DONOR BIOPSY EVALUATION LIVER TRANSPLANTATION PATHOLOGY Liver Transplantation

DONOR BIOPSY EVALUATION

LIVER TRANSPLANTATION PATHOLOGY

Liver Transplantation Pathology • Liver transplantation is currently the ultimate treatment of choice for previously incurable liver diseases, which fall into four categories – Cirrhosis – Acute liver failure with submassive or massive liver necrosis – Malignant liver tumors – Chronic metabolic or genetic diseases
Liver Transplantation Pathology Cirrhosis • Chronic Hepatitis – Due to chronic viral hepatitis C and B – Alcoholic and NASH – Account for the majority of cases • Cryptogenic cirrhosis – Due to precedent NASH • • PBC PSC AIH Cirrhosis is found in 70– 80 % of all patients receiving liver transplants
Liver Transplantation Pathology Acute liver failure • • Due to acetaminophen toxicity Fulminant viral hepatitis Drugs & toxins Account for 5– 10 % transplants
Liver Transplantation Pathology Malignant tumors • Account for 10 % of LTs Metabolic/genetic diseases • Account for <5 % of all LTs Failure of the first transplant • 15 % of liver transplantations Repeat transplantations on persons who already had two previous transplantations • 1– 2 % represent
Liver Transplantation Pathology • Problems facing pathologists in liver transplants – Quality of the graft which translates to graft viability – Graft preservation/reperfusion injury – Vascular & biliary duct injuries related to transplantation – Immune-mediated transplant rejection • Acute and chronic cell mediated rejection • Antibody mediated rejection – Complications of immunosuppressive therapy • Opportunistic infection & PTLD – Recurrence of the original or primary liver disease – Development of an additional liver disease that did not exist in the donor or the graft

QUALITY OF THE GRAFT

Quality of the Graft • Quality of the grafts obtained from deceased donors is assessed in frozen sections preferably using at least two core needle biopsies and a wedge biopsy specimen • The most important features evaluated in biopsies of donor livers – Fatty change – Necrosis – Fibrosis – Inflammation • These pathologic findings are interpreted in conjunction with clinical data provided about the potential donor

Quality of the Graft • To increase the number of potential donors special criteria have been developed for extended criteria donors(ECD) – Donor age >60 years of age – >40 % macrovesicular steatosis – Cold ischemia time >12 h – Donation after cardiac death (DCD) – Donor exposure risks • History of active injection drug abuse • Incarcerated status to mention just a few

Quality of the Graft Macrovesicular fatty change • Involving 10– 30 % hepatocytes can be acceptable for transplantation • Donor livers showing >30 % of macrovesicular fatty change are considered high risk and many times unsuitable for transplantation • Such livers are prone to preservation/reperfusion injury and the transplantation is associated with reduced graft survival • Livers showing moderate macrovesicular steatosis, in the range from 10 % to 30 %, are considered for transplantation on an individual basis in context of ECD • Most of the fat is cleared from hepatocytes during the first 3 weeks after transplantation
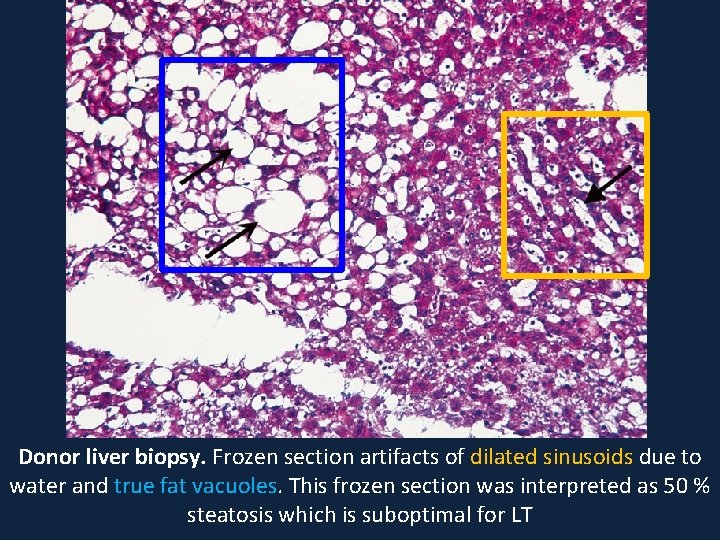
Donor liver biopsy. Frozen section artifacts of dilated sinusoids due to water and true fat vacuoles. This frozen section was interpreted as 50 % steatosis which is suboptimal for LT

Quality of the Graft Necrosis • If cellular necrosis involves >10 % of all hepatocytes the liver is considered unsuitable for transplantation • Subcapsular necrosis is common in wedge biopsies due to manipulation of the organ during harvesting and this it should be diagnosed only if present in core needle biopsies

Quality of the Graft Fibrosis • Donors are disqualified if the liver shows bridging fibrosis, incomplete or complete cirrhosis

