Dr Andrew Clark Associate Professor in Synthetic Chemistry


![Ln([Ao]/[A]) Kinetics to 80% Conversion (PS-PMDETA. Cu. Cl) Time (hrs) A. J. Clark, J. Ln([Ao]/[A]) Kinetics to 80% Conversion (PS-PMDETA. Cu. Cl) Time (hrs) A. J. Clark, J.](https://slidetodoc.com/presentation_image/ad7683af6c6aa6a594cb26aa158fc126/image-14.jpg)



- Slides: 65

Dr. Andrew Clark Associate Professor in Synthetic Chemistry Research areas Natural product isolation and total synthesis. Chemistry and biology of free radicals Development of synthetic methodology using copper, iron and ruthenium Functional Genomics / Chemical Genetics / Interactomics Use of plants in renewable plastics manufacture

Dr. Andrew Clark Chemistry Department, University of Warwick Coventry, UK +44 24 76523242 msrir@csv. warwick. ac. uk

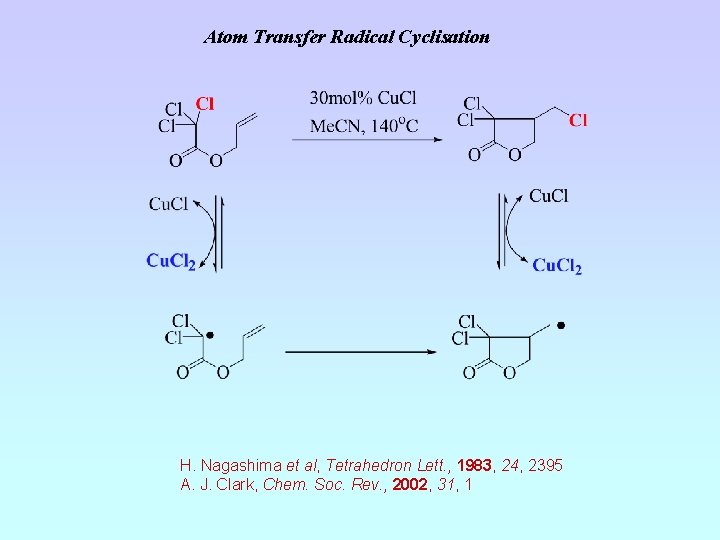
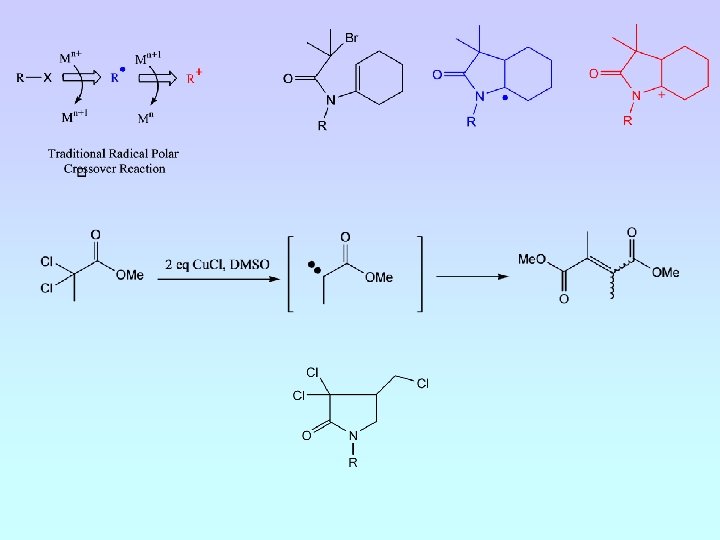
Atom Transfer Radical Cyclisation H. Nagashima et al, Tetrahedron Lett. , 1983, 24, 2395 A. J. Clark, Chem. Soc. Rev. , 2002, 31, 1

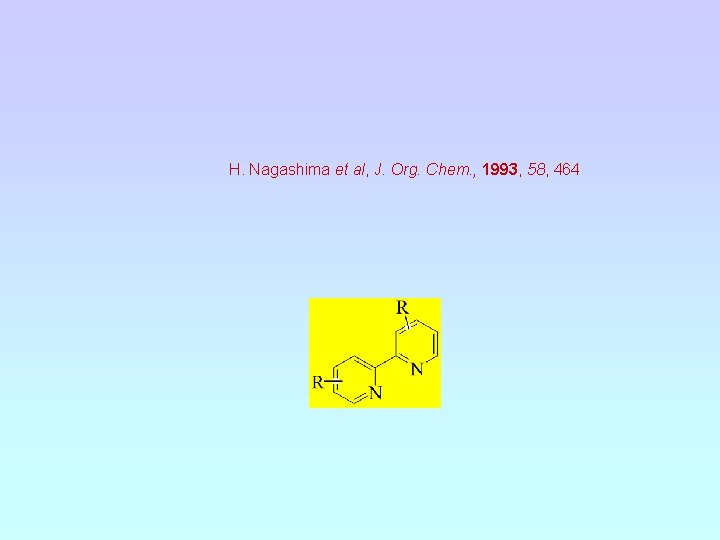
H. Nagashima et al, J. Org. Chem. , 1993, 58, 464


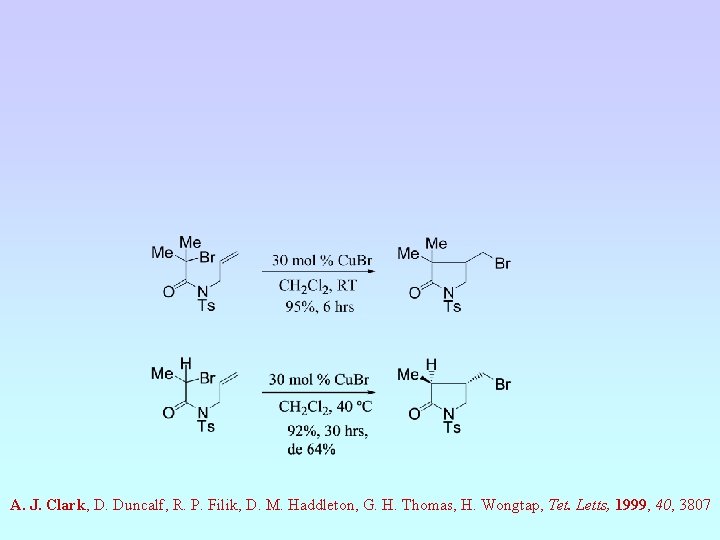
A. J. Clark, D. Duncalf, R. P. Filik, D. M. Haddleton, G. H. Thomas, H. Wongtap, Tet. Letts, 1999, 40, 3807

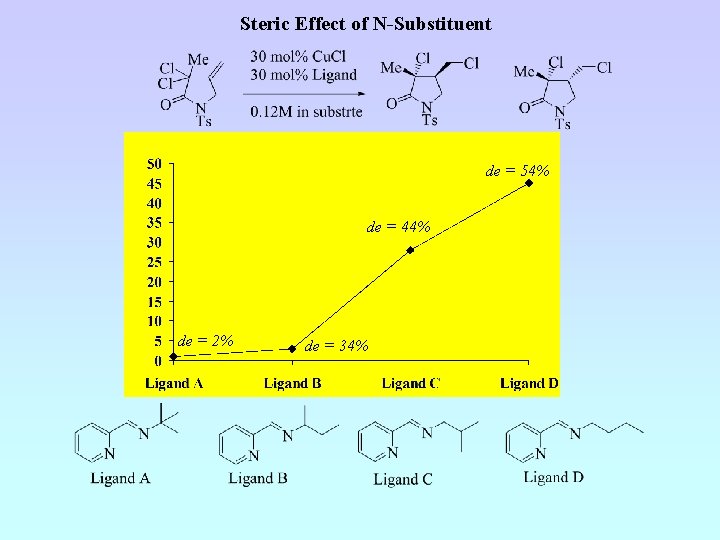
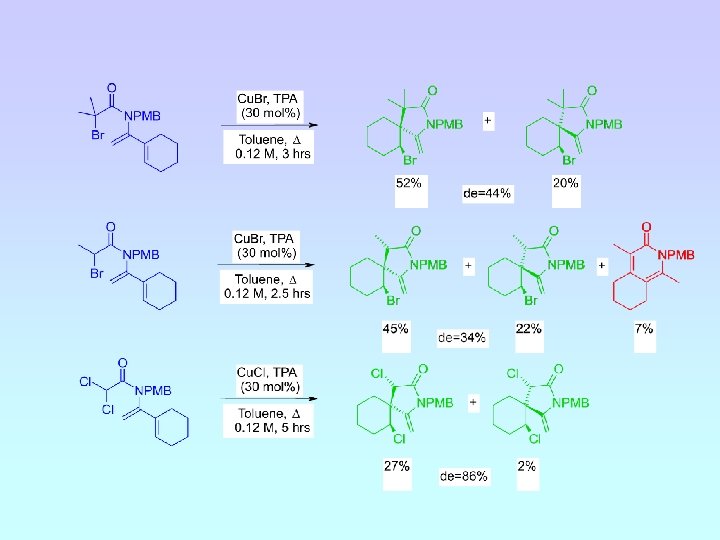
Steric Effect of N-Substituent de = 54% de = 44% de = 2% de = 34%

