Double Bond C 2 H 4 n An
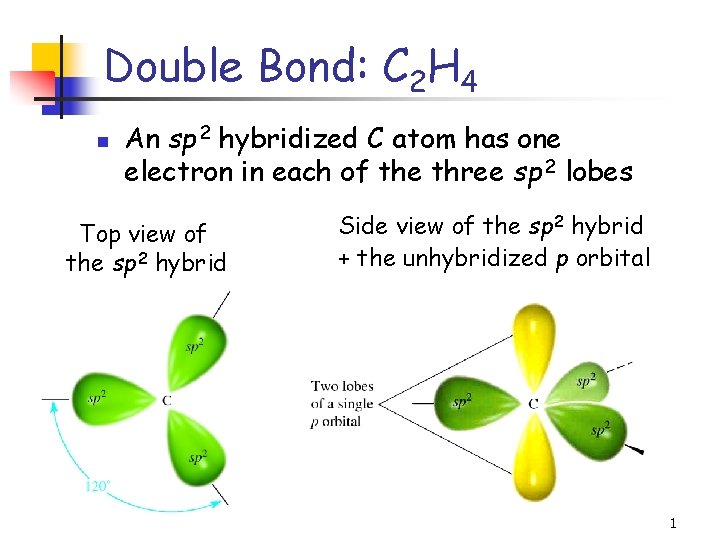
Double Bond: C 2 H 4 n An sp 2 hybridized C atom has one electron in each of the three sp 2 lobes Top view of the sp 2 hybrid Side view of the sp 2 hybrid + the unhybridized p orbital 1
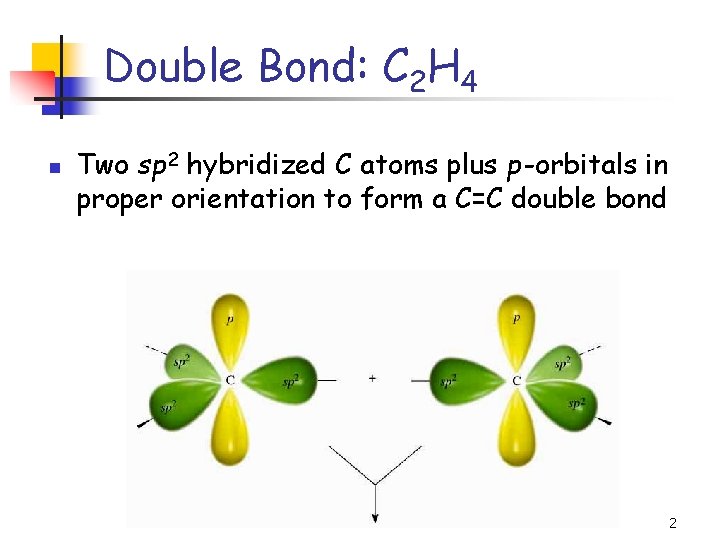
Double Bond: C 2 H 4 n Two sp 2 hybridized C atoms plus p -orbitals in proper orientation to form a C=C double bond 2

