variations of placenta previa. With a complete (total) placenta previa, the placenta covers the entire cervical os. a complete placenta previa presents the most serious risk. A partial previa describes a placenta that partially occludes the cervical os. A marginal previa is characterized by the encroachment of the placenta to the margin of the cervical os. Placenta accreta, percreta, and increta are placentas with abnormally fi rm attachments to the uterine wall. Unusual placental adherence may accompany a placenta previa.
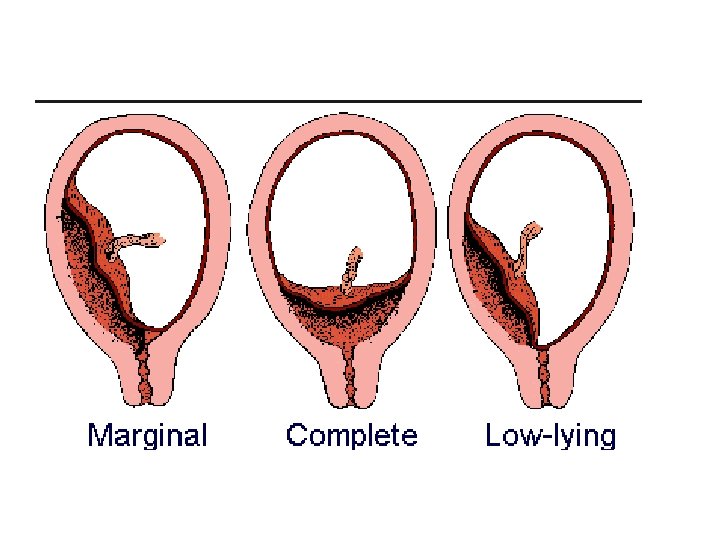

Placenta previa may be associated with conditions that cause scarring of the uterus such as a prior cesarean section, multiparity, or increased maternal age. A previa may also occur with a large placental mass as seen in multiple gestations and erythroblastosis. Other risk factors include smoking, cocaine use, a prior history of placenta previa, closely spaced pregnancies, African or Asian ethnicity, and maternal age greater than 35 years (Clark, 2004).

Signs and Symptoms The most common symptom is painless vaginal bleeding. This is believed to occur from small disruptions in the placental attachment during normal development and the subsequent stretching and thinning of the lower uterine segment during the third trimester. Initially, the bleeding is usually a small amount that stops as the uterus contracts to close the open blood vessels. However, bleeding can reoccur at any time and may be associated with profuse hemorrhage and shock that leads to signifi cant maternal and fetal mortality and morbidity. The blood is bright red (Cunningham et al. , 2005).

Diagnosis The timing of the diagnosis of placenta previa has undergone significant change in the last decade. Although thirdtrimester bleeding was often the first indicator of placenta previa, today, most cases of placenta previa are detected ante-natally before the onset of significant bleeding. The common practice of second-trimester abdominal ultrasound for the detection of fetal anomalies has led to this change. However, because most cases of placenta previa diagnosed in the second trimester tend to resolve as the uterus enlarges, management of placenta previa diagnosed in the second trimester differs from that for the same diagnosis made during the third trimester.

In patients diagnosed before 24 weeks’ gestation, a repeat ultrasound should be scheduled between 24 and 28 weeks’ gestation to confirm the diagnosis of placenta previa. However, if patients experience vaginal bleeding during this interval, they should be managed as presumed cases of placenta previa. Placenta previa should be suspected in all patients who present with bleeding

