Double Replacement 1 Hydrogen sulfide is bubbled through

Double Replacement 1. Hydrogen sulfide is bubbled through a solution of silver nitrate. H 2 S(g) + Ag. NO 3(aq) --> HNO 3(aq) + Ag 2 S(s) H 2 S + Ag+ + NO 3 - --> H+ +NO 3 - + Ag 2 S H 2 S + Ag+ H+ + Ag 2 S

2. An excess of sodium hydroxide solution is added to a solution of magnesium nitrate. OH- + Mg 2+ Mg(OH)2

3. Solutions of sodium iodide and lead nitrate are mixed. I- + Pb 2+ Pb. I 2

4. A solution of ammonia is added to a solution of ferric chloride. NH 3(g) + H 2 O(l) + Fe. Cl 3(aq) --> NH 4 Cl(aq) + Fe(OH)3(s) NH 3 + H 2 O + Fe 3+ + Cl- NH 4+ + Cl- + Fe(OH)3 NH 3 + H 2 O + Fe 3+ NH 4+ + Fe(OH)3

5. Solutions of silver nitrate and sodium chromate are mixed. + Ag + 2 Cr. O 4 Ag 2 Cr. O 4

6. Excess silver acetate is added to a solution of trisodium phosphate. Ag+ + PO 43 - Ag 3 PO 4

7. Manganese(II) nitrate solution is mixed with sodium hydroxide solution. Mn 2+ + OH- Mn(OH)2

8. A saturated solution of calcium hydroxide is added to a solution of magnesium chloride. 2+ Mg + OH Mg(OH)2

9. Hydrogen sulfide gas is added to a solution of cadmium nitrate. H 2 S + 2+ Cd Cd. S + + H

10. Dilute sulfuric acid is added to a solution of barium acetate. H+ + SO 42 - + Ba 2+ + C 2 H 3 O 2 Ba. SO 4 + HC 2 H 3 O 2

11. 11 A precipitate is formed when solutions of trisodium phosphate and calcium chloride are mixed. PO 43 - + Ca 2+ Ca 3(PO 4)2

12. A solution of copper(II) sulfate is added to a solution of barium hydroxide. Cu 2+ + SO 42 - + Ba 2+ + OH- Cu(OH)2 + Ba. SO 4

13. 13 Equal volumes of dilute equimolar solutions of sodium carbonate and hydrochloric acid are mixed. Na 2 CO 3(aq) + HCl(aq) --> Na. Cl(aq) + Na. HCO 3(aq) + 2+ 2 Na + CO 3 + H +Cl --> + + Na + Cl + Na + HCO 3 H+ + CO 32 - HCO 3 -

14. Solid barium peroxide is added to cold dilute sulfuric acid. Ba. O 2(s) + H 2 SO 4(aq) --> Ba. SO 4(s) + H 2 O 2(l) + +H 2 SO 4 Ba. O 2 + Ba. SO 4 + H 2 O 2

15. Excess hydrochloric acid solution is added to a solution of potassium sulfite. HCl(aq) + K 2 SO 3(aq) --> H 2 O(l) + SO 2(g) + KCl(aq) H+ + Cl- + K+ + SO 32 - --> + H 2 O + SO 2 + K + Cl H+ + SO 32 - H 2 O + SO 2
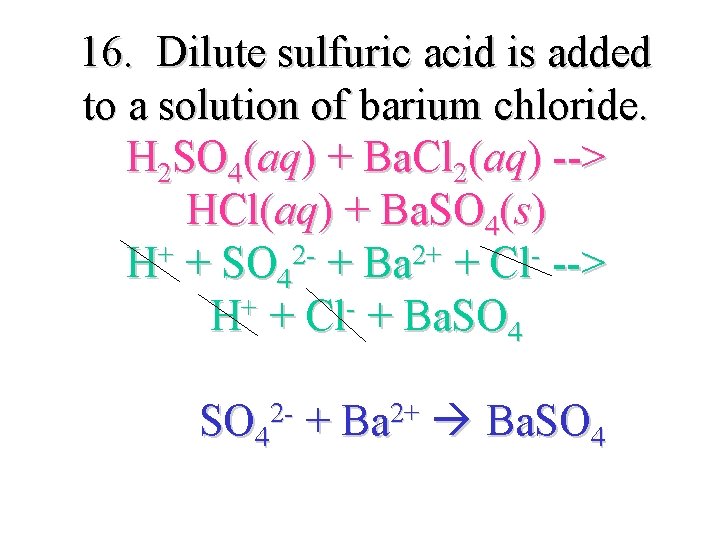
16. Dilute sulfuric acid is added to a solution of barium chloride. H 2 SO 4(aq) + Ba. Cl 2(aq) --> HCl(aq) + Ba. SO 4(s) + 22+ H + SO 4 + Ba + Cl --> H+ + Cl- + Ba. SO 42 - + Ba 2+ Ba. SO 4

17. A solution of sodium hydroxide is added to a solution of ammonium chloride. Na. OH(aq) + NH 4 Cl(aq) --> Na. Cl(aq) + NH 3(g) + H 2 O(l) Na+ + OH- + NH 4+ + Cl- --> + Na + Cl + NH 3 + H 2 O NH 4+ + OH- NH 3 + H 2 O

18. Dilute hydrochloric acid is added to a solution of potassium carbonate. HCl(aq) + K 2 CO 3(aq) --> H 2 O(l) + CO 2(g) + KCl(aq) H+ + Cl- + K+ + CO 32 - --> + H 2 O + CO 2 + K + Cl H+ + CO 32 - H 2 O + CO 2

19. Gaseous hydrogen sulfide is bubbled through a solution of nickel(II) nitrate. H 2 S(g) + Ni(NO 3)2(aq) --> HNO 3(aq) + Ni. S(s) H 2 S + Ni 2+ + NO 3 - --> + H + NO 3 + Ni. S H 2 S + Ni 2+ Ni. S + H+

20. A solution of sodium sulfide is added to a solution of zinc nitrate. Na 2 S(aq) + Zn(NO 3)2(aq) --> Na. NO 3(aq) + Zn. S(s) Na+ + S 2 - + Zn 2+ + NO 3 - --> Na+ + NO 3 - + Zn. S S 2 - + Zn 2+ Zn. S

21. Concentrated hydrochloric acid is added to solid manganese(II) sulfide. H+ + Mn. S H 2 S + Mn 2+

22. Solutions of tri-potassium phosphate and zinc nitrate are mixed. 3 PO 4 + 2+ Zn 3(PO 4)2

23. Dilute acetic acid solution is added to solid magnesium carbonate. HC 2 H 3 O 2 + Mg. CO 3 H 2 O + CO 2 + Mg 2+ + C 2 H 3 O 2 -

24. Gaseous hydrofluoric acid reacts with solid silicon dioxide. HF + Si. O 2 H 2 O + Si. F 4

25. Equimolar amounts of trisodium phosphate and hydrogen chloride, both in solution, are mixed. PO 43 - + H+ HPO 42 -
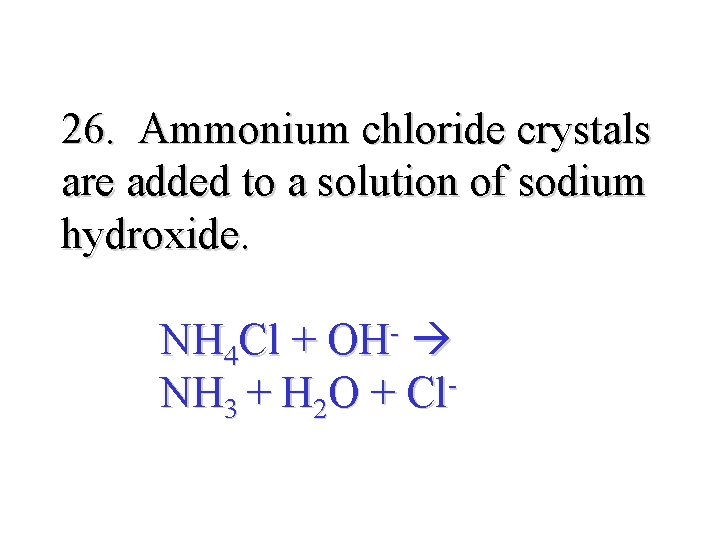
26. Ammonium chloride crystals are added to a solution of sodium hydroxide. NH 4 Cl + OH- NH 3 + H 2 O + Cl-