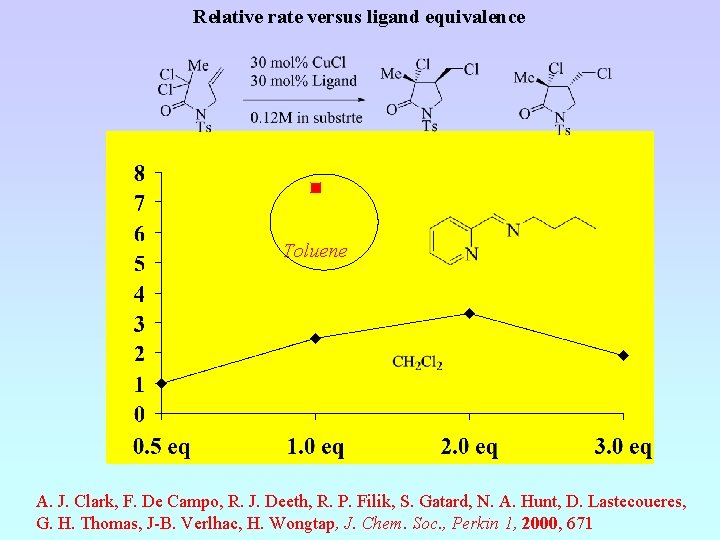
Relative rate versus ligand equivalence Toluene A. J. Clark, F. De Campo, R. J. Deeth, R. P. Filik, S. Gatard, N. A. Hunt, D. Lastecoueres, G. H. Thomas, J-B. Verlhac, H. Wongtap, J. Chem. Soc. , Perkin 1, 2000, 671

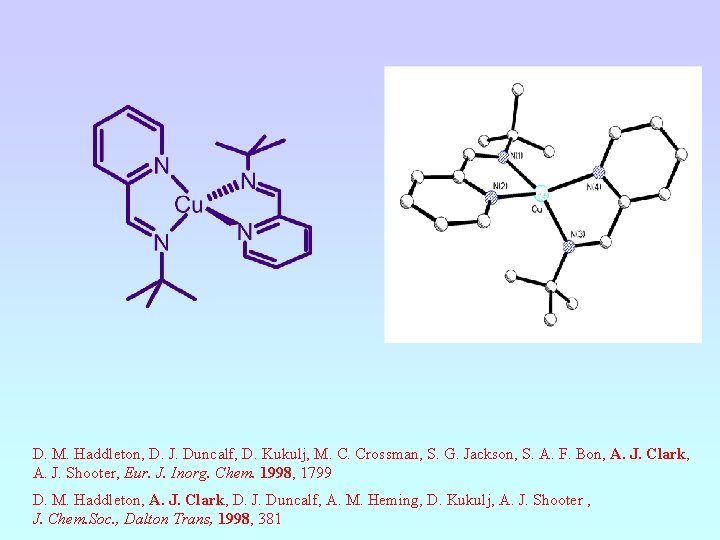
D. M. Haddleton, D. J. Duncalf, D. Kukulj, M. C. Crossman, S. G. Jackson, S. A. F. Bon, A. J. Clark, A. J. Shooter, Eur. J. Inorg. Chem. 1998, 1799 D. M. Haddleton, A. J. Clark, D. J. Duncalf, A. M. Heming, D. Kukulj, A. J. Shooter , J. Chem. Soc. , Dalton Trans, 1998, 381

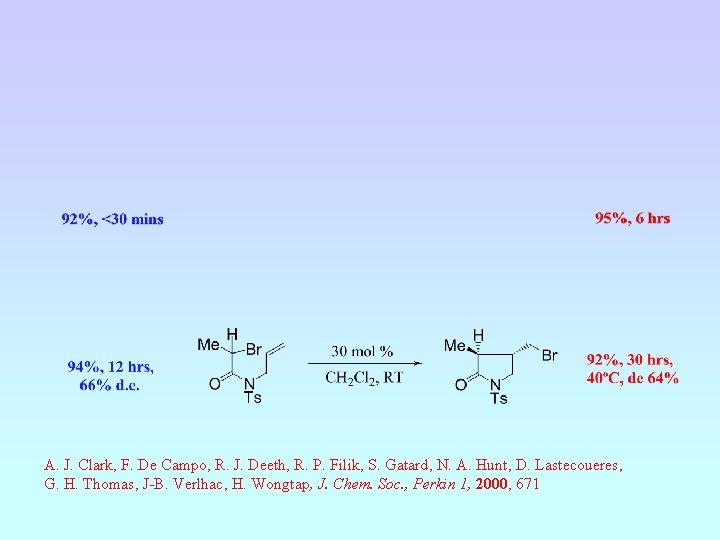
A. J. Clark, F. De Campo, R. J. Deeth, R. P. Filik, S. Gatard, N. A. Hunt, D. Lastecoueres, G. H. Thomas, J-B. Verlhac, H. Wongtap, J. Chem. Soc. , Perkin 1, 2000, 671

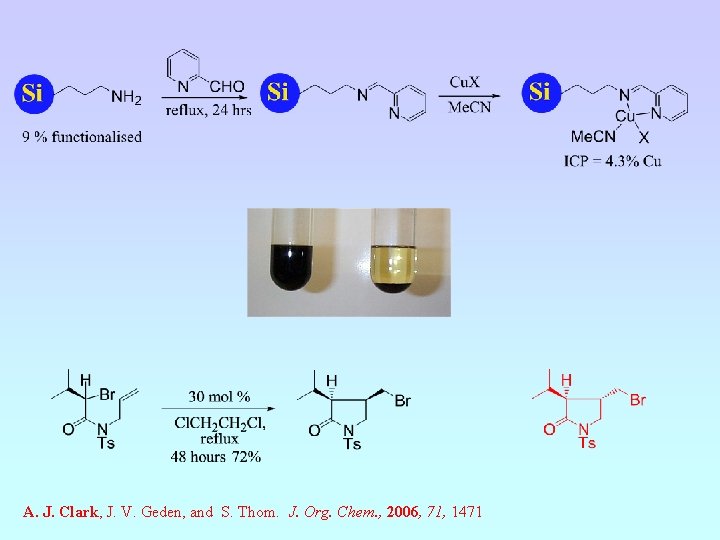
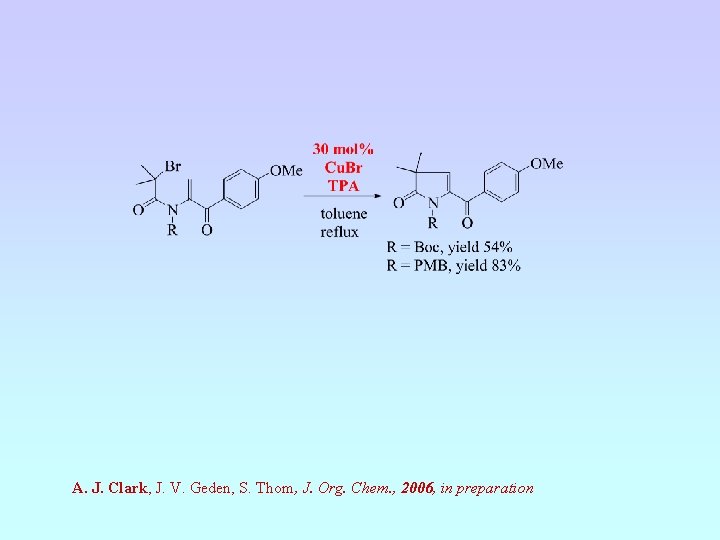
A. J. Clark, J. V. Geden, and S. Thom. J. Org. Chem. , 2006, 71, 1471