Double Bond: C 2 H 4 n n The portion of the double bond formed from the head-on overlap of the sp 2 hybrids is designated as a bond The other portion of the double bond, resulting from the side-on overlap of the p orbitals, is designated as a p bond 3
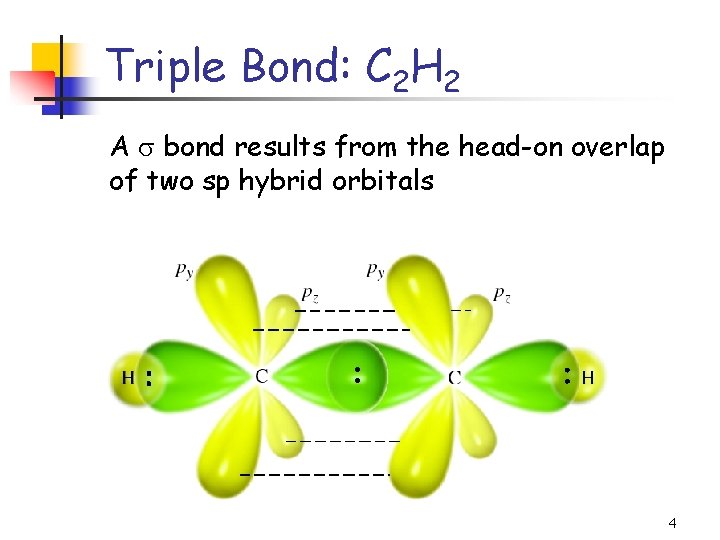
Triple Bond: C 2 H 2 A bond results from the head-on overlap of two sp hybrid orbitals 4
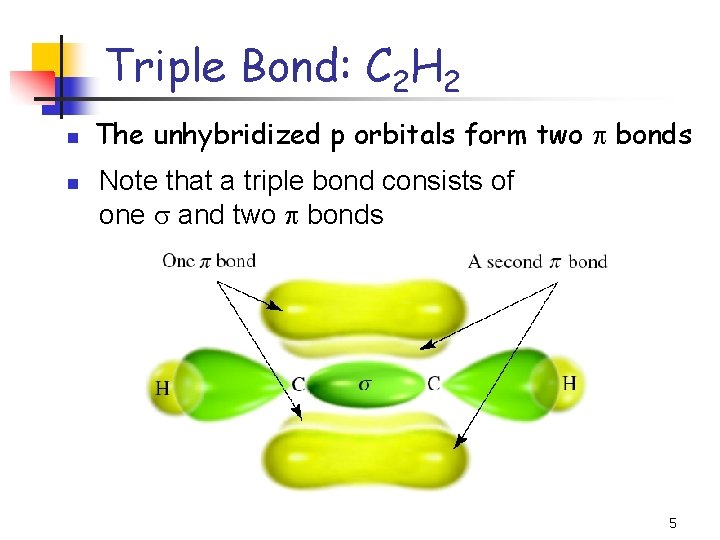
Triple Bond: C 2 H 2 n n The unhybridized p orbitals form two p bonds Note that a triple bond consists of one and two p bonds 5

CHAPTER 10 n Reactions in Aqueous Solutions I: Acids, Bases, and Salts 6

The Arrhenius Theory n n n Acids are substances that contain hydrogen and produce H+ cations in aqueous solutions Bases are substances that contain a hydroxyl group and produce OH– anions in aqueous solutions These two statements represent the Arrhenius theory of acids and bases 7

The BrØnsted-Lowry Theory n This theory is more general than the Arrhenius theory n n An acid is a proton donor (H+) A base is a proton acceptor NH 3(aq) + H 2 O(l) NH 4+(aq) + OH–(aq) base n acid Notice, that according to the Arrhenius theory NH 3 is not a base 8

The Hydronium Ion n n The protons (H+) are never present in solution by themselves Protons are always hydrated that is surrounded by several water molecules n We don’t know exactly how many H+(aq) is really H(H 2 O)n+ n Where n is a small integer We normally write the hydrated hydrogen ion as H 3 O+ and call it the hydronium ion 9

The BrØnsted-Lowry Theory n Acid-base reactions are the transfer of a proton from an acid to a base NH 3(aq) + HCl(aq) NH 4+(aq) + Cl–(aq) base acid HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl–(aq) acid base 10

Conjugate Acid-Base Pairs HF(aq) + H 2 O(l) H 3 O+(aq) + F–(aq) acid n n base acid base HF is a weak acid and it does not ionize completely in aqueous solutions This reaction also proceeds in the reverse direction (it is reversible) Two species that differ by a proton are called conjugate acid-base pairs Such conjugate pairs will exist for each acid-base reaction 11

Conjugate Acid-Base Pairs n n To find a conjugated base: + n Add 1 proton (H ) to the acid n Increase the total charge by 1 To find a conjugated acid: n Remove 1 proton from the base n Decrease the total charge by 1 H 2 O NH 3 CH 3 COOH CH 3 COO– 12