27. Hydrogen sulfide gas is bubbled through a solution of lead(II) nitrate. H 2 S + Pb 2+ Pb. S + H+

28. Solutions of silver nitrate and sodium chromate are mixed. Ag+ + Cr. O 42 - Ag 2 Cr. O 4

29. Solutions of sodium fluoride and dilute hydrochloric acid are mixed. F- + H+ HF

30. A saturated solution of barium hydroxide is mixed with a solution of iron(III) sulfate. Ba 2+ + OH- + Fe 3+ + SO 42 - Ba. SO 4 + Fe(OH)3

31. A solution of ammonium sulfate is added to a potassium hydroxide solution. NH 4+ + OH- NH 3 + H 2 O

32. A solution of ammonium sulfate is added to a saturated solution of barium hydroxide. NH 4+ + SO 42 - + Ba 2+ +OH- NH 3 + H 2 O + Ba. SO 4

33. Dilute sulfuric acid is added to solid calcium fluoride. H+ + SO 42 - + Ca. F 2 Ca. SO 4 + HF

34. Dilute hydrochloric acid is added to a dilute solution of mercury(I) nitrate. Cl- + Hg 22+ Hg 2 Cl 2

35. Dilute sulfuric acid is added to a solution of lithium hydrogen carbonate. + HCO 3 H 2 O + CO 2

36. Dilute hydrochloric acid is added to a solution of potassium sulfite. H+ + SO 32 - H 2 O + SO 2

37. 37 Carbon dioxide gas is bubbled through water containing a suspension of calcium carbonate. CO 2 + Ca. CO 3 + H 2 O HCO 3 - + Ca 2+

38. Excess concentrated sulfuric acid is added to solid calcium phosphate. H 2 SO 4 + Ca 3(PO 4)2 H 3 PO 4 + Ca. SO 4 2+ 2(or Ca + SO 4 )

39. Hydrogen sulfide gas is bubbled into a solution of mercury(II) chloride. H 2 S + 2+ Hg Hg. S + + H

40. Solutions of zinc sulfate and sodium phosphate are mixed. Zn 2+ + PO 43 - --> Zn 3(PO 4)2

41. Solutions of silver nitrate and lithium bromide are mixed. Ag+ + Br- --> Ag. Br

42. Solutions of manganese(II) sulfate and ammonium sulfide are mixed. Mn 2+ + S 2 - --> Mn. S

43. Excess hydrochloric acid solution is added to a solution of potassium sulfite. H+ + SO 32 - --> H 2 O + SO 2

Single. Replacement 1. A piece of aluminum metal is added to a solution of silver nitrate. Al(s) + Ag. NO 3(aq) --> Al(NO 3)3(aq) + Ag(s) Al + Ag+ + NO 3 --> 3+ Al + NO 3 + Ag Al + Ag+ Al 3+ + Ag

2. Aluminum metal is added to a solution of copper(II) chloride. Al(s) + Cu. Cl 2(aq) --> Cu(s) + Al. Cl 3(aq) Al + Cu 2+ + Cl- --> Cu + Al 3+ + Cl. Al + Cu 2+ Al 3+ + Cu
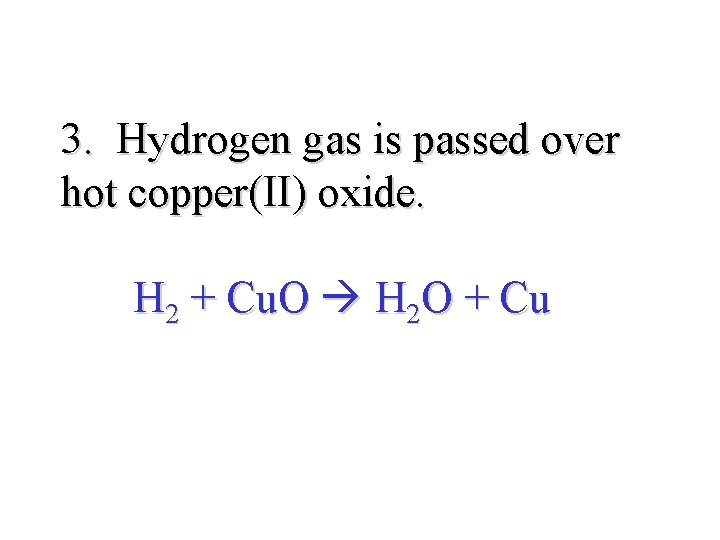
3. Hydrogen gas is passed over hot copper(II) oxide. H 2 + Cu. O H 2 O + Cu

4. Small chunks of solid sodium are added to water. Na + H 2 O Na+ + OH- + H 2

5. Calcium metal is added to a dilute solution of hydrochloric acid. Ca + H+ Ca 2+ + H 2

6. Magnesium turnings are added to a solution of iron(III) chloride. Mg + Fe 3+ Mg 2+ + Fe

7. Chlorine gas is bubbled into a solution of sodium bromide. Cl 2 + Br- Cl- + Br 2
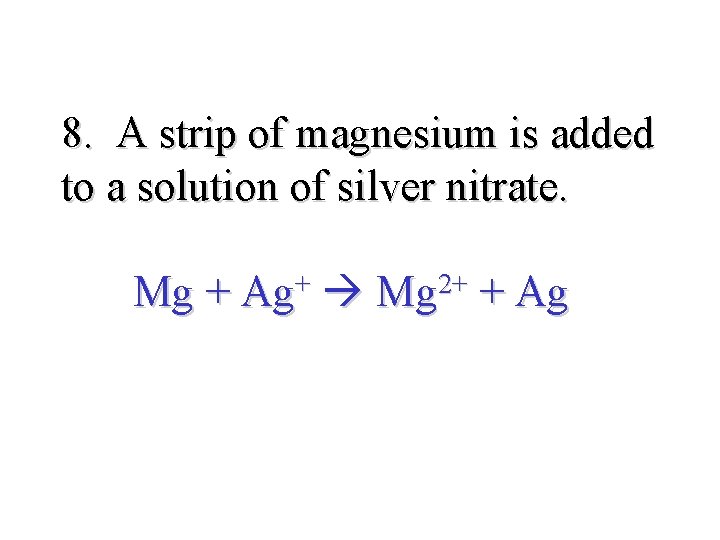
8. A strip of magnesium is added to a solution of silver nitrate. Mg + Ag+ Mg 2+ + Ag

9. Solid calcium is added to warm water. Ca + H 2 O Ca 2+ + OH- + H 2

10. Liquid bromine is added to a solution of potassium iodide. Br 2 + I- Br- + I 2

11. Chlorine gas is bubbled into a solution of potassium iodide. Cl 2 + I- Cl- + I 2

12. Lead foil is immersed in silver nitrate solution. Pb + + Ag 2+ Pb + Ag

13. Solid zinc strips are added to a solution of copper(II) sulfate. Zn + Cu 2+ Zn 2+ + Cu

14. Sodium metal is added to water. Na + H 2 O Na+ + OH- + H 2
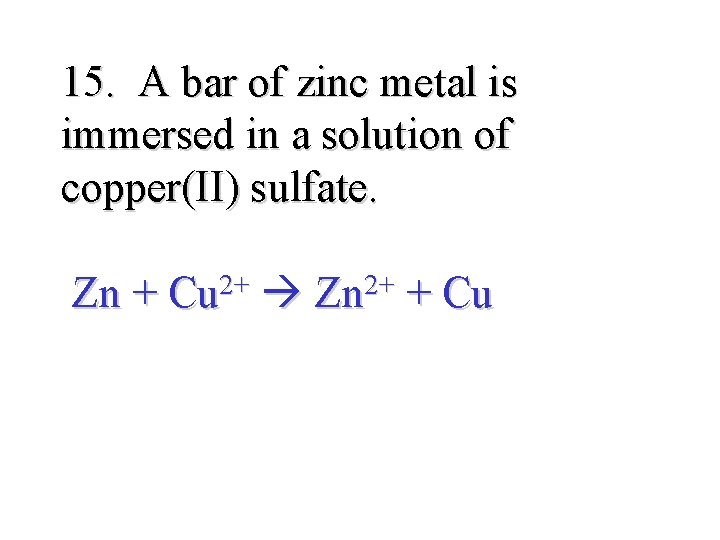
15. A bar of zinc metal is immersed in a solution of copper(II) sulfate. Zn + Cu 2+ Zn 2+ + Cu