PLACENTAL ABRUPTION Placenta abruption (abruptio placenta) is the premature separation of a normally implanted placenta from the uterine wall. An abruption results in hemorrhage between the uterine wall and the placenta. 15% occur during labor, and 30% are identified only on inspection of the placenta after delivery (Cunningham et al. , 2005).
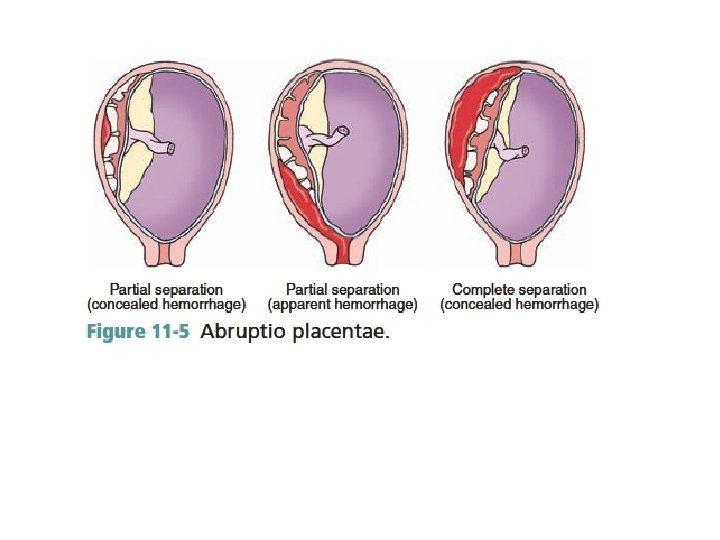

Etiology and Classifications At the initial point of placental separation, non clotted blood courses from the site of injury. The enlarging collection of blood may cause further separation of the placenta. Bleeding can be either concealed or revealed (apparent). A concealed hemorrhage occurs in 20% of cases and describes an abruption in which the bleeding is confined within the uterine cavity. The most common abruption is associated with a revealed or external hemorrhage, where the blood dissects downward toward the cervix (Fig. 11 -5). Placental abruption may be broadly classified into three grades that correlate with clinical and laboratory findings (Box 11 -2).

What causes placental abruption? The causes of placental abruption are not completely known. However, women are more at risk for this condition if they: v Smoke v Use cocaine during pregnancy v Age over 35 y v Have preeclampsia or hypertension v Are pregnant with twins or triplets v Have had a previous placental abruption v Experience trauma to the abdomen v Have abnormalities in the uterus

Signs and Symptoms Vaginal bleeding (although about 20% of cases will have no bleeding) Uterine tenderness Rapid contractions Abdominal pain Fetal heart rate abnormalities

Diagnosis Vaginal bleeding in the third trimester of pregnancy is the hallmark of placental abruption or placenta previa and should always prompt an investigation to determine its etiology. Diagnosis is made by clinical findings and, when available, ultrasound examination. Recent advances in ultrasound imaging and interpretation have greatly

Classifications of Abruptio Placenta Grade 1: Slight vaginal bleeding and some uterine irritability are usually present. Grade 2: External uterine bleeding is absent to moderate. The uterus is irritable and tetanic or very frequent contractions may be present. Grade 3: Bleeding is moderate to severe but may be concealed. The uterus is tetanic and painful. Maternal hypotension is frequently present and fetal death has occurred.
![Management The potential for rapid deterioration (hemorrhage, disseminated intravascular coagulation [DIC], fetal hypoxia) necessitates Management The potential for rapid deterioration (hemorrhage, disseminated intravascular coagulation [DIC], fetal hypoxia) necessitates](http://slidetodoc.com/presentation_image_h/2d48a874f59aefbb318112ec3a2c3b6e/image-35.jpg)
Management The potential for rapid deterioration (hemorrhage, disseminated intravascular coagulation [DIC], fetal hypoxia) necessitates delivery in some cases of placental abruption. However, most abruptions are small and noncatastrophic, and therefore do not necessitate immediate delivery. Certain actions, including hospitalization, laboratory studies, continuous monitoring, and ongoing patient support should be initiated when placental abruption is suspected

Care for the Patient Experiencing an Abruptio Placentae • Hospitalization • Intravenous placement with a large-bore catheter (16 -gauge) • Labwork: Includes CBC, coagulation studies (fibrinogen, PTT, platelet count, fibrin degradation products), type and screen for 4 units of blood Betamethasone may be given to the woman to promote fetal lung maturity when delivery is not imminent. • Rh(D)-negative patients should receive Rho. GAM to prevent isoimmunization. • Continuous evaluation of intake and output • Continuous electronic fetal monitoring • Delivery (cesarean or vaginal birth) may be initiated depending on the status of the mother and the fetus. • Nursing care is centered on continuous maternal–fetal assessment, with on-going information and emotional support for the patient and her family.