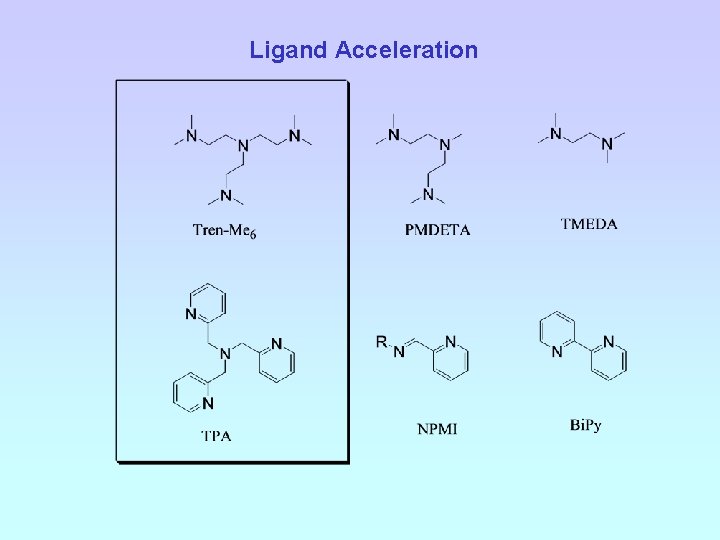
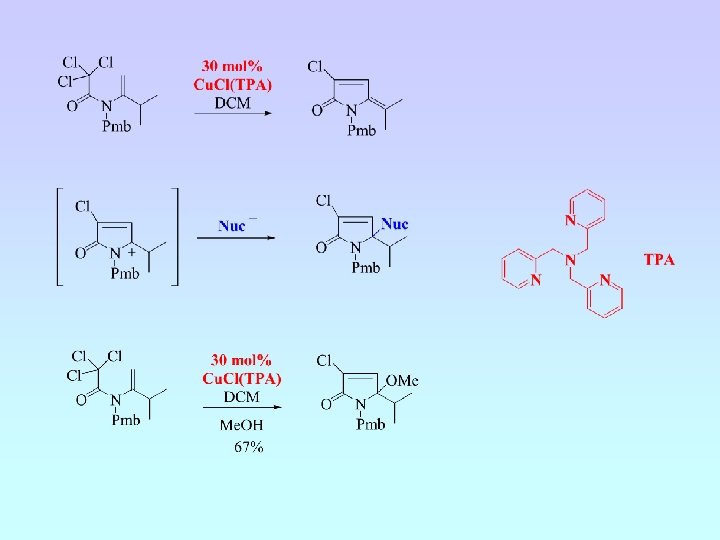
Ligand Acceleration

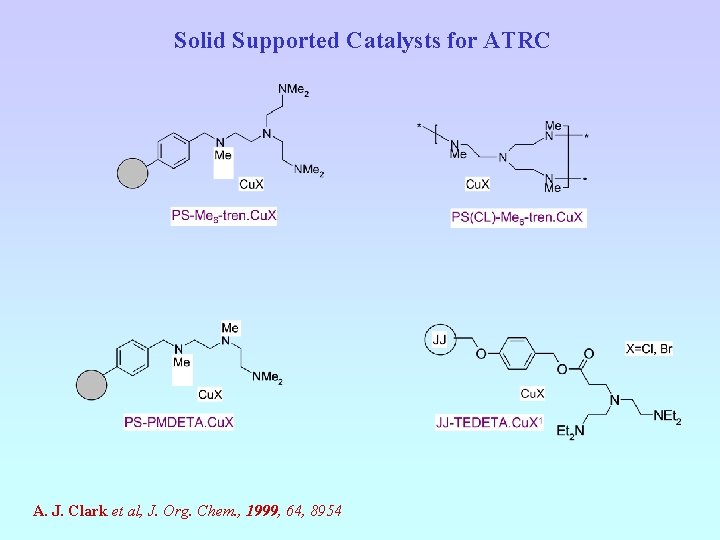
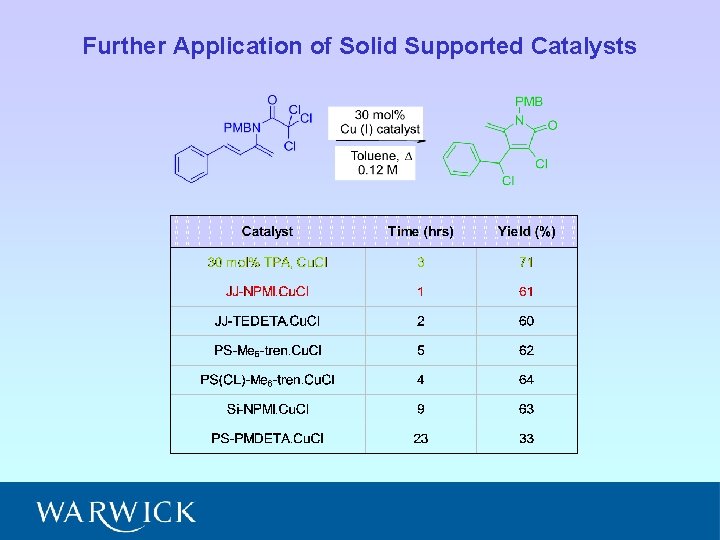
Solid Supported Catalysts for ATRC A. J. Clark et al, J. Org. Chem. , 1999, 64, 8954
![LnAoA Kinetics to 80 Conversion PSPMDETA Cu Cl Time hrs A J Clark J Ln([Ao]/[A]) Kinetics to 80% Conversion (PS-PMDETA. Cu. Cl) Time (hrs) A. J. Clark, J.](https://slidetodoc.com/presentation_image/ad7683af6c6aa6a594cb26aa158fc126/image-14.jpg)
Ln([Ao]/[A]) Kinetics to 80% Conversion (PS-PMDETA. Cu. Cl) Time (hrs) A. J. Clark, J. V. Geden, and S. Thom. J. Org. Chem. , 2006, 71, 1471

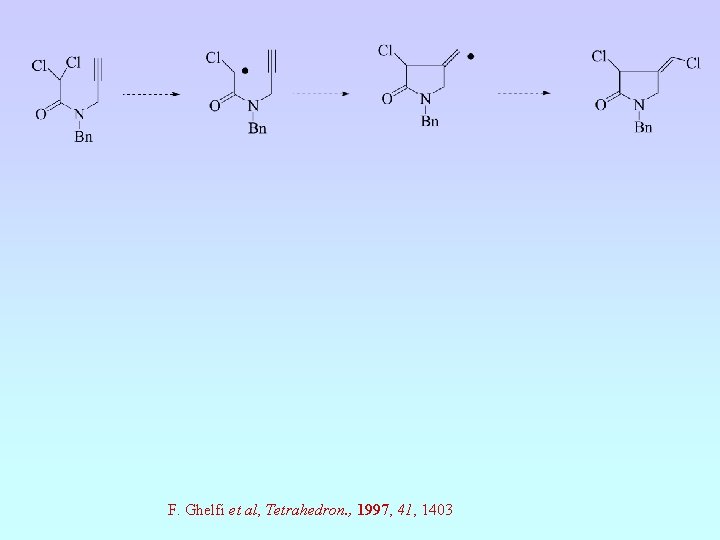
F. Ghelfi et al, Tetrahedron. , 1997, 41, 1403