16. A small piece of sodium metal is added to distilled water. Na + H 2 O --> + Na + OH + H 2

Combustion 1. Lithium metal is burned in air. Li + O 2 Li 2 O
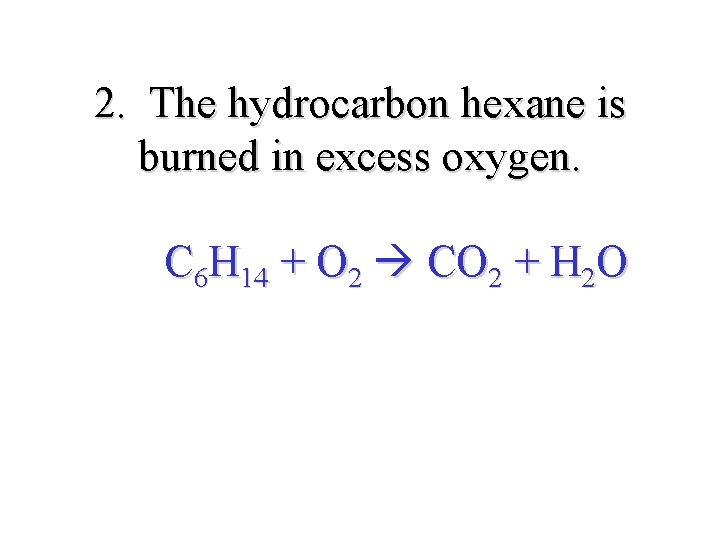
2. The hydrocarbon hexane is burned in excess oxygen. C 6 H 14 + O 2 CO 2 + H 2 O
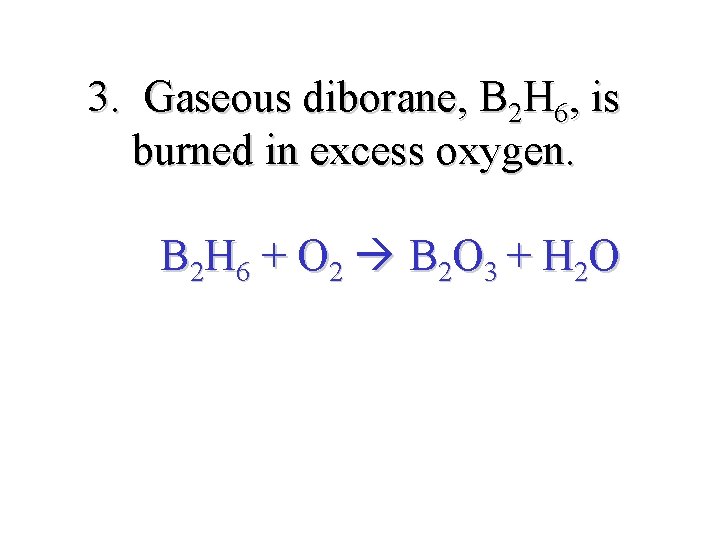
3. Gaseous diborane, B 2 H 6, is burned in excess oxygen. B 2 H 6 + O 2 B 2 O 3 + H 2 O

4. A piece of solid bismuth is heated strongly in oxygen. Bi + O 2 Bi 2 O 3

5. Solid zinc sulfide is heated in an excess of oxygen. Zn. S + O 2 Zn. O + SO 2

6. Propanol is burned completely in air. C 3 H 7 OH + O 2 CO 2 + H 2 O

7. Excess oxygen gas is mixed with ammonia gas in the presence of platinum. O 2 + NH 3 NO 2 + H 2 O
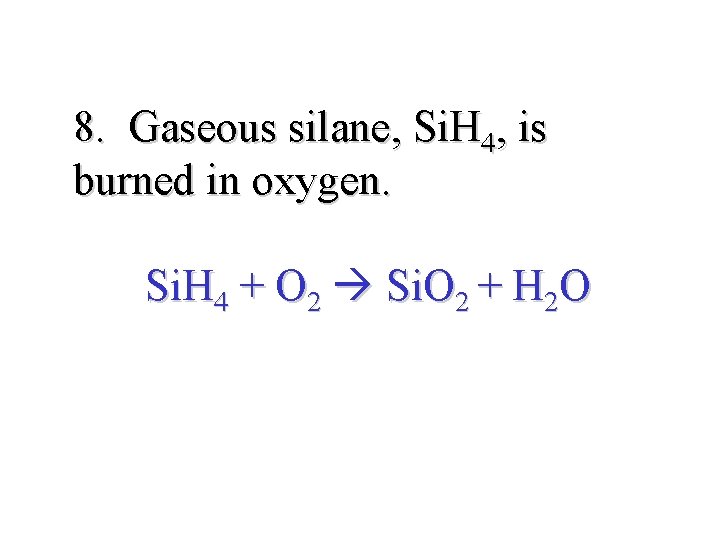
8. Gaseous silane, Si. H 4, is burned in oxygen. Si. H 4 + O 2 Si. O 2 + H 2 O

9. Ethanol is completely burned in air. C 2 H 5 OH + O 2 CO 2 + H 2 O

10. Solid copper(II) sulfide is heated strongly in oxygen gas. Cu. S + O 2 Cu. O + SO 2

11. Ethanol is burned in oxygen. C 2 H 5 OH + O 2 CO 2 + H 2 O

12. Carbon disulfide vapor is burned in excess oxygen. CS 2 + O 2 --> CO 2 + SO 2

Anhydrides 1. Excess water is added to solid calcium hydride. H 2 O + Ca. H 2 2+ Ca + OH or (Ca(OH)2) + H 2

2. Solid lithium hydride is added to water. Li. H + H 2 O Li+ + OH- + H 2

3. Liquid phosphorus trichloride is poured into a large excess of water. PCl 3 + H 2 O H 3 PO 3 + + H + Cl
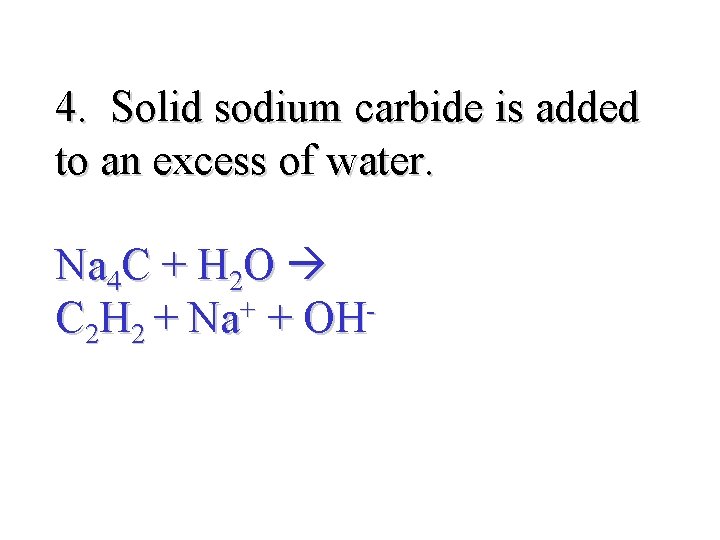
4. Solid sodium carbide is added to an excess of water. Na 4 C + H 2 O + C 2 H 2 + Na + OH