Preterm Labor Preterm labor (PTL) is defined as cervical changes and regular uterine contractions occurring between 20 and 37 weeks of pregnancy. Many patients present with preterm contractions, but only those who demonstrate changes in the cervix are diagnosed with preterm labor (ACOG, 2001).

Various Risk Factors Associated with Preterm Labor and Birth • History of preterm birth • Uterine or cervical anomalies • Multiple gestation • Hypertension • Diabetes • Obesity • Clotting disorders • Infection, especially urinary tract infections • Fetal anomalies • Premature rupture of membranes

• Vaginal bleeding • Late or no prenatal care • Smoking • Alcohol • Domestic violence • Age 17 years or 35 years • Low socioeconomic status • Stress • Long working hours with long periods of standing

Contraindications to the Use of Tocolytics in Preterm Labor • Significant maternal hypertension (eclampsia, severe preeclampsia, chronic hypertension) • Antepartum hemorrhage • Cardiac disease • Any medical or obstetric condition • Hypersensitivity to a specific tocolytic agent • Advanced cervical dilation • Fetal demise or lethal anomaly • Chorioamnionitis • In utero compromise • Acute: nonreassuring fetal heart rate pattern • Chronic: IUGR or substance abuse

Family Teaching Guidelines Preventing Prematurity Freda and Patterson (2004) suggest that nurses be proactive by educating women about preterm labor and teaching them how to recognize the warning signs and symptoms. ◆ Encourage all pregnant women to obtain prenatal care. ◆ Educate all pregnant women as to the signs and symptoms of preterm labor. ◆ Eliminate the term “Braxton-Hicks” from teaching (women may delay seeking treatment if they believe they are only experiencing Braxton-Hicks contractions). ◆ Ask all pregnant women if they have had any symptoms of preterm labor.

◆ Screen for vaginal and urogenital infections and treat appropriately. ◆ Teach women about the dangers of douching. ◆ Assess all pregnant women for intimate partner violence and intervene. ◆ Discuss stress levels early in pregnancy. ◆ Assess all pregnant women for nutritional status and weight gain in pregnancy and intervene as necessary. ◆ Assess for illicit drug use, and help the woman get into treatment. ◆ Encourage women who have preterm labor symptoms to drink fl uids, lie down for 1 hour, and go to the hospital for a vaginal exam if symptoms continue. ◆ Remind the woman with symptoms that she should not hesitate to call her provider repeatedly if her symptoms recur.

Premature Rupture of the Membranes To facilitate an understanding of premature rupture of the membranes (PROM), it is helpful to first define the various terms used: • Premature rupture of the membranes (PROM) is defi ned as rupture of the membranes before the onset of labor at any gestational age. • Preterm rupture of membranes is defined as rupture of the membranes before 37 completed weeks of gestation. It is a common cause of preterm labor, preterm delivery, and chorioamnionitis. • Preterm premature rupture of the membranes (PPROM) is defined as a combination of both terms— rupture occurs before the 37 th completed week of gestation and in the absence of labor. PPROM accounts for 25% of all cases of premature rupture of the amniotic membranes and is respon sible for 30% to 40% of all preterm deliveries (Cunningham et al. , 2005).

PATHOPHYSIOLOGY Premature rupture of the membranes is multifactorial. Choriodecidual infection or inflammation appears to play an important role in the etiology of PPROM, especially at early gestational ages. Other factors include decreased amniotic membrane collagen, lower socioeconomic status, cigarette smoking, sexually transmitted infections, prior preterm delivery, prior preterm labor during the current pregnancy, uterine distention (e. g. , multiple gestation, hydramnios), cervical cerclage, amniocentesis, and vaginal bleeding in pregnancy (Mercer, 2003). In many cases, the cause is not known.