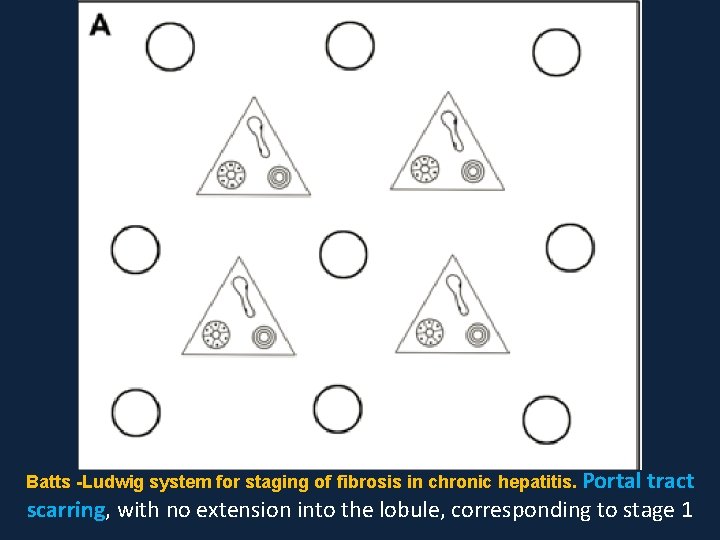
Batts -Ludwig system for staging of fibrosis in chronic hepatitis. Portal tract scarring, with no extension into the lobule, corresponding to stage 1
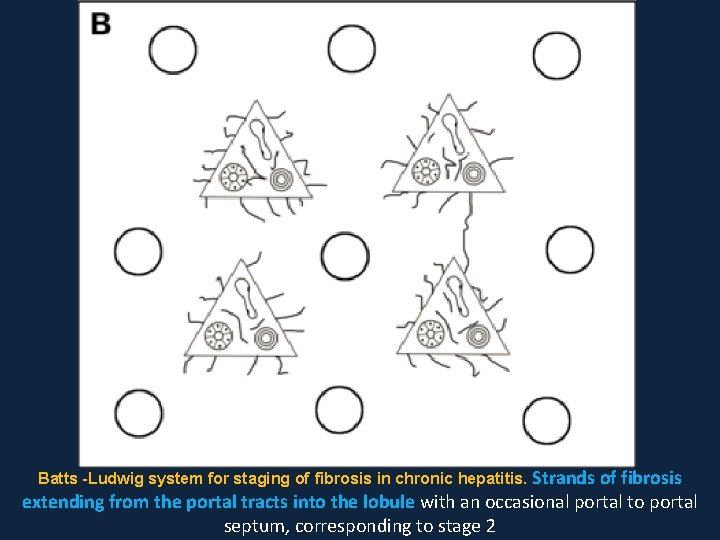
Batts -Ludwig system for staging of fibrosis in chronic hepatitis. Strands of fibrosis extending from the portal tracts into the lobule with an occasional portal to portal septum, corresponding to stage 2
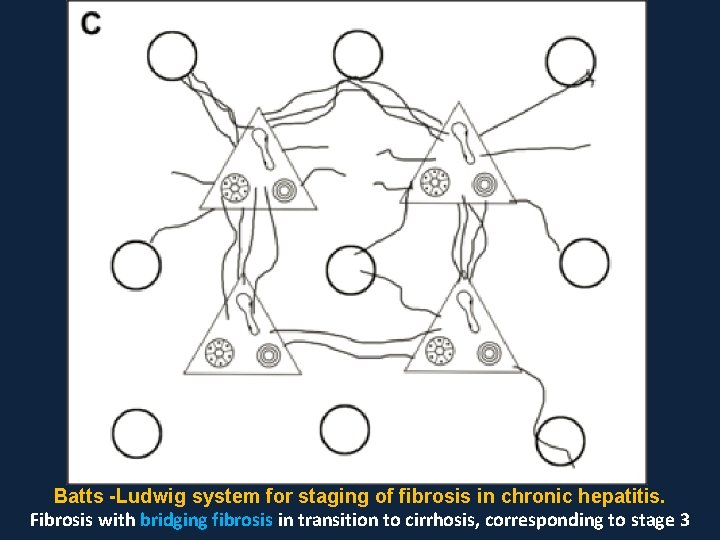
Batts -Ludwig system for staging of fibrosis in chronic hepatitis. Fibrosis with bridging fibrosis in transition to cirrhosis, corresponding to stage 3
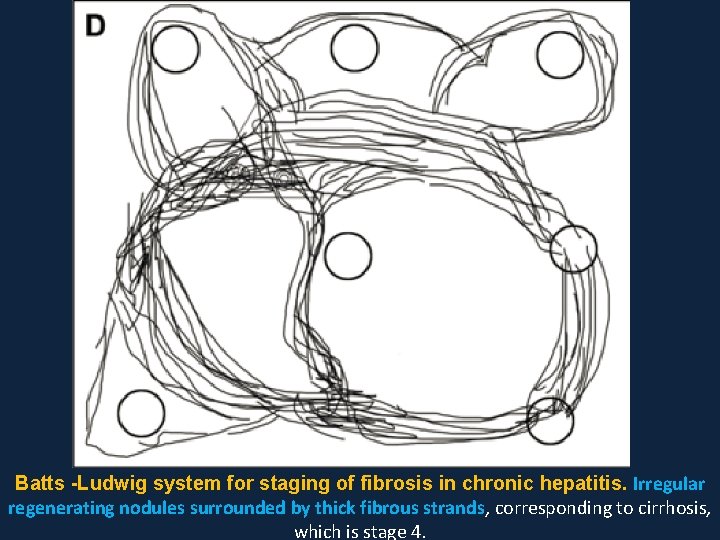
Batts -Ludwig system for staging of fibrosis in chronic hepatitis. Irregular regenerating nodules surrounded by thick fibrous strands, corresponding to cirrhosis, which is stage 4.

Quality of the Graft

Quality of the Graft

Quality of the Graft Inflammation • Donors are disqualified if the liver shows – Prominent portal inflammation or – Inflammation extending from the portal tracts into the lobules • Livers from donors infected with HCV can be considered for transplantation into HCV+ recipients if the inflammation is mild and the stage of fibrosis is <Ishak Stage 3/6 (no evidence of bridging fibrosis) • With the advent of new highly effective and well tolerated HCV therapies, there may be relaxation in use of grafts positive for HCV in the future

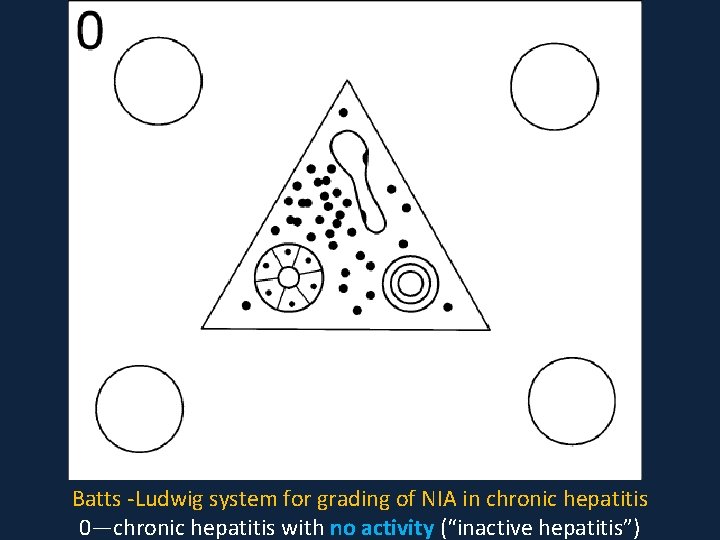
Batts -Ludwig system for grading of NIA in chronic hepatitis 0—chronic hepatitis with no activity (“inactive hepatitis”)
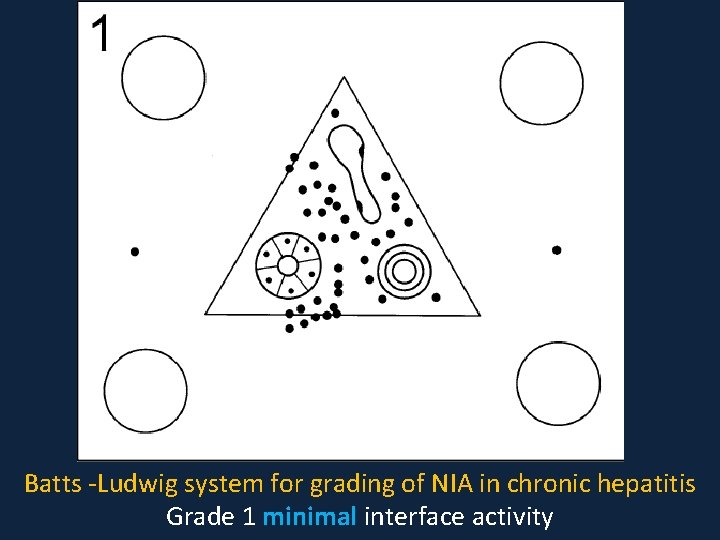
Batts -Ludwig system for grading of NIA in chronic hepatitis Grade 1 minimal interface activity
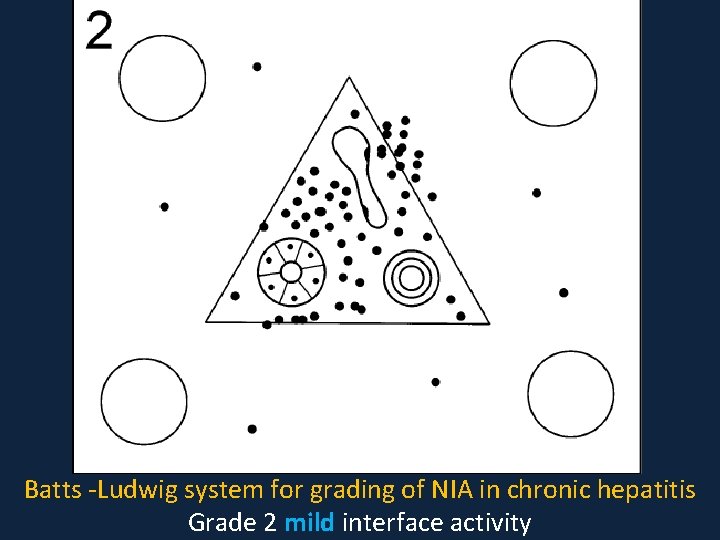
Batts -Ludwig system for grading of NIA in chronic hepatitis Grade 2 mild interface activity
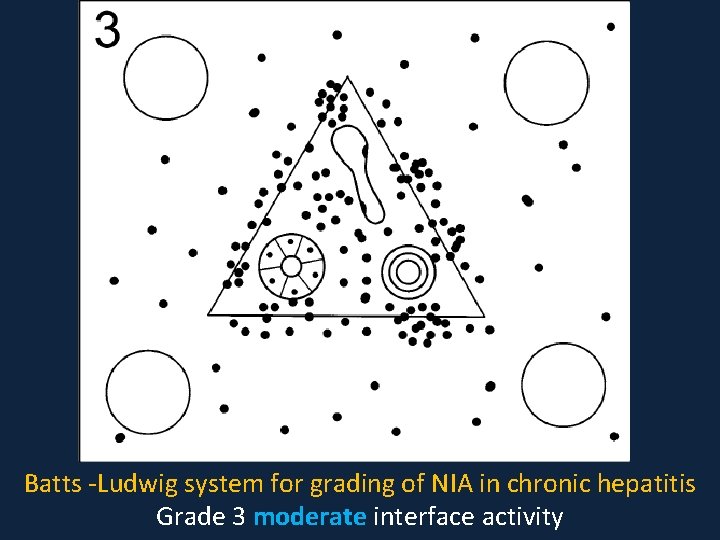
Batts -Ludwig system for grading of NIA in chronic hepatitis Grade 3 moderate interface activity
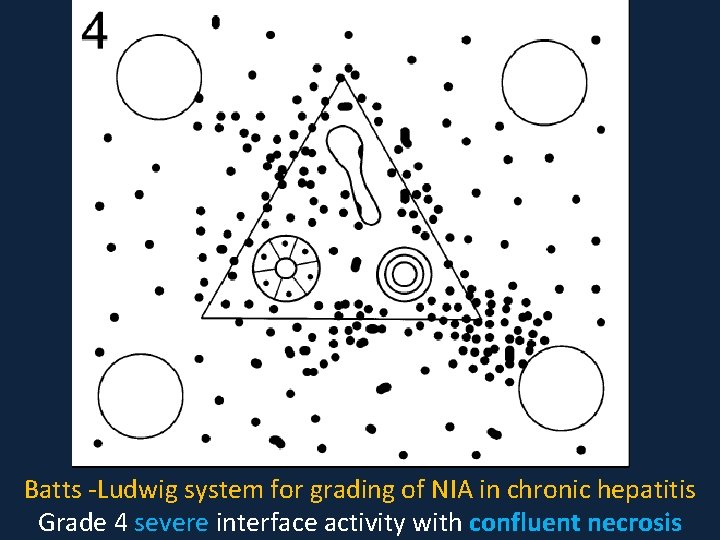
Batts -Ludwig system for grading of NIA in chronic hepatitis Grade 4 severe interface activity with confluent necrosis