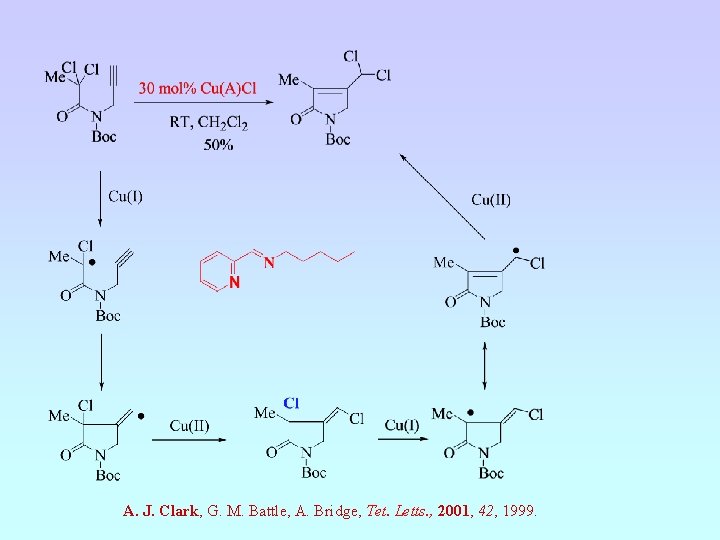
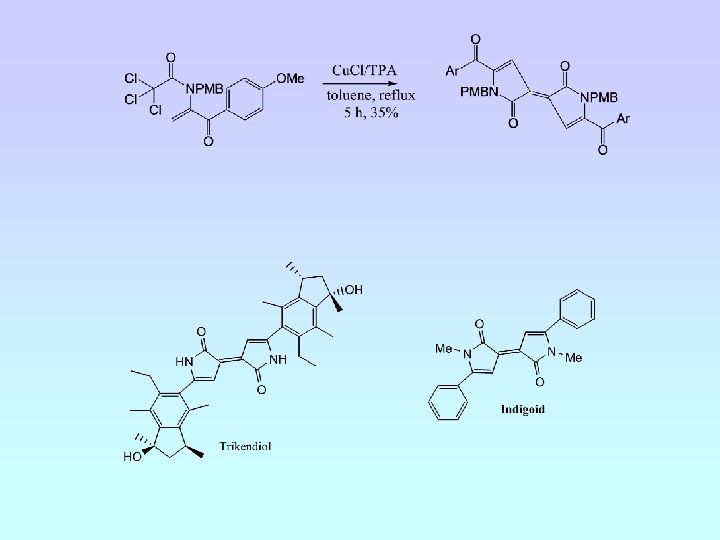
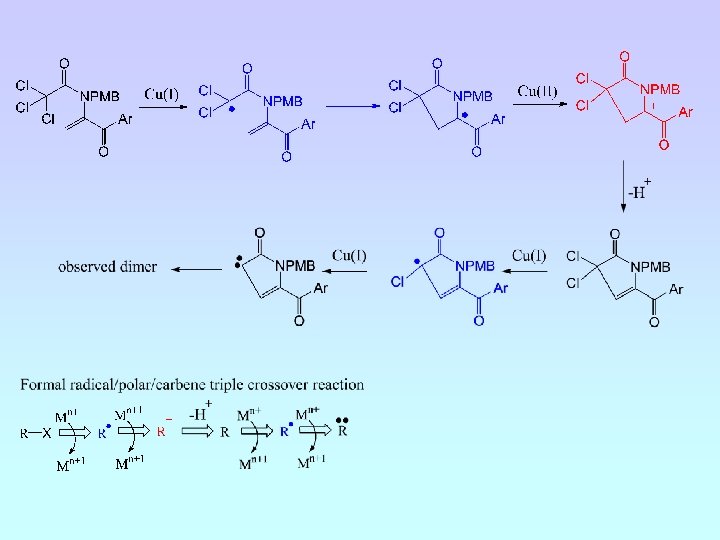
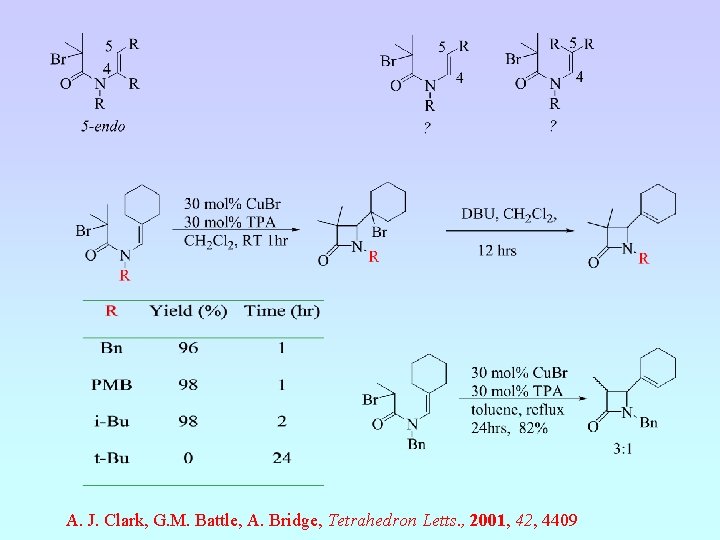
A. J. Clark, G. M. Battle, A. Bridge, Tet. Letts. , 2001, 42, 1999.

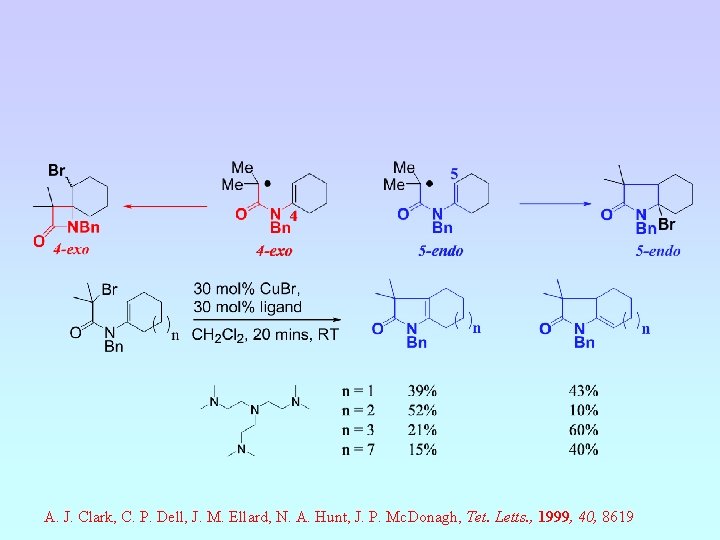
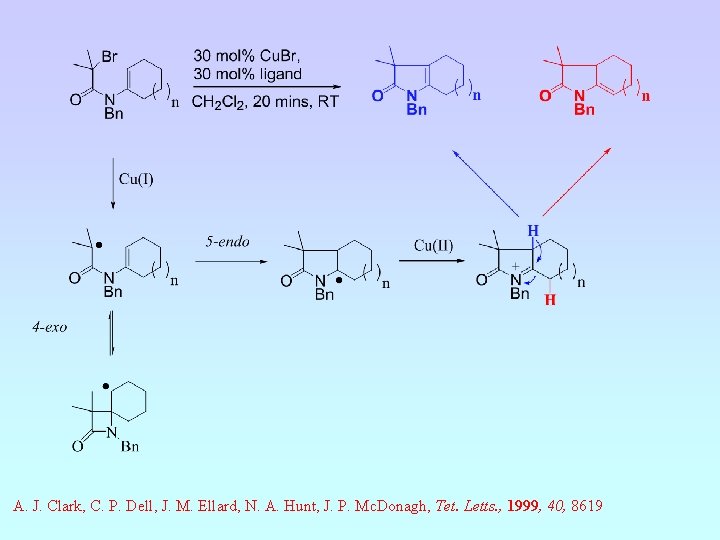
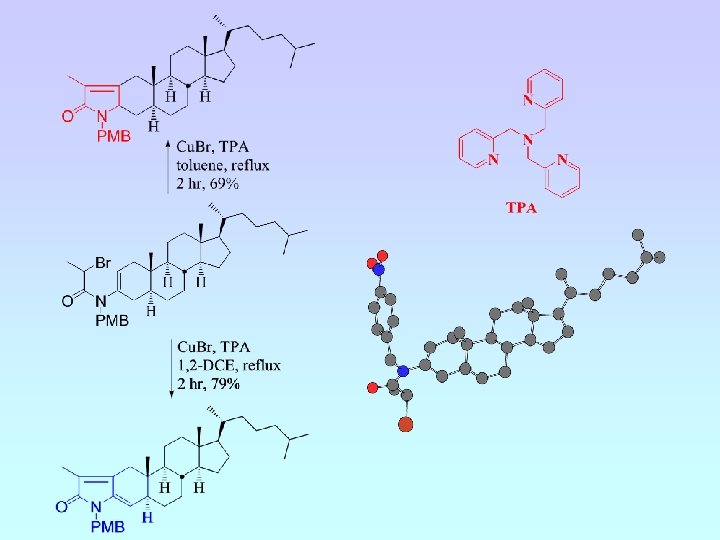
A. J. Clark, C. P. Dell, J. M. Ellard, N. A. Hunt, J. P. Mc. Donagh, Tet. Letts. , 1999, 40, 8619

