Doped and self doped double exchange in case
Doped and self doped double exchange in case of Cr. O 2 Group meeting 14 -06 -06 Muhammad Shahbaz Anwar

Outlines • Both manganites such as (La, Sr)Mn. O 3 or (La, Ca)Mn. O 3 and Cr. O 2 are half metallic. Can that be understood in one picture. • Superexchange and Double exchange. • Cr. O 2

Direct and Indirect Exchange • • Exchange interaction proceeds without the need for an intermediate atom. Very often it cannot be important mechanism in controlling magnetic properties as there is insufficient direct overlap between neighboring magnetic orbitals. • • A number of ionic solids, including some oxide and flurides, have magnetic ground state like Mn. O and Mn. F 2 but there is no direct exchange between Mn 2+ ions in each system. As direct interaction is short range interaction so there is a long range interaction is operating.
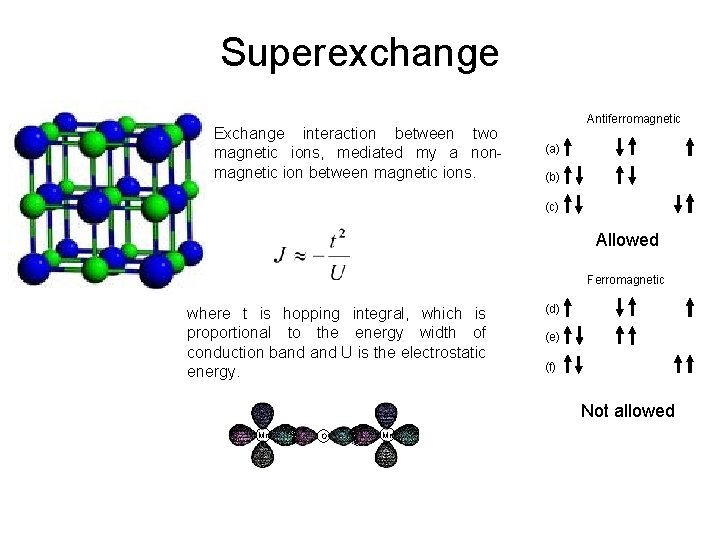
Superexchange Exchange interaction between two magnetic ions, mediated my a nonmagnetic ion between magnetic ions. Antiferromagnetic (a) (b) (c) Allowed Ferromagnetic where t is hopping integral, which is proportional to the energy width of conduction band U is the electrostatic energy. (d) (e) (f) Not allowed Mn O Mn