Quality of the Graft Comments • Livers from donors who have malignant tumors or have advanced atherosclerotic cardiovascular disease are not suitable for transplantation • Small vacuolar steatosis (previously known as “microvesicular steatosis”) does not adversely affect the fate of the graft and is not considered to be a risk factor unless severe • Most living donor livers are not biopsied prior to transplantation, unless the donors have some potential risk factors • The extent of macrovesicular steatosis in obese donors can be reduced by preoperative dietary regimens • Not all obese donors have fatty changes in more than 30 % hepatocytes

Donor Biopsy Evaluation • Nonspecific findings that do not preclude successful transplantation – Mild mononuclear portal inflammatory cell infiltrates – Bile ductular proliferation – Cholestasis
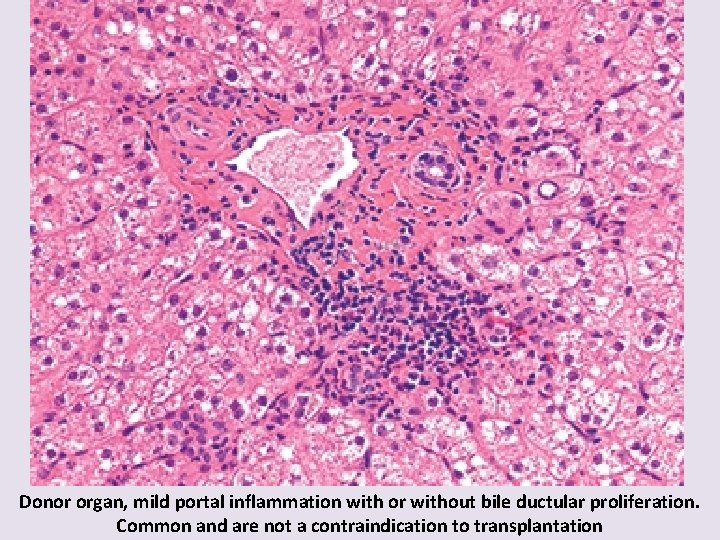
Donor organ, mild portal inflammation with or without bile ductular proliferation. Common and are not a contraindication to transplantation
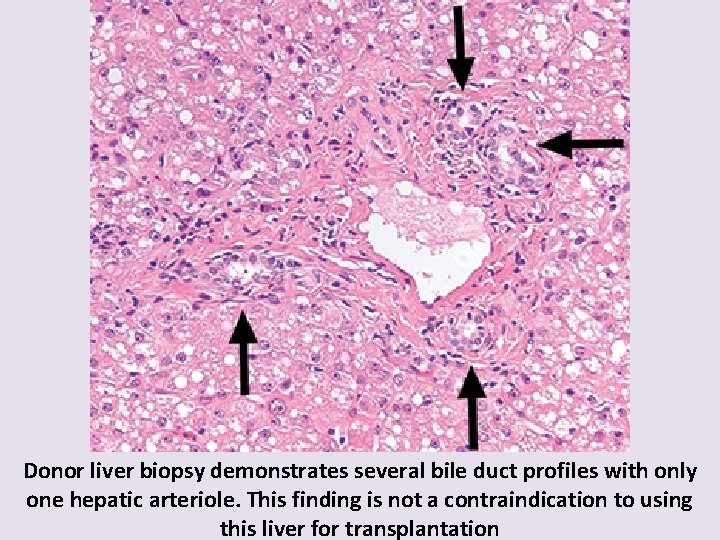
Donor liver biopsy demonstrates several bile duct profiles with only one hepatic arteriole. This finding is not a contraindication to using this liver for transplantation

LIVING DONORS

Living Donors • Disqualification rates based on biopsy findings alone vary from 3% to 21% • Most biopsies either have normal findings or show mild steatosis – 20% to 50% show, mostly mild, abnormalities

Living Donors / Macrovesicular steatosis • The most common histopathological abnormality • Present in 14% to 53% of potential living donors • Macrovesicular steatosis exceeding a certain, usually center-specific, threshold is the most common reason for donor disqualification

Living Donors / Macrovesicular steatosis • Most programs try to limit the severity of macrovesicular steatosis in living donors to <30% because this level of steatosis does not appear to adversely impact the postsurgical course of either donor or recipient • Other programs more conservatively limit macrovesicular steatosis to <10% or 20% • Diet modification and other treatments to reduce hepatic steatosis can be used to requalify potential donors

Living Donors • Other common findings in potential living donor biopsies include – Idiopathic low-grade chronic hepatitis – Nonnecrotizing granulomas – A variety other unexpected findings such as unexpected early stage PBC