DIAGNOSIS The diagnosis is based on the patient’s history of leaking vaginal fluid and the finding of a pooling of fluid on sterile speculum examination. Nitrazine and fern tests confirm the diagnosis of PROM. The level of amniotic fluid in the uterus may also be checked via an abdominal ultrasound examination. Leakage of amniotic fl uid is consistent with findings of oligohydramnios (decreased amniotic fluid).

MANAGEMENT • Gestational age should be established based on clinical history and prior ultrasound assessment when available. • Ultrasound should be performed to assess fetal growth, position, and residual amniotic fluid. The woman should be assessed for evidence of advanced labor, chorioamnionitis (intrauterine infection), abruptio placentae, and fetal distress.

• Patients with advanced labor, intrauterine infection, significant vaginal bleeding, or nonreassuring fetal testing are best delivered promptly, regardless of gestational age. There is further debate over the use of tocolytics, corticosteroids, and antibiotics in patients with PPROM. Tocolysis appears to be of little benefit in PPROM and may be harmful when chorioamnionitis is present. However, in many hospitals, tocolytic therapy is instituted for 48 hours, especially with earlier gestational ages, in order to administer a course of corticosteroids to enhance fetal lung maturity (Cunningham et al. , 2005).

Conservative management includes in patient observation unless the membranes reseal and the leakage of fluid stops. This approach initially consists of prolonged continuous fetal and maternal monitoring combined with modified bed rest to promote amniotic fluid accumulation and spontaneous membrane sealing. Delivery of the fetus should be accomplished if signs of infection are present: maternal temperature of 100. 4°F (38°C) or greater, foul-smelling vaginal discharge, elevated white blood count, uterine tenderness, and maternal and/or fetal tachycardia.

Without intervention, approximately 50% of patients who have ROM will go into labor within 24 hours. maintaining the pregnancy to gain further fetal maturity can be beneficial, prolonged PPROM has been correlated with an increased risk of v Chorioamnionitis v placental abruption v cord prolapse

Nursing interventions explaining to the patient that she will be on: v full or modified bed rest v vital signs will be checked at least every 4 hours to detect early signs of a developing infection v intermittent fetal monitoring is appropriate v Frequent ultrasound examinations are performed to assess amniotic v providing emotional support to the patient v encourage the woman and her family members to ask questions and express fears and concerns. v The woman should be assessed for evidence of advanced labor, chorioamnionitis (intrauterine infection), abruptio placentae, and fetal distress.

Hypertensive Disorders of Pregnancy Hypertensive disorders are the most common medical complication of pregnancy. The incidence of hypertensive disorders is between 5% and 10%, and this complication is the second leading cause of maternal death in the United States (embolic events are the leading cause) (Martin et al. , 2005). Hypertensive disorders contribute significantly to stillbirth and neonatal morbidity and mortality and can result in: § maternal cerebral hemorrhage § disseminated intravascular coagulation (DIC) § hepatic failureacute renal failure § pulmonary edema § adult respiratory distress syndrome § aspiration pneumonia § abruptio placentae (ACOG, 2002 a).

CLASSIFICATIONS AND DEFINITIONS Numerous attempts have been made to accurately describe pregnancy-related hypertensive disorders. Them NHBPEP, 2000 has recommended the following classifications: • Chronic hypertension • Preeclampsia–eclampsia • Chronic hypertension with superimposed preeclampsia • Gestational (or transient) hypertension

Chronic Hypertension Chronic hypertension is defined as hypertension that is present and observable before pregnancy or hypertension that is diagnosed before the 20 th week of gestation. Hypertension is defined as a blood pressure greater than 140/90 mm Hg. Hypertension for which a diagnosis is confirmed for the first time during pregnancy, and which persists beyond the 84 th day postpartum, is also classified as chronic hypertension (Roberts, 2004). Preeclampsia and Eclampsia Preeclampsia is a pregnancy-specific systemic syndrome clinically defined as an increase in blood pressure (140/90) after 20 weeks’ gestation accompanied by proteinuria (NHBPEP, 2000; Peters & Flack, 2004). This increase in blood pressure represents a change from the usual blood pressure findings during pregnancy. Under normal conditions, the blood pressure increases during the first trimester, decreases in the second trimester, and