Conjugate Acid-Base Pairs HF(aq) + H 2 O(l) acid 1 base 2 H 3 O+(aq) + F–(aq) acid 2 base 1 n HF is a weak acid and ionizes only slightly n It prefers to exist as HF(aq) rather than F–(aq) n n n This means that F–(aq) has higher affinity to the proton than H 2 O (it holds the proton stronger) Another way to put it is to say that F–(aq) is a stronger base than H 2 O H 3 O+(aq) gives up the proton easier than HF(aq) and therefore it is a stronger acid than HF(aq) (it is more prone to loosing the proton) 13

The BrØnsted-Lowry Theory n n n Important conclusion: n Weak acids have strong conjugate bases n Weak bases have strong conjugate acids The weaker the acid or base, the stronger the conjugate partner Another example: ammonia in water 14

Properties of Acids n n n Solutions of acids have a sour taste n Don’t taste them in the lab !!! They change the colors of many indicators n Acids turn blue litmus to red n Acids turn bromothymol blue from blue to yellow They react with metals to generate hydrogen gas, H 2 15

Displacement Reactions n Displacement reactions occur when one element displaces another element from a compound: Zn + H 2 SO 4 Zn. SO 4 + H 2 n n Total and net ionic equations: Not all the metals are capable of displacing hydrogen from an acid 16
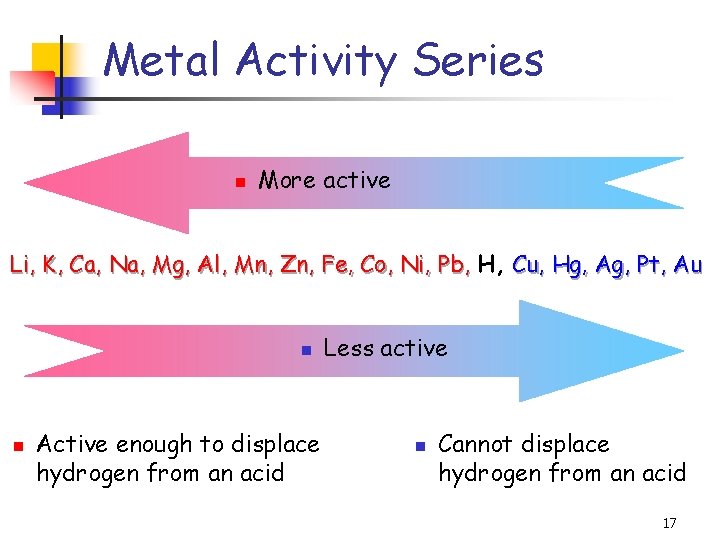
Metal Activity Series n More active Li, K, Ca, Na, Mg, Al, Mn, Zn, Fe, Co, Ni, Pb, H, Cu, Hg, Ag, Pt, Au n n Active enough to displace hydrogen from an acid Less active n Cannot displace hydrogen from an acid 17

Example 1 n Write reactions between the following metals and HCl solution. Write total and net ionic equations in each case n Fe, Na, Pt, Ni, Cu 18

Example 1 (continued) n Write reactions between the following metals and HCl solution. Write total and net ionic equations in each case n Fe, Na, Pt, Ni, Cu 19

Metal Activity Series n The more active metal will always displace the less active metal from the solution of its salt: Cu + 2 Ag. NO 3 Cu(NO 3)2 + 2 Ag 20

Example 2 n Write reactions between the following substances in aqueous solutions: n Zn + Cu. SO 4 n Hg + Fe(NO 2)3 n Mg + Hg(NO 3)2 n Al + Fe(NO 3)3 21

Example 2 (continued) 22

Assignments & Reminders n Go through the lecture notes n Read Chapter 10 completely n Read Section 4 -10 of Chapter 4 n n Monday (10/31) and Tuesday (11/1) – lecture quiz #5 (Chapter 8) Homework #5 is due by Monday (10/31) 23
- Slides: 23