Living Donors Iron deposits • Mild (1 to 2+ on a scale of 0 to 4) periportal hepatocellular iron deposits – Present in 17% of donors – M>>>F – A “normal” finding in males – Does not preclude transplantation
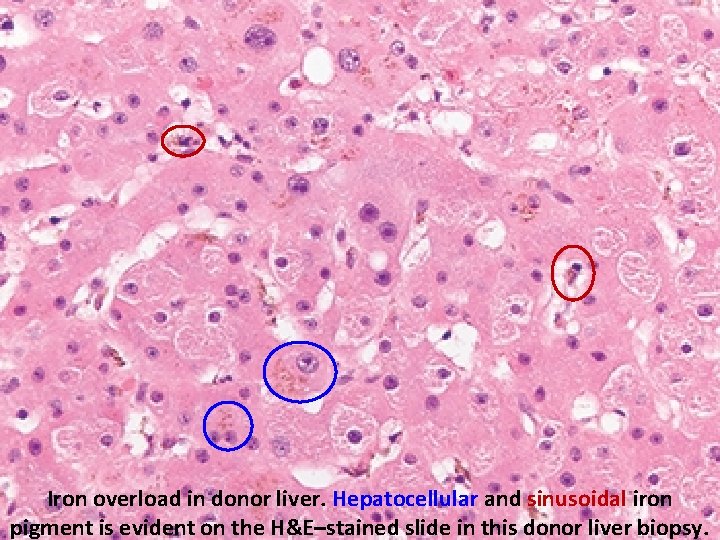
Iron overload in donor liver. Hepatocellular and sinusoidal iron pigment is evident on the H&E–stained slide in this donor liver biopsy.
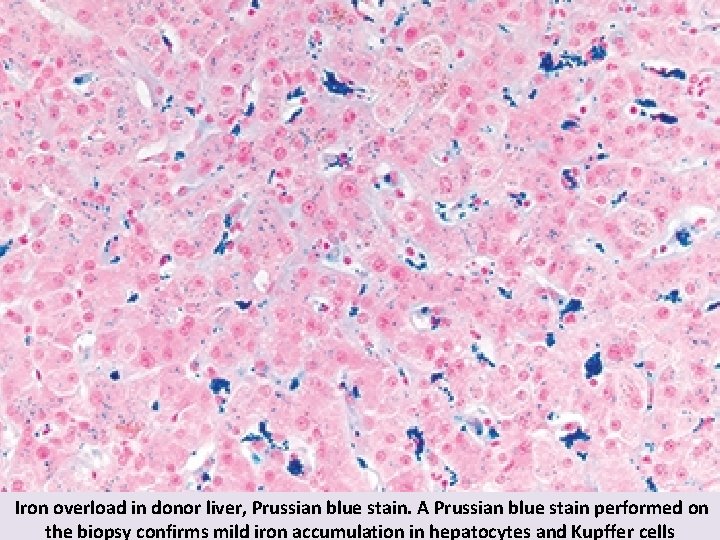
Iron overload in donor liver, Prussian blue stain. A Prussian blue stain performed on the biopsy confirms mild iron accumulation in hepatocytes and Kupffer cells

Living Donors Eosinophilia • Unexplained portal tract eosinophilia is occasionally seen • Does not appear to adversely affect the clinical course

DECEASED DONORS

Deceased Donors • Frozen section evaluation can assist in evaluation of nonideal or extended criteria donors (ECDs), defined by – Increased age (>60 years) – Macrovesicular steatosis (>40%) – Cold ischemia time exceeding 12 hours – Partial-liver allografts – Donation after cardiac death (DCD) – Hemodynamic instability – Use of vasopressors – Hypernatremia (>155 m. Eq /L) – HBV or HCV infection or anti–HBc antibody positivity – Liver mass – Fibrosis – Other focal lesions – History of cancer

Deceased Donors • Feng “donor risk score” based on a study of >20, 000 transplants – The score is based on differential weighing for • • Old age (>60 years) Anoxic and cerebrovascular causes of death Black race Short height DCD and split/partial grafts Regional or national sharing Cold ischemic time – The parameter scores are then summed, and higher total scores translate into greater risk and inversely correlate with 1 - and 3 -year survivals

Deceased Donors • Donor frozen section requests are most often prompted because of the – Gross appearance – Feel, or color of the donor liver – Known preexisting donor disease (e. g. , HCV) – A suspicious clinical history – Circumstances surrounding donor death or harvesting procedure • Tissue for frozen section evaluation should be representative and obtained fresh, preferably in the presence of the pathologist, who should also grossly inspect the donor liver

Deceased Donors • Number of the tissue samples – Three tissue samples if the gross appearance is uniform • Two 2 -cm 16 -gauge needle cores from the right & left lobes – Are used to stage fibrosis • One 2 -cm 2 subcapsular right lobe wedge biopsy – Helpful for evaluating arterial/arteriolar disease – Providing a larger sample for an assessment of steatosis

Deceased Donors • The fresh tissue samples should be immediately transported to the frozen section room – On a paper towel moistened with preservation solution or – In a plastic specimen container • Should be absolutely avoided – Storage in “physiologic” saline – Air drying – Placement of the tissue sample on an absorbent substrate
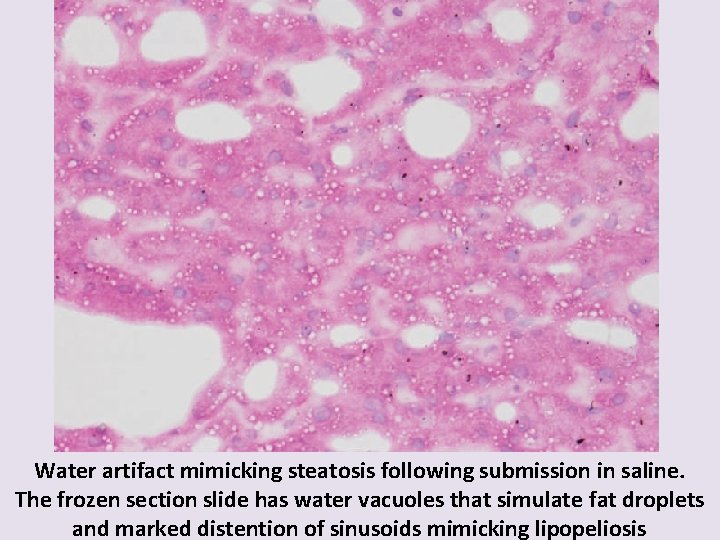
Water artifact mimicking steatosis following submission in saline. The frozen section slide has water vacuoles that simulate fat droplets and marked distention of sinusoids mimicking lipopeliosis
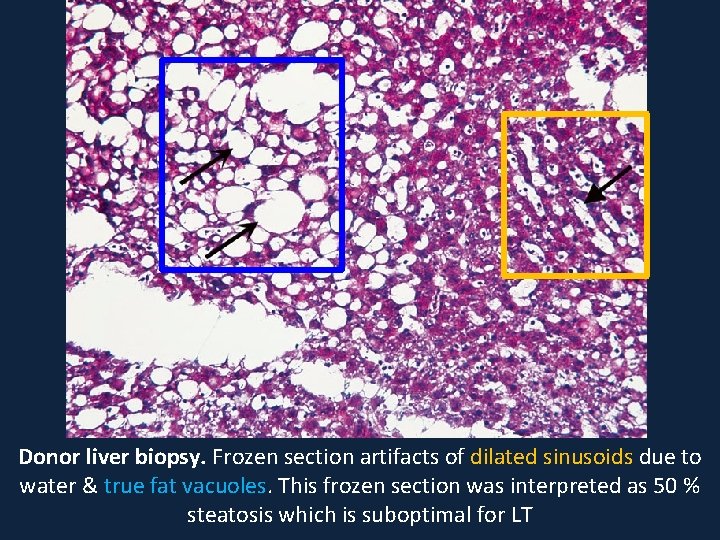
Donor liver biopsy. Frozen section artifacts of dilated sinusoids due to water & true fat vacuoles. This frozen section was interpreted as 50 % steatosis which is suboptimal for LT

