Dr Abdul latif Mahesar Pharmacology Department By the

Dr. Abdul latif Mahesar Pharmacology Department

By the end of this lecture, students should: why the drugs should be metabolized Recognize the importance of biotransformation Know the different sites for drug metabolism Define the major phase I and phase II metabolic reactions. Describe the modulation of liver microsomal enzymes by inducers and inhibitors Mention two drugs that are known as enzyme inducers and inhibitors.

To produce effects drugs need to get absorbed drugs which are highly lipophilic , unionized and non polar are absorbed maximum these drugs after producing effect should leave the body but due to above properties they are reabsorbed back in circulation drugs to get eliminated , their form must get changed. for this drugs get metabolized in the body

Drug Metabolism (Biotransformation) Definition Chemical reactions which occur in the body and lead to change of drugs from lipid soluble form to water soluble form that could easily excreted.

Importance of metabolism Detoxification of foreign chemicals Inactivation or termination of drug action Activation of prodrug

Organ sites of drug metabolism Ø Intestinal Mucosa and Lumen Ø Plasma Ø Kidney Ø Skin Ø Lung Ø Mainly and predominantly drugs are metabolized in LIVER

DRUGS ARE METABOLIZED BY THE ENZYME ACTIVITY through various enzymes in the body. BUT MAINLY THEY ARE METABOLIZED BY MICROSOMAL CYTOCHROME P 450 FAMILY OF ENZYMES

EXTRACELLULAR METABOLISM Intestinal Mucosa and Lumen Gut Mucosa Ø Ø Monoamine Oxidase (MAO). Sulphatase. Gut lumen (bacterial flora) Ø Ø Glucouronidase. Azoreductase ( N=N).
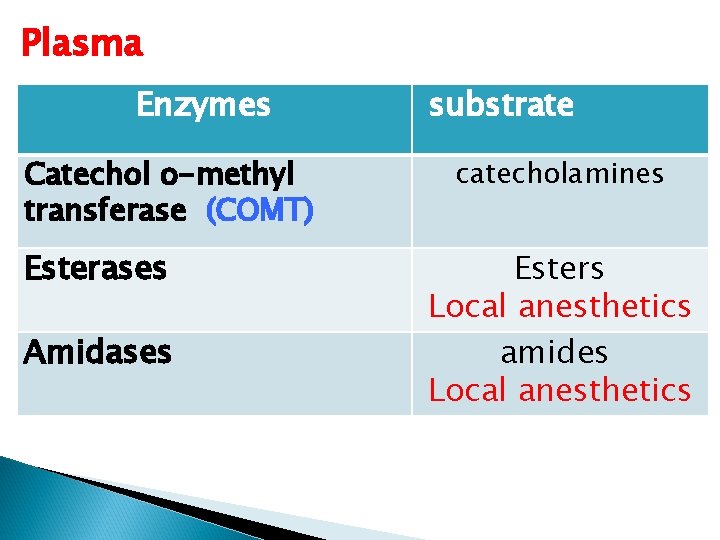
Plasma Enzymes Catechol o-methyl transferase (COMT) Esterases Amidases substrate catecholamines Esters Local anesthetics amides Local anesthetics
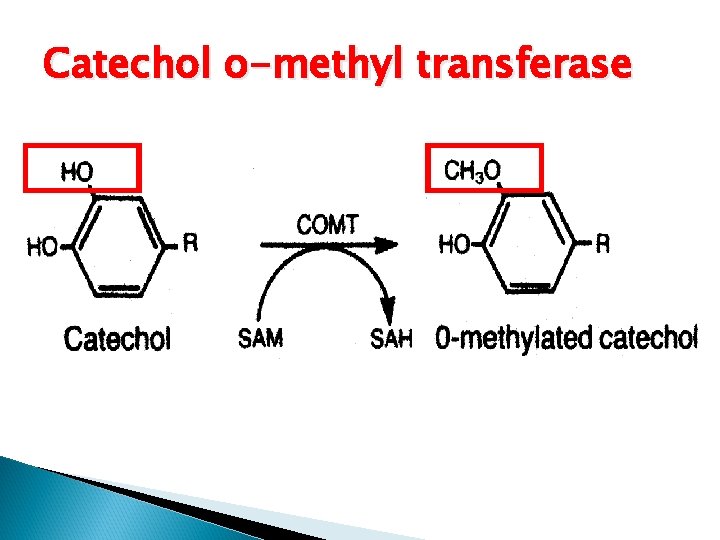
Catechol o-methyl transferase