5. Solid magnesium nitride is added to excess deuterium oxide. Mg 3 N 2 + D 2 O Mg(OD)2+ ND 3

6. Water is added to a sample of pure phosphorus tribromide. H 2 O + PBr 3 H 3 PO 3 + H+ + Br-

7. Water is added to a sample of pure sodium hydride. H 2 O + Na. H Na+ + OH- + H 2

8. Dinitrogen trioxide gas is bubbled into water. N 2 O 3 + H 2 O HNO 2
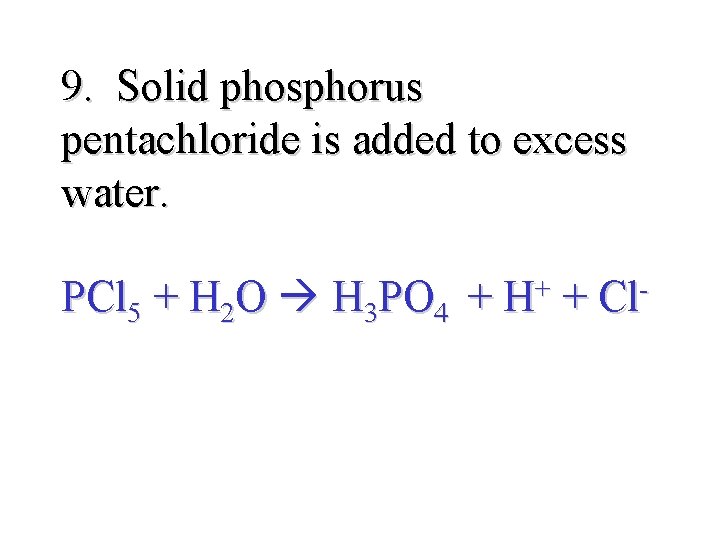
9. Solid phosphorus pentachloride is added to excess water. PCl 5 + H 2 O H 3 PO 4 + H+ + Cl-

10. Solid dinitrogen pentoxide is added to water. N 2 O 5 + H 2 O H+ + NO 3 -

11. Sulfur trioxide gas is added to excess water. SO 3 + H 2 O H+ + SO 42 -

12. Solid sodium oxide is added to water. Na 2 O + H 2 O Na+ + OH-

13. Phosphorus(V) oxytrichloride is added to water. POCl 3 + H 2 O H 3 PO 4+ H+ + Cl-

14. Water is added to a sample of solid magnesium nitride. H 2 O + Mg 3 N 2 Mg(OH)2 + NH 3

15. Solid potassium oxide is added to water. K 2 O + H 2 O + K + OH

16. Solid sodium cyanide is added to water. Na. CN + H 2 O HCN + Na+ + OH-

17. Trisodium phosphate crystals are added to water. Na 3 PO 4 + H 2 O Na+ + OH- + HPO 42 - ( or H 2 PO 4 -)

18. Solid lithium oxide is added to excess water. Li 2 O + H 2 O Li+ + OH-

19. Solid barium oxide is added to distilled water. Ba. O + H 2 O Ba 2+ + OH-

20. 20 Solid calcium hydride is added to distilled water. Ca. H 2+ H 2 O 2+ Ca + OH or (Ca(OH)2) + H 2

21. Phosphorous(V) oxide powder is sprinkled over distilled water. P 2 O 5 + H 2 O --> H 3 PO 4
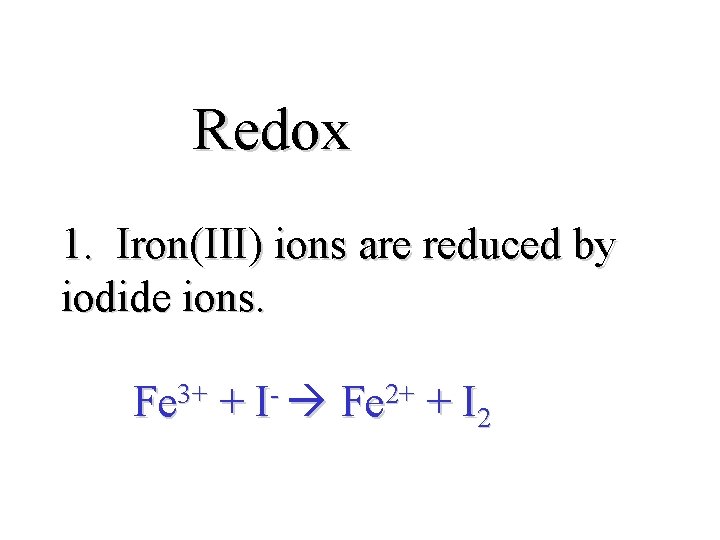
Redox 1. Iron(III) ions are reduced by iodide ions. 3+ Fe + I 2+ Fe + I 2

2. Potassium permanganate solution is added to concentrated hydrochloric acid. Mn. O 4 - + H+ + Cl- 2+ Mn + Cl 2 + H 2 O
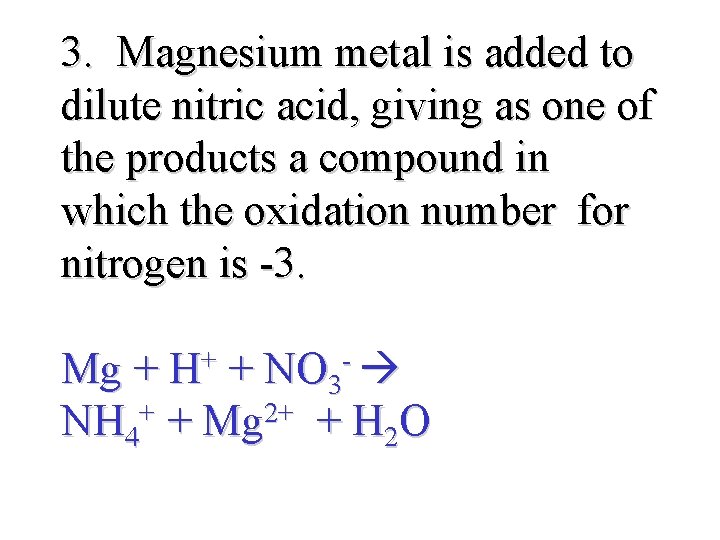
3. Magnesium metal is added to dilute nitric acid, giving as one of the products a compound in which the oxidation number for nitrogen is -3. Mg + H+ + NO 3 - NH 4+ + Mg 2+ + H 2 O
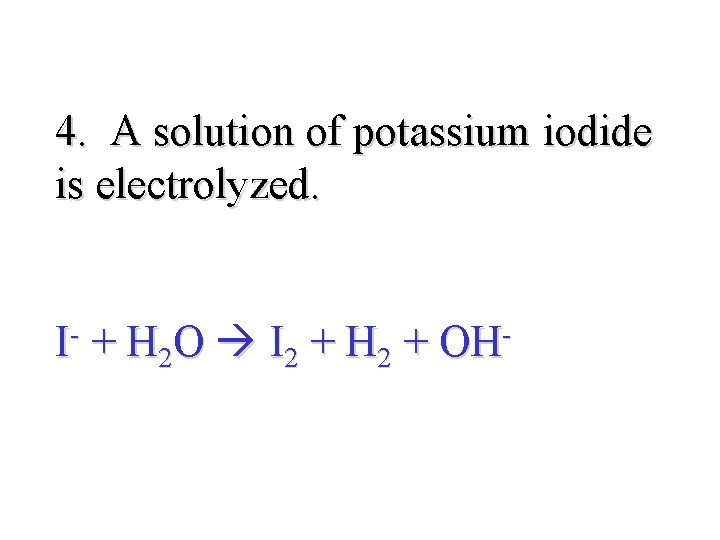
4. A solution of potassium iodide is electrolyzed. I + H 2 O I 2 + H 2 + OH

5. Potassium dichromate solution is added to an acidified solution of sodium sulfite. Cr 2 O 72 - + SO 32 - + H+ 3+ 2 Cr + SO 4 + H 2 O