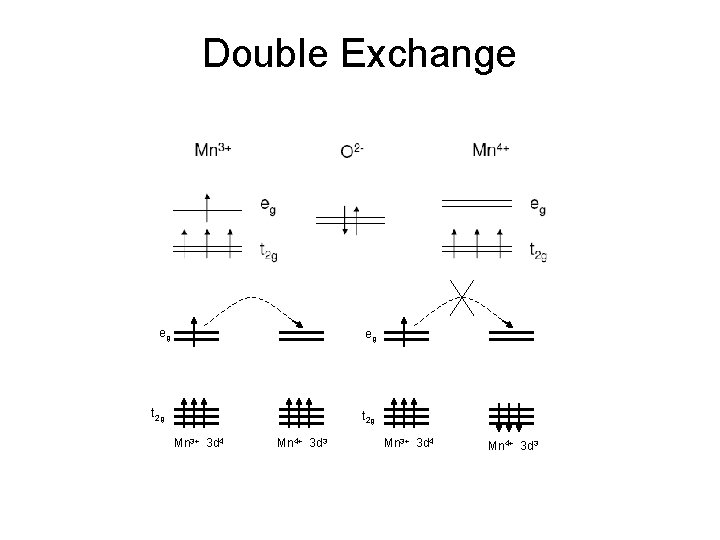
Double Exchange eg eg t 2 g Mn 3+ 3 d 4 Mn 4+ 3 d 3
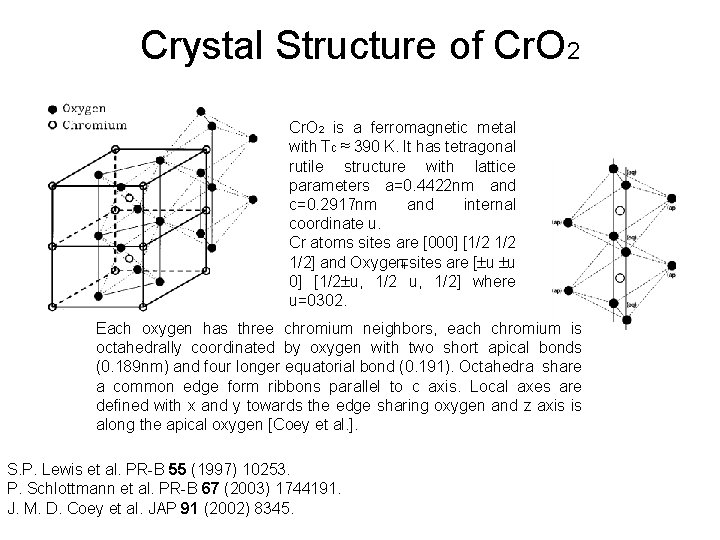
Crystal Structure of Cr. O 2 is a ferromagnetic metal with Tc ≈ 390 K. It has tetragonal rutile structure with lattice parameters a=0. 4422 nm and c=0. 2917 nm and internal coordinate u. Cr atoms sites are [000] [1/2 1/2] and Oxygen sites are [ u u 0] [1/2 u, 1/2] where u=0302. Each oxygen has three chromium neighbors, each chromium is octahedrally coordinated by oxygen with two short apical bonds (0. 189 nm) and four longer equatorial bond (0. 191). Octahedra share a common edge form ribbons parallel to c axis. Local axes are defined with x and y towards the edge sharing oxygen and z axis is along the apical oxygen [Coey et al. ]. S. P. Lewis et al. PR-B 55 (1997) 10253. P. Schlottmann et al. PR-B 67 (2003) 1744191. J. M. D. Coey et al. JAP 91 (2002) 8345.
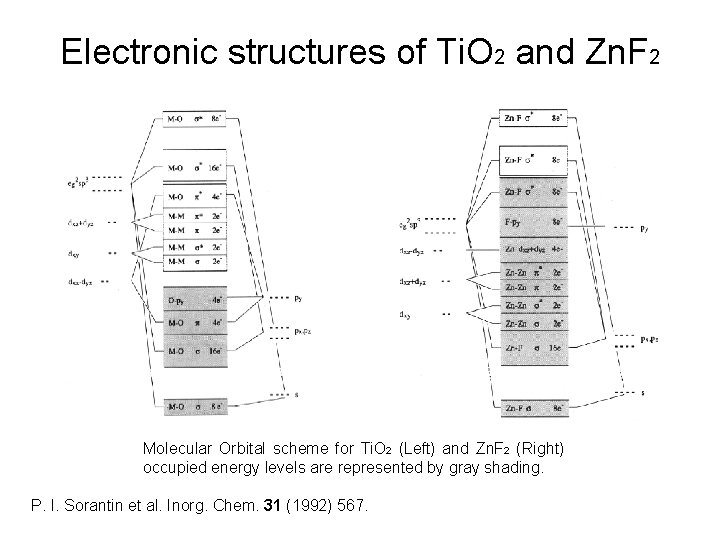
Electronic structures of Ti. O 2 and Zn. F 2 Molecular Orbital scheme for Ti. O 2 (Left) and Zn. F 2 (Right) occupied energy levels are represented by gray shading. P. I. Sorantin et al. Inorg. Chem. 31 (1992) 567.