Chronic Hypertension with Superimposed Preeclampsia According to Roberts (2004), the following criteria are necessary to establish a diagnosis of superimposed preeclampsia: 1. Hypertension and no proteinuria early in pregnancy (prior to 20 weeks’ gestation) and new-onsetproteinuria, (defined as the urinary excretion of 0. 3 g of protein in a 24 -hour specimen) 2. 3. 4. 5. Hypertension and proteinuria before 20 weeks’ gestation: • A sudden increase in protein—urinary excretion of 0. 3 g protein or more in a 24 -hour specimen, or two dipstick test results of 2 (100 mg/d. L), with no evidence of urinary tract infection A sudden increase in blood pressure in a woman whose blood pressure has been well Controlled Thrombocytopenia (platelet count lower than 100, 000/mm 3) An increase in the liver enzymes alanine transaminase (ALT) or aspartate transaminase (AST) to abnormal levels

Gestational (or Transient) Hypertension This is a non specific term used to describe the woman who has a blood pressure elevation detected for the first time during pregnancy, without proteinuria. This term is used only until a more specific diagnosis can be assigned postpartum. If preeclampsia does not develop (e. g. , protein does not become present in the urine), and the woman’s blood pressure falls into a normal range by 12 weeks postpartum, the diagnosis of transient hypertension of pregnancy can be made (Roberts, 2004).