6. Solutions of potassium iodide, potassium iodate, and dilute sulfuric acid are mixed. I + IO 3 + + H H 2 O + I 2

7. A solution of tin(II) sulfate is added to a solution of iron(III) sulfate. 2+ Sn + 3+ Fe 4+ Sn + 2+ Fe

8. Metallic copper is heated with concentrated sulfuric acid. Cu + H 2 SO 4 Cu 2+ + SO 2 + H 2 O

9. Manganese(IV) oxide is added to warm, concentrated hydrobromic acid. Mn. O 2 + H+ + Br- Mn 2+ + Br 2 + H 2 O

10. Chlorine gas is bubbled into cold dilute sodium hydroxide. Cl 2 + OH- Cl. O- + Cl- + H 2 O

11. Solid iron(III) oxide is heated in excess carbon monoxide. Fe 2 O 3 + CO Fe + CO 2

12. Hydrogen peroxide solution is added to acidified potassium iodide solution. H 2 O 2 + + H + I H 2 O + I 2

13. Hydrogen peroxide is added to an acidified solution of potassium dichromate. H 2 O 2 + H+ + Cr 2 O 72 - 3+ H 2 O + Cr + O 2

14. Sulfur dioxide gas is bubbled through an acidified solution of potassium permanganate. SO 2 + H+ + Mn. O 4 - 2+ 2 Mn + H 2 O + SO 4

15. A solution containing tin(II) ions is added to an acidified solution of potassium dichromate. Sn 2+ + H+ + Cr 2 O 72 - 3+ 4+ Cr + Sn + H 2 O

16. Solid silver sulfide is warmed with dilute nitric acid. Ag 2 S + H+ + NO 3 - + Ag + S + NO 2 + H 2 O

17. A dilute solution of sulfuric acid is electrolyzed between platinum electrodes. H 2 O H 2 + O 2

18. Pellets of lead are dropped into hot sulfuric acid. Pb + H+ + SO 42 - 2+ H 2 O + SO 2 + Pb (or Pb. SO 4 )

19. Potassium permanganate solution is added to a solution of oxalic acid, H 2 C 2 O 4 , acidified with a few drops of sulfuric acid. + Mn. O 4 + H 2 C 2 O 4 + H Mn 2+ + CO 2 + H 2 O

20. Powdered iron is added to a solution of iron(III) sulfate. Fe + Fe 3+ Fe 2+

21. A concentrated solution of hydrochloric acid is added to powdered manganese dioxide and gently heated. H+ + Cl- + Mn. O 2 Cl 2 + Mn 2+ + H 2 O

22. A strip of copper metal is added to a concentrated solution of sulfuric acid. Cu + H+ + SO 42 - Cu 2+ + SO 2 + H 2 O

23. Copper(II) sulfide is oxidized by dilute nitric acid. Cu. S + H+ + NO 3 - 2+ Cu + NO + H 2 O + S

24. A solution of copper(II) sulfate is electrolyzed using inert electrodes. 2+ Cu + H 2 O O 2 + + H + Cu

25. A solution of potassium iodide is added to an acidified solution of potassium dichromate. I- + H+ + Cr 2 O 72 - I 2 + Cr 3+ + H 2 O
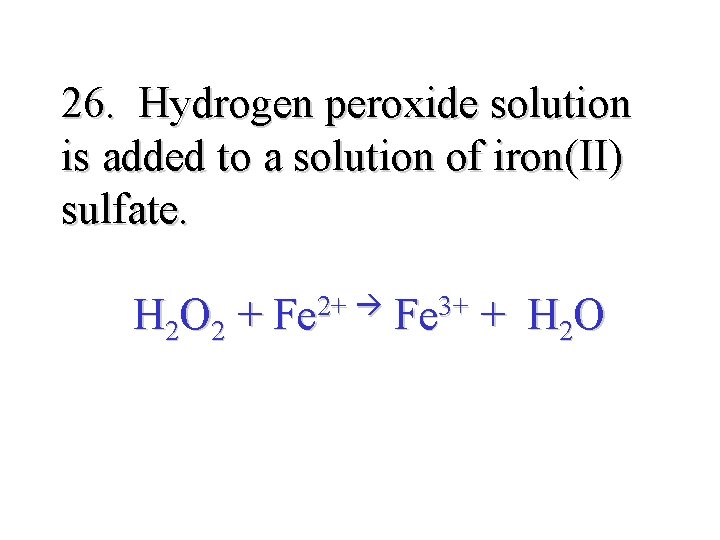
26. Hydrogen peroxide solution is added to a solution of iron(II) sulfate. H 2 O 2 + 2+ Fe 3+ Fe + H 2 O

27. Solid silver is added to a dilute nitric acid (6 M) solution. Ag + H+ +NO 3 - + Ag + NO + H 2 O

28. A solution of formic acid, HCOOH, is oxidized by an acidified solution of potassium dichromate. + H 2 Cr 2 O 7 HCOOH + + 3+ H 2 O + Cr + CO 2

29. A piece of iron is added to a solution of iron(III) sulfate. Fe + Fe 3+ Fe 2+

30. An acidified solution of potassium permanganate is added to a solution of sodium sulfite. H+ + Mn. O 4 - + SO 32 - Mn 2+ + SO 42 - + H 2 O

31. A solution of tin(II) chloride is added to a solution of iron(III) sulfate. 2+ Sn + 3+ Fe 4+ Sn + 2+ Fe

32. Concentrated hydrochloric acid solution is added to solid manganese(IV) oxide and the reactants are heated. + + Mn. O 2 2+ Mn + Cl 2 + H 2 O + H Cl

34. Potassium permanganate solution is added to an acidic solution of hydrogen peroxide. Mn. O 4 - + H+ + H 2 O 2 Mn 2+ + O 2 + H 2 O

35. Solid copper is added to a dilute nitric acid solution. Cu + H+ + NO 3 - Cu 2+ + NO + H 2 O
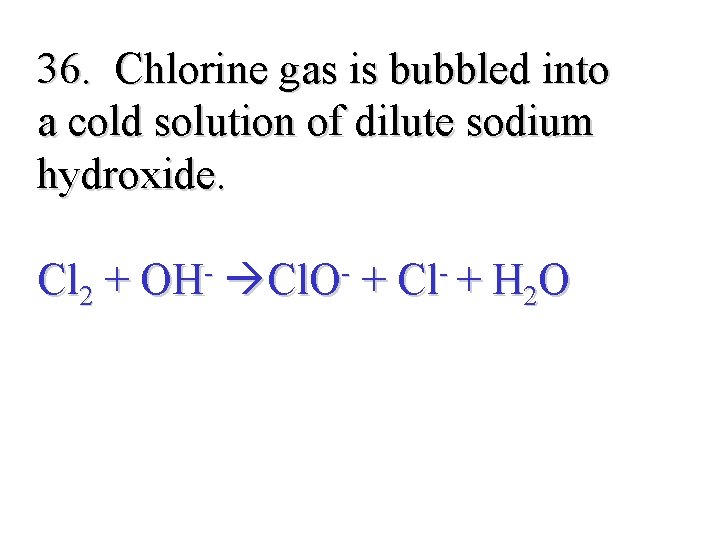
36. Chlorine gas is bubbled into a cold solution of dilute sodium hydroxide. Cl 2 + OH- Cl. O- + Cl- + H 2 O

37. A solution of potassium permanganate is mixed with an alkaline solution of sodium sulfite. Mn. O 4 - + OH- + SO 32 - SO 42 - + H 2 O + Mn. O 42 - (or Mn. O 2)