Deceased Donors • Air drying and storage in physiologic saline can cause hepatocytes to appear shrunken/necrotic, leading to overestimation of ischemic injury • Absorbent substrates blot fat out of the tissue, resulting in false underestimation of the extent of fatty infiltration

Deceased Donors • Difficultly cutting the frozen section – Should alert the pathologist to the possibility of a steatotic donor liver – Can be correlated with the gross appearance • Necrosis – Recognition of hepatocytes in various stages of necrosis or apoptosis can be enhanced by staining several sections for increasing lengths of time in eosin – This approach enhances contrast between • Viable & • Damaged/nonviable hepatocytes – More hypereosinophilic and often show early nuclear karyorrhexis

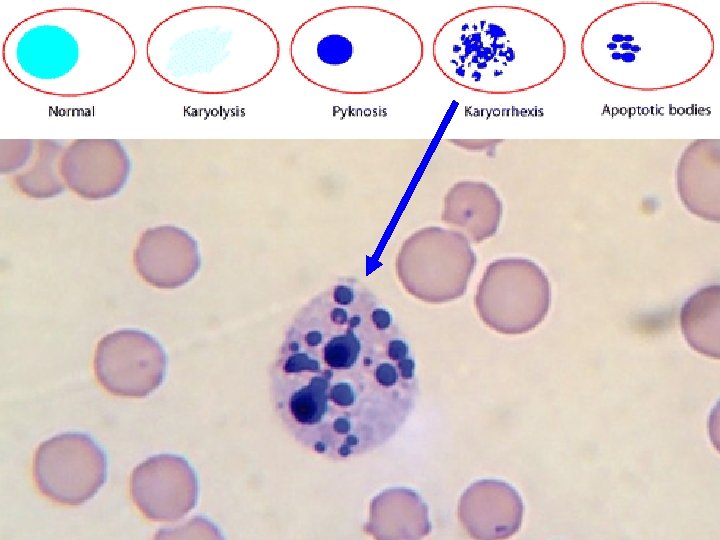

Deceased Donors • Any histopathological findings are then correlated with the donor history and laboratory values • Partial or fragmented clinical histories are common and can be substantially misleading • The pathologist should request additional information and/or samples if biopsy findings do not correlate with the given history or events

Deceased Donors • Donor biopsy evaluation is only one test • The pathologist is unable to predict the adequacy of organ function after transplantation based on frozen section light microscopic evaluation before the operation in the absence of significant histopathological findings

Deceased Donors • Reasons for donor organ disqualification vary by center • Most institutions will refuse donors that are found – To be positive for certain infections (e. g. , HIV, rabies) – Have a recent or even remote history of a highrisk malignancy

Deceased Donors • Biopsy findings that disqualify organs are also center specific but often include – Hepatocyte coagulative-type necrosis involving >25% of amply sampled parenchyma – Severe macrovesicular steatosis – Advanced intrahepatic atherosclerosis – Definite evidence of bridging fibrosis
Deceased Donors • Polarization microscopy can be used to quickly, easily, and accurately assess liver fibrosis without special stains • It is helpful therefore to equip frozen section room microscopes with this capability

Deceased Donors • A grossly fatty appearance is the most common reason for requesting frozen section evaluation of a deceased donor liver • Experienced donor surgeons are usually able to accurately estimate steatosis severity before biopsy evaluation • Gross estimates can be misleading – In donors with small vacuolar steatosis – When the donor liver is poorly illuminated during examination