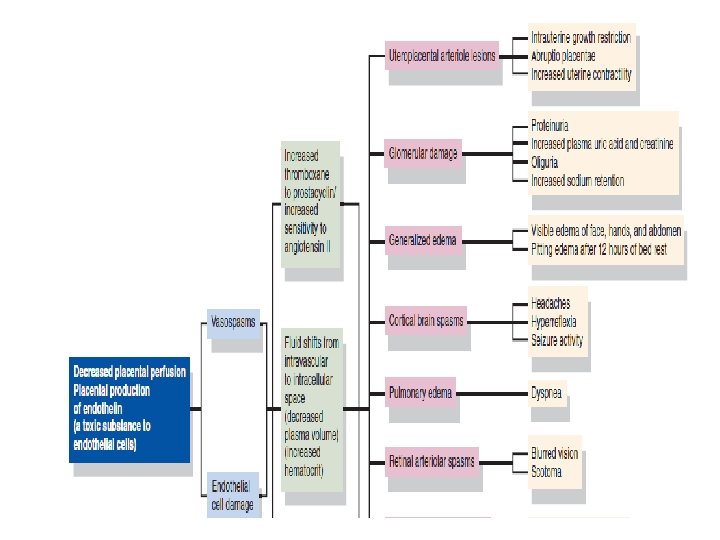
Preeclampsia PATHOPHYSIOLOGY The normal physiological adaptations to pregnancy are altered in the woman who develops preeclampsia. Preeclampsia is a multisystem, vasopressive disease process that targets the cardiovascular, hematologic, hepatic, renal, and central nervous systems. Preeclampsia is associated with a clinical spectrum of events that range from mild to severe with a potential endpoint of eclampsia. Patients do not suddenly “catch” severe preeclampsia or develop eclampsia but rather progress in a fairly predictable course through the clinical spectrum. In most cases, the progression is relatively slow, and the disorder may never proceed beyond mild preeclampsia. in other situations, the disease can progress more rapidly, and change from a mild to a severe form in a matter of days or weeks. In the most serious cases, the progression can be rapid: mild preeclampsia evolves to severe preeclampsia or eclampsia over hours or days (Roberts, 2004). Hence, the nurse must alert the patient to signs and symptoms that signal a worsening condition and continuously assess the patient for any change.
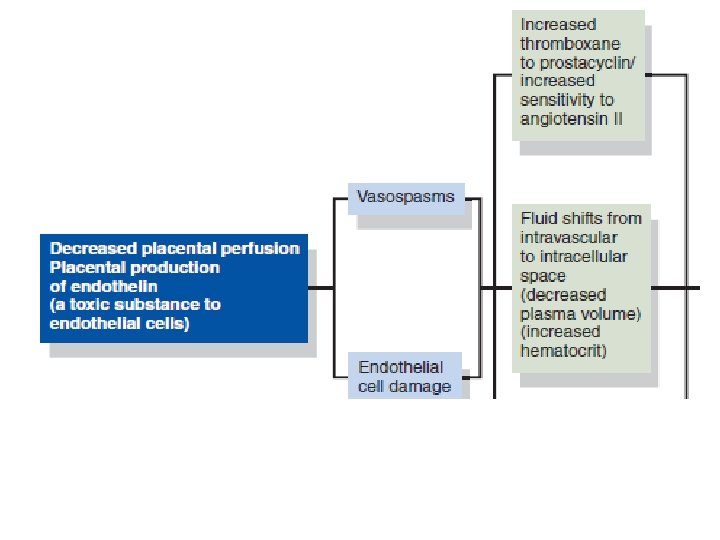
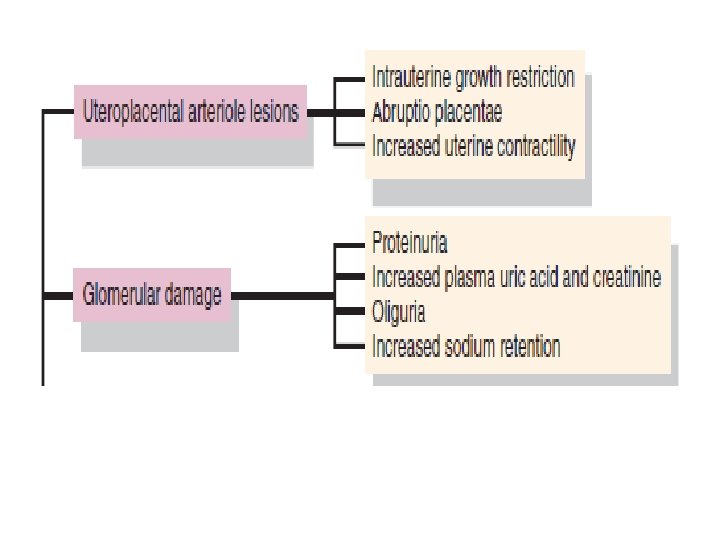

Risk Factors for Preeclampsia • Primigravida (6– 8 times greater risk) • Age extremes (17 years and 35 years) • Diabetes • Preexisting hypertension • Multiple gestation (5 times greater risk) • Fetal hydrops (10 times greater risk) • Hydatidiform mole (10 times greater risk) • Preeclampsia in a previous pregnancy • Family history • Obesity • Immunological factors • Chronic renal disease • Rh incompatibility • African-American ethnicity