Cellular sites of drug metabolism § Cytoplasm § Mitochondria § Lysosomes § MICROSOMES mainly

Cytoplasm e. g. Alcohol dehydrogenase: reduction of alcohol. Ethanol acetaldehyde acetic acid. CH 3 CH 2 OH CH 3 CHO CH 3 COOH. Mitochondria § Monoamine oxidase enzyme (MAO): oxidation of catecholamines § Acetylation by N-acetyl transferase: - Introduction of acetyl group (CH 3 COO )

Microsomes Microsomal enzyme system = mixed function oxidase = mono-oxygenase = Cytochrome P-450.

Types of hepatic metabolic reactions Two phases of hepatic metabolic reactions: Phase I reactions include: Oxidation. Reduction. Hydrolysis. Phase II reactions include Conjugation reactions
In phase 1 reaction relatively a reactive group such as hydroxyl (OH) group is introduced in to the drug molecule or a functional group get appeared to which conjugating group is attached such as glucuronide(Phase II). Both stages usually reduces the lipid solubility and thus increases the elimination this usually occurs in livier
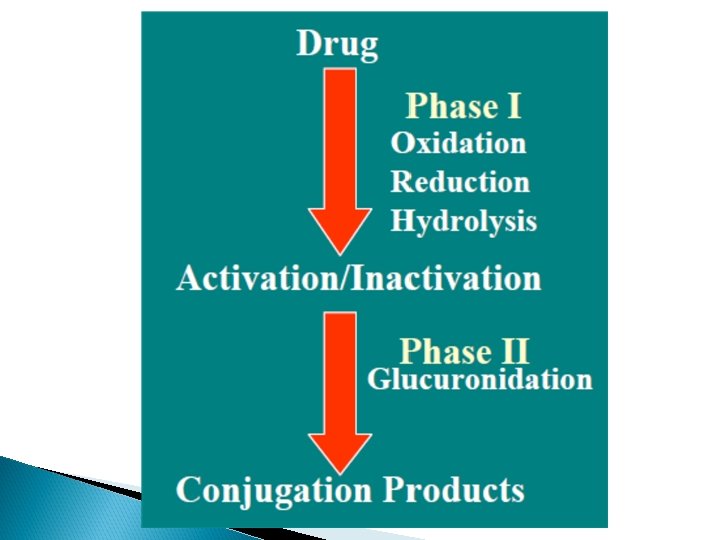

Hepatic drug metabolizing enzymes including cytochrome P 450 are present in endoplasmic reticulum of the cell. CYP 450 enzymes are haem proteins. It is a large family of distinct enzymes. They differ from each other in amino acid sequence , so the activity There about 74 families but mainly involved in drug metabolsim are CYP 1, CYP 2, CYP 3

Oxidation Reactions Microsomal oxidation Oxidation by cytochrome P 450 enzymes present in microsomes Non microsomal oxidation Oxidation by soluble enzymes in cytosol or mitochondria of cells as oxidases and dehydrogenases.

Non-microsomal Oxidation Dehydrogenases ◦ Alcohol dehydrogenase ◦ Aldehyde dehydrogenase Oxidases ◦ Monoamine oxidase (MAO): metabolism of catecholamines ◦ Xanthine oxidase: metabolism of xanthine Hypoxanthine uric acid accumulation GOUT
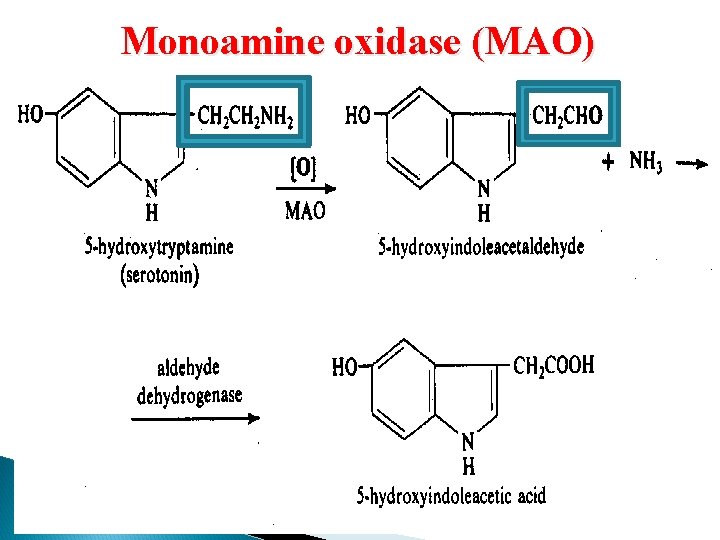
Monoamine oxidase (MAO)