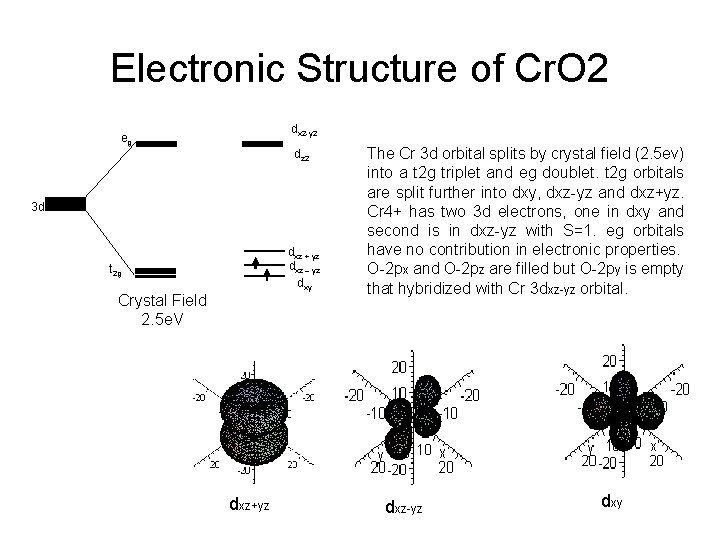
Electronic Structure of Cr. O 2 dx 2 -y 2 eg dz 2 3 d dxz + yz dxz – yz dxy t 2 g Crystal Field 2. 5 e. V dxz+yz The Cr 3 d orbital splits by crystal field (2. 5 ev) into a t 2 g triplet and eg doublet. t 2 g orbitals are split further into dxy, dxz-yz and dxz+yz. Cr 4+ has two 3 d electrons, one in dxy and second is in dxz-yz with S=1. eg orbitals have no contribution in electronic properties. O-2 px and O-2 pz are filled but O-2 py is empty that hybridized with Cr 3 dxz-yz orbital. dxz-yz dxy
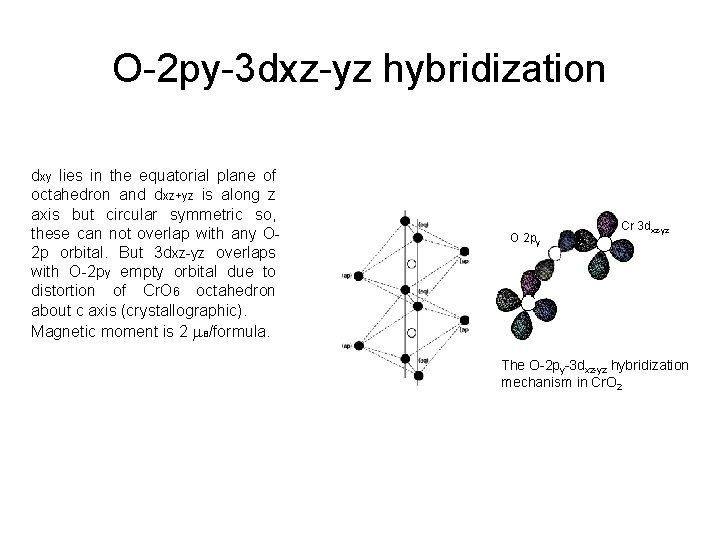
O-2 py-3 dxz-yz hybridization dxy lies in the equatorial plane of octahedron and dxz+yz is along z axis but circular symmetric so, these can not overlap with any O 2 p orbital. But 3 dxz-yz overlaps with O-2 py empty orbital due to distortion of Cr. O 6 octahedron about c axis (crystallographic). Magnetic moment is 2 B/formula. O 2 py Cr 3 dxz-yz The O-2 py-3 dxz-yz hybridization mechanism in Cr. O 2
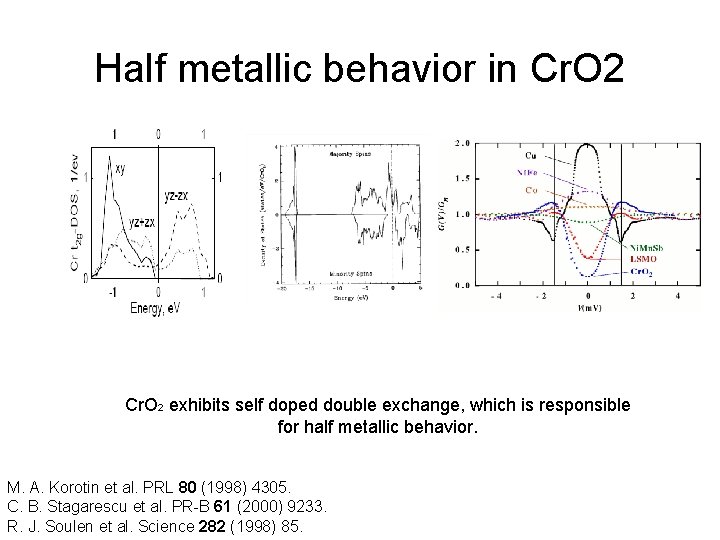
Half metallic behavior in Cr. O 2 exhibits self doped double exchange, which is responsible for half metallic behavior. M. A. Korotin et al. PRL 80 (1998) 4305. C. B. Stagarescu et al. PR-B 61 (2000) 9233. R. J. Soulen et al. Science 282 (1998) 85.
- Slides: 10