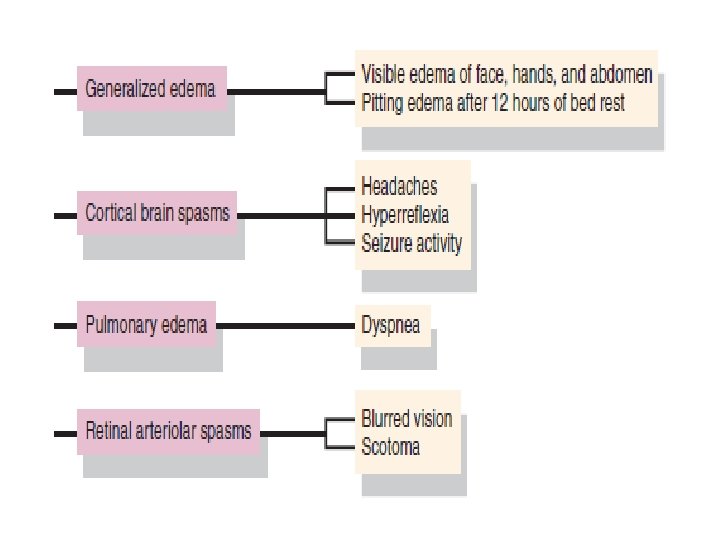
Home Management of the Pregnant Patient with a Hypertensive Disorder Before discharge, it is important to ascertain that the home environment is conducive to rest and the patient will be able to rest frequently throughout the day. It is essential that the patient can verbalize understanding of the importance of keeping all prenatal appointments and that she must immediately notify her physician or midwife at the first appearance of: ◆ higk Blood pressure ◆ Visual changes ◆ Epigastric pain ◆ Nausea and vomiting ◆ Bleeding gums ◆ Headaches ◆ Increasing edema, especially of the hands and face ◆ Decreasing urinary output ◆ Decreased fetal movement ◆ “Just not feeling right”
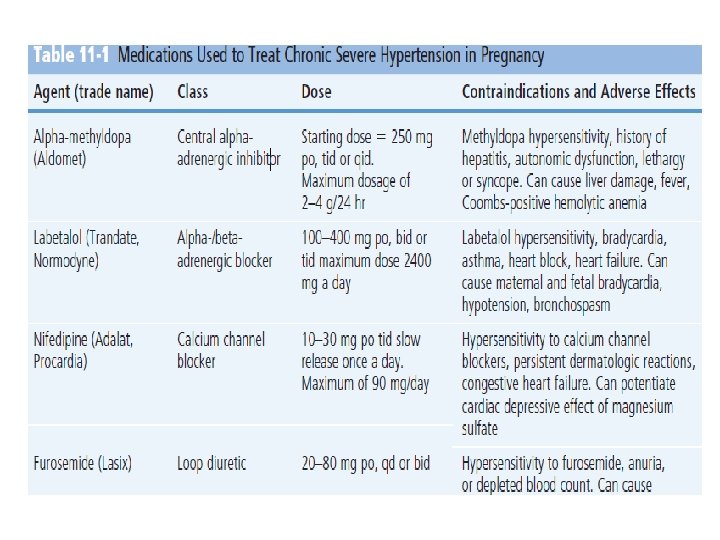


Nursing Implications: Remember that this is a very potent, high alert drug! Explain purpose and side effects of the medication to the patient and her companion. Explain that she may feel very warm and become fl ushed and experience nausea and vomiting, visual blurring, and headaches. Magnesium sulfate must never be abbreviated (i. e. , Mg. SO 4 is not acceptable) and requires a written order by the physician for administration. Always use an infusion pump for administration and run the medication piggyback, not as the main line. Monitor pulse, blood pressure, respirations, and ECG frequently throughout parenteral administration. Respirations should be at least 16/min before each dose. Monitor neurological status before and throughout therapy. Institute seizure precautions Keep the room quiet and darkened to decrease the likelihood of triggering seizure activity Patellar reflexes should be tested before each parenteral dose of magnesium sulfate. If absent, no additional dose should be administered until a positive response returns. Monitor intake and output. Urine output should be maintained at a level of at least 100 m. L/4 hr.

• Correct maternal acidemia. Blood gas analysis allows monitoring of oxygenation and p. H status. Respiratory acidemia is possible after a seizure. • Avoid polytherapy. Maternal respiratory depression, respiratory arrest, or cardiopulmonary arrest is more likely in women who receive polytherapy to arrest a seizure. Remember that anticonvulsants are respiratory depressants and may interact. • Be sure to check the fetus or fetuses (all must be accounted for). After a seizure there may be loss of FHR variability and bradycardia on the fetal monitoring tracing. • Check the patient for ruptured membranes, contractions, and cervical dilation. • Prepare for delivery as indicated. • Support the patient and her family. This is a very frightening event for them and they will need reassurance and to be kept aware of the plan of care and the well-being of their baby