Reduction reactions ØMicrosomal reduction ◦ Nitrobenzene Aniline. NO 2 NH 2 ØNon microsomal reduction ◦ Chloral hydrate Trichloroethanol (active). CCl 3 - CHO CCl 3 –CH 2 OH

Hydrolysis Ø All are non microsomal Ø Hydrolysis occurs by enzymes that are (esterases or amidases) Ø Drugs (substrates) affected are either esters or amides

Hydrolysis Ø Esters as acetylcholine (neurotransmitter). Acetylcholine + acetate. esterase R 1 -COOR 2 R 1 COOH + R 2 OH. Ø Amides as lidocaine (used as local anesthetic) amidase R 1 - CONH - R 2 R 1 - COOH + R 2 NH 2.

Phase I reactions can result in Inactivation of drug (termination of action) q Conversion of active drug to another active metabolite. q Conversion of drugs to toxic metabolites. Paracetamol hepatotoxic metabolite q Activation of pro-drug q Product might undergo phase II. q

Phase II Conjugation Reactions Conjugation of metabolite coming from (phase I) with endogenous substance as methyl group, acetyl group, sulphate, amino acid or glucouronic acid to produce conjugate that is water soluble and easily excreted.

Types of conjugation reactions Conjugation reaction Enzyme required glucouronide conjugation Glucouronyl transferase _ Acetylation (CH 3 COO ) Sulphation (SO 4 __ ) N-acetyl transferase Sulfo transferase Methylation ( CH 3 ) methyl transferase Amino acids conjugation Glycine conjugation

Phase II reactions: All are non microsomal except glucouronidation Glucouronide conjugation is a microsomal process (the most common). Deficieny of glucouronyl transferase enzyme in neonates may result into toxicity with chloramphenicol (Gray baby syndrome).

Characteristics of Phase II Products Usually pharmacologically inactive. Polar more water soluble. more readily excreted in urine.

Factors affecting metabolism Age rate of metabolism in neonates Nutrition Genetic Variation Diseases ( rate of metabolism in liver diseases) Gender Degree of Protein Binding Enzyme Induction & inhibition

Enzyme Induction & inhibition Ø Activities of liver microsomal enzymes may be changed by administration of some drugs. Ø Liver microsomal enzymes inducers: drugs that increase activities of liver microsomal enzymes. Ø Liver microsomal enzymes inhibitors drugs that decrease activities of liver microsomal enzymes.

Microsomal inducers Microsomal inhibitors Alcohol Cigarette smoking Phenobarbitone hypnotic Phenytoin (antiepileptic) Rifampicin (Anti TB) Grisofulvin (antifungal) Grape fruits Cimetidine Erythromycin (antibiotic) Ketoconazole (antifungal)

Enzyme induction may result in: Ø increase metabolism of the inducer drug. Ø Tolerance : decrease in its pharmacological action Ø Drug interactions: increase the metabolism and excretion of co-administered drugs e. g. phenytoin & oral contraceptives. Ø Failure of contraceptive may lead to pregnancy if combined with phenytoin. Ø

Enzyme inhibition may Delay the metabolism and excretion of the inhibitor drug and co-administered drugs. Prolong the action of the inhibitor drug & coadministered drugs. e. g. erythromycin & warfarin. Inhibition of warfarin metabolism may lead to increase its anticoagulant effect.
- Slides: 33