38. Solid sodium dichromate is added to an acidified solution of sodium iodide. Na 2 Cr 2 O 7 + H+ + I- 3+ + Cr + I 2 + H 2 O + Na
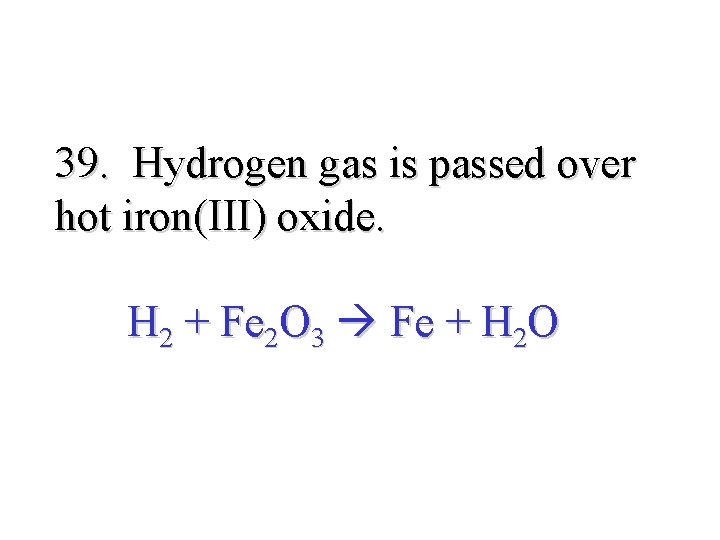
39. Hydrogen gas is passed over hot iron(III) oxide. H 2 + Fe 2 O 3 Fe + H 2 O

40. Solutions of potassium iodide and potassium iodate are mixed in acid solution. I + IO 3 + + H I 2 + H 2 O

41. Hydrogen peroxide is added to an acidified solution of sodium bromide. H 2 O 2 + H+ + Br- Br 2 + H 2 O

42. 42 Chlorine gas is bubbled into a cold, dilute solution of potassium hydroxide. Cl 2 + OH Cl + Cl. O + H 2 O
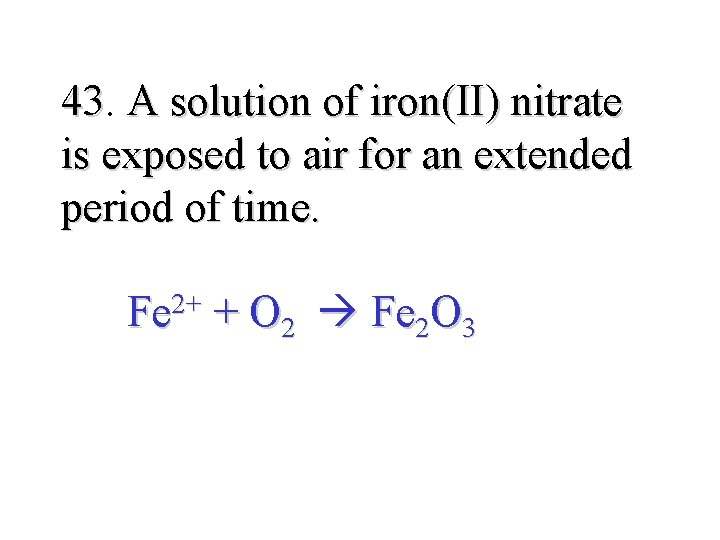
43. 43 A solution of iron(II) nitrate is exposed to air for an extended period of time. Fe 2+ + O 2 Fe 2 O 3

44. A stream of chlorine gas is passed through a solution of cold, dilute sodium hydroxide. Cl 2 + OH Cl + Cl. O + H 2 O

45. A solution of tin(II) chloride is added to an acidified solution of potassium permanganate. Sn 2+ + H + + Mn. O 4 - --> Mn 2+ + Sn 4+ + H 2 O

46. A concentrated solution of hydrochloric acid is added to solid potassium permanganate. + H + Cl 2 + Cl + Mn. O 4 --> Mn 2+ + H 2 O

47. A solution of potassium dichromate is added to an acidified solution of iron(II) chloride. 2+ 2+ Cr 2 O 7 + H + Fe Cr 3+ + Fe 3+ + H 2 O -->

Acid-Base Neutralizations 1. Solutions of ammonia and hydrofluoric acid are mixed. NH 3 + H 2 O --> NH 4+ + OHOH- + HF--> H 2 O + FNH 3 + HF NH 4+ + F-

2. Hydrogen sulfide gas is bubbled through a solution of potassium hydroxide. H 2 S + OH- H 2 O + S 2 -

3. A solution of sulfuric acid is added to a solution of barium hydroxide until the same number of moles of each compound has been added. 2 H+ + SO 42 - + Ba 2+ + 2 OH- 2 H 2 O + Ba. SO 4

4. A solution of sodium hydroxide is added to a solution of sodium dihydrogen phosphate until the same number of moles of each compound has been added. OH- + H 2 PO 4 - HPO 42 - + H 2 O

5. Dilute nitric acid is added to crystals of pure calcium oxide. Ca. O + H 2 O --> Ca 2+ + 2 OH 2 H+ + 2 OH- --> 2 H 2 O + H + Ca. O H 2 O + 2+ Ca

6. Equal volumes of 0. 1 -molar sulfuric acid and 0. 1 -molar potassium hydroxide are mixed. 2 H+ + SO 42 - + K+ + OH---> H 2 O + H+ + K+ + SO 42+ H +OH H 2 O

7. A solution of ammonia is added to a dilute solution of acetic acid. NH 3 + H 2 O --> NH 4+ + OHOH- + HC 2 H 3 O 2 --> H 2 O + C 2 H 3 O 2 NH 3 + HC 2 H 3 O 2 NH 4+ + C 2 H 3 O 2 -

8. Excess sulfur dioxide gas is bubbled through a dilute solution of potassium hydroxide. SO 2 + H 2 O --> H 2 SO 3 2 H 2 SO 3 + 2 OH --> 2 H 2 O + SO 3 2 SO 2 + OH H 2 O + SO 3

9. Sulfur dioxide gas is bubbled into an excess of a saturated solution of calcium hydroxide. SO 2 + H 2 O --> H 2 SO 3 2+ H 2 SO 3 + Ca + 2 OH --> Ca. SO 3 + 2 H 2 O SO 2 + Ca 2+ + OH- Ca. SO 3 + H 2 O

10. A solution of sodium hydroxide is added to a solution of calcium hydrogen carbonate until the number of moles of sodium hydroxide added is twice the number of moles of the calcium salt. 2+ 2 OH +Ca + 2 HCO 3 2 H 2 O + Ca. CO 3

11. Equal volumes of 0. 1 M hydrochloric acid and 0. 1 M sodium monohydrogen phosphate are mixed. H+ + 2 HPO 42 - H 2 PO 4 -

12. Hydrogen sulfide gas is bubbled through excess potassium hydroxide solution. H 2 S + OH- H 2 O + S 2 -

13. Ammonia gas and carbon dioxide gas are bubbled into water. NH 3 + CO 2 + 2 H 2 O --> NH 4+ + 2 OH- + H 2 CO 3 --> H 2 O + CO 32 NH 3 + CO 2 + H 2 O NH 4+ + CO 32 -

14. Carbon dioxide gas is bubbled through a concentrated solution of sodium hydroxide. CO 2 + H 2 O --> H 2 CO 3 + 2 OH- --> CO 32 - + 2 H 2 O CO 2 + OH- CO 32 - + H 2 O
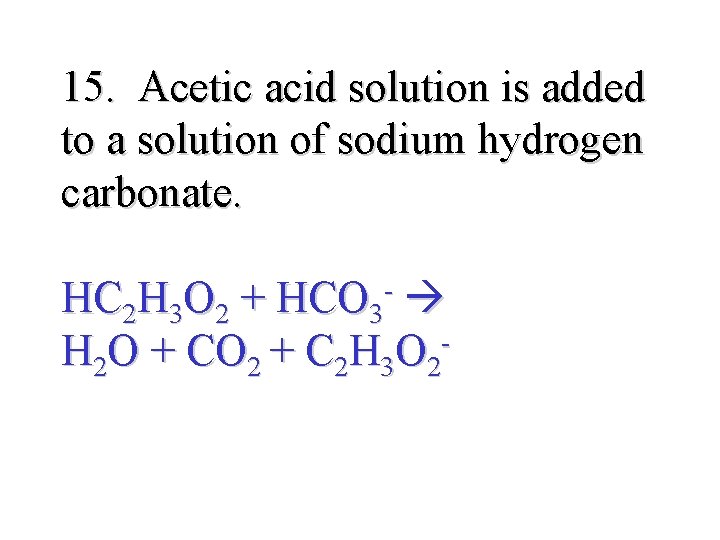
15. Acetic acid solution is added to a solution of sodium hydrogen carbonate. HC 2 H 3 O 2 + HCO 3 - H 2 O + CO 2 + C 2 H 3 O 2