Deceased Donors • Large vacuolar or macrovesicular steatosis – Fat globules>nuclear diameter – Displacement of the hepatocyte nucleus to the periphery of the cell • Small vacuolar or microvesicular steatosis – Multiple tiny fat globules – Usually substantially<nuclear diameter – Nucleus remains centrally placed
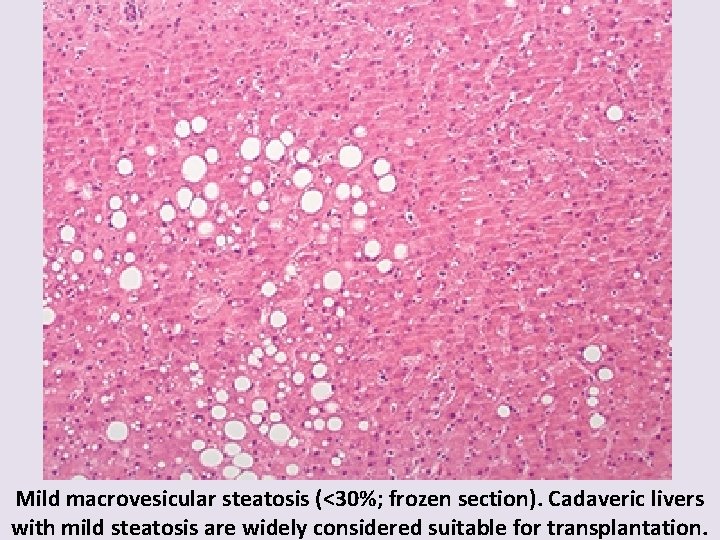
Mild macrovesicular steatosis (<30%; frozen section). Cadaveric livers with mild steatosis are widely considered suitable for transplantation.
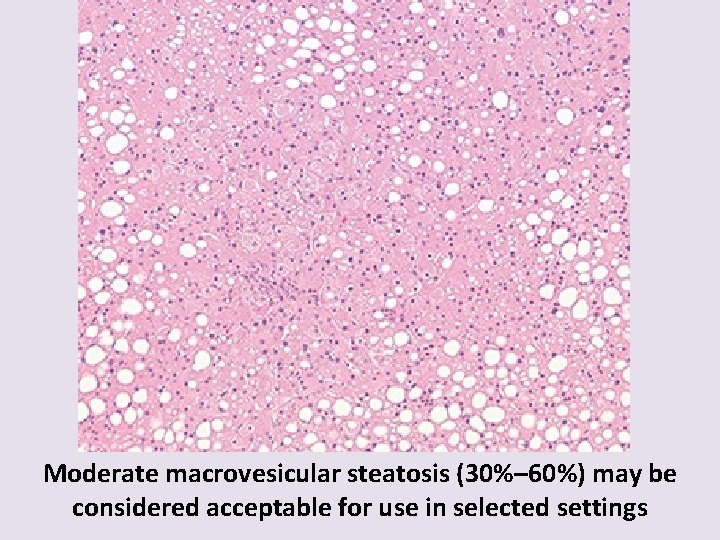
Moderate macrovesicular steatosis (30%– 60%) may be considered acceptable for use in selected settings
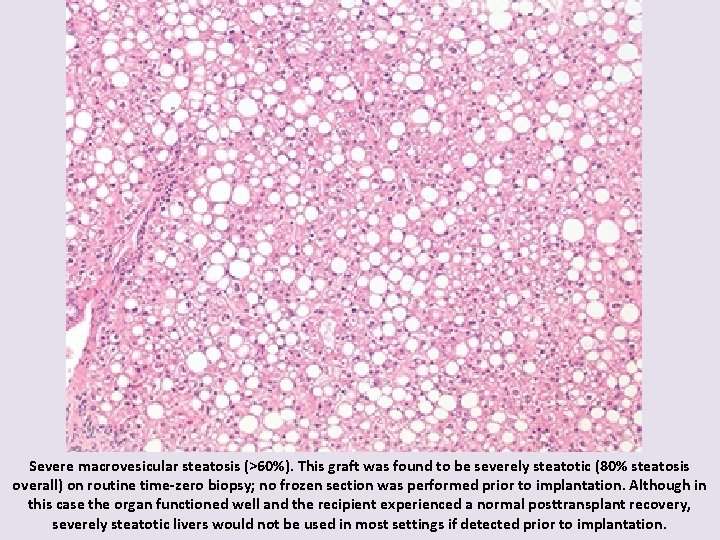
Severe macrovesicular steatosis (>60%). This graft was found to be severely steatotic (80% steatosis overall) on routine time-zero biopsy; no frozen section was performed prior to implantation. Although in this case the organ functioned well and the recipient experienced a normal posttransplant recovery, severely steatotic livers would not be used in most settings if detected prior to implantation.
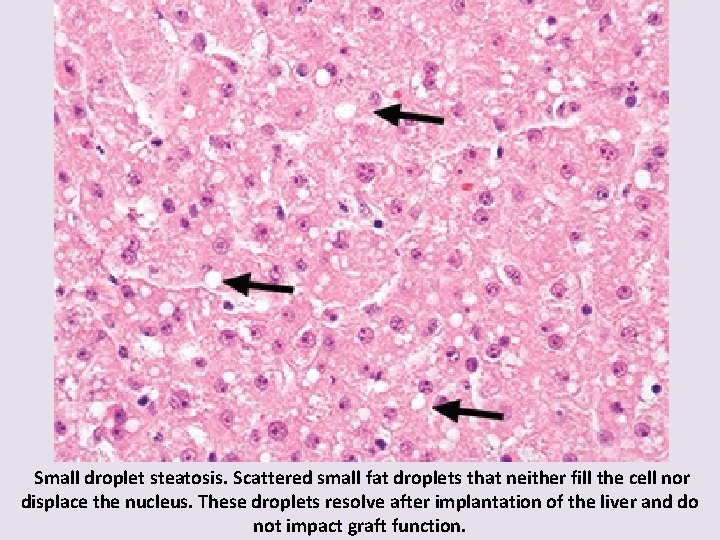
Small droplet steatosis. Scattered small fat droplets that neither fill the cell nor displace the nucleus. These droplets resolve after implantation of the liver and do not impact graft function.
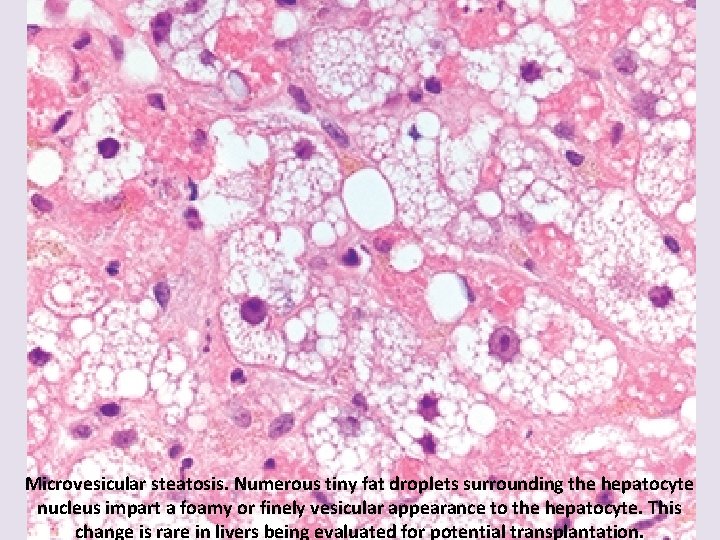
Microvesicular steatosis. Numerous tiny fat droplets surrounding the hepatocyte nucleus impart a foamy or finely vesicular appearance to the hepatocyte. This change is rare in livers being evaluated for potential transplantation.
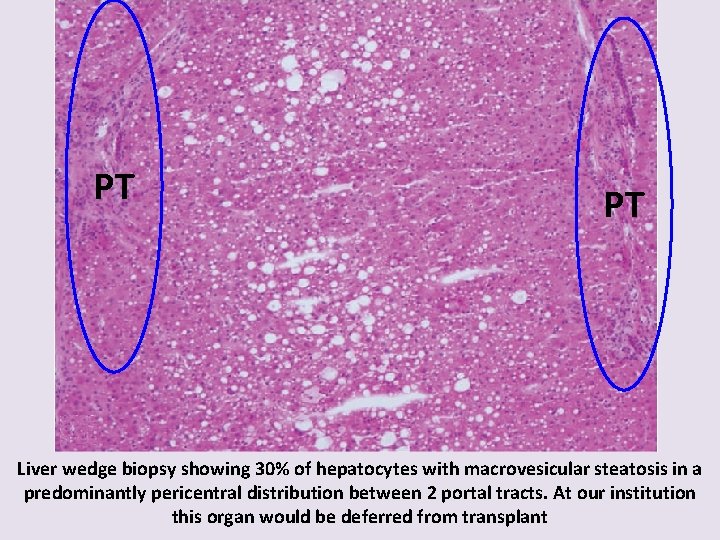
PT PT Liver wedge biopsy showing 30% of hepatocytes with macrovesicular steatosis in a predominantly pericentral distribution between 2 portal tracts. At our institution this organ would be deferred from transplant

Deceased Donors • It can be difficult in some cases to decide whether the fats vacuoles are large (macro-) or small (micro) because the fat globules vary substantially in size • Use nuclear size as a rough gauge to make the distinction between microsteatosis and macrosteatosis

Deceased Donors • Macrovesicular steatosis (>30%) – Increases susceptibility to preservation-reperfusion injury – Impairs regeneration – Is associated with decreased graft survival • Small vacuolar microvesicular steatosis – Often found after a short period of warm ischemia & other insults – Usually does not adversely affect outcome • One study, associated “high-grade” small vacuolar or microvesicular steatosis with delayed graft function

Deceased Donors • The severity of macrovesicular steatosis can be roughly estimated on H & E– stained slides alone • Fat stains – Are not necessary • Automated morphometric analysis – Is not really practical in the frozen section setting • Comparison of the microscopic findings to a standardized diagram illustrating various percentages of macrovesicular steatosis – Improves accuracy and reproducibility of steatosis scoring



Deceased Donors • Most studies confirmed the reproducibility of evaluating donor macrovesicular steatosis by pretransplant frozen section evaluation • Cutoff values <30% can decrease reproducibility • Other modalities used to assess steatosis – Clinical and biochemical parameters – Hepatic CT in conjunction with other noninvasive clinical data, and MRI

Deceased Donors • Use of steatotic donor livers is controversial and varies by center • Most centers disqualify donor livers when macrovesicular steatosis >50% of hepatocyte volume because it has been reliably associated with increased risk for early dysfunction and failure

Deceased Donors • This practice has been questioned, particularly if other risk factors such as cold ischemic time (CIT) or complications are absent or have been mitigated • Scoring macrovesicular steatosis – Evenly distributed range for scoring macrovesicular steatosis • Mild < 30% • Moderate = 30%-60% • Severe>60%) – A scale that more closely reflects the triage algorithm for ECD

Deceased Donors • Algorithm for consideration of multiple factors – Mild donor macrovesicular steatosis (<10%) • Does not at all influence the decision-making process – Moderate macrovesicular steatosis (10% to 30%) • Usually still used for transplantation • But other factors are also taken into consideration in the decision-making process such as ECD characteristics – >30%, or severe steatosis • Are used only under special circumstances – Cold ischemic time is kept to a minimum & – Very few or no other ECD risk factors » The outcome in such situations can be comparable to nonsteatotic donor livers