A. J. Clark, C. P. Dell, J. M. Ellard, N. A. Hunt, J. P. Mc. Donagh, Tet. Letts. , 1999, 40, 8619


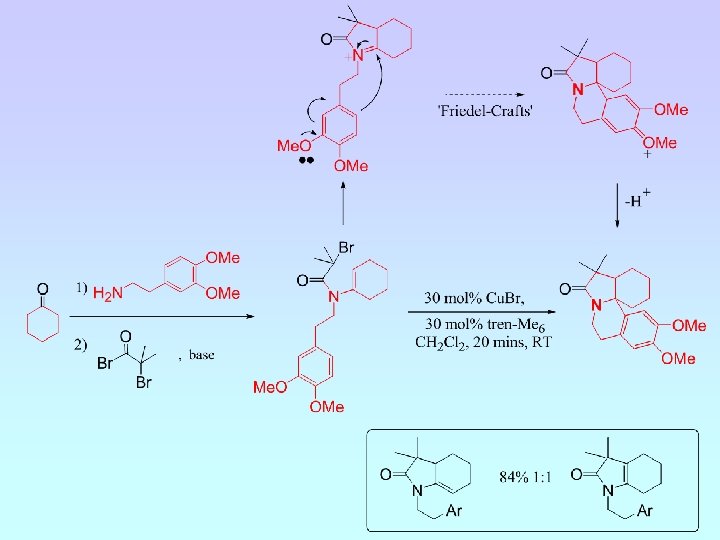
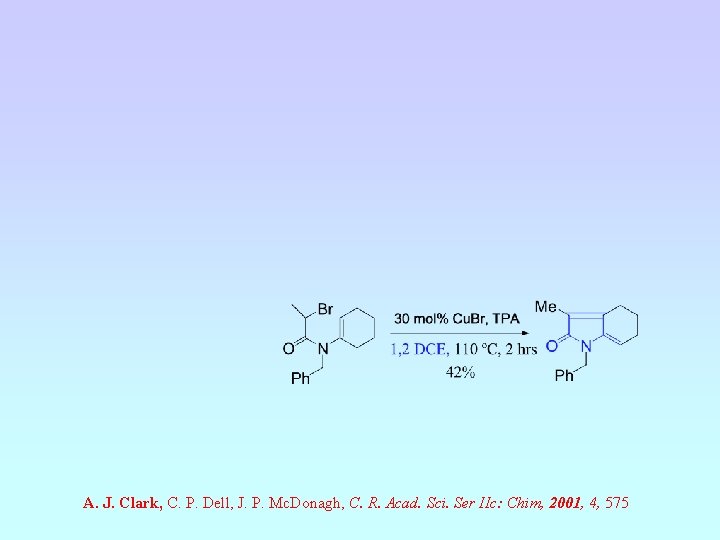
A. J. Clark, C. P. Dell, J. P. Mc. Donagh, C. R. Acad. Sci. Ser IIc: Chim, 2001, 4, 575


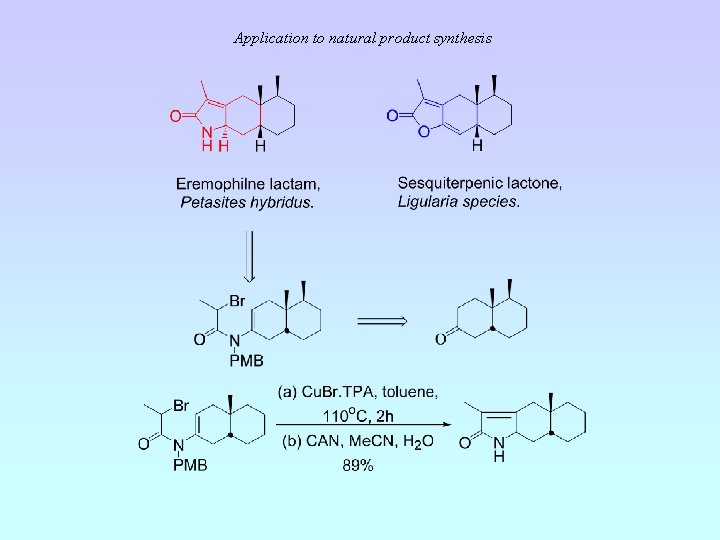
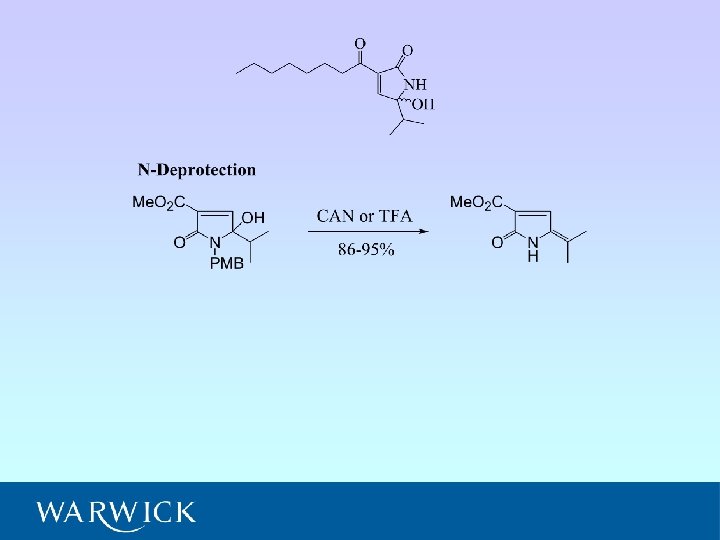
Application to natural product synthesis


A. J. Clark, J. V. Geden, S. Thom, J. Org. Chem. , 2006, in preparation



A. J. Clark, G. M. Battle, A. Bridge, Tetrahedron Letts. , 2001, 42, 4409










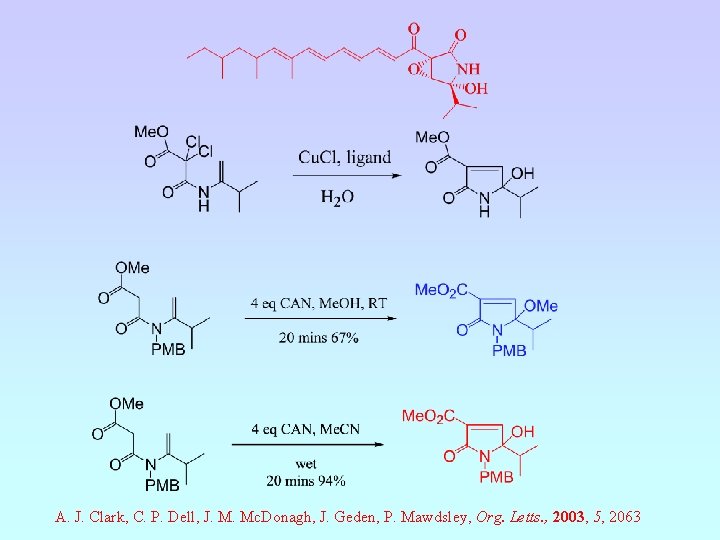
A. J. Clark, C. P. Dell, J. M. Mc. Donagh, J. Geden, P. Mawdsley, Org. Letts. , 2003, 5, 2063


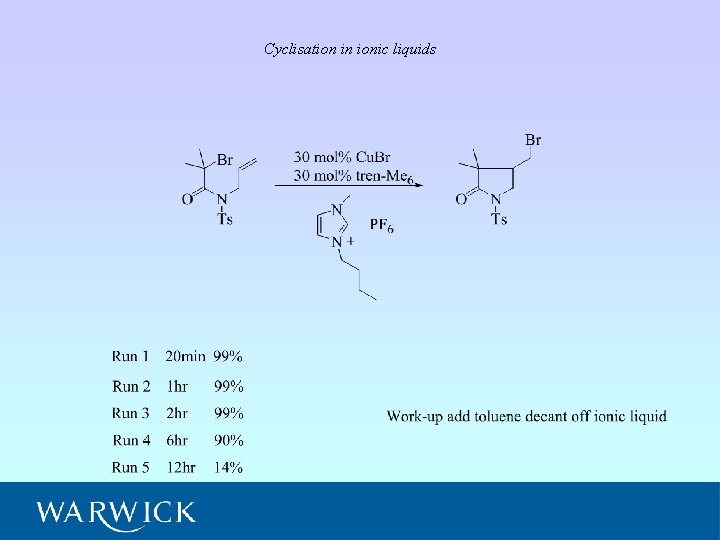
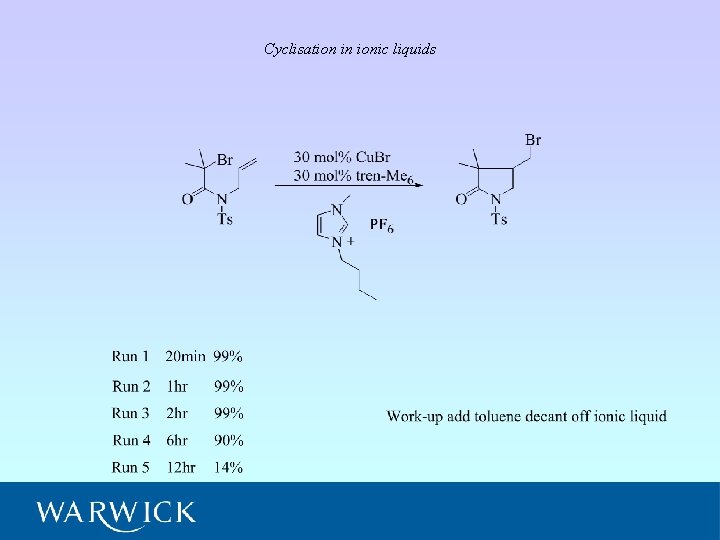
Cyclisation in ionic liquids