16. Excess potassium hydroxide solution is added to a solution of potassium dihydrogen phosphate. OH- + H 2 PO 4 - H 2 O + PO 43 -
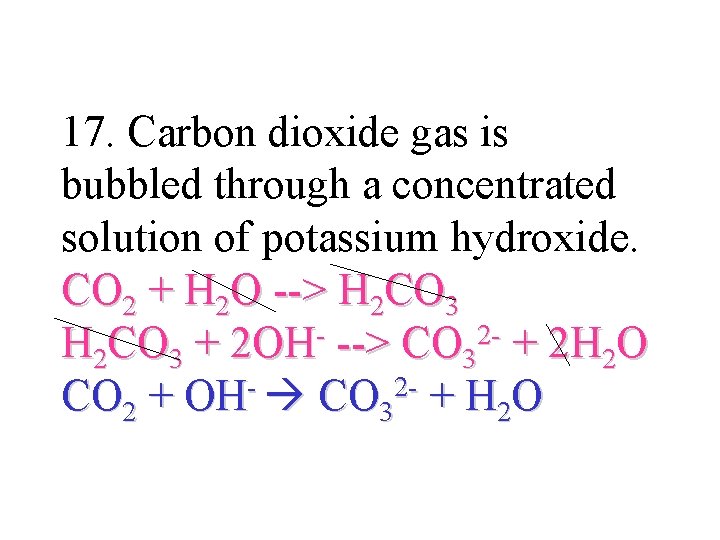
17. Carbon dioxide gas is bubbled through a concentrated solution of potassium hydroxide. CO 2 + H 2 O --> H 2 CO 3 2 H 2 CO 3 + 2 OH --> CO 3 + 2 H 2 O CO 2 + OH- CO 32 - + H 2 O

Complex Ions 1. Concentrated (15 M) ammonia solution is added in excess to a solution of copper(II) nitrate. NH 3 + Cu 2+ [Cu(NH 3)4]2+

2. An excess of nitric acid solution is added to a solution of tetraaminecopper(II) sulfate. H+ + [Cu(NH 3)4]2+ NH 4+ + Cu 2+

3. Dilute hydrochloric acid is added to a solution of diamminesilver(I) nitrate. H+ + Cl- + [Ag(NH 3)2]+ Ag. Cl + NH 4+

4. Solid aluminum nitrate is dissolved in water. Al(NO 3)3 + H 2 O +3 [Al(H 2 O)6] + NO 3

5. A suspension of copper(II) hydroxide is treated with an excess of ammonia water. Cu(OH)2 + NH 3 [Cu(NH 3)4]2+ + OH-
![6. A solution of diamminesilver(I) chloride is treated with dilute nitric acid. [Ag(NH 3)2]+ 6. A solution of diamminesilver(I) chloride is treated with dilute nitric acid. [Ag(NH 3)2]+](http://slidetodoc.com/presentation_image_h2/9c3766b91f00a5a02a1b100bb364845a/image-161.jpg)
6. A solution of diamminesilver(I) chloride is treated with dilute nitric acid. [Ag(NH 3)2]+ + Cl- + H+ NH 4+ + Ag. Cl

7. An excess of concentrated ammonia solution is added to freshly precipitated copper(II) hydroxide. NH 3 + Cu(OH)2 [Cu(NH 3)4]2+ + OH-

8. Excess dilute nitric acid is added to a solution containing the tetraaminecadmium(II) ion. H+ + [Cd(NH 3)4]2+ NH 4+ + Cd 2+

9. An excess of ammonia is bubbled through a solution saturated with silver chloride. NH 3 + Ag+ [Ag(NH 3)2]+

10. Solid aluminum oxide is added to a solution of sodium hydroxide. Al 2 O 3 + OH- [Al(OH)4](may have water as a reactant)

11. A concentrated solution of ammonia is added to a solution of zinc iodide. NH 3 + 2+ Zn 2+ [Zn(NH 3)4]
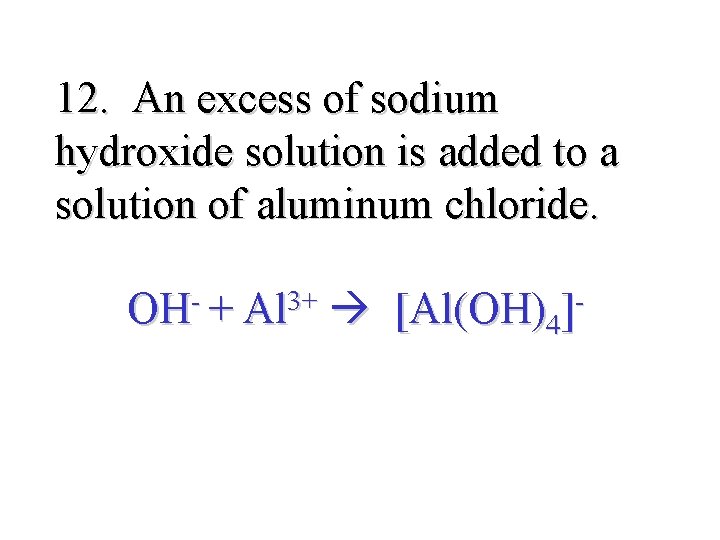
12. An excess of sodium hydroxide solution is added to a solution of aluminum chloride. OH- + Al 3+ [Al(OH)4]-

13. A concentrated solution of ammonia is added to a solution of copper(II) chloride. NH 3 + Cu 2+ [Cu(NH 3)4]2+

14. Excess concentrated sodium hydroxide solution is added to solid aluminum hydroxide. OH- + Al(OH)3 [Al(OH)4]-

15. Excess concentrated ammonia solution is added to a suspension of silver chloride. NH 3 + Ag. Cl + [Ag(NH 3)2] + Cl

16. Pellets of aluminum metal are added to a solution containing an excess of sodium hydroxide. Al + OH- [Al(OH)4]-

17. A suspension of zinc hydroxide is treated with concentrated sodium hydroxide solution. Zn(OH)2 + OH- [Zn(OH)4]2 -
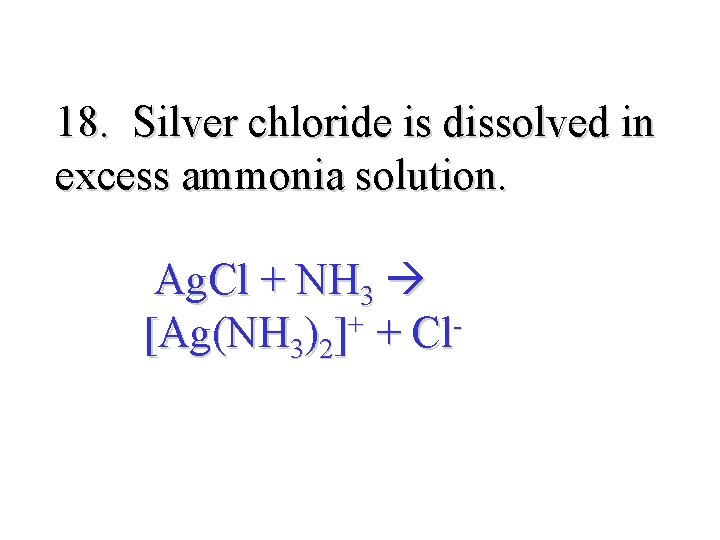
18. Silver chloride is dissolved in excess ammonia solution. Ag. Cl + NH 3 [Ag(NH 3)2]+ + Cl-