Deceased Donors • Nonsteatosis ECD factors – With histopathological manifestations • Advanced age (>60 years) • HCV infection • Cardiovascular instability/ischemic injury – Without histopathological manifestations • • Black race Short stature Cerebrovascular cause of death Hypernatremia (>155 m. Eq/L) Cold ischemia time >12 hours Partial-liver allografts DCD status

Macrovesicular Steatosis • The posttransplant effects of steatosis? – Greater susceptibility to ischemia – Buildup of fat deposits in the hepatocytes results in an increased cell volume • May cause sinusoidal obstruction • More frequently found in the elderly – Ischemia/reperfusion injury of donor livers with severe macrovesicular steatosis may cause lysis of steatotic cells • The liberated lipid droplets may then form cystic spaces in liver sinusoids (lipopeliosis) – This results in damage to the graft and loss of function by disrupting hepatic microcirculation, with resultant foci of parenchymal necrosis

Macrovesicular Steatosis • There is some debate whether livers with less severe forms of macrovesicular steatosis are acceptable for transplant • Some studies have encouraged greater caution before approving even mildly macrosteatotic livers – Livers may maintain functionality – Become more susceptible to bleeding & fibrinolysis following transplantation

Macrovesicular Steatosis • Rate of primary nonfunction in 390 frozen section biopsy specimens – Primary nonfunction • 13% of grafts with >30% macrovesicular steatosis • 2. 5% of livers without steatosis • Donor livers that have >30% macrovesicular steatosis should be deferred – The decision is ultimately left to the transplant surgeon

Macrovesicular Steatosis • Fresh biopsies are necessary for making an accurate assessment • If a significant accumulation of artifacts are present and are misinterpreted, they could result in the exclusion of an otherwise acceptable organ

Mcrovesicular Steatosis • Causes of misinterpretation of severity of steatosis at frozen section – Underestimation of steatosis • Air drying – Quickly diminishes the amount of fat detected • Placing the biopsy on a towel or gauze – Can cause fat to leach out of the hepatocytes – Overestimation of steatosis • Placing the biopsy in saline – Freezing of water droplets in the tissue

Deceased Donors Warm ischemia • Should be limited optimally to <20 minutes • Even under ideal circumstances DCD donors are still more susceptible to ischemic cholangiopathy, which usually develops several weeks to months after transplantation

Deceased Donors Warm ischemia • Frozen section evaluation is rarely helpful in evaluating DCD donors because – Organ reperfusion is required for histopathological manifestations of necrosis and inflammation to be recognized – The compartment most susceptible to injury, the biliary tree, is not practically accessible for needle biopsy evaluation

Deceased Donors Necrosis • Necrosis in donor biopsies has negatively impacted recipient outcome in some but not all studies • An algorithmic approach or reproducible method of quantifying the necrosis has not been reported and might account for the different observations • Donor liver is usually disqualified if necrosis involves – >20% of hepatocytes & – Diffusely involves both the wedge and needle cores

Deceased Donors Necrosis • Immediately subcapsular necrosis with neutrophilic inflammation is common in donor organs and should not be included in the assessment • The extent of necrosis should be correlated with serial liver injury tests in the donor • Caution is urged when necrosis on donor biopsy findings is combined with rising ALT & AST • The biopsy findings might not accurately represent the final extent of the necrosis

Deceased Donors HCV-positive donors • Many centers use mildly diseased HCV+ donors to prolong the life of a HCV+ recipient with end-stage HCV–induced liver failure • Rarely use mildly diseased HCV+ donors in HCVrecipients with fulminant hepatic failure • Graft and patient survival outcomes have been minimally impacted by donor HCV status, but more rapid progression of fibrosis can occur

Deceased Donors HCV-positive donors • All HCV+ donors are subjected to frozen section biopsy analysis • Those with nonbridging fibrosis (<3/6, Ishak scale) are offered to potential recipients after informed consent • Other groups report one stage lower fibrosis cutoff (<2/6) • The advent of new, more effective, anti-HCV therapy will likely significantly alter this process

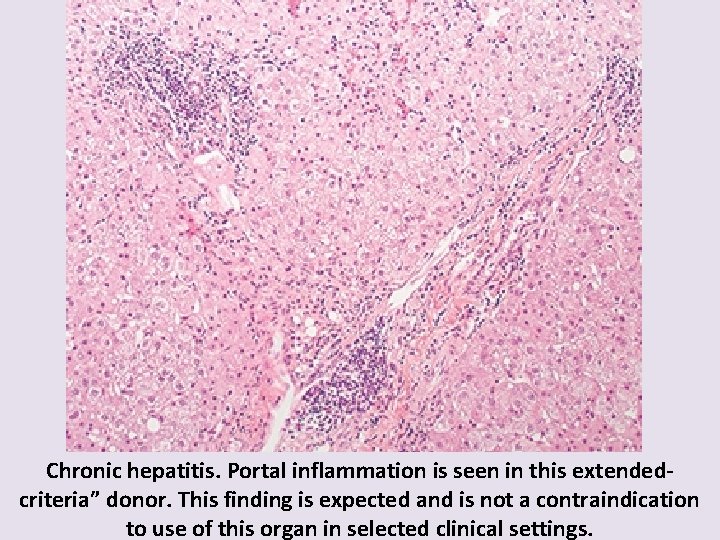
Chronic hepatitis. Portal inflammation is seen in this extendedcriteria” donor. This finding is expected and is not a contraindication to use of this organ in selected clinical settings.

Deceased Donors HBV-positive donors • Anti-HBc–positive donors can transmit HBV to naive and unvaccinated recipients • The risk is lower in vaccinated anti-HBc+ recipients and can be further reduced by anti-HBV medications and passive antibodies • Donor biopsy evaluation is generally not helpful in this circumstance because the vast majority of biopsies have normal findings in the absence of other diseases

Deceased Donors Inadvertently transferred diseases • A variety of neoplastic, infectious, and metabolic diseases have been inadvertently transferred from donors to recipients – Cancers – Amyloidosis – Hemochromatosis – Fungal diseases – Viral diseases – Parasitic diseases

Deceased Donors Inadvertently transferred diseases • Metabolic diseases can be intentionally or unintentionally transferred with the donor organ in “domino” transplants with the expectation that the latency period between transplantation and recipient disease onset is gained life years – Familial amyloid polyneuropathy – Oxalosis – Possibly α 1 -antitrypsin deficiency

Deceased Donors Inadvertently transferred diseases • Revisiting any donor data in recipients with unexpected complications can potentially provide valuable insights

• A host of other unusual and unexpected findings, including alpha-1 antitrypsin deficiency, amyloidosis, histoplasmosis, and varying degrees of iron overload, have also been reported in living and cadaveric donor livers • While a few cases of “recurrent” iron overload have been reported with use of hemochromatotic donor livers, in other reports organs with iron and other pathologies such as alpha-1 antitrypsin deficiency have been used successfully