Further Application of Solid Supported Catalysts

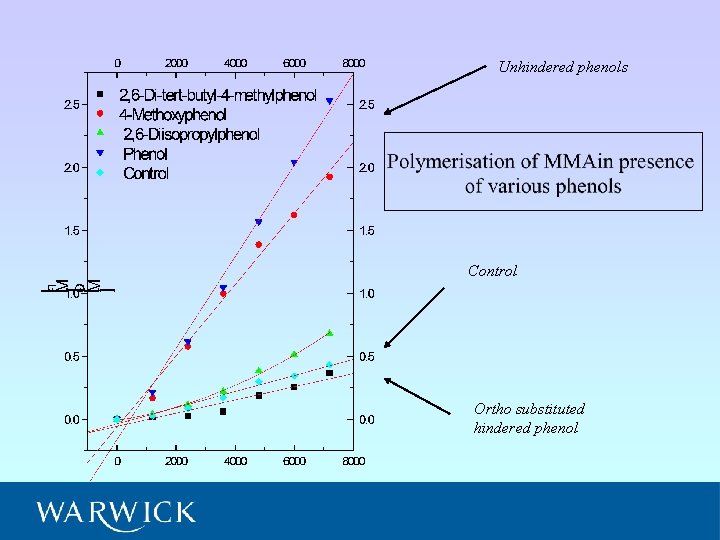
Unhindered phenols Control Ortho substituted hindered phenol

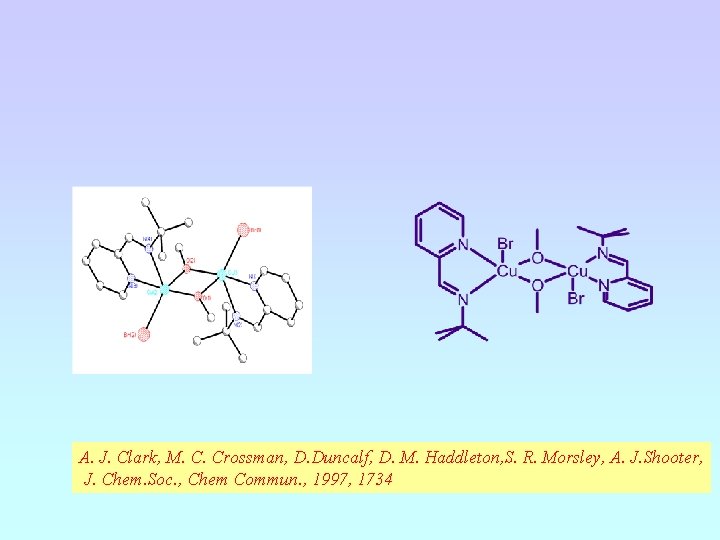
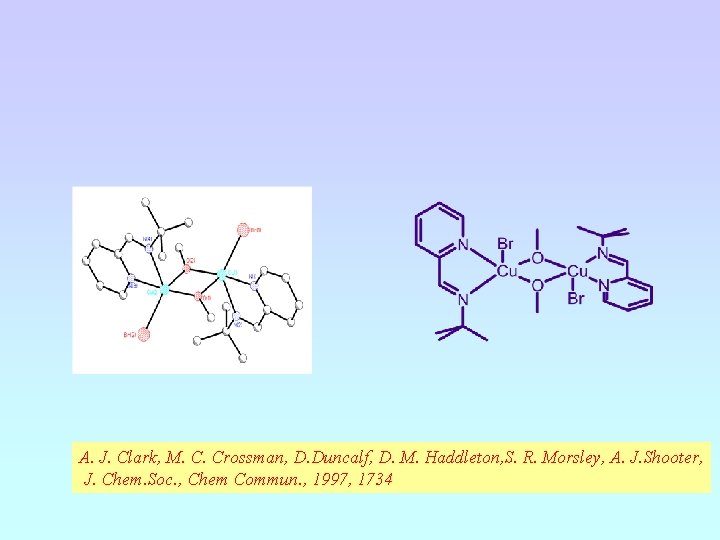
A. J. Clark, M. C. Crossman, D. Duncalf, D. M. Haddleton, S. R. Morsley, A. J. Shooter, J. Chem. Soc. , Chem Commun. , 1997, 1734

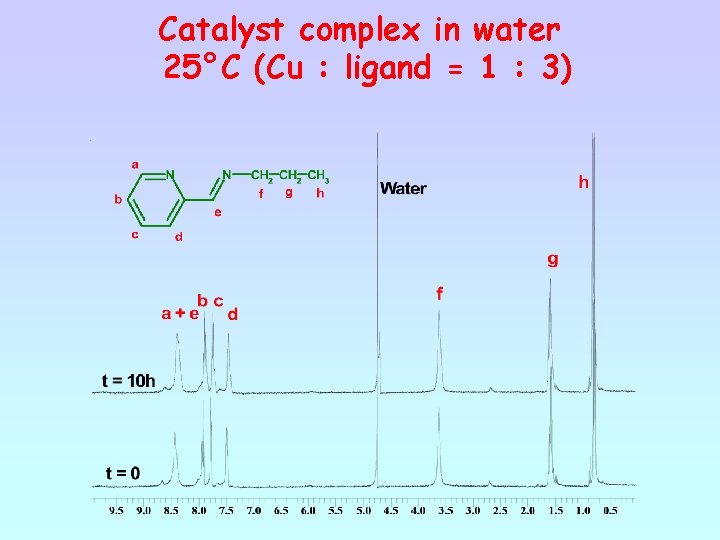
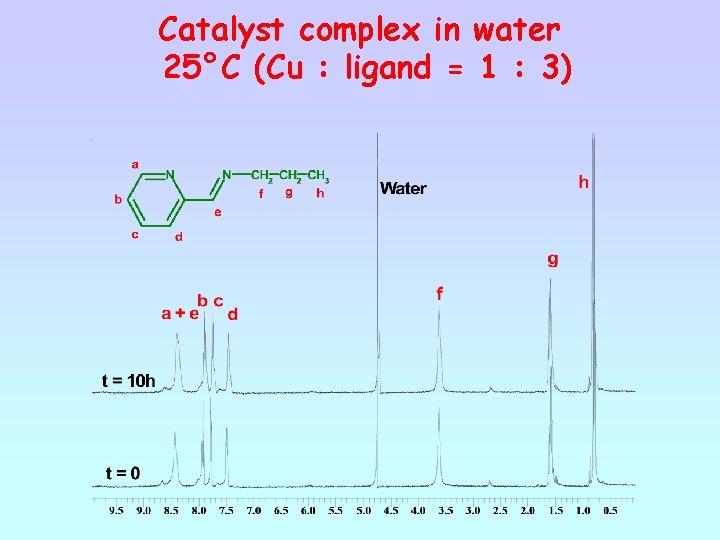
Catalyst complex in water 25°C (Cu : ligand = 1 : 3)