19. Sodium hydroxide solution is added to a precipitate of aluminum hydroxide in water. OH- + Al(OH)3 [Al(OH)4]-

20. A drop of potassium thiocyanate is added to a solution of iron(III) chloride. SCN + 3+ Fe 2+ Fe. SCN

21. A concentrated solution of ammonia is added to a suspension of zinc hydroxide. NH 3 + Zn(OH)2 2+ [Zn(NH 3)4] + OH

22. Excess concentrated potassium hydroxide solution is added to a precipitate of zinc hydroxide. Zn(OH)2 + OH- [Zn(OH)4]2 -

23. A solution of ammonium thiocyanate is added to a solution of iron(III) chloride. SCN- + Fe 3+ --> Fe. SCN 2+

24. Excess sodium cyanide is added to a solution of silver nitrate. CN- + Ag+ --> Ag(CN)2 -
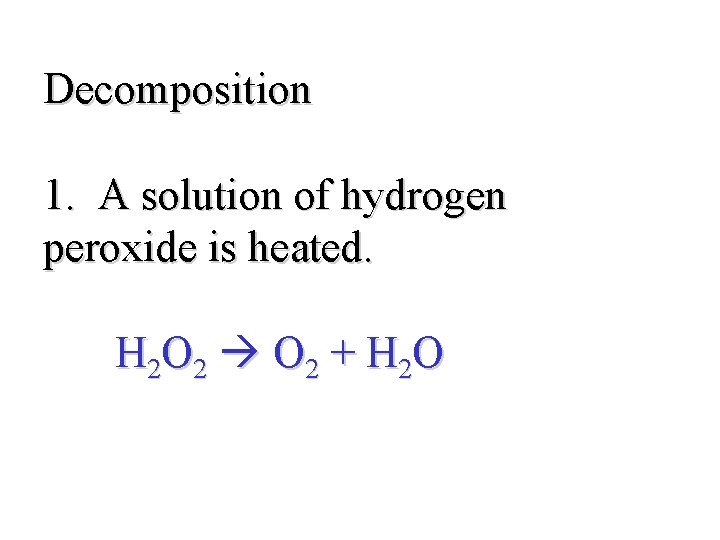
Decomposition 1. A solution of hydrogen peroxide is heated. H 2 O 2 + H 2 O

2. Solid magnesium carbonate is heated. Mg. CO 3 Mg. O + CO 2
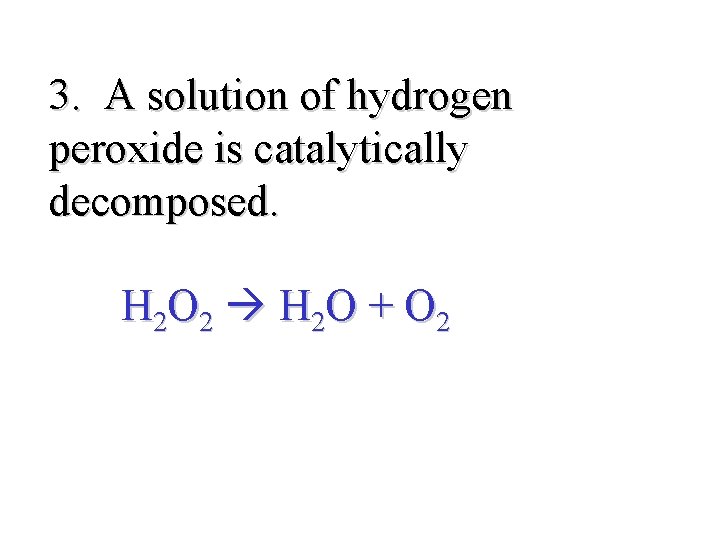
3. A solution of hydrogen peroxide is catalytically decomposed. H 2 O 2 H 2 O + O 2

4. Solid potassium chlorate is heated in the presence of manganese dioxide as a catalyst. KCl. O 3 --> KCl + O 2

5. Sodium hydrogen carbonate is dissolved in water. Na. HCO 3 + Na + HCO 3

6. Solid ammonium carbonate is heated. (NH 4)2 CO 3 CO 2 + NH 3 + H 2 O

Addition 1. The gases boron trifluoride and ammonia are mixed. BF 3 + NH 3 F 3 BNH 3

2. A mixture of solid calcium oxide and solid tetraphosphorus decaoxide is heated. Ca. O + P 4 O 10 Ca 3(PO 4)2

3. Solid calcium oxide is exposed to a stream of carbon dioxide gas. Ca. O + CO 2 Ca. CO 3

4. Solid calcium oxide is heated in the presence of sulfur trioxide gas. Ca. O + SO 3 Ca. SO 4

5. Calcium metal is heated strongly in nitrogen gas. Ca + N 2 Ca 3 N 2

6. Excess chlorine gas is passed over hot iron filings. Cl 2 + Fe Fe. Cl 3
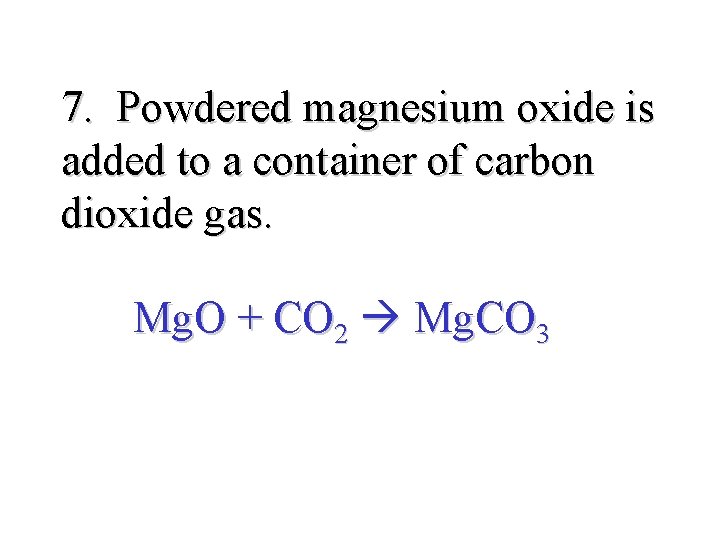
7. Powdered magnesium oxide is added to a container of carbon dioxide gas. Mg. O + CO 2 Mg. CO 3

8. A piece of lithium metal is dropped into a container of nitrogen gas. Li + N 2 Li 3 N
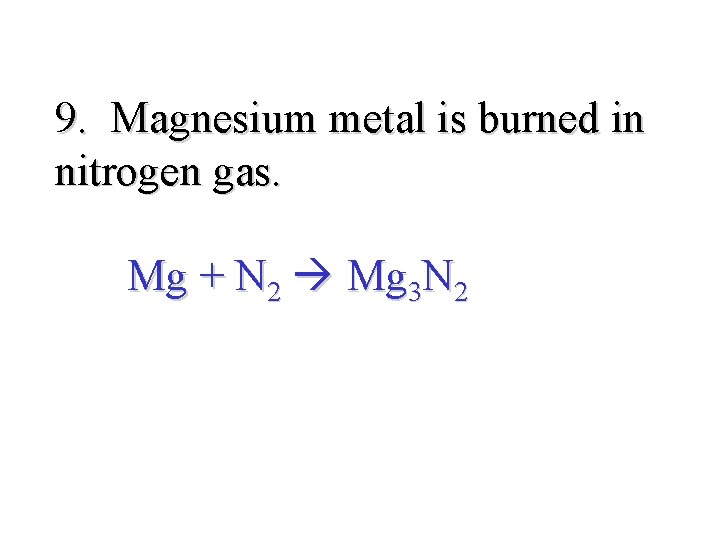
9. Magnesium metal is burned in nitrogen gas. Mg + N 2 Mg 3 N 2

10. Sulfur dioxide gas is passed over solid calcium oxide. SO 2 + Ca. O Ca. SO 3

11. Samples of boron trichloride gas and ammonia gas are mixed. BCl 3 + NH 3 --> H 3 NBCl 3
- Slides: 196