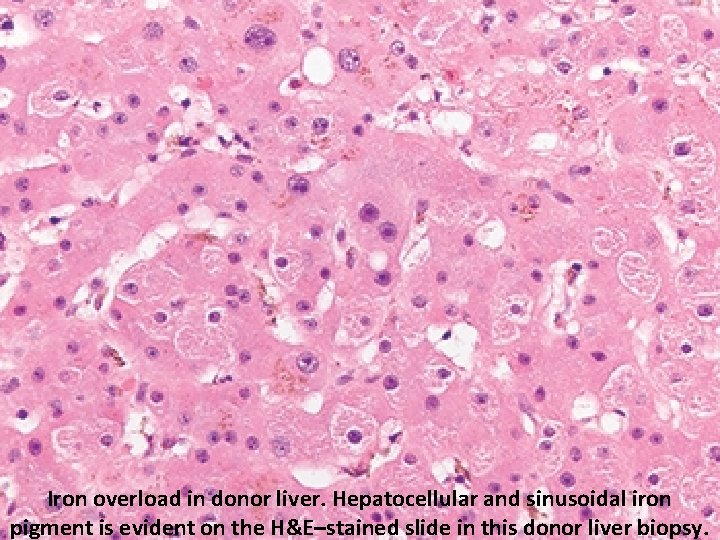
Iron overload in donor liver. Hepatocellular and sinusoidal iron pigment is evident on the H&E–stained slide in this donor liver biopsy.
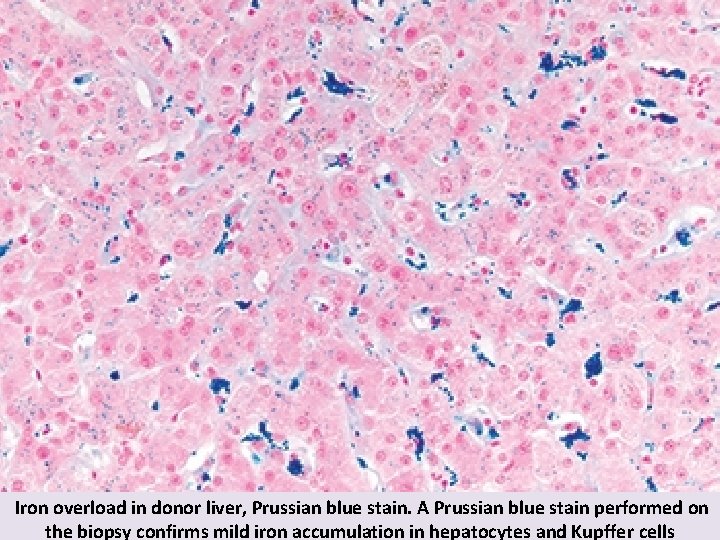
Iron overload in donor liver, Prussian blue stain. A Prussian blue stain performed on the biopsy confirms mild iron accumulation in hepatocytes and Kupffer cells
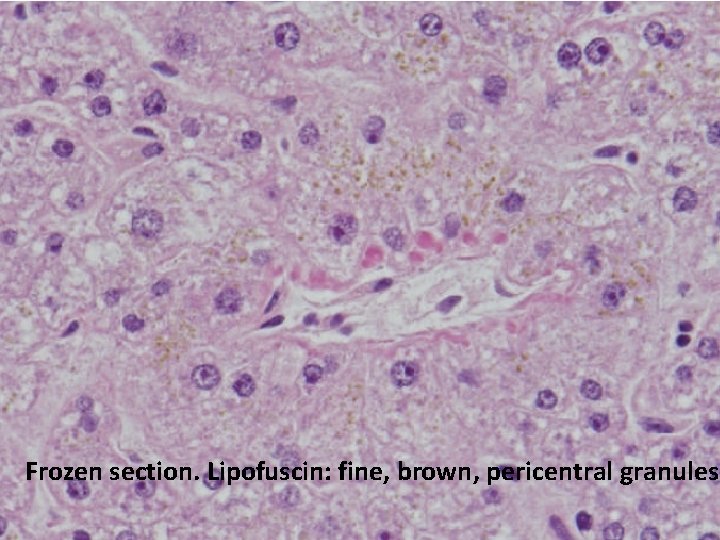
Frozen section. Lipofuscin: fine, brown, pericentral granules
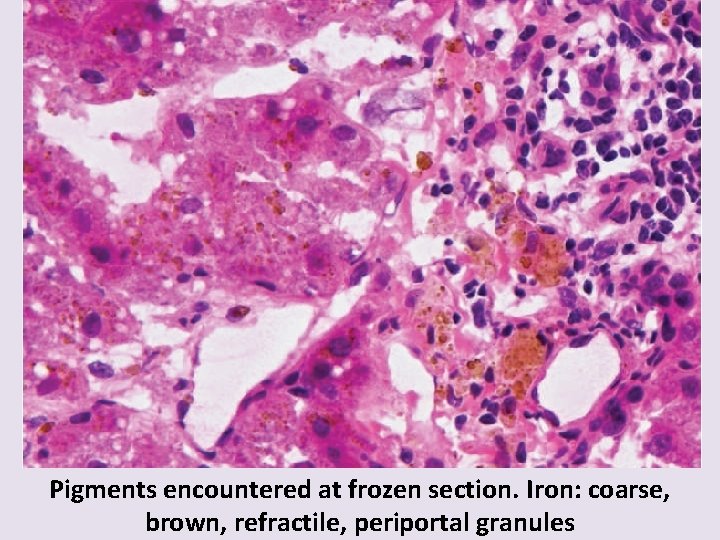
Pigments encountered at frozen section. Iron: coarse, brown, refractile, periportal granules
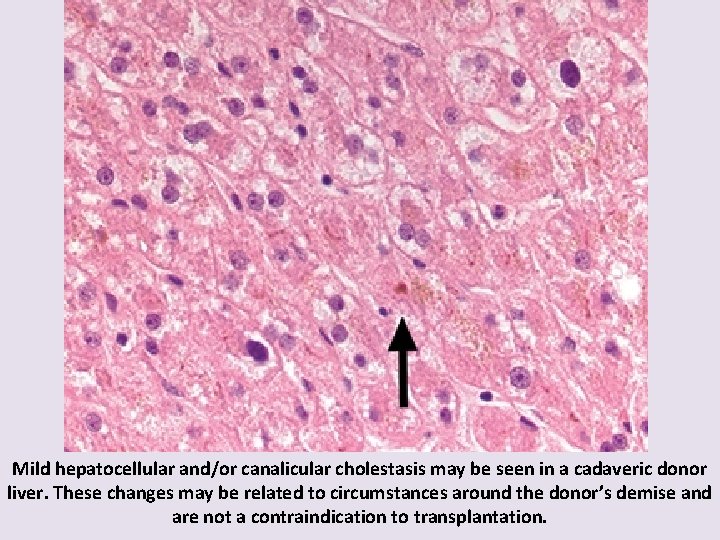
Mild hepatocellular and/or canalicular cholestasis may be seen in a cadaveric donor liver. These changes may be related to circumstances around the donor’s demise and are not a contraindication to transplantation.
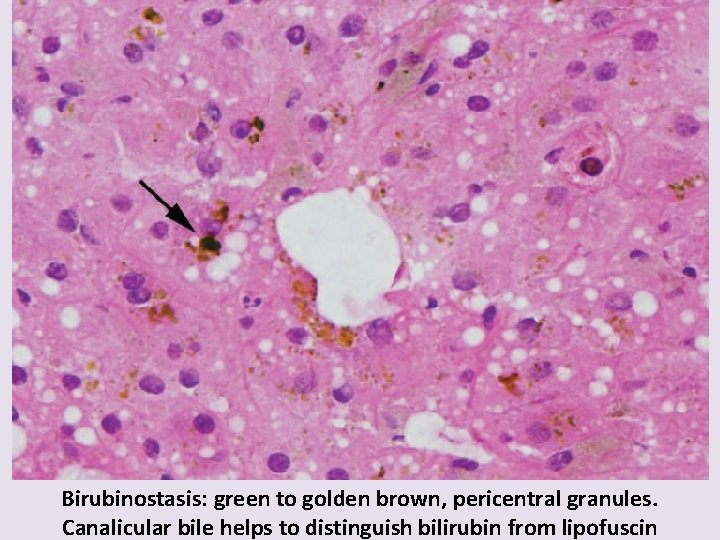
Birubinostasis: green to golden brown, pericentral granules. Canalicular bile helps to distinguish bilirubin from lipofuscin







- Slides: 108