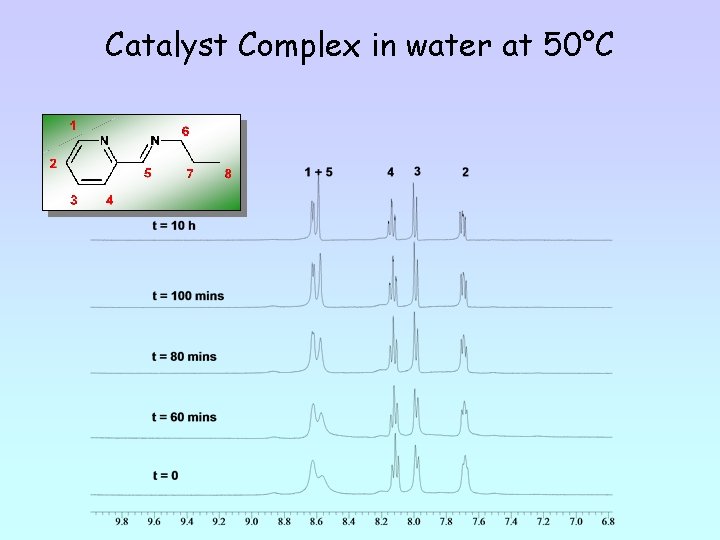
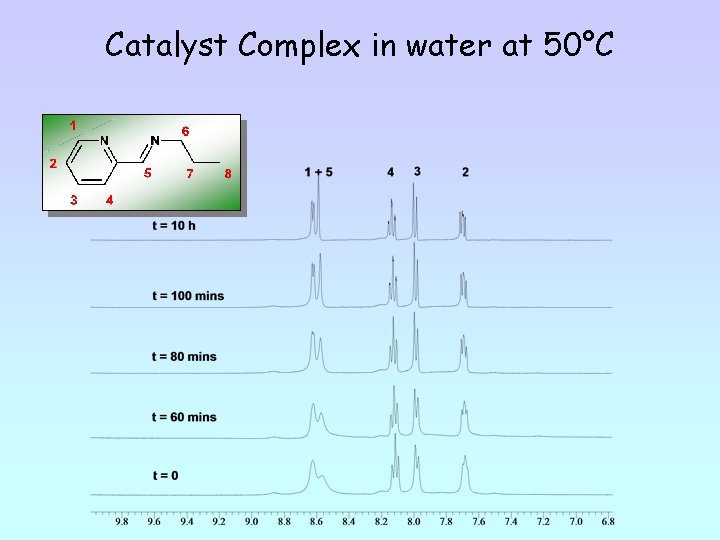
Catalyst Complex in water at 50°C


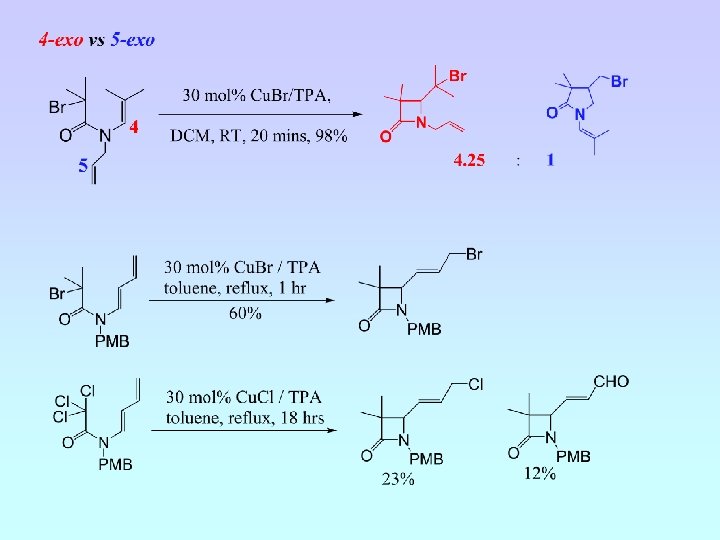
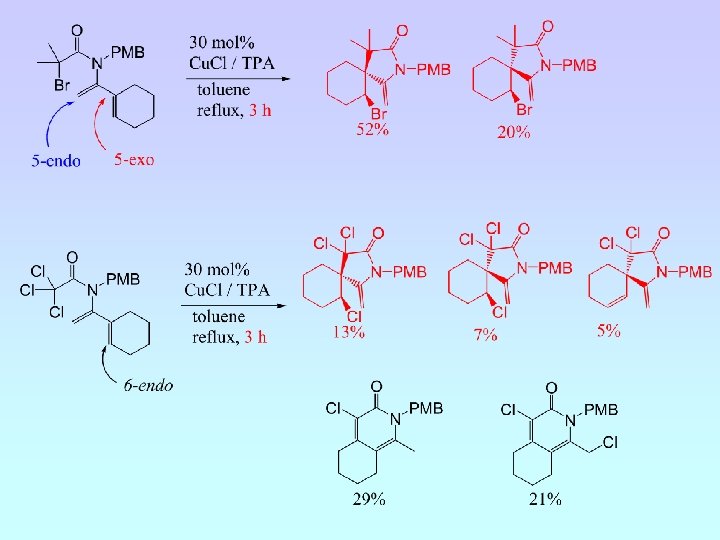
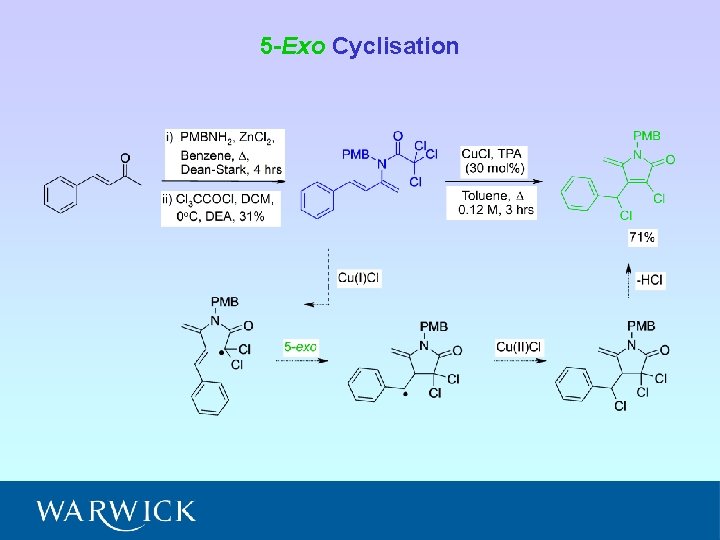
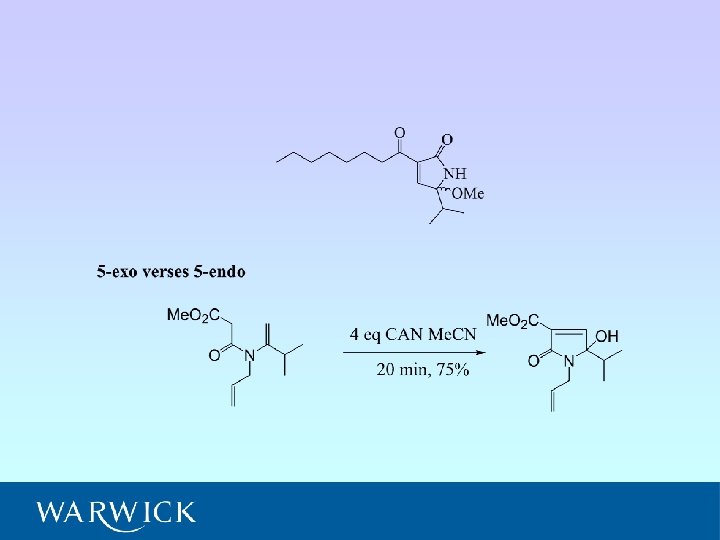
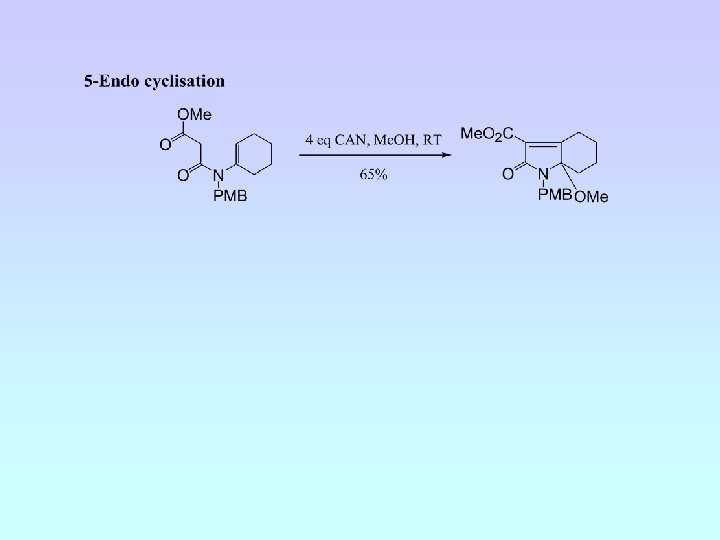
5 -Exo Cyclisation