critical nursing action Care of the Pregnant Patient Post-seizure • Do not attempt to shorten or abolish the initial seizure. Attempts to administer anticonvulsants intravenously without secure venous access can lead to phlebitis and venous thrombosis. • Prevent maternal injury. • Maintain adequate oxygenation; administer oxygen via face mask at 10 L/min • Minimize the risk of aspiration. Position the patient on her side to facilitate drainage. Suction equipment should be ready and working. • Give adequate magnesium sulfate to control seizures. As soon as possible following the seizure, venous access should be secured with a 4 - to 6 -g loading bolus of magnesium sulfate given over 15– 20 minutes. If the patient seizes following the loading dose, another 2 -g bolus may be given intravenously, over 3– 5 minutes. •

Correct maternal acidemia. Blood gas analysis allows monitoring of oxygenation and p. H status. Respiratory acidemia is possible after a seizure. • Avoid polytherapy. Maternal respiratory depression, respiratory arrest, or cardiopulmonary arrest is more likely in women who receive polytherapy to arrest a seizure. Remember that anticonvulsants are respiratory depressants and may interact. • Be sure to check the fetus or fetuses (all must be accounted for). After a seizure there may be loss of FHR variability and bradycardia on the fetal monitoring tracing. • Check the patient for ruptured membranes, contractions, and cervical dilation. • Prepare for delivery as indicated. • Support the patient and her family. This is a very frightening event for them and they will need reassurance and to be kept aware of the plan of care and the well-being of their baby •

HELLP Syndrome HELLP is an acronym for: Hemolysis, Elevated Liver enzymes and Low Platelets Due to the arteriolar vasospasms in the cardiovascular system that occur in preeclampsia, the circulating red blood cells (RBCs) are destroyed as they try to navigate through the constricted vessels (Hemolysis). Vasospasms decrease blood fl ow to the liver, resulting in tissue ischemia and hemorrhagic necrosis (Elevated Liver enzymes). In response to the endothelial damage caused by the vasospasms (small openings develop in the vessels), platelets aggregate at the site and a fi brin network is set up, leading to a decrease in the circulating platelets (Low Platelets). HELLP syndrome is a serious complication of preeclampsia that can manifest itself at any time during pregnancy and the puerperium, but like preeclampsia, it is rare before 20 weeks’ gestation. However, unlike preeclampsia, HELLP syndrome occurs more often in Caucasians, multiparas, and in women older than 35 years. One third of all cases of HELLP syndrome occur during postpartum, and only 80% of these patients are diagnosed with preeclampsia before delivery (Sibai et al. , 2005).

HELLP syndrome is actually a laboratory diagnosis for a variant of severe preeclampsia. The primary presentation is consistent with hepatic dysfunction evidenced by fi ndings from the patient’s liver function tests (ACOG, 2002 a; Poole, 2004 b). HELLP syndrome is characterized by rapidly deteriorating liver function and thrombocytopenia. Liver capsule distention often produces epigastric pain. Though rare, liver rupture is one of the most ominous consequences of severe preeclampsia/HELLP syndrome, with a reported maternal death rate of more than 30%. The precise cause of liver rupture is unknown, but the prevailing theory postulates that the increased hepatic pressure leads to rupture. It is theorized that endothelial dysfunction with intravascular fi brin deposits and hepatic sinusoidal obstruction leads to intrahepatic vascular congestion, increased intrahepatic pressure, and distention of Glisson’s capsule. This pathological process progresses tothe development of a subcapsular hepatic hematoma and subsequent liver rupture (ACOG, 2002 a; Cunningham et al. , 2005).

Therapy for HELLP syndrome centers on improving the platelet count by transfusion of fresh-frozen plasma or platelets and delivery as soon as feasible by vaginal or cesarean birth. Intrapartum nursing care involves continuous maternal–fetal monitoring. Measurement of central venous pressure or pulmonary arterial wedge pressure (Swan–Ganz catheter) may be required to monitor fl uid status accurately when pulmonary edema or acute renal failure are present (ACOG, 2002 a).

THANKS A LOT
- Slides: 72