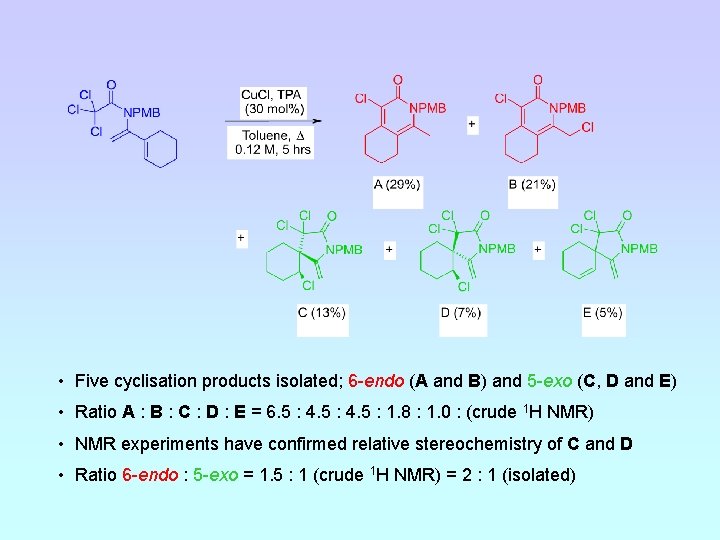
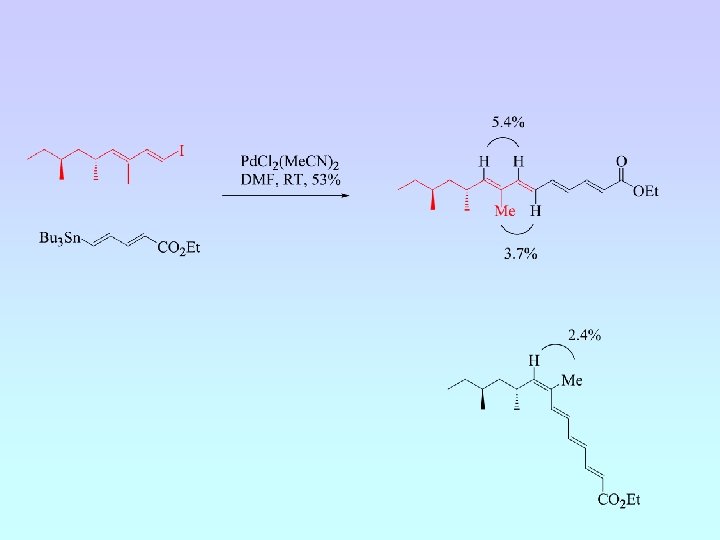
• Five cyclisation products isolated; 6 -endo (A and B) and 5 -exo (C, D and E) • Ratio A : B : C : D : E = 6. 5 : 4. 5 : 1. 8 : 1. 0 : (crude 1 H NMR) • NMR experiments have confirmed relative stereochemistry of C and D • Ratio 6 -endo : 5 -exo = 1. 5 : 1 (crude 1 H NMR) = 2 : 1 (isolated)

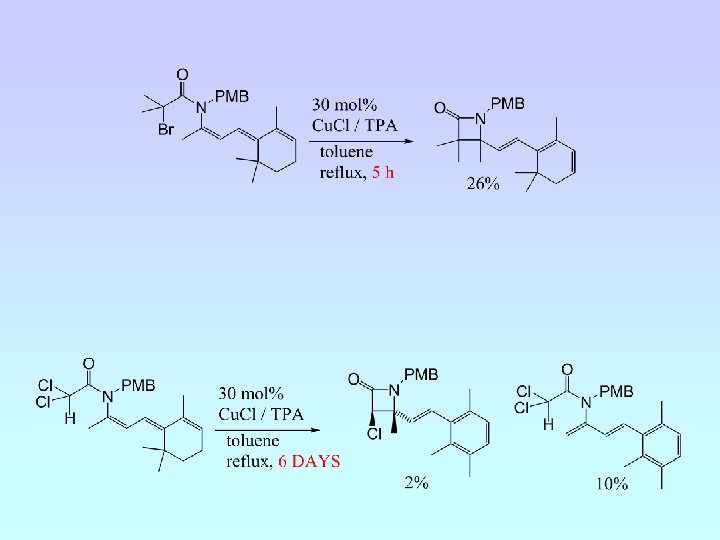
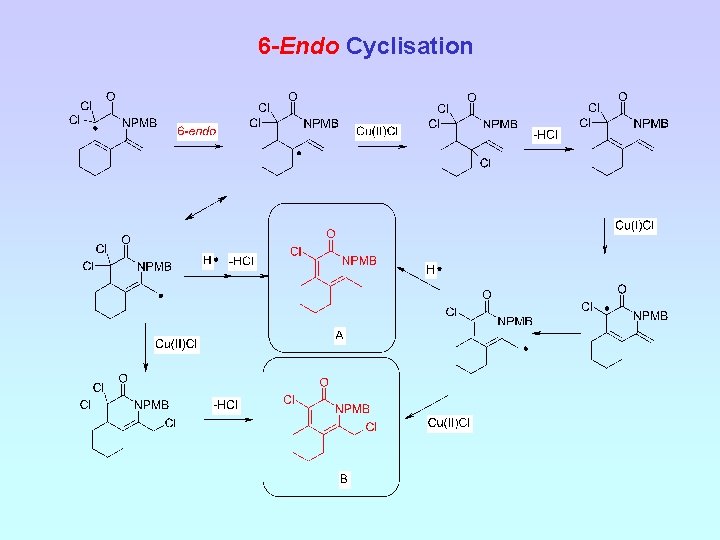
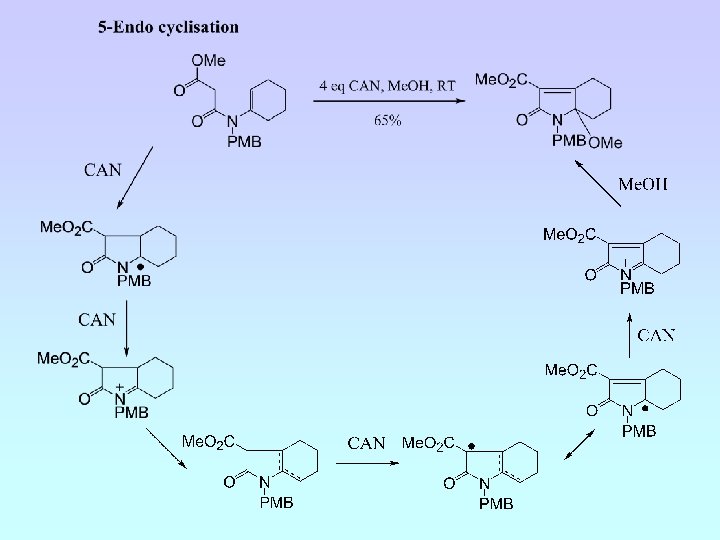
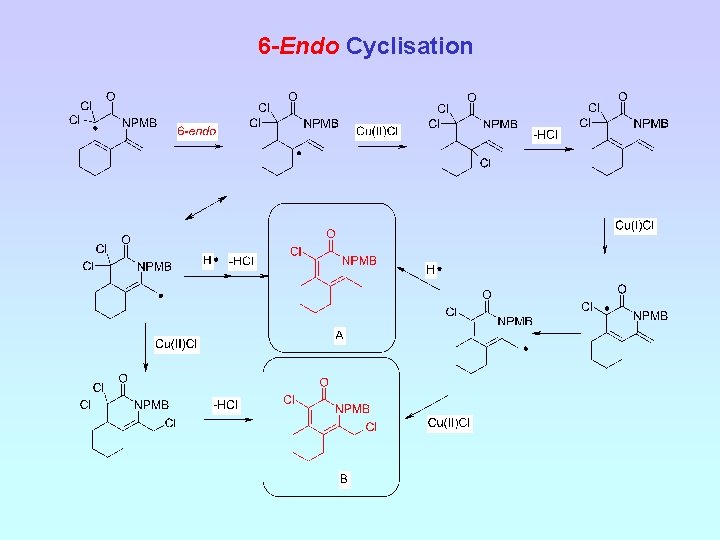
6 -Endo Cyclisation







6 -Endo Cyclisation






Cyclisation in ionic liquids

A. J. Clark, M. C. Crossman, D. Duncalf, D. M. Haddleton, S. R. Morsley, A. J. Shooter, J. Chem. Soc. , Chem Commun. , 1997, 1734

Catalyst complex in water 25°C (Cu : ligand = 1 : 3)

Catalyst Complex in water at 50°C
